Abstract
A novel method for preparing Ni2P/Al2O3(L) catalysts in an N2 atmosphere by decomposition of hypophosphites was proposed, and Ni2P/Al2O3(T) catalyst was synthesized by the temperature programmed reduction method in a H2 atmosphere for comparison. These prepared catalysts were washed with deionized water to remove impurities. The X-ray diffraction, N2-adsorption specific surface area measurements, CO uptake, and X-ray photoelectron spectroscopy were applied to characterize these catalysts. The activities of the Ni2P/Al2O3 catalysts prepared with the two different methods were tested in the dibenzothiophene hydrodesulfurization reaction.
Introduction
With the increasingly stringent environmental regulations in recent years, hydrodesulfurization (HDS), as the most used method to remove sulfur (S) in the petroleum refining industry, has attracted much attention.1,2 The efficiency of the HDS method was highly influenced by the applied catalysts. 3 Thus, the academic and industrial communities have tested a series of catalysts, such as noble metal catalysts, Mo or MoS2 catalysts, and transition metal phosphides catalysts. Among these, Ni2P with high activity and easy access has been reported as the most promising catalyst for the HDS process. 4
The traditional method for Ni2P preparation was a temperature programmed reduction (TPR) in an H2 atmosphere.5,6 However, a heat treatment method with hypophosphite in an N2 atmosphere has roused great interest. 7 Compared with the Ni2P catalyst synthesized with hypophosphites in an N2 atmosphere, the synthesis of Ni2P from hypophosphites in H2 atmosphere was expensive and complex.
In this article, a simple and novel heat treatment method for synthesizing Ni2P catalyst was proposed. The Ni2P/Al2O3(L) catalyst was synthesized by direct heating to the preparation temperature with NaH2PO2 and NiCl2·6H2O in a flow of N2 . For comparison, Ni2P/Al2O3(T) catalyst was prepared with NaH2PO2 in an H2 atmosphere with the TPR method. The effects of the preparation method on the structure and dibenzothiophene (DBT) hydrodesulfurization (HDS) catalytic activity of the prepared catalysts and washed catalysts were investigated.
Experimental
Preparation of catalysts
The catalyst precursor was prepared via impregnating nickel chloride (NiCl2·6H2O) and sodium hypophosphite (NaH2PO2) on an Al2O3 support (initial P/Ni molar ratio = 2, Ni = 10 wt.%). In a typical process, 0.44 g of NiCl2·6H2O and 0.60 g of NaH2PO2 were dissolved in 20 mL of deionized water. The 2.0-g Al2O3 support was impregnated with the above solution for 12 h and then evaporated at 80°C. The resulting precursor was reduced by different methods. The precursor obtained by directly heating to 300°C and being kept for 2 h in an N2 atmosphere was denoted as Ni2P/Al2O3(L). This precursor was heated to 300°C at a rate of 2°C/min and kept for 2 h in flowing H2 (100 mL/min) to form the catalyst denoted as Ni2P/Al2O3(T). Both catalysts were passivated at room temperature in an O2/N2 mixture (1.0 mol% of O2) at 20 mL/min for 1 h. The catalysts were then washed with deionized water 3 times to remove impurities. The washed catalysts were denoted as Ni2P/Al2O3(L-W) and Ni2P/Al2O3(T-W), respectively. Washing the catalysts could expose much more active site and thus increase the surface area, which is beneficial to reveal the intrinsic reaction.
Characterization of catalysts
X-ray diffraction (XRD) data were collected on a D/max-2200 PC X-ray diffractometer using Cu Kα radiation. The 2θ scans covered the range 10–80°. The typical physicochemical properties were analyzed using a Quantachrome Autosorb-1-MP analyzer. CO chemisorption uptake was performed in a Micromeritics ASAP 2010 apparatus. X-ray photoelectron spectroscopy (XPS) spectra were recorded with a ESCALAB MKII spectrometer equipped with a hemispherical analyzer using monochromatic Mg Kα radiation (true = 1253.6 eV).
Catalytic activities
The catalytic activity was tested with an HDS reaction in a fixed-bed reactor with 0.5 wt.% DBT in decalin. Catalytic activity was measured at 320°C, 4.0 MPa, hydrogen/oil ratio of 500 (V/V), and weight hourly space velocities (WHSVs) of 8.0 h−1. The products were analyzed by an Agilent-6890N gas chromatograph equipped with a flame ionization (FID) detector using a commercial HP-5 column.
Results and discussion
XRD
Figure 1 shows the XRD patterns of the Al2O3 support, Ni2P/Al2O3(L) and Ni2P/Al2O3(T) catalysts. The broad peaks at 2θ = 37.5°, 45.5°, and 66.1° belong to Al2O3. The sharp peak at 40.6° belongs to Ni2P (PDF: 03-0953). No other peak related to Ni or P appeared, indicating that both methods could produce a pure Ni2P active phase. The average size of the Ni2P crystallite (Dc) calculated with the Scherrer equation is shown in Table 1. The average size of Ni2P/Al2O3(L) catalyst was 18.4 nm, that is smaller than that for Ni2P/Al2O3(T) catalyst (22.6 nm). This result indicated that the heat treatment method forming a Ni2P phase with the strongly reducing gas PH3 was beneficial to produce a smaller average crystallite size. 8
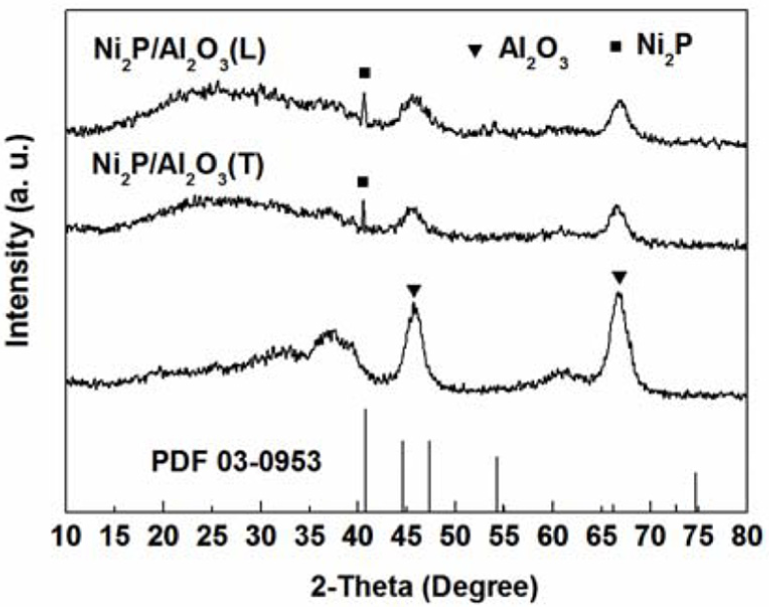
XRD patterns of the support and catalysts.
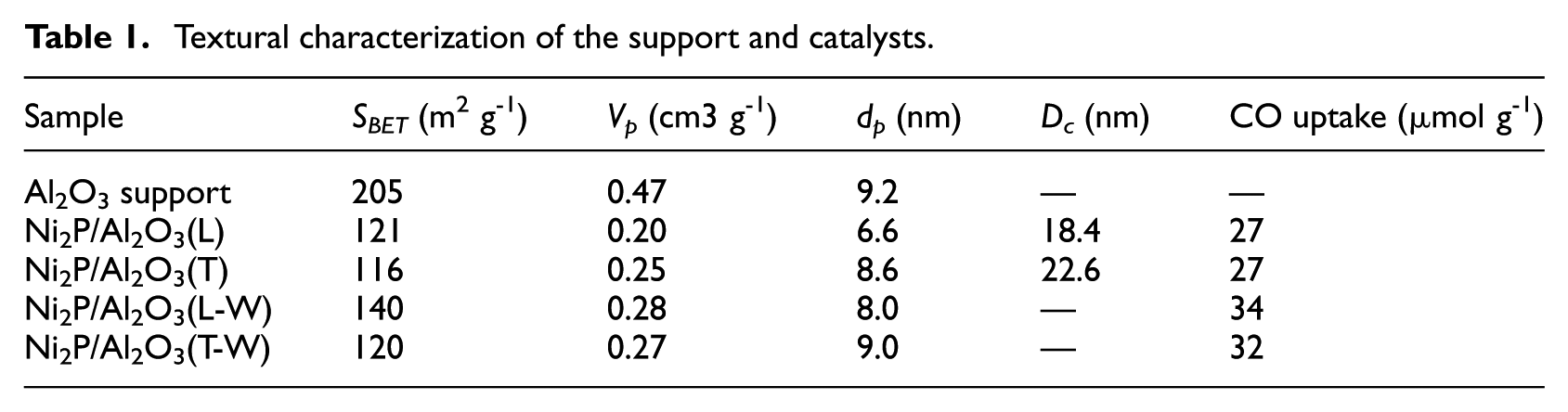
Textural characterization of the support and catalysts.
BET
Table 1 shows the Brunauer–Emmett–Teller (BET) results for the Al2O3 support and catalysts. The surface area (SBET) and pore volume (Vp) of the Al2O3 support were 205 m2 g−1 and 0.47 cm3 g−1, respectively; however, the SBET and Vp values of the Ni2P/Al2O3 catalysts decreased significantly because of the blockage of pores by loading the nickel phosphide phase. The SBET of the Ni2P/Al2O3(L) was 121 m2 g−1, higher than that for the Ni2P/Al2O3(T) catalyst (116 m2 g−1). This result may be caused by the run over of PH3—releasing pores blockage. The SBET and Vp of Ni2P/Al2O3(L) and Ni2P/Al2O3(T) showed an increase after washing. What deserves our attention was that the SBET of Ni2P/Al2O3(L) catalyst had a significant increase from 121 to 140 m2 g−1after washing. These results indicate that the support pores had been blocked by the reaction by-products such as P2O3 and/or H3PO3, which were dissolved in the washing process to increase SBET and Vp. 9
CO uptake
The CO molecules are mainly adsorbed at Ni-active sites; therefore, the CO uptake measurements were applied to “titrate” the number of Ni-active sites exposed on the catalyst surface and their dispersion on the catalysts.10,11 The CO uptakes of the catalysts are listed in Table 1. The CO uptakes of Ni2P/Al2O3(L) and Ni2P/Al2O3(T) were both 27 μmol g−1, which indicated that the two catalysts prepared from heat treatment and the TPR method had exposed similar Ni sites. Also, the CO uptakes of the two catalysts had increased significantly after washing. The CO uptakes of Ni2P/Al2O3(L-W) and Ni2P/Al2O3(T-W) were 34 and 32 μmol g−1, respectively. This result may be caused by excluding excess phosphorus atoms as well as increasing SBET after washing.
XPS
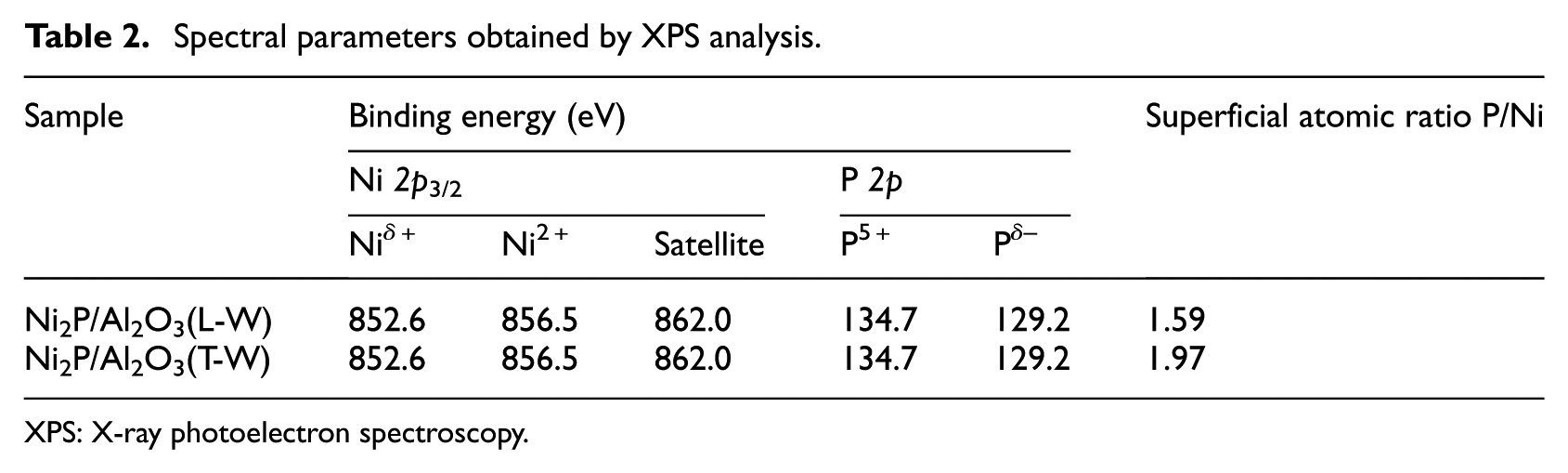
The XPS spectra of the Ni (2p) and the P (2p) regions of the washed catalysts are shown in Figure 2. The surface compositions and the corresponding binding energies are reported in Table 2. In all samples, the spectra showed Niδ+ (0 < δ < 2) and Pδ− (0 < δ < 1) species associated with the Ni2P phase. The binding energies of the Ni 2p (856.5 eV) and P 2p (134.7 eV) belong to Ni2+ and P5+ species, respectively. The peaks at 852.6 and 129.2 eV are attributed to Niδ+ (0 < δ < 2) and Pδ− (0 < δ < 1) species, respectively. 12 We found that the peak intensity of the Niδ+ and Pδ− for Ni2P/Al2O3(L-W) was higher than that for Ni2P/Al2O3(T-W), which suggested that the exposure of the Ni2P phase on Ni2P/Al2O3(L-W) was higher than that for Ni2P/Al2O3(T-W). This result was in agreement with the CO uptake discussed in the “CO uptake” section.
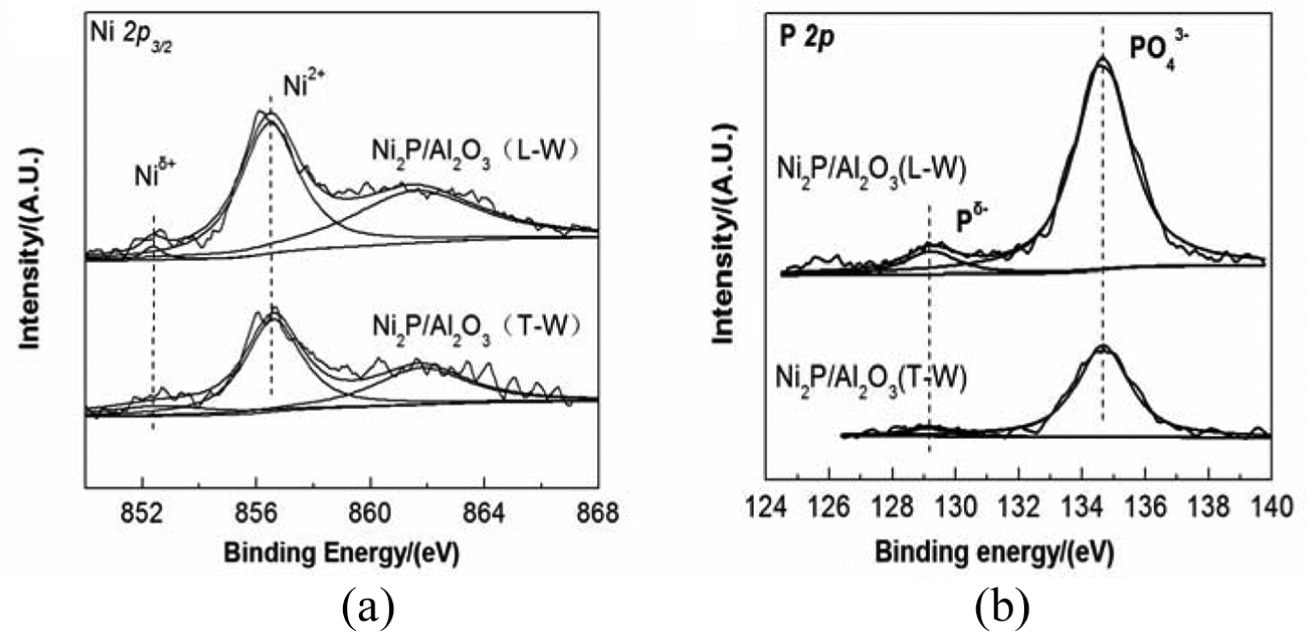
XPS spectra of the catalysts.
Spectral parameters obtained by XPS analysis.
XPS: X-ray photoelectron spectroscopy.
The surface P/Ni atomic ratios calculated from XPS analyses are shown in column 7 on Table 2. The theoretical P/Ni ratio was 1/2 for the two catalysts. But the catalysts exhibited higher P/Ni than 1/2, which was caused by the enrichment of phosphorous on the surface of the catalysts. The superficial P/Ni ratio of Ni2P/Al2O3(L-W) catalyst was 1.59, which was lower than that of Ni2P/Al2O3(T-W). This result may be caused by the release of PH3 during the heat treatment process. 13 This was in accordance with the CO uptake results and peak intensities.
HDS activity
The catalytic activity and stability test of the catalysts were evaluated with the HDS of DBT, and the results are given in Figure 3. The DBT conversion over the Ni2P/Al2O3(L) and Ni2P/Al2O3(T) were 83.9% and 81.4% after the stream became stable, respectively. The activities increased after washing; those for Ni2P/Al2O3(L-W) and Ni2P/Al2O3(T-W) were 94.5% and 86.1%, respectively. This result may be caused by the better dispersion and smaller size of the Ni2P phase; on the other hand, the higher surface area and lower surface P-rich were also beneficial to the catalytic activity.
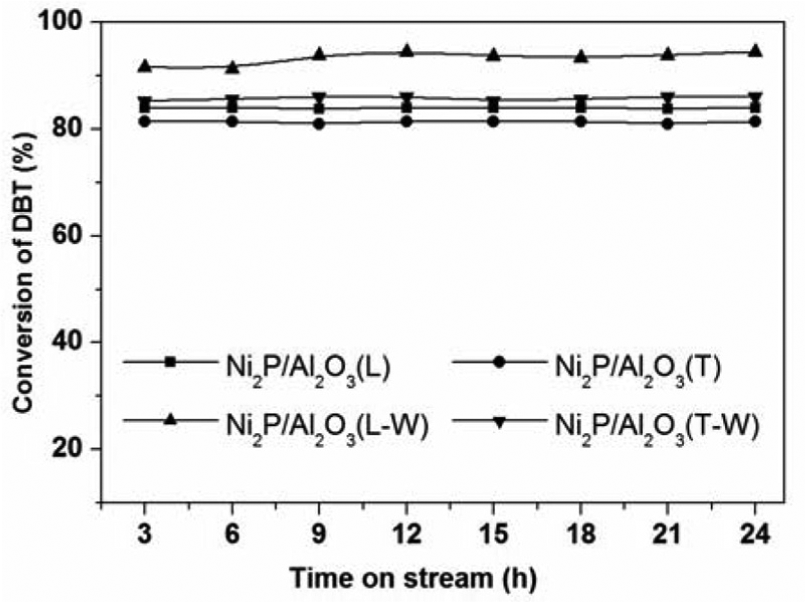
HDS activity of the catalysts.
The HDS selectivity over the catalysts is shown in Figure 4. The products detected were biphenyl (BP) and cyclohexylbenzene (CHB). The selectivity for BP was much higher than that for CHB. According to the literature, 14 the BP is produced from DBT desulfurization (DDS) pathway at Ni(I) sites, and CHB is converted from DBT hydrogenation (HYD) pathway at Ni(II) sites. Therefore, the Ni sites on the prepared catalysts were mainly Ni(I) sites. The heat treatment method in an N2 atmosphere could lead to many more Ni(I) sites than the TPR method. These activity reaction results have great significance to the design and modification of Ni2P catalysts.
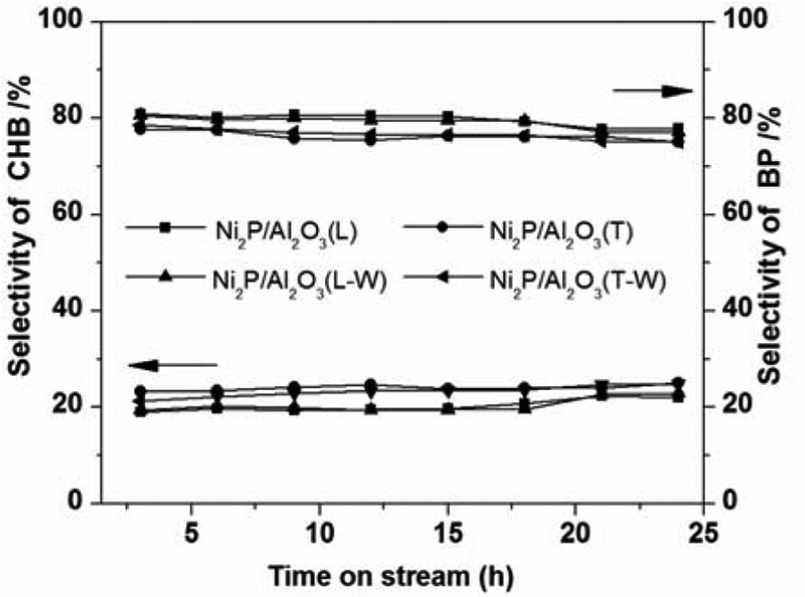
HDS selectivity of the catalysts.
Conclusion
Ni2P/Al2O3(L) catalyst with high HDS activity has been prepared in an N2 atmosphere by decomposition of NaH2PO2 with NiCl2·6H2O. XRD analysis has shown that the Ni2P/Al2O3(L) catalyst has developed a pure Ni2P phase. The crystallite size of Ni2P/Al2O3(L) was smaller than that of Ni2P/Al2O3(T) catalyst according to Scherrer’s equation. BET results showed that Ni2P/Al2O3(L) exhibited a larger surface area than Ni2P/Al2O3(T) catalyst. Also the surface area shows an obvious increase after washing with deionized water. XPS results indicated that the enrichment of P on the surface decreases on Ni2P/Al2O3(L-W) catalyst, which exposed more nickel atoms, leading to a higher CO uptake. The highest DBT HDS activity was 94.5%, as achieved over Ni2P/Al2O3(L-W) at a condition of 320°C, 4.0 MPa, H2/oil = 500 (V/V), and WHSV = 8.0 h−1.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge Education Department of Liaoning Province (Project No.: LJZ2016002) and Natural Science Foundation of Liaoning Province (Project No.: 20180550432) for financial support to this work.
