Abstract
A series of Ti-incorporated bulk Ni2P catalysts was prepared by means of temperature-programmed reduction, and the role of metallic Ti on the structure and catalytic activity of the Ni2P catalysts was studied. For this purpose, bulk Ni2P catalysts with metal Ti contents of 0.005 wt%, 0.01 wt%, and 0.02 wt% were synthesized. X-ray diffraction, CO uptake, Brunauer–Emmett–Teller measurements, and X-ray photoelectron spectroscopy were utilized to characterize the catalysts. Addition of titanium could increase the surface area and promote the formation of small, highly dispersed Ni2P particles. The Ti0.02-Ni2P system with a Ti molar fraction of 0.02 showed the highest hydrodesulfurization activity of 99.6%, which was an increase of 44% compared with that found for the bulk Ni2P.
Introduction
Hydrodesulfurization (HDS) is a catalytic chemical process most widely used, among all the sulfur removal methods, to remove sulfur atoms from sulfur-containing molecules present in oil feedstocks. 1 It has been realized that current commercial HDS catalysts are insufficient to meet the required standards,2,3 and according to the restrictions imposed by SO x regulations and the continuing decline in the quality of petroleum raw materials, HDS has become one of the most important processes in the refining industry. 4
Recently, the transition metal phosphides, as a new class of materials, have attracted wide attention as a new generation of HDS catalysts for their high HDS activity for fuels5,6 and high thermal stabilities. The observed activity levels decrease as follows: Ni2P > WP > MoP > CoP > Fe2P. Accordingly, Ni2P has aroused particular attention due to its high activity in the HDS of dibenzothiophene (DBT). 6
In order to improve the catalytic performance, a third component has been added to the Ni2P catalyst. 7 Our previous research showed that yttrium, 8 zinc, neodymium, 9 and calcium as elements can increase the surface area of the catalyst, resulting in smaller crystal size and better dispersion of active catalytic particles. It was found that the HDS activity of co-precipitation Ni2P catalysts can be enhanced by the addition of Ti. Silva-Rodrigo et al. 10 reported that NiMo/MCM-41 catalysts incorporated with Ti display high activity in HDS. Ti species can enhance the thiophene conversion; moreover, it can improve its stability under hydrothermal conditions. Consequently, it is of great interest to investigate the influence of the Ti content on the performance of Ni2P catalysts for the HDS of refractory sulfur compounds such as DBT. Ti was completely added to the catalyst skeleton to form weak Lewis acid sites, producing a larger pore, mesoporous, structure. The changes to the framework were significantly advantageous in enhancing the catalytic properties of catalysts for transesterification of fuels. 11 The increase in Ti content leads to a rise in the amount of framework Ti sites and can thus modify the acidity of the catalysts.
In the present work, a series of Ti-incorporated Ni2P catalysts was prepared by the temperature-programmed reduction (TPR) method, and the effect of Ti content on the HDS performance of bulk Ni2P was studied using DBT as the model compound.
Experimental
Preparation of catalysts
The Ni2P catalysts were prepared by the co-precipitation method according to Song and co-workers8,9 Briefly, the oxidic precursor of bulk Ni2P was prepared with excess phosphorus, the mole ratio of nickel and phosphorus being 1. 12 Redundant phosphorus was used to effect the loss of phosphorus during the reduction stage. In a typical preparation, 2.52 g Ni(NO3)2·6H2O and 1.77 g (NH4)2HPO4 were dissolved in deionized water. The mixture was stirred moderately while the water was evaporated to yield a solid product. The resulting solid was heated at 120°C for 12 h and calcined at 500°C for 3 h to obtain the oxidic precursor. The oxidic precursor is denoted as PNi2P. The different dosages of Ti(OC4H9)4 were dissolved in ethylene glycol (the concentrations being 0.000424, 0.000085, and 0.00017 mol L−1, respectively) and then were impregnated into the PNi2P to form solid product. Then, the precursors were reduced and passivated using the method described elsewhere. 9 The corresponding catalysts are denoted as Ti x -Ni2P, where the subscript x is the molar fraction of Ti with respect to Ni2P and has values of 0.005, 0.01 and 0.02. For comparison, a bulk Ni2P catalyst was prepared following the method described above.
Characterization of catalysts
X-ray diffraction (XRD) was used to analyze the catalyst samples, and analysis was carried out on a D/Max 2200 PC X-ray diffractometer and needs manufacturer using Cu Kα radiation under setting conditions of 40 kV, 30 mA and a scanning range from 10 to 80° at a rate of 10°min-1.
The typical physicochemical properties of the catalysts were analyzed by the Brunauer–Emmett–Teller (BET) method using Micromeritics adsorption equipment model NOVA 2000e. All samples were outgassed at 473 K until the vacuum pressure was 6 mm Hg. The adsorption isotherms for nitrogen were measured at 77 K.
The X-ray photoelectron spectroscopy (XPS) spectra were acquired using an ESCALAB MKII spectrometer under vacuum. XPS measurements were performed using monochromatic Mg Kα radiation (E = 1253.6 eV) and the spectrometer was equipped with a hemispherical analyzer operating at a fixed pass energy of 40 eV. The recorded photoelectron binding energies were referenced against the C 1s contamination line at 284.8 eV.
Catalytic activity test
The HDS of DBT was measured using a feed consisting of a decalin solution of DBT (1 wt%) in a flowing fixed-bed reactor at high pressure. The conditions of the HDS reaction were 613 K, 3.0 MPa, weight hourly space velocity (WHSV) = 1.5 h−1, and a hydrogen/oil ratio of 700 (V/V). The catalyst was pressed into small pieces, crushed, then and sieved to 30–60 mesh. In the reaction, sampling of liquid products was started 2 h after the steady reaction conditions had been achieved. Liquid samples were collected every hour and a flame ionization detector gas chromatograph with a GC-14C–60 column was used to analyze the samples. According to the ratio of converted DBT to initial DBT, the total conversion was calculated. The turnover frequency (TOF) values of the samples containing nickel phosphide were calculated using equation (1) 13
where F is the molar rate of DBT fed into the fixed-bed reactor (mol s−1), W is the weight of catalyst (g), X is the conversion of DBT (%), and M is the number of moles of sites loaded on the catalyst which was determined by the CO uptake.
Results and discussion
XRD
XRD patterns of samples with different Ti loadings are shown in Figure 1. For all the samples, the peaks at 2θ = 40.7, 44.6, 47.3, 54.1, and 54.9° are assigned to the Ni2P phase. For all samples, what can be inferred is that the active phase formed was mainly Ni2P. Weak peaks of Ni5P4 were observed in the bulk Ni2P sample. However, for samples with Ti, the peaks of Ni5P4 disappeared, indicating that the addition of Ti into the bulk Ni2P could inhibit the formation of the Ni5P4 phase. With increasing content of Ti, the Ni2P peaks of Ti x -Ni2P became wider, which indicates that Ti can promote the formation of a smaller Ni2P active phase. The value of the average crystallite size (Dc) Dc was calculated from Scherrer’s equation and the results are listed in Table 1. As can be seen from Table 1, the Dc values of all the Ti x -Ni2P samples were smaller than those found for the bulk Ni2P. In addition, Dc of the Ti x -Ni2P decreased with increase of the Ti loading. This demonstrated that the incorporation of Ti can promote the formation of smaller particles of Ni2P.
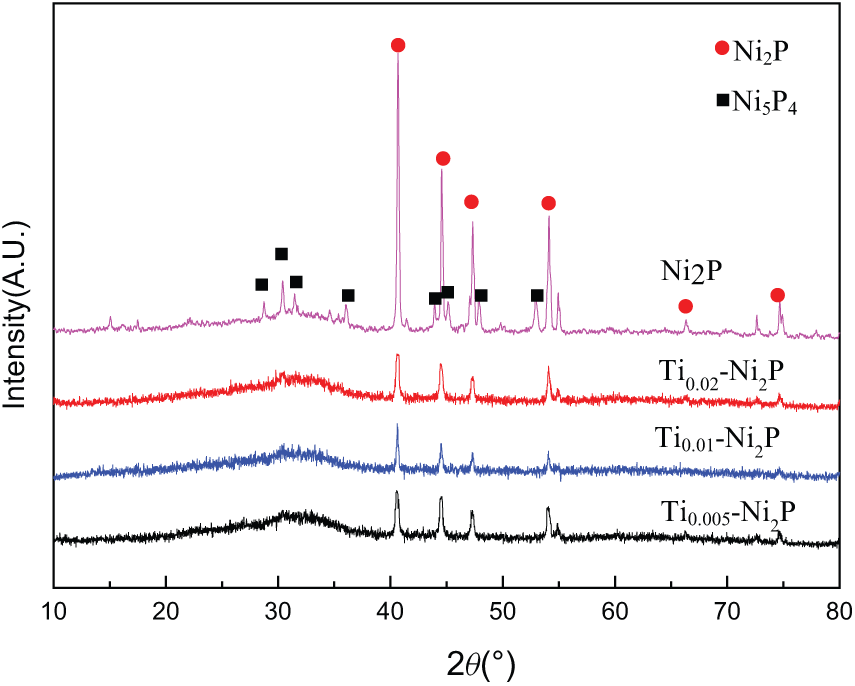
XRD patterns of bulk Ni2P and Ti x -Ni2P catalysts.
Properties and HDS catalytic performance of the bulk Ni2P and Ti x -Ni2P catalysts.
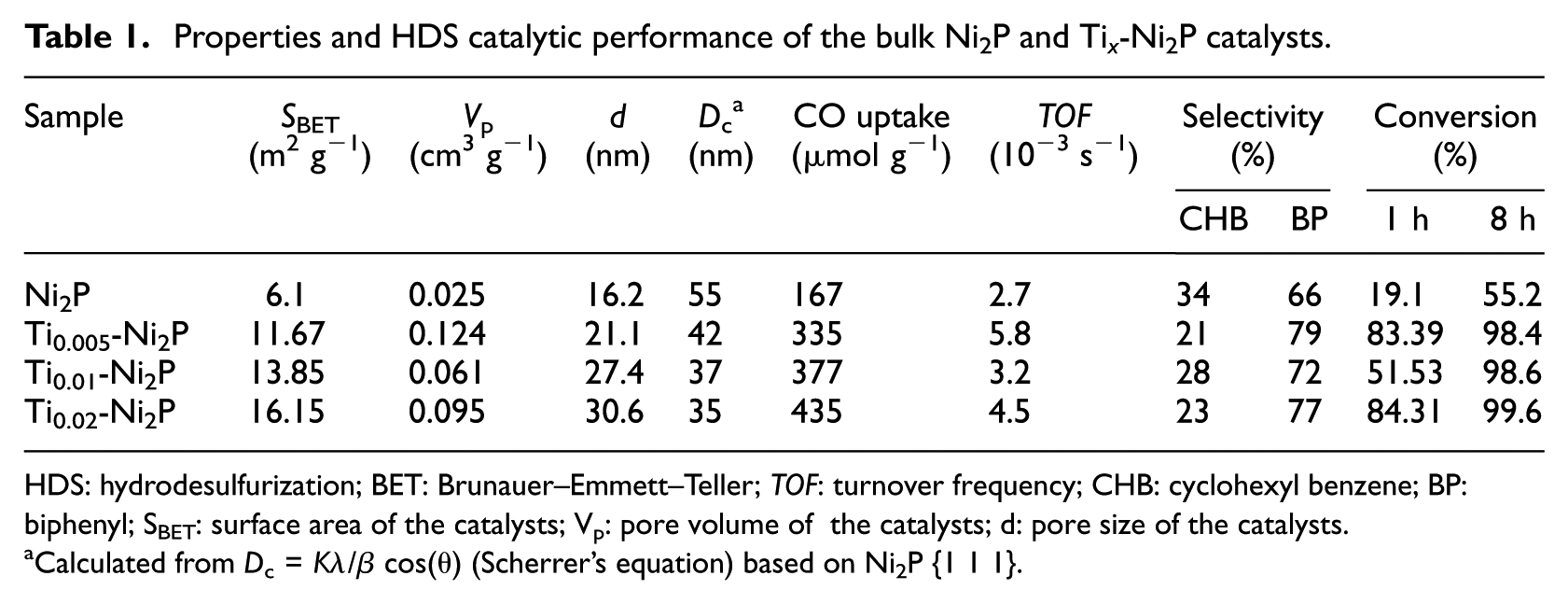
HDS: hydrodesulfurization; BET: Brunauer–Emmett–Teller; TOF: turnover frequency; CHB: cyclohexyl benzene; BP: biphenyl; SBET: surface area of the catalysts; Vp: pore volume of the catalysts; d: pore size of the catalysts.
Calculated from Dc = Kλ/β cos(θ) (Scherrer’s equation) based on Ni2P {1 1 1}.
BET
The textural characterizations of Ni2P and Ti x -Ni2P catalysts are summarized in Table 1. The BET surface area and porosity measurements were performed by N2-adsorption analysis. The surface area and pore volume of the Ni2P catalysts were 6.1 m2 g−1 and 0.025 cm3 g−1, respectively. 14 As the Ti content increased, the specific surface area of Ti x -Ni2P increased, and the pore diameter showed the same trend while the diameter of the particles (Dc) decreased, indicating that Ti metal promoted the formation of smaller and more highly dispersed Ni2P particles in the catalyst. The surface area of the Ti0.02-Ni2P catalyst was the greatest, being almost twice that found for bulk Ni2P. As for the pore volume, that of the Ti0.005-Ni2P catalyst was the greatest, being approximately five times more than that found in bulk Ni2P.
CO uptake
The CO uptakes for the Ni2P and Ti x -Ni2P catalysts were measured at room temperature, and the results are presented in Table 1 (the CO molecules attached to the phosphorus sites can be ignored 15 ). As shown in Table 1, the CO uptake of the bulk Ni2P was 167 μmol g−1. What is notable is that the CO uptakes of the Ti x -Ni2P catalysts were markedly higher than the CO uptake of the bulk Ni2P catalyst. Thus, the CO uptake of Ti0.02-Ni2P was 435 μmol g−1 which was approximately three times that of bulk Ni2P. The values for Ti0.005-Ni2P and Ti0.01-Ni2P were 335 and 377 μmol g−1, respectively, which showed that incorporating Ti into Ni2P can improve the dispersion of active Ni2P particles due to the smaller size (XRD analysis) and higher surface area (BET analysis) of Ti x -Ni2P. This will be discussed in detail in the XPS analysis section.
XPS
The XPS spectra of all the catalysts are shown in Figure 2, and the binding energies and the P/Ni atomic ratios are shown in Table 2. As in previous studies,16,17 for all catalysts, the Ni 2p3/2 core-level spectrum consists of two components, of which one is assigned to Nid+ in the Ni2P phase and is centered at 851.4–853.5 eV and the second is at 855.4–856.7 eV. As a consequence of superficial passivation, the interaction of Ni2+ ions with phosphate ions corresponded to the core-level spectrum, along with broad satellite peaks at almost 6 eV higher than the binding energy of the parent signal,17,18 and this structured peak is ascribed to a divalent species. Simultaneously, other broad peaks centered at the high binding energy side can be assigned to the Ni 2p signal from oxidized Ni species. 19
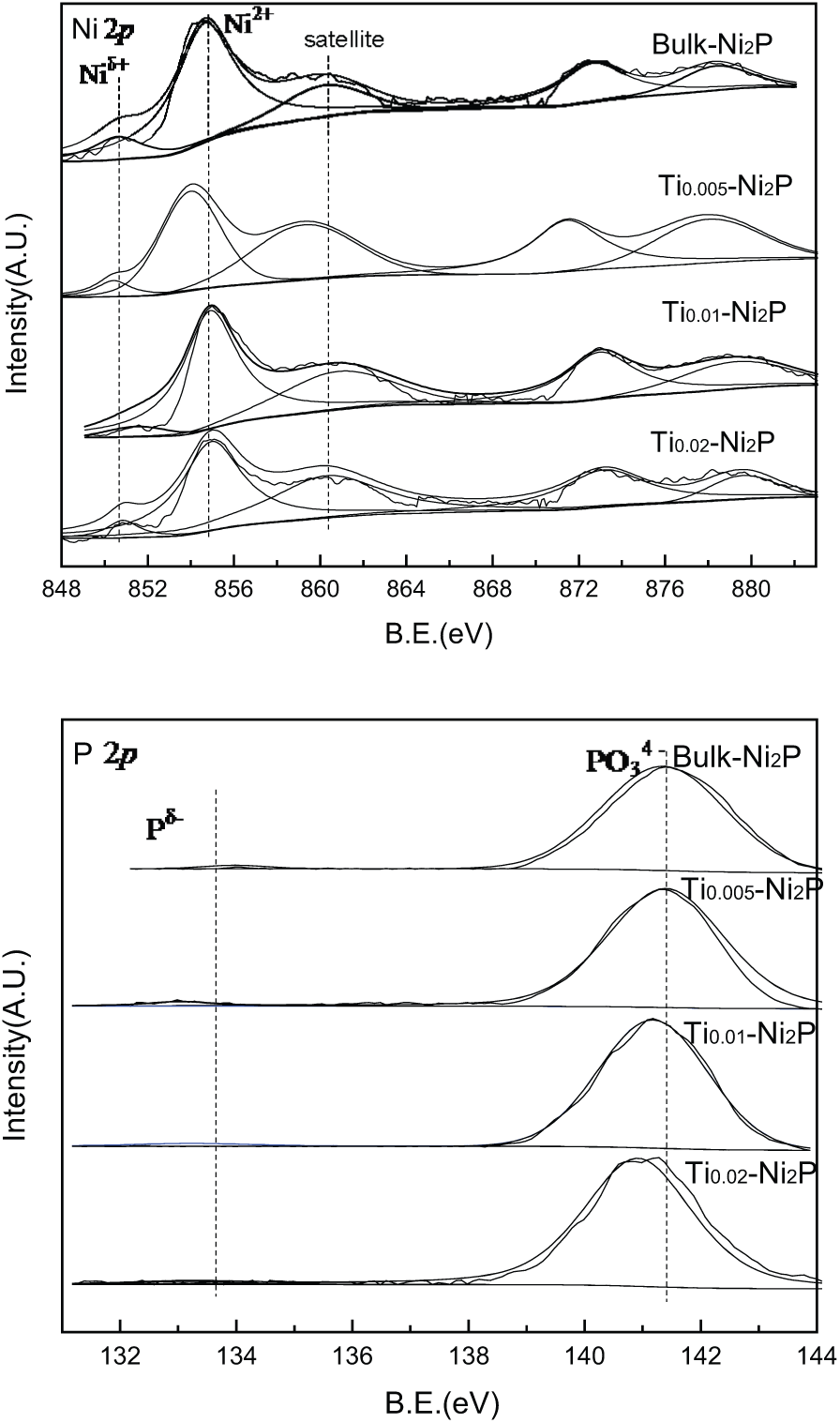
XPS spectra of Ni2P and Ti x -Ni2P catalysts.
Spectral parameters obtained by XPS analysis for bulk Ni2P and Ti x -Ni2P catalysts.
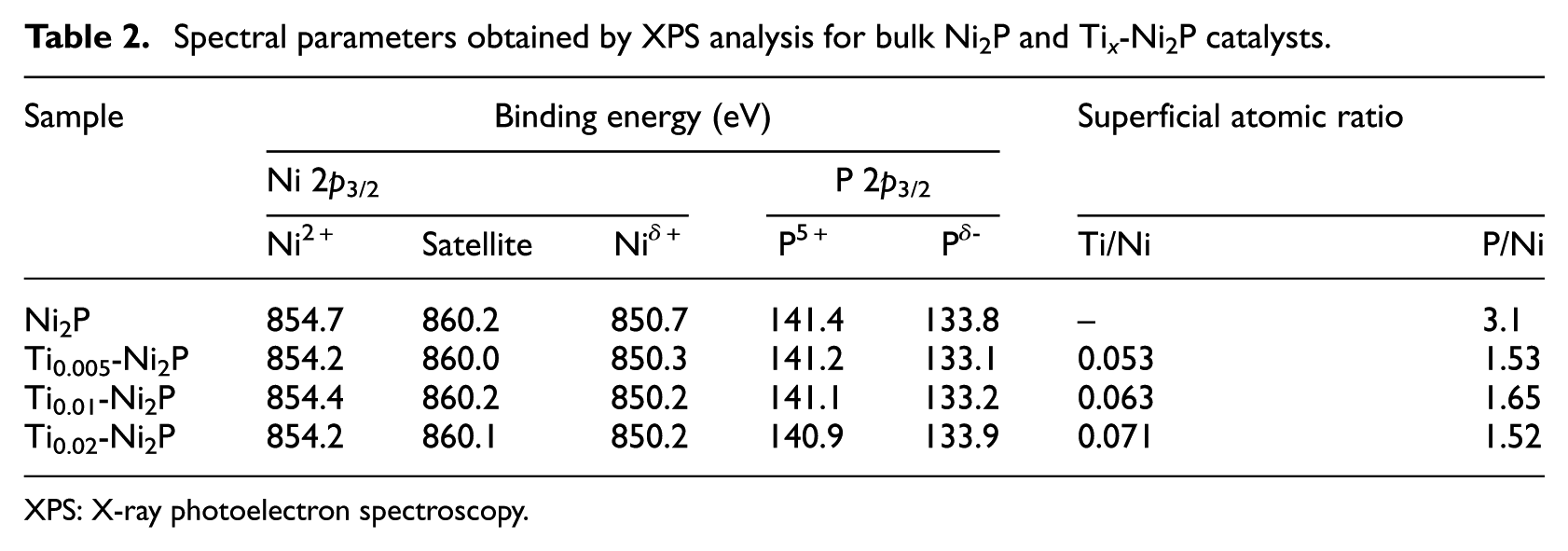
XPS: X-ray photoelectron spectroscopy.
As can be seen from Figure 2, the peaks centered at 132.8–133.7 eV can be assigned to Pδ− species in the Ni2P phase,
20
and the peak at 140.3–141.7 eV can be attributed to phosphate (P5+) arising from superficial oxidation of the Ni2P particles.21,22 As can be seen from Figure 2 and Table 2, with Ti species incorporated into bulk Ni2P, the binding energy of Ni2+ and P5+ shifts slightly to a lower value, indicating that there is an interaction between the Ni2P particles and the Ti species. In addition, the increase of electron density leads to a slight stabilization of the Ni 3d levels and a small Ni–to–P charge transfer.
23
This special effect is assists the reduction of
To be specific, the electrons could be readily transferred and injected from Ti species into the Ni species through the conduction band, because the Ti3+ ion is an electron donor and Ni2P is an excellent electron conductor. The intensity of the peaks located around 853.0 eV (Figure 2) decreases with incorporation of Ti, while the intensity of the peaks located around 857.4 eV increases with incorporation of Ti. 24 XPS analysis for the Ni2P and Ti x -Ni2P catalysts was utilized to calculate the surface Ti and phosphorus with Ni atomic ratios, respectively (Table 2). The P/Ni atomic ratios of the Ti x -Ni2P samples was lower than that found for bulk Ni2P, indicating that the addition of Ti to Ni2P can inhibit the enrichment of phosphorus on the surface of the catalysts. The data obtained demonstrated that there were more Ni sites exposed on the surface of the samples, which was confirmed by the CO uptake analysis (see Table 1, column 6), which may also affect the HDS process of the catalyst.
HDS activity
Generally, two parallel reaction pathways participate in the DBT HDS reaction: (1) direct desulfurization (DDS) and (2) hydrogenation (HYD). 4 The formation of biphenyl (BP) and a small amount of BP further transformed into cyclohexyl benzene (CHB) occurs in the DDS pathway, and HYD yields mainly CHB. Due to the inappreciable amount of BP transformed into CHB, the DDS selectivity is almost equivalent to the selectivity for BP, and the HYD selectivity could be represented by the CHB selectivity. The conversions of DBT via HDS over Ni2P and Ti x -Ni2P catalysts are shown in Figure 3. The conversion of DBT via the HDS process over the bulk Ni2P catalyst was only 4.2% after 1 h. The DBT conversion over the Ti0.005-Ni2P catalyst was 51.5% after 1 h. As the content of Ti increased, the DBT conversion increased further. For Ti0.01-Ni2P and Ti0.02-Ni2P catalysts, the DBT conversion was about 83% after 1 h. Over time, the HDS activity of all catalysts conversion rates increased gradually and eventually stabilized. The DBT conversion over bulk Ni2P catalyst was 55.2% after 8 h, while all the Ti x -Ni2P catalysts showed almost 100% conversion of DBT after 8 h, which is approximately two-fold that shown by the bulk Ni2P. Upon incorporating the Ti into the Ni2P catalysts, the DBT conversions increased dramatically. Generally, the smaller Ni2P particles and better dispersion would result in higher HDS activity of the catalyst. The marked increase of HDS activity for the different loadings of Ti x -Ni2P catalysts might due to the smaller particles and the more widely dispersed active Ni2P phase in these catalysts. This observation suggests that a high dispersion of Ni2P, as can be inferred from Figure 1, can be beneficial in enhancing the HDS activity.20,25
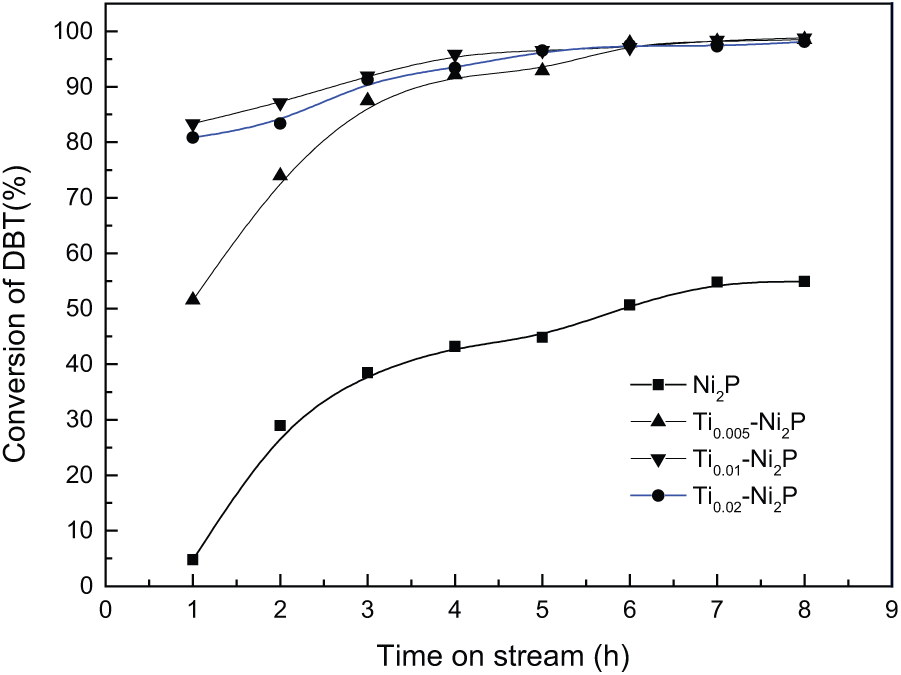
HDS activity of the Ni2P and Ti x -Ni2P catalysts. Temperature = 340 °C; pressure = 3.0MPa; H2/oil ratio = 700 (V/V); WHSV = 1.5 h−1.
HDS selectivity
To investigate further, the effect of Ti on the HDS process, its selectivity data has also been studied. The ratios of DBT to BP and DBT to CHB are given in columns 8 and 9 of Table 1. BP and CHB statistics in the case of in the case of Ni2P and Ti x -Ni2P catalysts are shown in Figure 4.
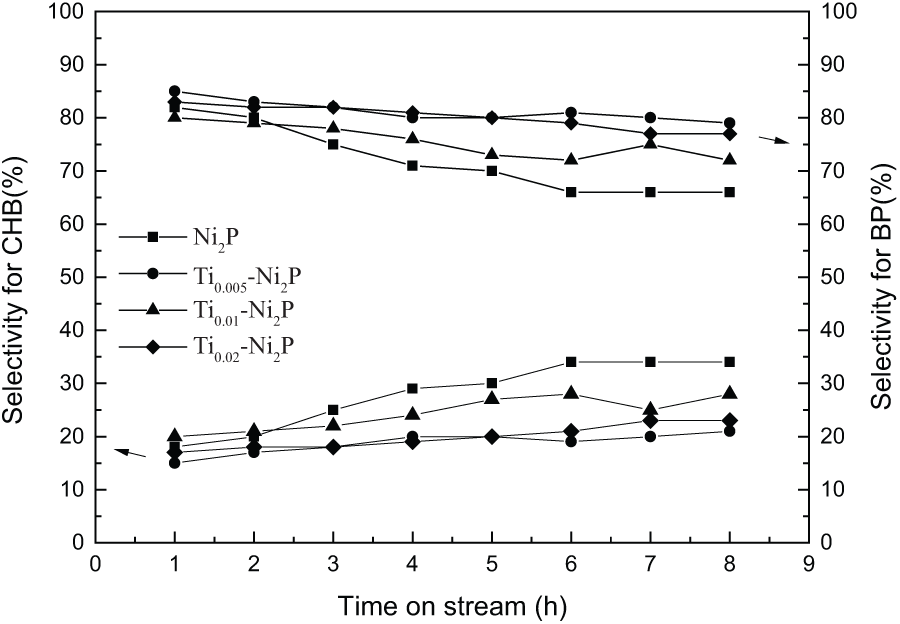
HDS selectivity with the Ni2P and Ti x -Ni2P catalysts. Temperature = 340 °C; pressure = 3.0 MPa; H2/oil ratio = 700 (V/V); WHSV = 1.5 h−1.
For all of the samples, the output of BP is much higher than that of CHB, which means that DBT is primarily removed through the DDS pathway over all the catalysts. 26 At the outset, the selectivity for BP and CHB were approximately the same for all the catalysts. However, the selectivity for BP with bulk Ni2P decreased faster than with Ti x -Ni2P with time on stream. Therefore, the selectivity for BP with Ti x -Ni2P was higher than what was found with the bulk Ni2P after 8 h. Among all the samples, Ti0.005-Ni2P showed the maximum value, the selectivity for BP being increased approximately 15% compared with bulk Ni2P, which indicates the DDS route was enhanced by the addition of Ti. 27
Conclusion
Ti-modified unsupported Ni2P catalysts with higher HDS activity have been prepared successfully. XRD analysis indicated that incorporating Ti into the bulk Ni2P was capable of inhibiting the formation of the Ni5P4 phase, thereby forming the more active Ni2P phase. The crystallite size (Dc) of Ni2P decreased when the loading of Ti increased, and the Dc of Ti0.02-Ni2P catalyst has the lowest value. Besides, the BET analysis results of Ti-incorporated Ni2P catalyst showed a markedly larger surface area, resulting in the large influence of Ti metal on the surface area of the Ni2P catalyst and a better dispersion of the active phase.
In addition, the Ti species was enriched on the surface of the promoted nickel phosphide indicating the existence of an electronic effect caused by the surface Ti species, which might result in electron enrichment on the surface Ni and P species. This electron-enriched surface is suggested to be one of the reasons to account for the enhanced HDS activity of Ti x -Ni2P. The Ti x -Ni2P catalyst exhibited the highest DBT HDS activity of 99.6%, which was an increase of 44% when compared with that found for the bulk Ni2P.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21276048) and the Natural Science Foundation of Heilongjiang Province (ZD201201).

