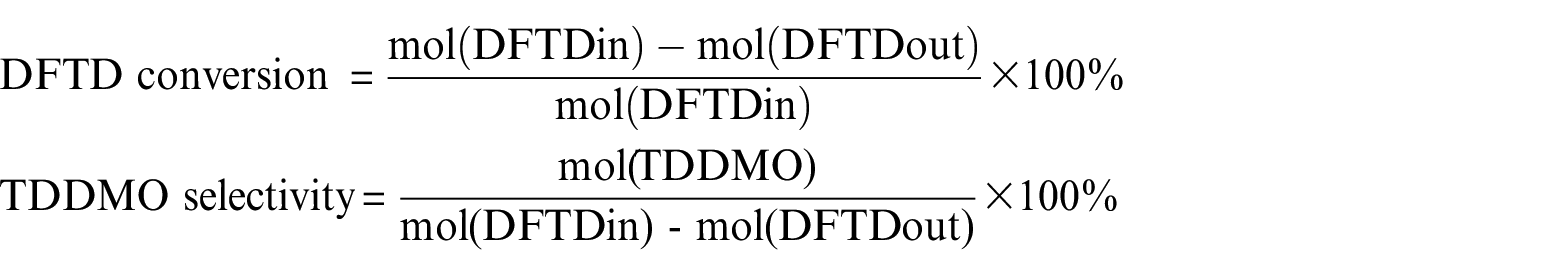Abstract
A series of magnetically separable catalysts based on Ru–Ni bimetallic compounds supported on Fe3O4 nanoparticles was prepared by the co-precipitation method. These catalysts were evaluated for diformyltricyclodecanes hydrogenation reactions, achieving 97% tricyclodecanedimethylol selectivity at 98% diformyltricyclodecanes conversion under mild conditions. The catalyst could be easily recovered by using the magnetic property of the iron oxide support. The catalysts were characterized with X-ray photoelectron spectroscopy, X-ray diffraction, and temperature-programmed reduction. These complementary characterization results suggested that the superior catalytic activity may be derived from the delicate synergy between Ru and Ni species.
Introduction
Tricyclodecanedimethylol (TDDMO) as an important intermediate can be potentially used in the polymer industry, pharmaceutical industry, etc. 1 TDDMO-derived polyester composites, which could be synthesized from the TDDMO esterification reaction, have found widespread use in water-based dispersants, paints, lubricants, etc.2–5 because of their colorimetric and low-viscosity properties, and coating containing TDDMO-derived polyester as an additive can exhibit excellent adhesive performance, even under extreme conditions such as high/low temperature and acidic/basic environment.
Considering the superior potential of TDDMO-based materials in practical use, much research has been focused on the synthesis of TDDMO which could be classified into two categories as follows: (1) a one-step approach in which dicyclopentadiene (DCPD) was converted into the diol in a “one-pot” manner with dialdehyde as an intermediate formed in situ and (2) a two-step approach in which DCPD first underwent a hydroformylation reaction step to produce the corresponding dialdehyde and subsequent hydrogenation reactions.
Shibatani et al. 6 studied the catalytic conversion of DCPD into TDDMO in the presence of a Co-based catalyst and phosphine ligand, using aliphatic alkane as solvent at 150–200°C, and under pressures of 70–150 atm. The “one-step” synthesis of TDDMO was successfully realized with a selectivity of over 60% at almost complete DCPD conversion. However, this method is difficult to apply in industry because of the very high reaction pressure. Furthermore, the products are difficult to separate from the homogeneous system, and the target product overall yield is low because of the undesired side reaction. Ma et al. 7 carried out DCPD hydroformylation catalyzed by Co3O4-supported gold nanoparticles in a heterogeneous system. Although the TDDMO selectivity was over 87.3% and this method seemed a promising pathway, industrized production was disadvantaged due to the high cost of precious metals.
GB Patent 750144 8 reported that the oxo reaction of DCPD was carried out in the presence of a cobalt compound at 140°C, under 180 atm reaction pressure to obtain diformyltricyclodecanes (DFTD), and then the DFTD was hydrogenated in the presence of a nickel catalyst to produce the diol. However, the selectivity for DFTD was only 67.4%, which led to a low overall yield of TDDMO. In another study, Podolean et al. 9 reported that the oxo reaction of DCPD as carried out in the presence of a rhodium-based catalyst to obtain DFTD, and then the hydrogenation of DFTD was carried out in the presence of the same catalyst at 240°C. Although the selectivity for TDDMO was over 93%, this method was disadvantaged due to the very high reaction temperature. In a recent study, Wang et al. 10 have reported Co–Rh bimetallic compounds supported on a Fe3O4 nanoparticles catalyst being tested for the DCPD hydroformylation reaction, achieving >98% DFTD selectivity at >99% DCPD conversion under mild conditions. Encouraged by these results, we planned to use the “two-step” method to synthesize TDDMO by developing an efficient catalytic process for the conversion of DFTD into TDDMO.
However, recycling of heterogeneous catalysts using common methods such as filtration and centrifugation are tedious and time-consuming, and these separation processes inevitably lead to the loss of catalyst. Recently, magnetic separation for catalyst recycling that overcomes the disadvantages caused by the traditional separation methods has received considerable attention.11–16 In the present work, we chose Fe3O4 as the support to investigate the hydrogenation activity and selectivity of a Ru–Ni bimetallic catalyst.
For the 10%Ru/Fe3O4 catalyst, in spite of the DFTD conversion of 93%, only 74% TDDMO selectivity could be achieved at 140°C and 4 MPa for 4 h. Under the same reaction conditions, using a 10% Ni/Fe3O4 catalyst, 92.7% TDDMO selectivity with 89% DFTD conversion could be achieved. Inspired by these two complementary results, we planned to develop the Ru–Ni bimetallic catalyst which enhanced not only the conversion of DFTD but also the selectivity for TDDMO. Fortunately, the conversion of DFTD was enhanced to 98%; meanwhile, the selectivity was enhanced to 97% using the bimetallic Ru–Ni/Fe3O4 catalyst, even at a very low Ru loading. Catalysis characterization including X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and temperature-programmed reduction (TPR) was carried out to characterize the Ni/Fe3O4 catalysts modified with ruthenium.
Materials and methods
All chemicals were of analytical grade and used without further purification.
Catalyst preparation
The Ru–Ni/Fe3O4 catalyst was prepared by the co-precipitation method. Specifically, Fe(NO3)3·9H2O (42 g), RuCl3·4H2O (0.2069 g), and Ni(NO)3·6H2O (3.5676 g) were dissolved in 150 mL distilled water. This solution was added dropwise to a 660 mL Na2CO3 solution (0.47 M) under vigorous stirring for 1.5 h. The reaction mixture was further stirred for 2 h. After filtration and washing to pH 7.0 with 1000–1500 mL of distilled water, the resultant precipitate was dried at 120°C for 16 h and calcined at 400°C in air for 4 h. The solid obtained was then reduced under H2 flow at 400°C for 2 h. Finally, around 8 g of 1wt%Ru–9wt% Ni/Fe3O4 catalyst was obtained. The obtained catalyst was ground to powder and denoted as 1%Ru–9%Ni/Fe3O4 (or 1Ru) for further use and characterization.
Catalyst characterization
XRD analysis
XRD was carried out using a Siemens D/max-RB powder X-ray diffractometer. Diffraction patterns were recorded over a 2θ range of 15–70° in a step increment of 0.04° with a count time of 1 s.
TPR
A FINESORB-3010 instrument equipped with a thermal conductivity detector was used to perform TPR. In a typical measurement, the catalyst (about 100 mg) was heated under dry air at 383 K for 1 h before analysis. The TPR profile was recorded while heating the sample from room temperature to 673 K at a ramping rate of 10 K min−1 under an H2/Ar (10% v/v) flow.
XPS
XPS analysis was performed using a VG-ESCALAB 210 instrument equipped with an Mg anode and a multi-channel detector. Charge referencing was measured against adventitious carbon (C1s, 285.0 eV). The surface speciation was determined from the peak areas of the corresponding lines using a Shirley-type background and empirical cross-section factors for XPS.
Reaction testing
Hydrogenation of DFTD to TDDMO was carried out in a 200 mL stainless steel autoclave reactor with an inserted glass liner. In a typical experiment, Ru–Ni/Fe3O4 catalyst (0.2 g), tetrahydrofuran (20 mL), DFTD (5 g), and PPh3 (0.2 g) were introduced into the autoclave and stirred. Then, the reactor was purged with H2 thrice, and the pressure was adjusted to an initial value 4 MPa at ambient temperature. The mixture was then stirred with a stirring rate of 650 rpm which was found to be adequate for overcoming diffusion limitations, and the temperature was elevated to 140°C and held for 2 h.
Reaction product analysis
The reaction effluents were analyzed by a gas chromatography (Shimadzu 2014) instrument equipped with a flame ionization detector using the area normalization method. The definitions of DFTD conversion and TDDMO selectivity are
where mol(DFTDin) is the introduced DFTD in moles, mol(DFTDout) is the unreacted remaining DFTD in moles when the sample was taken for the analysis and mol(TDDMO) is the amount of TDDMO in moles produced in the reaction
Results and discussion
Results of DFTD hydrogenation over different catalysts
The results for hydrogenation of DFTD over various catalysts are summarized in Table 1. For the 10%Ru/Fe3O4 catalyst (entry 1), in spite of the DFTD conversion of 93%, only 74% TDDMO selectivity could be achieved at 140°C and 4 MPa for 4 h. Compared with the 10%Ru/Fe3O4 catalyst, the 10%Ni/Fe3O4 catalyst was less active towards DFTD hydrogenation but it could dramatically improve the TDDMO selectivity from 74% to 92.7% (entries 1 and 2). A comparison of entries 1 and 2 shows that the ruthenium sites are conducive to the conversion of DFTD, and the nickel sites contribute to selectivity for the product TDDMO under the same experimental conditions. Inspired by these results, a series of other Ru–Ni bimetallic catalysts was prepared with increasing Ru loading. It can be observed in entries 3–8 that conversion of DFTD gradually increased with increasing Ru loading up to the highest figure of 98%, then there is a very slight drop to 94% which is still higher than 93%. This suggests that Ru–Ni bimetallic catalysts can improve the conversion of DFTD compared with the monometallic catalyst Ru/Fe3O4 under the same conditions and that there is a synergetic effect between Ru and Ni bimetallic catalysts.
Effect of Ru loading on the tricyclodecanedimethylol (TDDMO) synthesis.

DFTD: diformyltricyclodecanes.
Experimental conditions: 140°C and 4 MPa H2 for 4 h; 5 g DFTD; 0.2 g catalyst; 0.2 g PPh3.
0.1 g PPh3.
0.3 g PPh3.
As shown in entries 2–4, the TDDMO selectivity unexpectedly increased up to the highest figure of 97% as the Ru/Ni ratio increased from 0:10 to 1:9, indicating that adding an appropriate amount of Ru could greatly improve the TDDMO selectivity and proving again that there is a synergetic effect between Ru and Ni for the reaction, not simply due to the physical addition of Ru and Ni. When the Ru loading continues to increase, the TDDMO selectivity gradually decreases. This suggested that the optimal loading of Ru in the bimetallic catalyst was found to be 1%Ru–9%Ni/Fe3O4 in the range tested. This catalyst was chosen for further studies.
In order to explore the effect of ligands for DCPD hydrogenation, experiments were also performed with the results being summarized in entries 4, 9, and 10. When the amount of PPh3 was increased from 0.1 to 0.3 g, the TDDMO selectivity initially increased and then decreased, indicating that an appropriate amount of PPh3 had a positive impact on TDDMO selectivity and that a catalytically active site is proposed as a multi-component Ru–Ni–(PPh3)y complex.
XRD, TPR, and XPS results
The XRD profiles of 10%Ru/Fe3O4, 10%Ni/Fe3O4 and a series of Ru–Ni/Fe3O4 catalysts are shown in Figure 1. The results showed that the characteristic diffraction peaks of Fe3O4 were observed for all samples.10,17 For metallic Ru–Ni/Fe3O4 catalysts, the XRD results indicated that Ni species were highly dispersed on the iron oxide as the characteristic diffraction peaks of Ni and Ru could not be discerned.
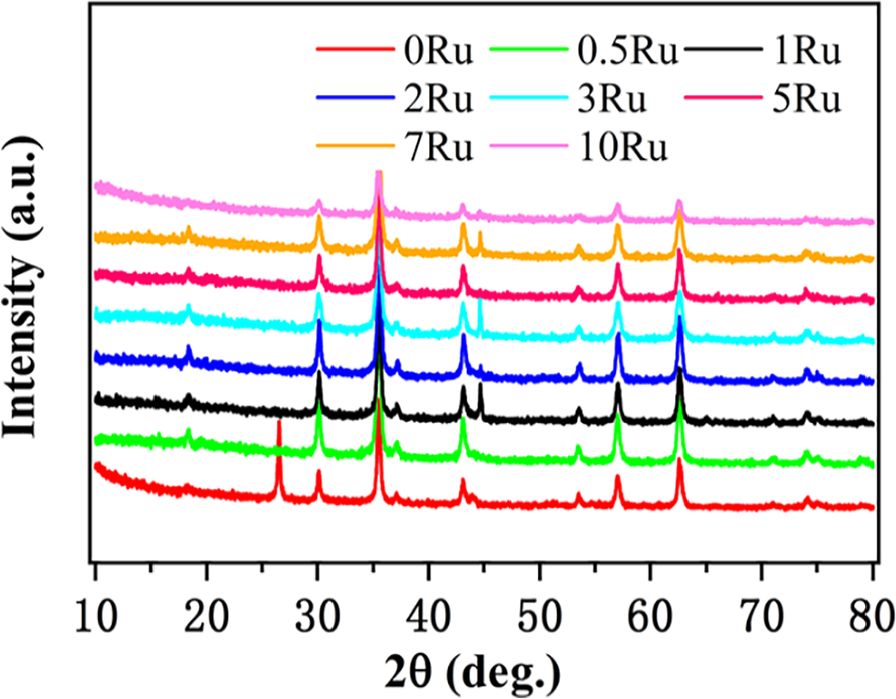
XRD patterns of the catalysts.
TPR was used to probe the Rh oxidation state in the ruthenium-modified Ni/Fe3O4 catalyst precursors (before reduction, existing as oxides). The results are summarized in Figure 2. Three obvious reduction peaks at 294, 338, and 548°C were found for 10%Ni/Fe2O3. A broad peak at 548°C can be ascribed to a subsequent reduction of Fe3O4 and ultimately involves the formation of metallic iron. 18 Compared with the reduction peaks of Ni/Fe2O3, the NiO x reduction peaks in Ru–Ni/Fe3O4 could not be discerned. This phenomenon was probably superimposable with the broad FeO x reduction peak. The RuO x reduction peaks of the ruthenium-modified materials were shifted to higher temperatures relative to those of the Ru/Fe3O4 catalyst. The extent of peak shift became greater with the increase of ruthenium loading. In addition, the area under the curve (AUC) also varied with the ruthenium loading. When the Ru/Ni ratio in Ru–Ni bimetallic catalysts was increased from 0:10 to 7:3, the AUC decreased, indicating that the ruthenium surface atoms were blocked by the added Ni atoms. These results also showed that the RuO x reducibility was easily altered. Thus, the higher activities of the ruthenium-modified Ni/Fe3O4 catalysts were attributed to the presence of ruthenium surface species that were induced and stabilized by the ruthenium atoms.
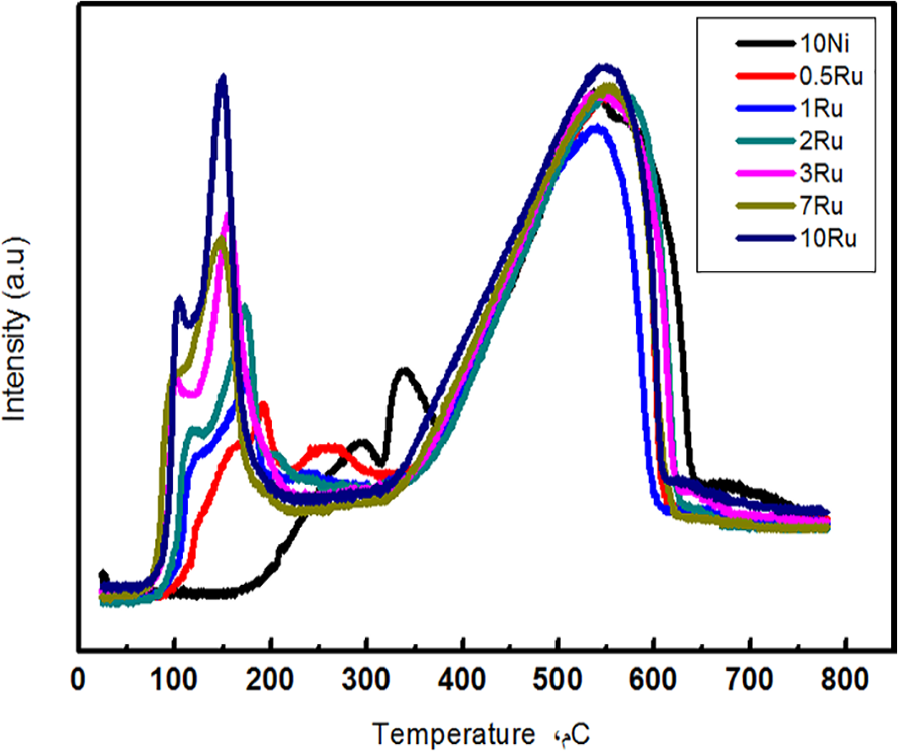
TPR profiles of various catalysts.
Conclusion
In conclusion, a Ru–Ni bimetallic catalyst supported on magnetic Fe3O4 nanoparticles was evaluated based on its catalytic performance for hydrogenation of DFTD to TDDMO, a value-added fine chemical. The catalyst could be easily isolated using an external magnetic field, facilitating the practical application of such catalyst materials. For the reaction of DFTD, a monometallic Ru/Fe3O4 catalyst was conducive to the conversion of the reaction, and a Ni/Fe3O4 catalyst contributed to the selectivity for the product TDDMO. However, not only was the conversion of DFTD greatly increased to 98% but the selectivity for TDDMO was remarkably enhanced to 97% using a bimetallic Ru–Ni/Fe3O4 catalyst, even at a very low Ru loading.
XRD results revealed that the Ru and Ni species were highly dispersed on the magnetic iron oxide matrix. The TPR results indicated that there is a synergetic effect between ruthenium and nickel species and that the added ruthenium likely enhanced the catalytic performance by giving rise to a more reactive nickel surface species.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work has been financially supported by the Chinese Government “Thousand Talent” Program (Y42H291501) and the National Natural Science Foundation of China (U1139302).