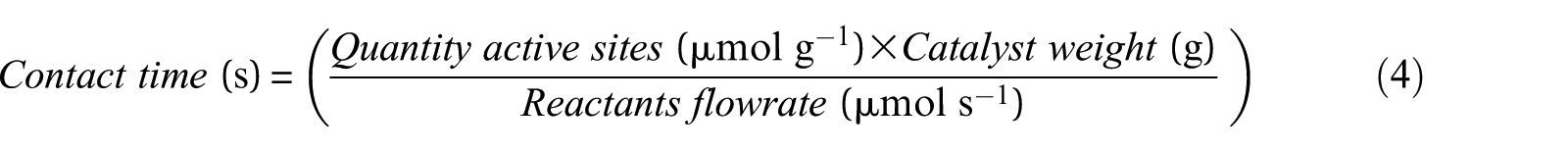Abstract
A nickel phosphide hydrodeoxygenation catalyst (Ni2P-O/MCM-41) was prepared using a new synthetic method. The as-prepared catalyst was evaluated in the hydrodeoxygenation of benzofuran, and the effects of reaction temperature, pressure, and the H2/liquid ratio were investigated. A pseudo first-order model was employed to describe the reaction kinetics of benzofuran hydrodeoxygenation over the Ni2P-O/MCM-41 catalyst. The reaction rate constants (k1–k5) at different temperatures were determined according to this model. At 533 K, the conversion of 2-ethylphenol in to ethylbenzene began to increase dramatically, and the yield of O-free product, ethylcyclohexane, started to increase rapidly. At 573 K, 3.0 MPa, and a H2/liquid ratio of 500 (V/V), the conversion of benzofuran over Ni2P-O/MCM-41 reached 93%, and the combined yield of O-free products was 91%. Contact time analysis indicated that demethylation was not favored over the Ni2P-O/MCM-41 catalyst.
Introduction
Biomass, the most abundant and inexpensive renewable resource, has attracted much attention as an alternative to fossil fuels in recent years.1,2 Fast pyrolysis is the simplest and most cost-effective process for conversion of biomass to liquid fuel, which is a mixture of oxygenated compounds known as bio-oil.2,3 The high oxygen content of bio-oil, usually 20–50 wt%, results in numerous problems when it is used in standard equipment that has been constructed for use with oil-derived fuels; therefore, the upgrade of bio-oil is necessary.3,4 Hydrodeoxygenation (HDO) is the most common method for upgrading bio-oil, involving the removal of oxygen with hydrogen over HDO catalysts.2,5 There are more than 200 types of oxygen-containing compounds in bio-oil, which leads to significant challenges for mechanistic and kinetic studies of HDO, so studies are usually conducted using model compounds. 6
Various HDO catalysts have been studied, including those based on sulfides, noble metals, base metals, and transition metal phosphides. Supported sulfide catalysts have served as hydrodesulfurization (HDS) and hydrodenitrogenation (HDN) catalysts for decades, and the reaction mechanisms and kinetics over sulfide catalysts are well understood. 2 Sulfide catalysts are also efficient for HDO but require the addition of sulfur to the feed to maintain the sulfide form of the catalyst, which negates the superiority of bio-oil for its low-sulfur content. 7 Noble metal catalysts perform better than sulfide catalysts in terms of hydrocarbon yield and deoxygenation level. 8 However, the reaction mechanisms and kinetics still lack explicit understanding. 2 Transition metal phosphide catalysts have attracted much attention because of their high activity and stability in HDO.9,10 Of the tested metal phosphides, including Ni2P, WP, Co2P, MoP, and NiMoP, Ni2P was the most promising catalyst.11,12 The activity of Ni2P catalysts has also been found to be influenced by the method of synthesis.13,14
Ni2P has been reported to have high activity in the HDO of furan, benzofuran (BF), anisole, guaiacol, and 2-methyltetrahydrofuran (2-MTHF).1,5,10 The reaction routes of model compounds over Ni2P catalysts have been investigated, but kinetics and active sites have received little attention. 2 Iino et al. 5 have studied the kinetics during HDO of 2-MTHF over a Ni2P/SiO2 catalyst. Their studies determined the reaction routes and calculated rate constants for each step using a Langmuir–Hinshelwood mechanism. Cho et al. 15 proposed a more realistic kinetic description based on a rake mechanism. Fitting of the results using a rake mechanism conforms to a simpler first-order model.
In this study, a Ni2P-O/MCM-41 catalyst was successfully prepared using the new synthetic method reported by our group. 1 The prepared catalyst was evaluated for the HDO of BF. The effects of reaction temperature, pressure, and the H2/liquid ratio were investigated further. The reaction rate constants at different temperatures for each reaction step were determined according to the proposed mechanism.
Experimental
Preparation of catalyst
The catalyst precursor was prepared by impregnating the MCM-41 support with an ammonium hypophosphite (NH4H2PO2) and nickel chloride (NiCl26H2O) solution for 12 h. After the water was evaporated, the impregnated solid was dried at 333 K. The precursor was then ground with a mortar and pestle, pelletized using a press, crushed, and then sieved to achieve appropriate particle sizes. The precursor was placed in a fixed-bed reactor at 673 K in a flow of H2, held for 2 h, then naturally cooled to 373 K, and held for 1 h in flowing air (20 mL min–1). The catalyst obtained with Ni loading of 8.8 wt% and an initial Ni/P molar ratio of 1/2 was named as Ni2P-O/MCM-41.
Characterization of catalyst
X-ray diffraction (XRD) analysis was carried out on a D/max-2200/PC X-ray diffractometer (Rigaku Company) using Cu Kα radiation, with a scan range from 10 to 80° at a rate of 10°min–1. The typical physico-chemical properties of the catalysts were analyzed by the Brunauer–Emmett–Teller (BET) method using Micromeritics adsorption equipment type NOVA 2000e. The CO uptake measurement was adopted to titrate the nickel atoms of the catalyst surface and measurements were performed on a Micromeritics ASAP 2010 apparatus under static volumetric conditions. After pretreatment in a continuous N2 flow, CO pulses were injected into the N2, and the CO uptake was measured using a thermal conductivity detector until there was no further CO uptake after consecutive injections.
Catalytic activities
The reaction was performed in a flowing high-pressure fixed-bed catalytic reactor, using a feed consisting of a decalin solution of BF (2 wt%). The conditions of the HDO reaction were 493–573 K, 1.5–3.5 MPa, weight hourly space velocity (WHSV) = 2.0–16.0 h−1, and a hydrogen/liquid ratio of 300–700 (V/V). The temperature was raised to 573 K at a rate of 3 K min–1, then altered as follows: 573, 553, 533, 513, 493, 513, 533, 553, and 573 K, with each temperature maintained for 3 h. The samples of reaction product were obtained after steady reaction conditions had been achieved for 6 h. The feed and reaction products were analyzed using a gas chromatograph (Shimadzu GC-14C) equipped with a capillary column (PH-1, 60 m × 0.25 mm) together with a flame ionization detector.
The conversion of BF (XBF), yields of O-free compounds (XO-free), and selectivity (Si) were defined as follows
where nBF,0 and nBF are the moles of BF in the feed and product respectively, ni is the moles of product i. nO-free product is the moles of product of O-free product. nall product is the moles of all product. XO-free percentages show how much BF is converted into hydrocarbons without oxygen.
Contact time values of different reactant flow rates were calculated using equation (4). The quantity of active sites was measured by CO chemisorption
Results and discussion
Catalyst characterization
The Ni2P-O/MCM-41 catalyst used in this work has been studied previously. 1 To assist the reactivity and kinetic studies, the characterization results are briefly specified in this section. The XRD patterns of the Ni2P-O/MCM-41 catalyst showed that a pure Ni2P phase was obtained and no other phase related to Ni or P was seen. The average size of the Ni2P crystallites calculated by the Scherrer equation was 20 nm. The surface area and pore volume of the MCM-41 support were 1051 m2 g−1 and 0.84 cm3 g−1, and for Ni2P-O/MCM-41 they were 509 m2 g−1 and 0.37 cm3 g−1, respectively. The decreases in surface area and pore volume of Ni2P-O/MCM-41 were caused by the blocking of pores by Ni2P particles. The pore size distributions obtained by a Barrett–Joyner–Halenda analysis lay in the range 2.5–3.5 nm. The CO uptake of the Ni2P-O/MCM-41 catalyst was determined as 53 mmol g−1. The detailed characterization results can be seen in Song et al. 1
Reactivity measurements
Effect of reaction time
The prepared Ni2P-O/MCM-41 catalyst was examined in the HDO of BF for 25 h. Observation and measurement started from the input of the BF stream. The conversion of BF decreased from 97 to 88% within 5 h, then displayed remarkable stability for 24 h. The main products detected were ethylcyclohexane (ECH, 65%), methylcyclohexane (MCH, 10%), 2-ethylphenol (2-EtC6H4OH, 9%), ethylbenzene (EB, 6%), and2,3-dihydrobenzofuran (2,3-DHBF, 5%). Trace products observed were phenol (PhOH, 3.2%), methylbenzene (MB, 1.5%), and benzene (B, 0.5%). The product ratios did not change with reaction time, and therefore the Ni2P-O/MCM-41 catalyst did not suffer deactivation in the range of 5–24 h (Figure 1).
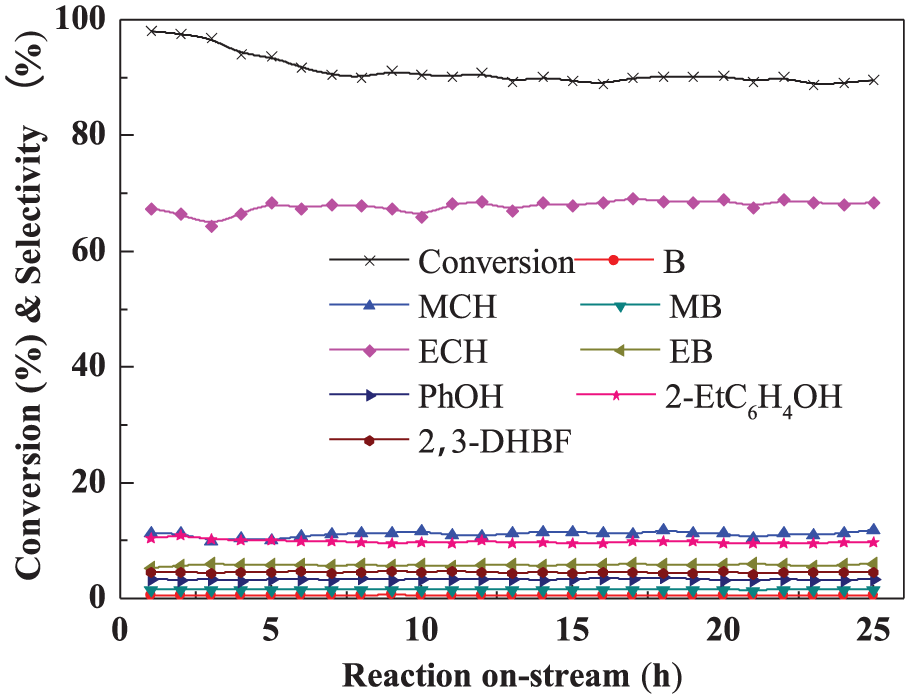
Effect of reaction on-stream on conversion of BF and product selectivities.
Effect of temperature
Different reaction temperatures (493, 513, 533, 553, and 573 K) were examined as shown in Figures 6 and 7 in Song et al. 1 In order to assist kinetic analysis, similar experiments were carried out and further analysis of the reaction mechanism was conducted. The conversion of BF increased rapidly with increasing reaction temperature. At lower reaction temperatures (<533 K), the main products were oxygen-containing intermediates, such as 2,3-DHBF and 2-EtC6H4OH, indicating that the conversion of 2-EtC6H4OH into EB by dehydration was slow at lower temperatures. The yield of O-free products increased gradually at higher reaction temperatures. However, for reaction temperatures >533 K, the proportion of ECH increased dramatically accompanied by decreased proportions of 2,3-DHBF and 2-EtC6H4OH, indicating that the conversions of 2,3-DHBF into 2-EtC6H4OH, 2-EtC6H4OH into EB, and EB into ECH increased significantly above 533 K. The lower proportions of EB and MCH compared with ECH suggested that the conversion of ECH into MCH was slow, even at a higher temperature. Formation of EB by dehydration was followed by rapid conversion into ECH. However, only a small amount of ECH was converted into MCH by demethylation, leading to increased quantities of ECH in the product. Similar results have been reported by Moon et al. 16 in a study of guaiacol HDO over a Ni2P/SiO2 catalyst.
Effect of pressure
The HDO reaction requires the participation of H2. When H2/liquid = 500, the partial pressure of the liquid can be ignored, so the H2 partial pressure is equal to the total pressure P. 17 The effect of pressure was studied, and the results are shown in Figure 2. As the pressure was increased from 1.5 to 3.5 MPa, the conversion of BF increased from 18 to 98%, and the yield of O-free products increased from 19 to 91%. Correspondingly, the proportion of ECH increased rapidly, while the quantities of other O-free products increased gradually at higher pressures. This indicated that higher pressure was beneficial to formation of ECH by BF HDO. The effect of pressure on the selectivity for ECH showed a similar trend to the effect of temperature.
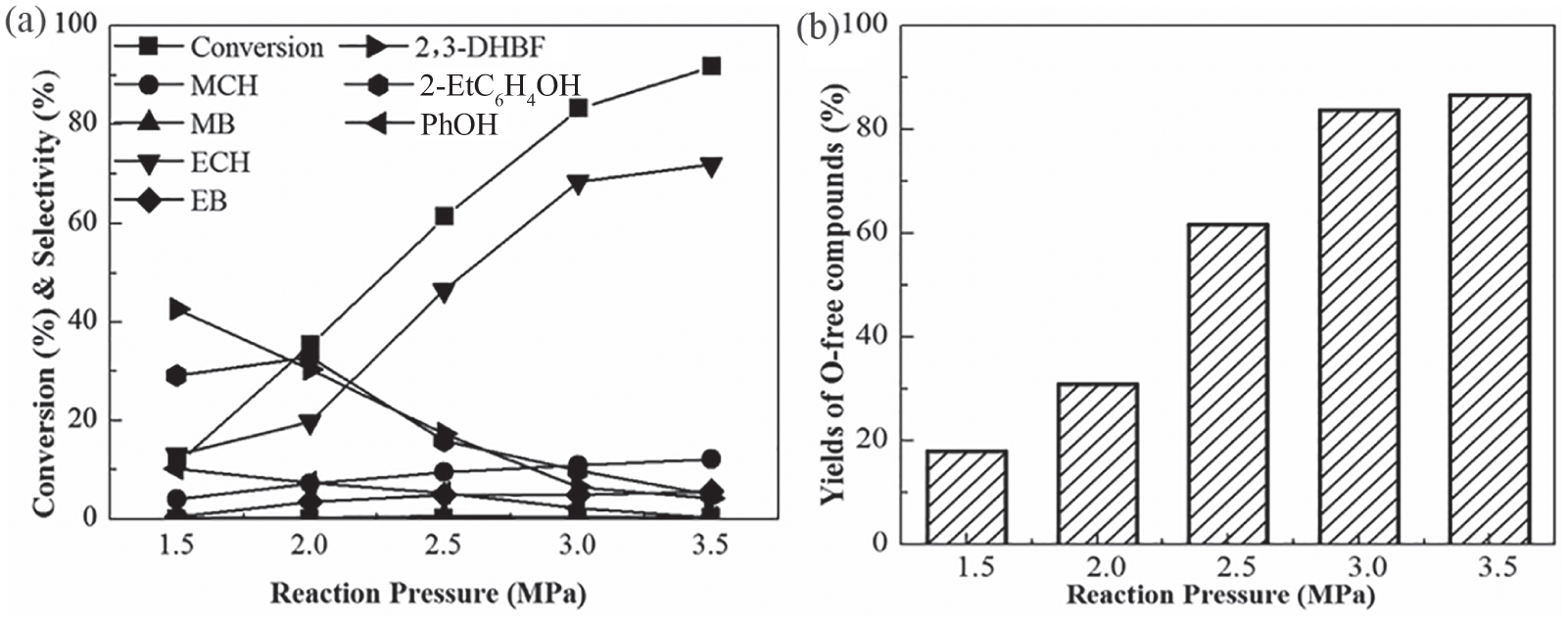
(a) Effect of pressure on conversion of BF and major product selectivities and (b) yield of O-free products of BF HDO over Ni2P-O/MCM-41catalysts. T = 573 K, WHSV = 4.0 h−1, and H2/liquid ratio = 500 (V/V).
Effect of H2/liquid ratio
To study the effect of the volume ratio of H2/liquid on the HDO of BF over the Ni2P-O/MCM-41 catalyst, HDO reactions were conducted at volume ratios of 300, 400, 500, 600, and 700. The results are shown in Figure 3. The conversion of BF increased rapidly as the H2/liquid ratio increased initially and then stabilized, reaching 93% at H2/liquid = 500 (V/V). The proportions and yields of O-free compounds showed a similar trend to the effects of temperature and pressure. The yield of ECH reached 67%, and the yield of total O-free compounds reached 91% at an H2/liquid ratio of 500 (V/V) and then stabilized. Similar results were reported by Wang et al., 18 who found that the conversion of 3-methylthiophene reached a maximum at an H2/oil ratio of 400 (V/V) and then stabilized during HDS over a Ni2P/TiO2-Al2O3 catalyst.
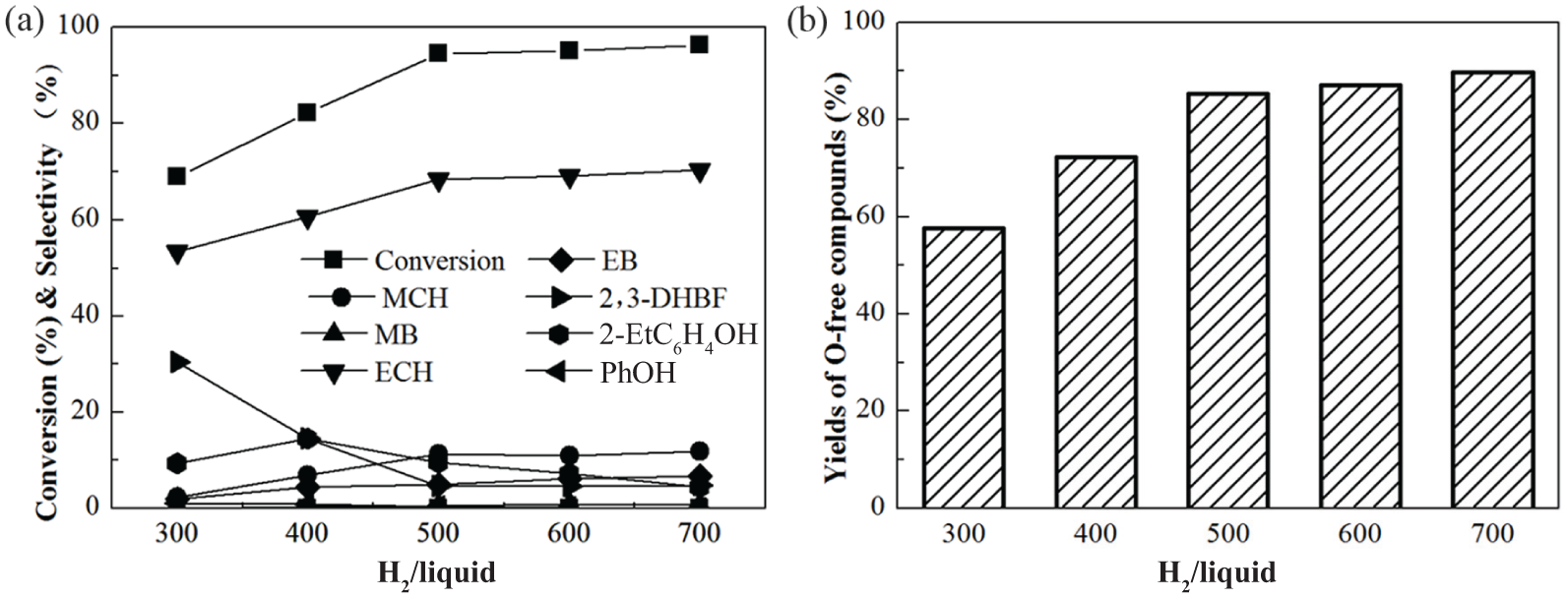
(a) Effect of H2/liquid ratio on conversion of BF and major product selectivities and (b) yield of O-free products of BF HDO over Ni2P-O/MCM-41catalysts. T = 573 K, P = 3.0 MPa, and WHSV = 4.0 h−1.
Contact time analysis
Figure 4(a) shows the effect of contact time on the conversion of BF and the major product yields. The conversion of BF initially increased rapidly as the contact time was increased, then increased slowly at a contact time of 3 s, finally reaching a conversion of 99% at a contact time of 12 s. Similar results were observed by Cho et al. 15 in their study of 2-MTHF HDO over a Ni2P/SiO2 catalyst. The conversion of 2-MTHF and the yield of O-free products increased with increasing contact time. As shown in Figure 4(a), the proportions of the oxygen-containing products 2,3-DHBF, 2-EtC6H4OH, and PhOH decreased with increased contact time as expected, while the proportion of the main O-free product ECH increased significantly. The proportion of MCH also increased with increasing contact time but to a lesser extent, suggesting that the transformation of ECH to MCH by demethylation was a rate-limiting step. The proportions of the trace products B and MB were almost invariant. This indicates that demethylation was not favored over the Ni2P-O/MCM-41 catalyst, since B and MB are both formed by demethylation. Liu et al. 19 studied BF HDO over supported Pd, Pt, and alloyed Pt–Pd catalysts and also found that the deethylation reaction was not favored.
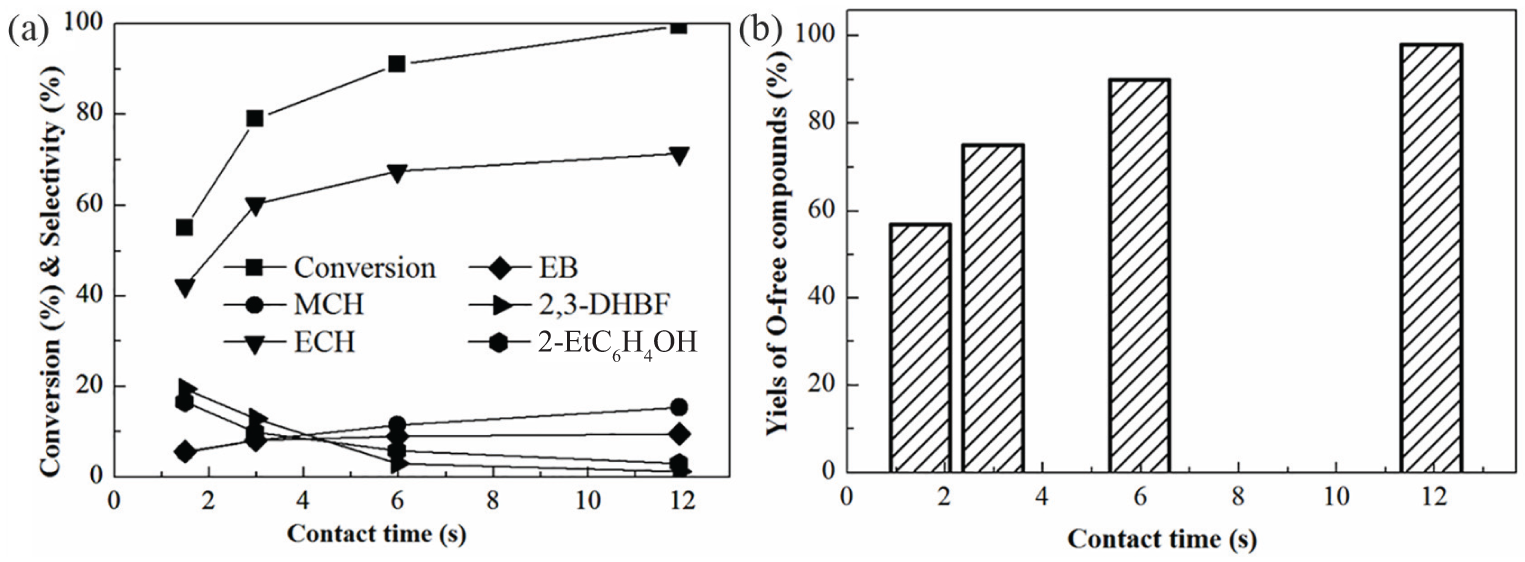
Effect of time on (a) conversion of BF and major product selectivities and (b) yield of O-free products of BF HDO over Ni2P-O/MCM-41catalysts. T = 573 K, P = 3.0 MPa, and H2/liquid = 500 (V/V).
Kinetic analysis
From contact time analysis and our previous study, 1 the overall reaction network for BF HDO over Ni2P-O/MCM-41 is proposed as shown in Scheme 1. The network has one major pathway and two minor pathways. The first step in the major pathway involves hydrogenation of the furan ring, leading to the formation of 2,3-DHBF. The 2,3-DHBF is then converted into 2-EtC6H4OH by C–O bond cleavage of the heterocyclic ring via hydrogenolysis. EB is obtained from 2-EtC6H4OH by dehydration, then converted into ECH by hydrogenation of the benzene ring, followed by demethylation of ECH to MCH. In the minor pathway, EB is transformed into B and MB via hydrogenation of the ethyl group. B can also be formed by dehydration of PhOH, which is formed from 2-EtC6H4OH by deethylation.
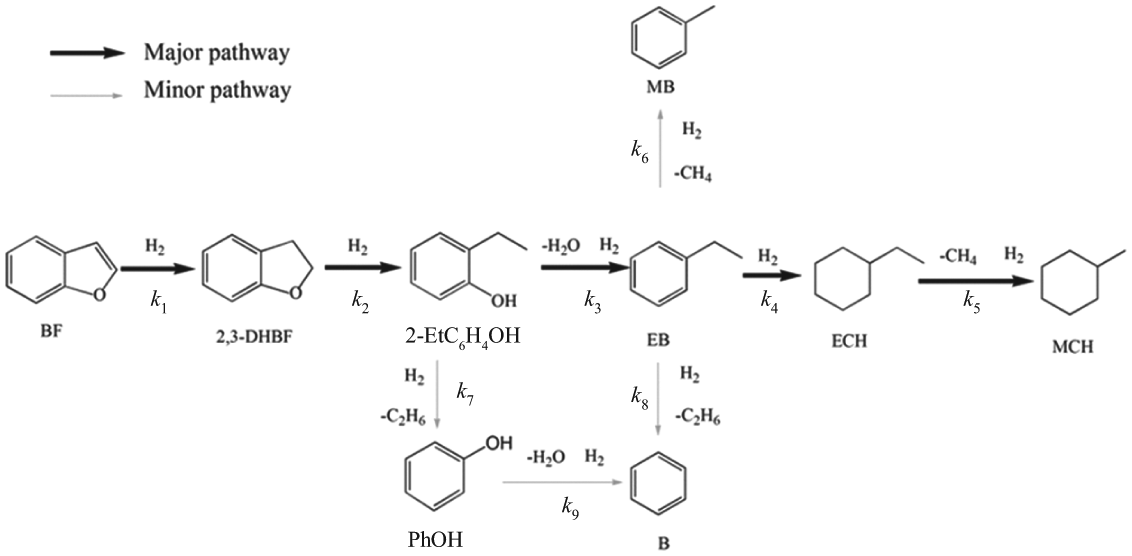
Reaction pathways for HDO of BF over the Ni2P-O/MCM-41 catalyst.
Reaction rate constants, ki, are defined for each reaction step in the reaction network as shown in Scheme 1. A pseudo first-order model, which has frequently been used for the HDO reaction, 2 was used to investigate the kinetics of HDO over the Ni2P-O/MCM-41 catalyst. Considering that the volume ratio of H2/liquid was 500 in this study, the amount of H2 was very much larger than that of BF, so the concentration of H2 was considered to remain unchanged during the reaction. 5 Since the yields of MB, PhOH, and B (<3.2%) were very low, the corresponding reaction rate constants (k6–k9) were too low to calculate or analyze. The kinetic rate law equations for each reaction step in the major pathway (k1–k5) are given in Table 1. C1, C2, C3, C4, C5, and C6 denote concentrations of BF, 2,3-DHBF, 2-EtC6H4OH, EB, ECH, and MCH, respectively. Similar pseudo first-order models and mathematical calculations were reported by Iino et al. 5 in their kinetic studies on 2-MTHF HDO over a Ni2P/SiO2 catalyst.
Kinetic rate law equations for each reaction step.
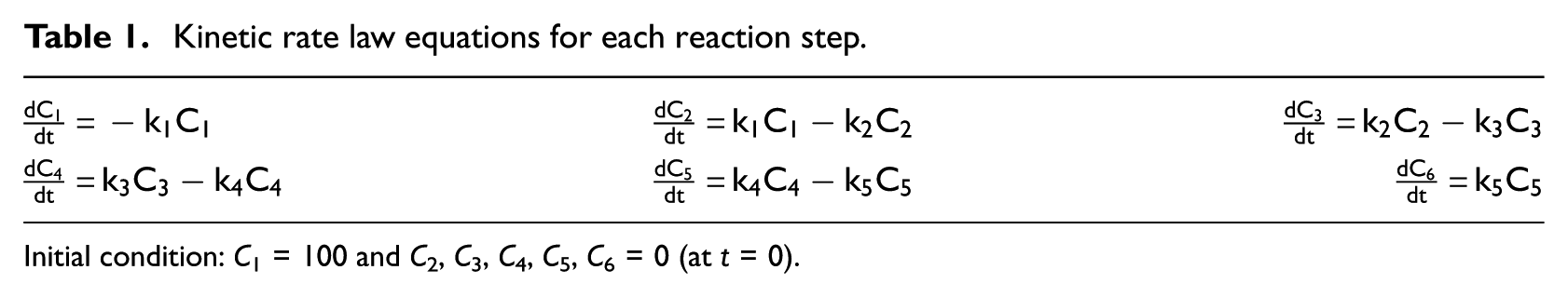
Initial condition: C1 = 100 and C2, C3, C4, C5, C6 = 0 (at t = 0).
The kinetic analysis was conducted at different temperatures (493, 513, 533, 553, and 573 K) and different contact times (1.49, 2.98, 5.97, and 11.93 s). The rate constants (k1–k5) were calculated by solving the kinetic rate law equations in Table 1 using MATLAB, and the results are shown in Table 2. The residual sum of squares (RSS) for the reaction rate constants were less than 0.12, demonstrating that the experimental data satisfied the pseudo first-order model well.
Reaction rate constants of each step of BF HDO at different temperatures.
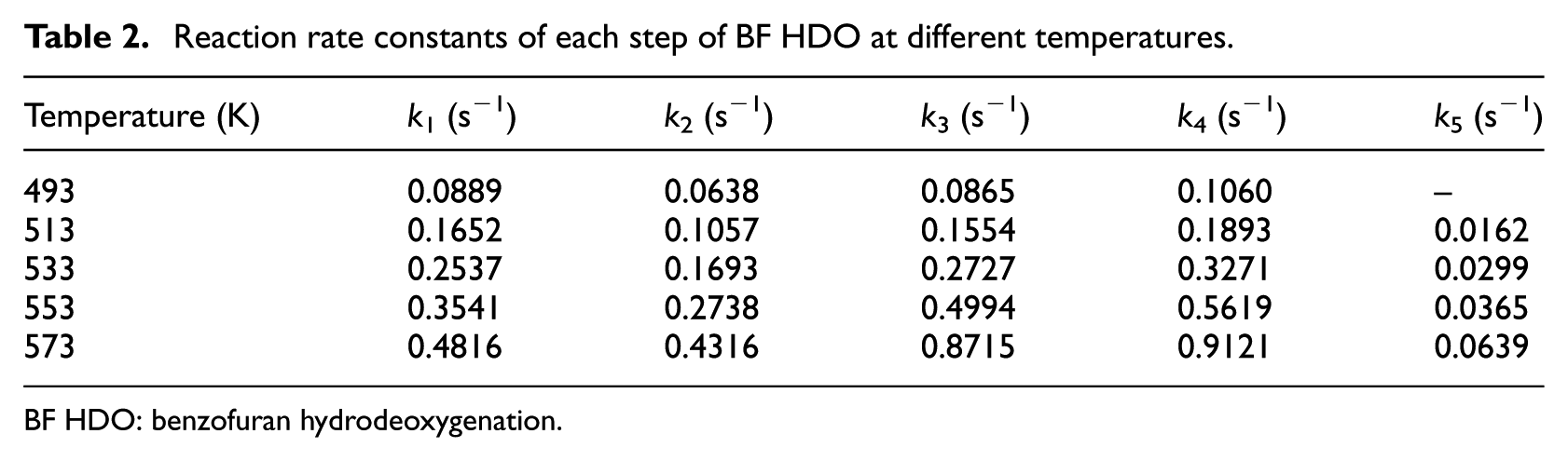
BF HDO: benzofuran hydrodeoxygenation.
It can be seen from Table 2 that k5 was much smaller than the other rate constants, which was consistent with previous results. Such a small value of k5 further confirmed that the demethylation reaction was unfavorable over the Ni2P-O/MCM-41 catalyst. The value of k4 was higher than the other rate constants, indicating that hydrogenation of the benzene ring of EB was facile. All reaction rate constants increased with increasing reaction temperature, which obeys the Arrhenius equation within the temperature range utilized.
Conclusion
The Ni2P-O/MCM-41 catalyst was successfully prepared using a temperature-programmed reduction method and then modified with oxygen. The prepared Ni2P-O/MCM-41 catalyst was evaluated in the HDO of BF, and the effects of temperature, pressure, H2/liquid ratio, and contact time were studied. The results showed that the conversion of BF and the proportions of O-free products increased with increasing temperature and pressure. However, the conversion of BF and proportions of O-free products increased rapidly with increasing H2/liquid ratio initially, and then stabilized at ratios higher than 500 (V/V). At reaction temperatures <533 K, the dehydration of 2-EtC6H4OH to EB was slow, and, consequently, the main products obtained were oxygen-containing intermediates (such as 2,3-DHBF and 2-EtC6H4OH). At 533 K, the conversion of 2-EtC6H4OH into EB began to increase dramatically, and the yield of the O-free product ECH started to increase rapidly. The reaction rate constants (k1–k5) at different temperatures were calculated using a pseudo first-order model, and the results showed that the model was suitable for describing the reaction kinetics of BF HDO over the Ni2P-O/MCM-41 catalyst. The results showed that the main O-free product was ECH and that demethylation was not favored over the Ni2P-O/MCM-41 catalyst. At 573 K, 3.0 MPa, and H2/liquid = 500 (V/V), the conversion of BF over Ni2P-O/MCM-41 reached 93% with a yield of O-free products of 91%.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.