Abstract
The kinetics of carbon monoxide methanation over Ni-Mo-SiO2 catalyst were studied. The model was developed based on catalyst tests carried out in a fixed-bed reactor at a reaction temperature varied from 300 °C to 450 °C under a pressure from 0.1 to 1.5 MPa with a weight hourly space velocity of 60,000 mL h−1 g−1. An orthogonal design method was adopted to select test points with temperature, pressure, and feed compositions as factors. Based on the experimental observations, a Langmuir–Hinshelwood equation kinetic model was formulated and its parameters were estimated by fitting the experimental data implemented in MATLAB. The activation energy for the formation of CH4 was 45.4 kJ mol−1 and comparing the experimental and model-predicted data showed that the proposed model gives a reasonable fit with an average absolute relative deviation of ±9.8%.
Keywords
Introduction
Since the crude oil crisis of the 1970s, the synthesis of synthetic natural gas (SNG, syngas) from coal has attracted increasing attention in both academia and industry. 1 The methanation of CO, namely the production of CH4 from the reaction between CO and H2, is important in satisfying the increasing demand for SNG. It SNG is usually produced by gasification of coal followed by a methanation process, which is a key element in SNG production. 2 Nickel-based catalysts have been generally employed in industry for the methanation process because of their relatively low cost and high activity. 2 On nickel-based catalysts, syngas is converted to CH4 and H2O accompanied with the water gas shift reaction (WGS reaction) as a side reaction. The kinetics of CO methanation on nickel-based catalysts have been studied, being mostly based on Langmuir–Hinshelwood (L-H) adsorption2,3 or an empirical approach.4,5 The kinetic model meets the needs for optimizing the reaction process and designing industrial reactors.
Whether the L-H kinetic model or the power equations model is used, the estimation of each parameter in the kinetic equation needs to be based on experimental data. For the chemical reaction process, the power equations kinetic model is mostly used for homogeneous reactions, while the L–H model is used for heterogeneous catalytic processes.6,7
Two main mechanisms have been proposed in the literature for the methanation of CO:
The first involves adsorbed CO dissociating to adsorbed carbon and oxygen, which are hydrogenated to CH4 and H2O.8,9
The second mechanism involves an oxygenated species, such as a COHx complex. 10
In our previous work, 11 we prepared a Ni-Mo-SiO2 catalyst by hydrothermal synthesis method and it showed good catalytic performance in the methanation reaction of syngas. The catalytic performance of Ni/SiO2, Ni-SiO2, and Ni-Mo-SiO2 catalysts at 0.1 MPa and 250–550 °C was investigated, CO was completely converted (>99%) over Ni-1.0%Mo-SiO2 and Ni-2.0%Mo-SiO2 catalysts at 300 °C, and was maintained in the range of 300 °C–500 °C. In contrast, for Ni/SiO2 and Ni-SiO2 catalysts, CO conversions were 40% and 17% at 300 °C, respectively. For Ni-Mo-SiO2 catalysts, the CH4 yields were above 90% at 350–450 °C, and the highest CH4 yield was about 95% at 400 °C. In contrast, Ni/SiO2 and Ni-SiO2 catalysts showed the best CH4 yield of 85% and 83% at 400 °C, respectively. 11 For a new catalytic system, such as Ni-0.5%Mo-SiO2 catalyst, a kinetic model elucidating the rate mechanistic step is essential for the design of a reactor and to predict the reactor performance confidently. Therefore, the study of the intrinsic kinetics of methanation has important guiding significance for engineering scaleup and reactor selection.
Due to the gas–solid heterogeneous catalytic reaction for syngas methanation, the L–H equation was adopted to study the intrinsic kinetics of syngas methanation on Ni-0.5%Mo-SiO2 catalyst in the fixed-bed reactor (MA11205, Micromeritics Company) and yielded the corresponding kinetic parameters. The effectiveness of the kinetics obtained was further verified by comparing the calculated results with the experimental results under varying reaction conditions.
Experimental
Catalyst preparation
The Ni-0.5%Mo-SiO2 catalyst was prepared by our hydrothermal method, and the detailed synthetic steps are reported in our previous work. 11 Ni(NO3)2·6H2O and (NH4)6Mo7O24·4H2O were used as the metal precursor salts. Hexadecyl trimethyl ammonium bromide and tetraethylorthosilicate (TEOS) were used as the surfactant and silica sources, respectively. Ni-0.5%Mo-SiO2 was prepared by the hydrothermal synthesis method according to the procedure of Xin and Zhang. 12 Typically, CTABr and NaOH were dissolved in 100 mL of deionized water with vigorous stirring at 40 °C. Likewise, Ni(NO3)2·6H2O and (NH4)6Mo7O24·4H2O were dissolved in 10 mL of deionized water, respectively. TEOS and the solutions of Ni and Mo salts were slowly dropped into the surfactant–water solution at the same time. The mixture was stirred vigorously for 2 h to obtain a gel which was transferred to an autoclave and crystallized statically at 120 °C for 24 h. After being cooled to room temperature, the resulting product was further filtered under reduced pressure better, washed with deionized water, and dried at 100 °C overnight. After calcination at 550 °C for 6 h with a heating rate of 1 °C min−1 in air, the Ni-Mo-incorporated SiO2 catalyst was obtained and designated as Ni-Mo-SiO2, in which the nickel molar content was 10%.
Experimental design
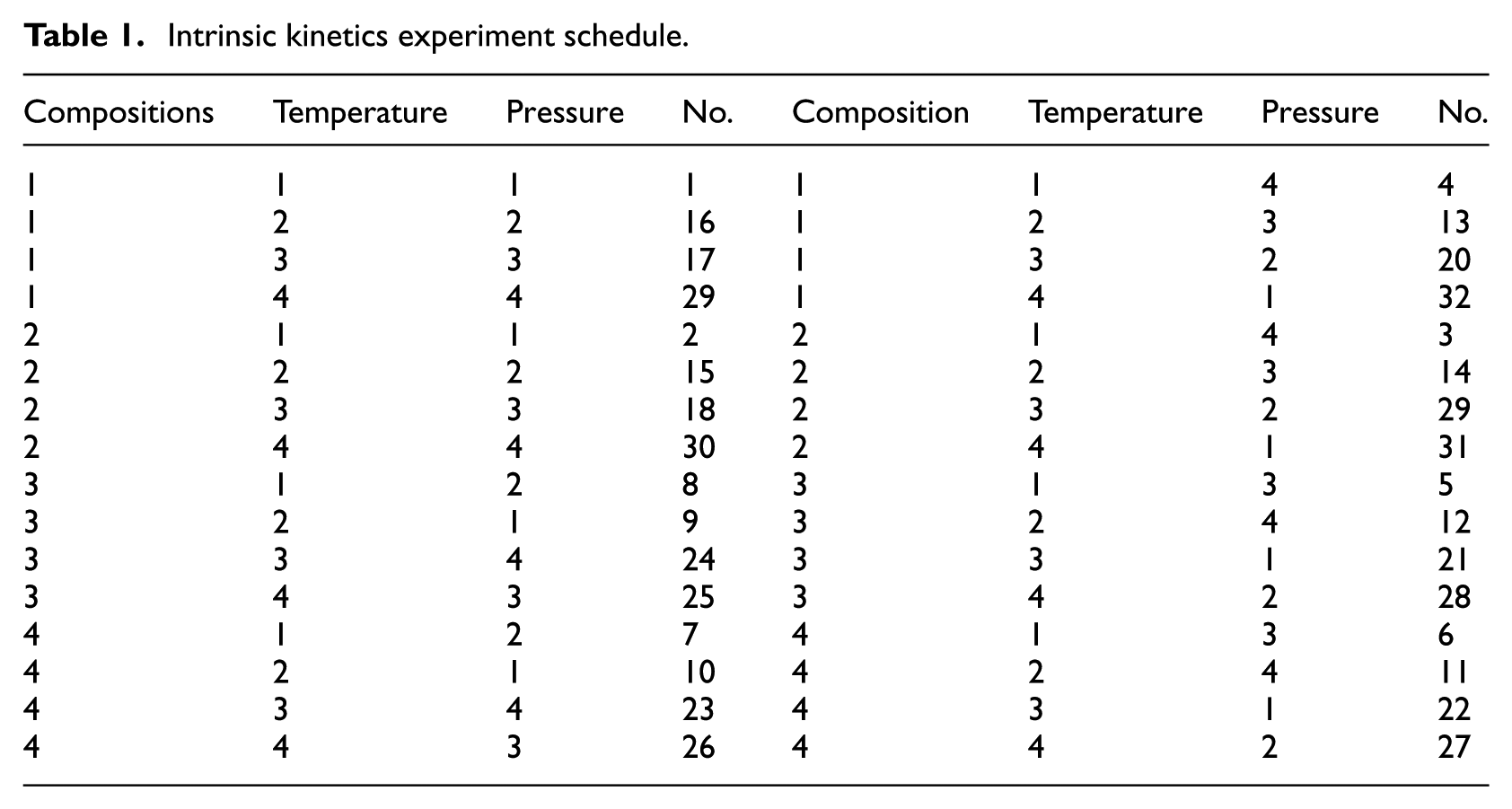
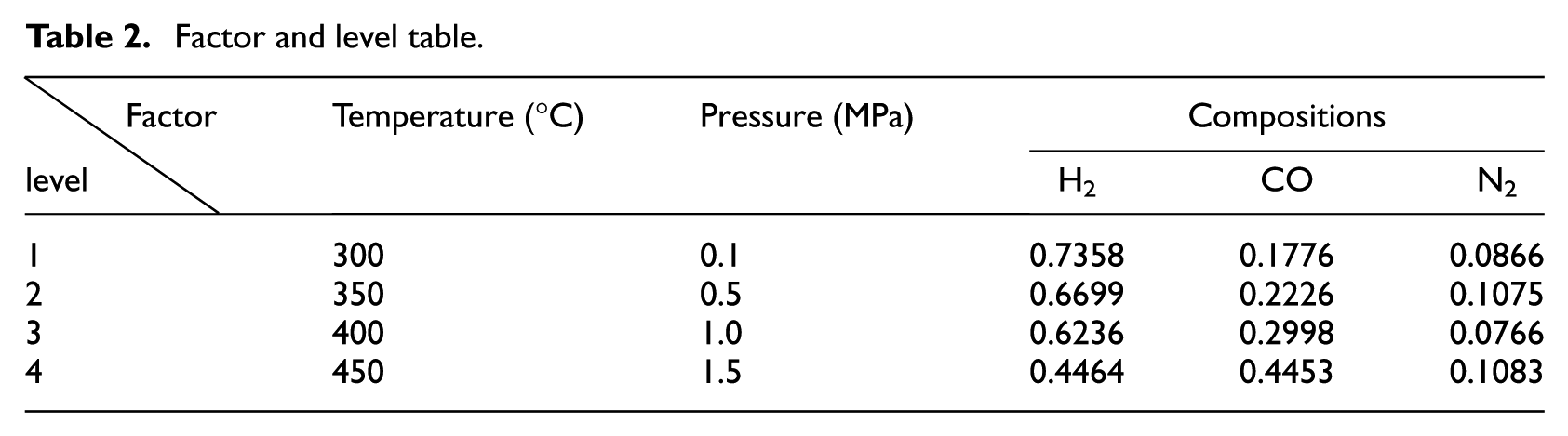
In order to reduce the number of experiments, and obtain more information, we adopted an orthogonal design method to select test points with temperature, pressure, and feed compositions as factors. The orthogonal table of L32(49) 13 was employed by each factor with four levels to complete the overall design of the experiment (see Tables 1 and 2).
Intrinsic kinetics experiment schedule.
Factor and level table.
Pre-experiment checks
Before the reaction system was tested, the instruments, meters, equipment, and piping were checked and corrected.
Tightness experiment
When the experimental equipment was established, the system pressure was increased to 1.5 MPa, and leak detection of the system was performed with suds, and no leakage point was found. The system pressure decreased by 0.01 MPa in 20 h with feed gas off.
Blank experiment
No catalyst was used in the reactor and only blank quartz sand was used. The other conditions of the system were consistent with the kinetic test. The blank experiment was carried out with the raw material gas under the dynamic reaction condition. The results showed that the reaction tube and gas pipeline and inert filler quartz sand had no catalytic effect on the raw material gases. Therefore, any catalysis of other substances in the system except the catalyst was not considered.
Elimination of the influence of internal and external diffusion
The elimination of an internal diffusion effect was carried out at atmospheric pressure in a pressurized microreactor with a reaction tube with inner diameter of 8 mm and a catalyst dosage of 0.10 g. Prior to reaction, the catalyst was reduced with pure H2 (50 mL min−1) at 600 °C, then the temperature was dropped to 350 °C under a N2 atmosphere in 0.5 h, and then the raw gas (n(H2): n(CO): n(N2) = 3:1:1, total weight hourly space velocity (WHSV) = 30,000 mL h−1 g−1) was introduced. Gas products were analyzed by gas chromatograph with a thermal conductivity (TCD) detector (Tianmei, GC-7900) and trace amounts of CO and CO2 were detected by flame ionization detector (FID) through the methanation reformer. The particle sizes of the catalyst were 40, 60, 80, and 100 mesh, respectively.
The elimination of an external diffusion effect was carried out at atmospheric pressure in a pressurized microreactor with a reaction tube with inner diameter of 8 mm and a catalyst dosage of 0.10 g. Prior to reaction, the catalyst was reduced with pure H2 (50 mL min−1) at 600 °C, then the temperature was dropped to 350 °C under a N2 atmosphere in 0.5 h, and then the raw gas (n(H2): n(CO): n(N2) = 3:1:1) was introduced. Gas products were analyzed by gas chromatograph with the TCD detector and trace amounts of CO and CO2 were detected by FID detection through the methanation reformer. The particle size of the catalyst were 80–100 mesh and total gas WHSV values were 15,000, 30,000, 45,000, 60,000 and 90,000 mL h−1 g−1, respectively.
Checkout of constant temperature zone
Determination of the axial temperature distribution of the whole reaction tube was carried out by moving the thermocouple in the thermocouple sleeve to select an isothermal section loading catalyst of about 100 mm. In order to improve the isothermal property of the catalyst bed, the catalyst was diluted with quartz sand and its particle size was consistent with that of the catalyst (Vquartz sand/Vcatalyst = 2:1). The average particle size of 80–100 mesh catalyst, dp = 0.16 mm, reactor diameter dt = 8 mm, catalyst bed height H = 40 mm, dt/dp = 50 > 10, and H/dp = 250 > 100, so it met the requirements of the intrinsic reaction kinetics test.
Kinetics experiment
The activity of Ni-0.5%Mo-SiO2 catalyst in the stability test of 100 h showed no obvious loss. 11 Therefore, it could be considered that the activity was stable in the kinetic test. The Ni-0.5%Mo-SiO2 catalyst was first heated to 600 °C in a N2 atmosphere, then reduced for 2 h at 600 °C in a pure H2 atmosphere (50 mL min−1), then cooled to the reaction temperature in a N2 atmosphere and then it was switched to the feed gas (n(H2): n(CO): n(N2) = 3:1:1) for the activity test. According to the principle of changing the pressure and composition under isothermal conditions, the experimental points were designed according to the orthogonal table, and the experimental order is shown in Table 2, column 4.
After adjusting the reaction conditions, the experimental data were measured when the reaction was stable. The interval between the two reaction conditions was 3 h, which ensured that the gas exchange was complete and three parallel samples were taken at each reaction condition. When the key component contents of CO and CH4 of the outlet showed no change, it was considered that the experimental point had reached the steady state with the average value as the final result. The first test point was repeated after completing all experimental design points and the concentration of CH4 and CO showed no change, indicating that the intrinsic kinetics were tested in the stable range of catalytic activity of the catalyst.
To calculate CO conversion and CH4 selectivity, the outlet gases were collected after half an hour of steady-state operation at each temperature. The calculation formulae were described as follows
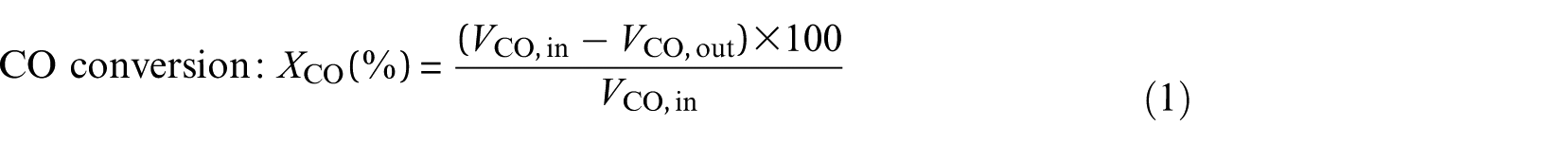
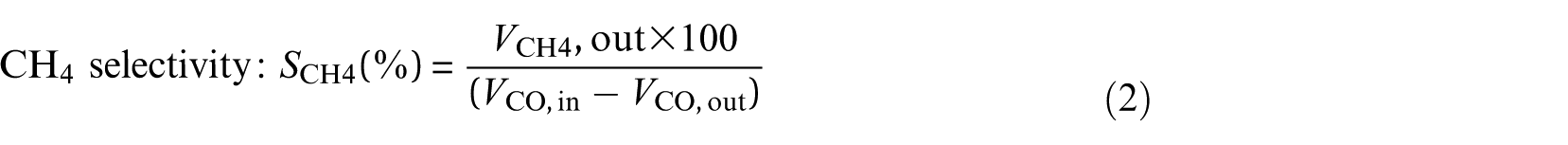

Results and discussion
Physicochemical properties of catalyst
The physical properties of the Ni-0.5%Mo-SiO2 catalyst were characterized using N2 physical adsorption–desorption instruments. The surface area, pore size, and pore volume of the catalyst derived from N2 desorption isotherms are shown in Table 3. The catalyst had a surface area of 436 m2 g−1 and a relatively small pore volume of 0.87 cm3 g−1. In addition, the compositions of bulk and surface metals on the catalysts were calculated by inductively coupled plasma spectrometry (ICP) and X-ray photo electron spectra (XPS) measurement, and the results are shown in Table 3. It can be seen from Table 3 that the nickel and molybdenum contents of the catalysts measured by ICP are close to the theory design values, indicating that all the nickel and molybdenum were successfully incorporated into the catalysts.
Physicochemical property of Ni-5%Mo-SiO2 catalyst.

Calculated by the BET equation.
BJH desorption pore volume.
Calcultaed by ICP.
Calcultaed by XPS.
Effect of catalyst particle size on catalyst activity
The catalytic activities of catalysts with different particle sizes were measured at atmospheric pressure and 350 °C with the same amount of catalyst and gas velocity, and the effect of particle size of the catalyst on conversion of CO is shown in Figure 1. Due to the presence of internal diffusion resistance, the gas concentration at the outer surface of the catalyst is different from that in the capillary hole of the catalyst particles. For a thicker capillary, the inner diffusion resistance and the concentration difference were smaller. When the catalyst was prepared, the pore size of catalyst could not be changed. However, if the catalyst’s capillary pipe becomes shorter and more internal surfaces are exposed, then the internal diffusion resistance of the catalyst will be smaller. Therefore, changing the catalyst particle size is an effective way to eliminate the internal diffusion effect. As shown in Figure 1, when the particle size of catalyst was less than 60 mesh (0.28 mm), the conversion of CO remained almost unchanged, indicating that there was no internal diffusion effect. Therefore, the particle size of the catalyst used in the determination of intrinsic kinetic data was selected as 80–100 mesh.
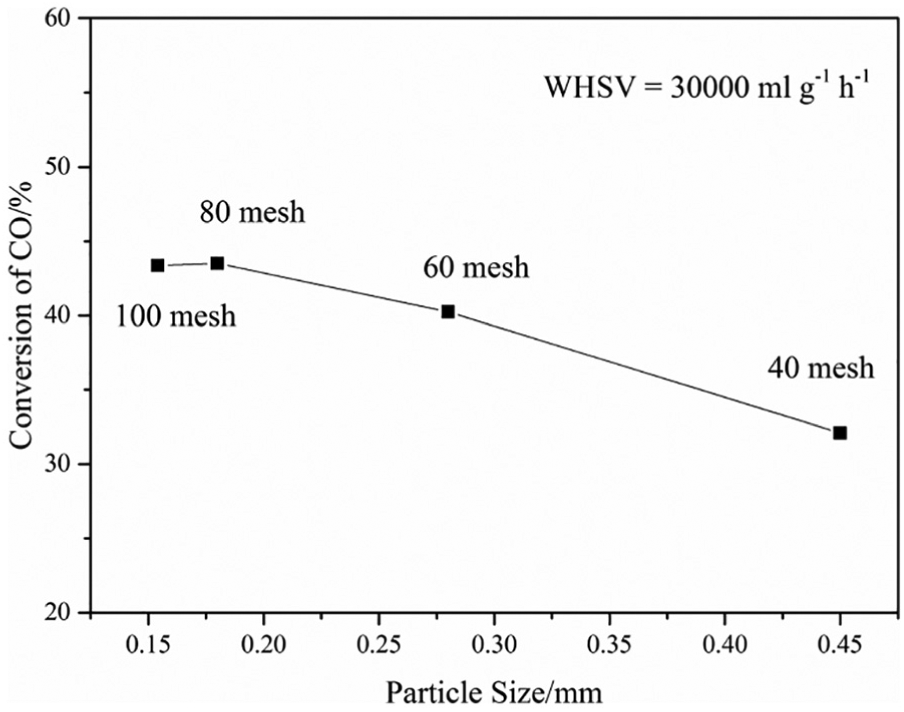
Effect of particle size of the catalyst on conversion of CO.
Effect of gas velocity on catalyst activity
The catalytic activities of catalysts under different gas velocities (WHSV = 15,000, 30,000, 45,000, 60,000, 90,000 mL h−1 g−1) were measured at atmospheric pressure and 350 °C with the same amount of catalyst (100 mg) and the same particle size (80–100 mesh) and the effect of gas velocity on conversion of CO is shown in Figure 2. When the total WHSV was greater than 45,000 mL h−1 g−1, the CO conversion remained at around 40.5%, which indicated that the level of gas turbulence increased with increasing space velocity of the feed gas, which greatly reduced the resistance of the gas film, thus eliminating the influence of external diffusion. Therefore, the particle size of the catalyst selected in the kinetic experiment was 80–100 mesh, and the space velocity was about 60,000 mL h−1 g−1.
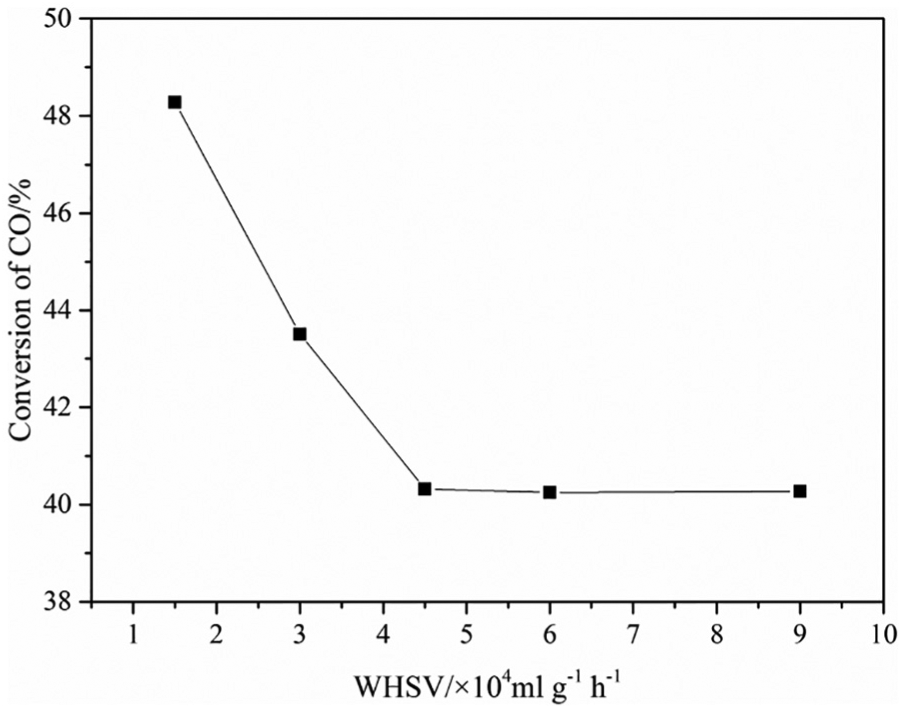
Effect of WHSV of the total gas on conversion of CO.
Characteristics of reaction system
The methanation process is mainly composed of the following reaction equations
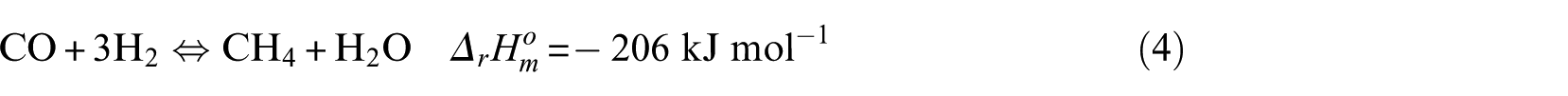
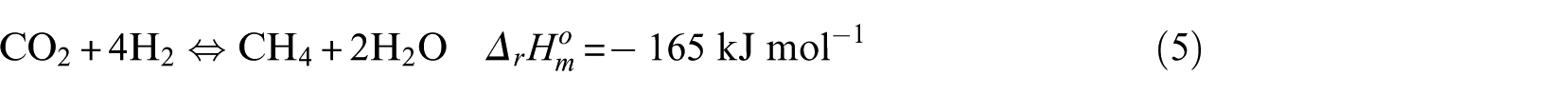

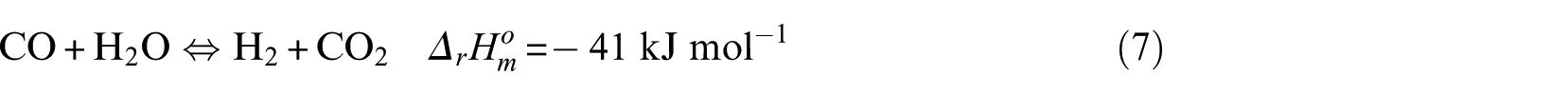

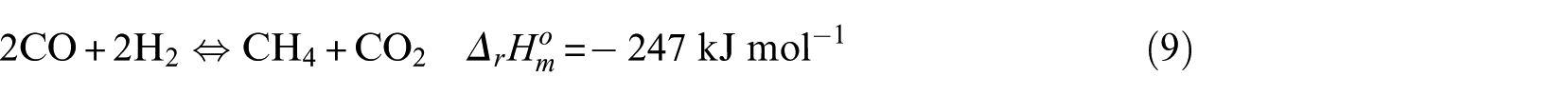
In the kinetic experiment, the Ni-0.5%Mo-SiO2 catalyst showed no obvious carbon deposition behavior under the test conditions, so it can be concluded that CO disproportionation (equation (6)) and the CH4 cracking reaction (equation (8)) did not occur. The key components were CO and CH4, and the critical component score was 2. According to the reaction characteristics of Ni-0.5%Mo-SiO2 catalyst, the methanation reaction (equation (4)) was selected as the main reaction.
According to the relationship between stoichiometry and thermodynamics, the characteristic analysis of methanation reaction system14–17 is as follows:
The methanation reaction and the water steam shift reaction are all exothermic reactions, and the reaction equilibrium constant decreases with increase in the reaction temperature; the methanation reaction is a gas molar number decreasing reaction, and high pressure and low temperature are favorable for the formation of CH4.
The methanation reaction system is a strongly exothermic process, and the adiabatic temperature of the gas rises by 60 °C∼70 °C with conversion of 1% CO to CH4.
Equation (6) and equation (8) refer to the carbon deposition reaction, and the deactivation of the catalyst is mainly due to the occurrence of carbon deposition. Equation (6) is the main reaction at low reaction temperature; equation (8) is the main reaction at high reaction temperature, the carbon deposition reaction will not occur in the temperature range of 260 °C∼490 °C and n(H2): n(CO) > 2.
Results of kinetic experiments
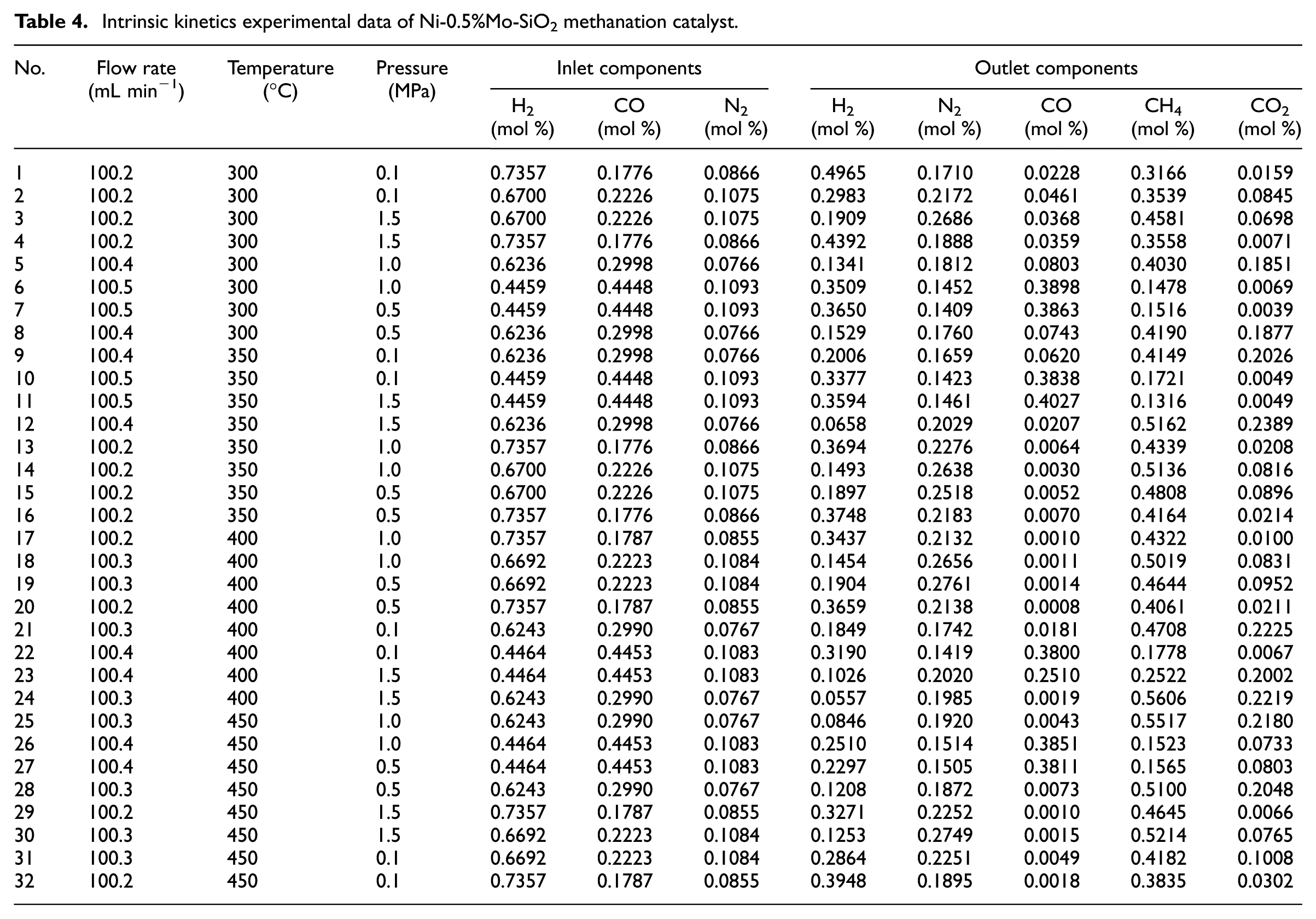
The activity of Ni-0.5%Mo-SiO2 catalyst in the stability test of 100 h showed no obvious loss. 11 Therefore, it could be considered that the activity was stable in the kinetic test. The Ni-0.5%Mo-SiO2 catalyst was first heated to 600 °C in a N2 atmosphere, then reduced for 2 h at 600 °C in a pure H2 atmosphere (50 mL min−1), and then cooled to the reaction temperature in a N2 atmosphere. After adjusting the reaction conditions, the experimental data were measured when the reaction was stable. The interval between the two reaction conditions was 3 h, which ensured that the gas exchange was complete and three parallel samples were taken for each reaction condition. When the key component contents of CO and CH4 of the outlet showed no change, it was considered that the experimental point had reached the steady state with the average value as the final result. The results of the kinetic test are shown in Table 4.
Intrinsic kinetics experimental data of Ni-0.5%Mo-SiO2 methanation catalyst.
Parameter estimation of the intrinsic kinetic equation model
Checkout of experimental data
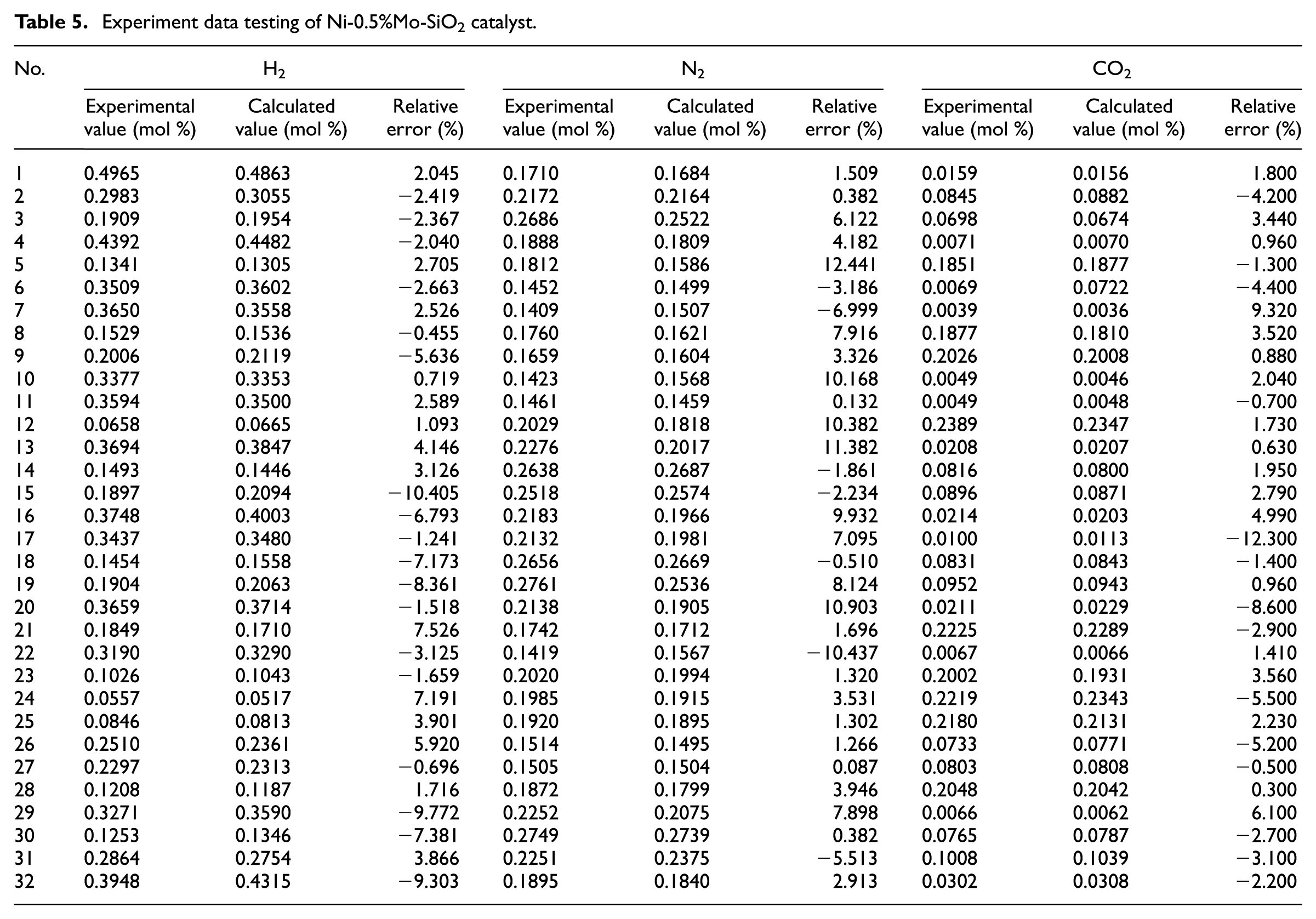
The material balance of the experimental data was calculated and deduced with CH4 and CO as the key components. The composition and experimental values of the dry basis composition of H2, N2, and CO2 calculated are listed in Table 5, and their relative errors were less than 10.4%, 12.4%, and 9.3%, respectively. It was demonstrated that the experimental results were basically reliable, and the experimental data obtained could be used for the establishment of the intrinsic kinetic model.
Experiment data testing of Ni-0.5%Mo-SiO2 catalyst.
Conversion between dry and wet basis of outlet
The wet basis composition at the outlet of the reactor could be obtained by using the conversion relationship between the dry basis and the wet basis. The results are shown in Table 6.
Wet basis composition of the reactor outlet.
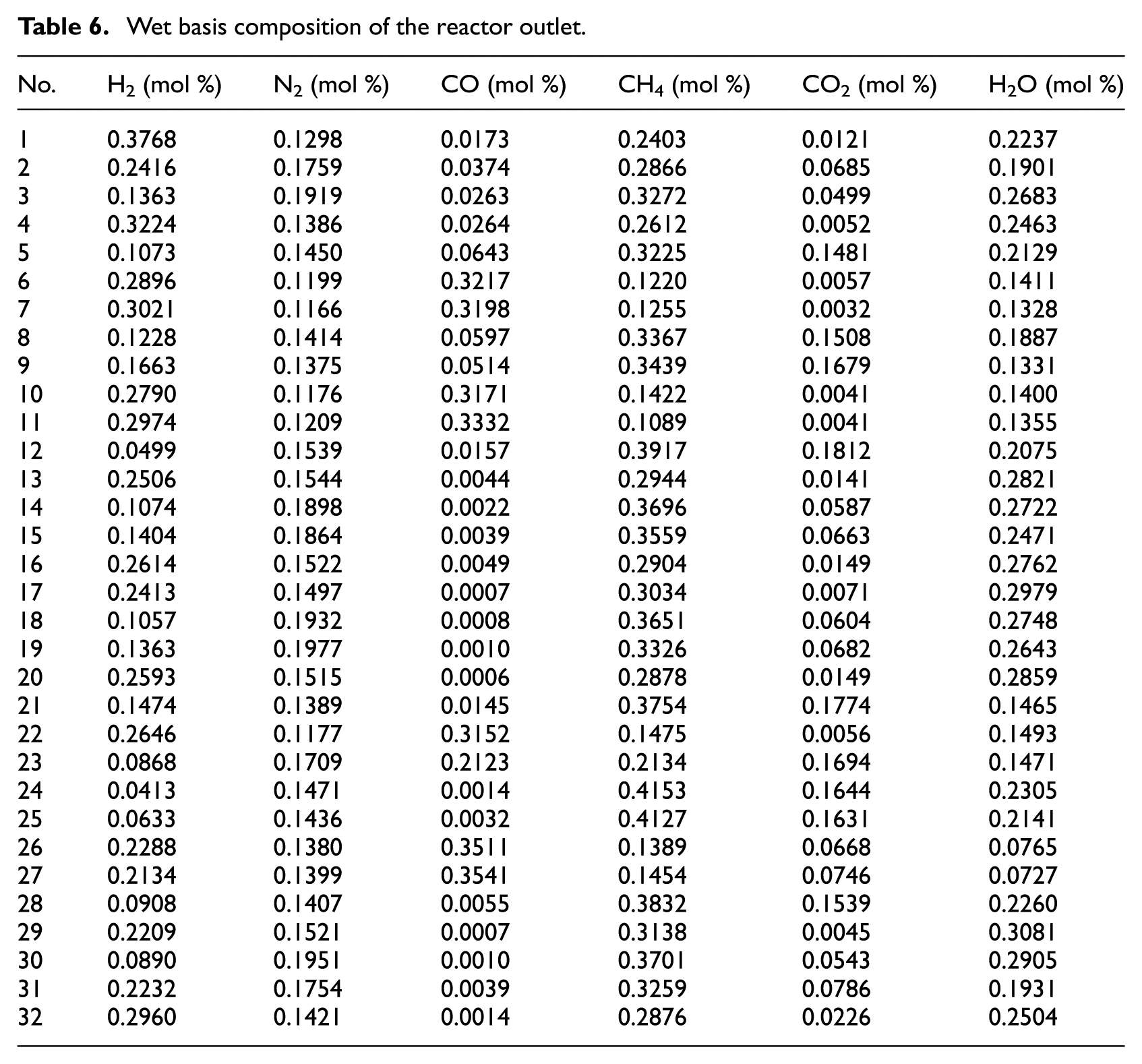
Parameter estimation
Since the methanation reaction was a heterogeneous gas–solid reaction, the L–H model was used as a kinetic model for the Ni-0.5%Mo-SiO2 methanation catalyst
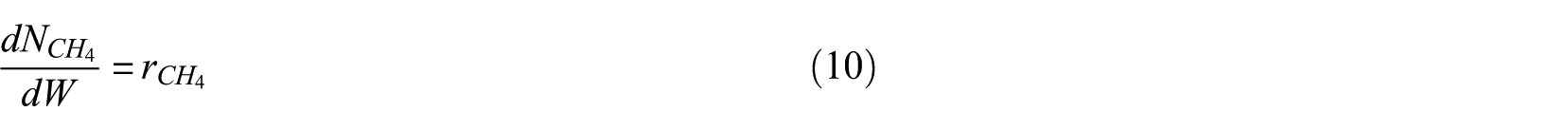

In general, the reaction rate constant k and the adsorption equilibrium constant K can be expressed by the Arrhenius and van’t Hoff formulae


where k0 and K0 are pre-exponential factors, and Ea and ΔH are the activation energy and reaction enthalpy, respectively.
In order to facilitate parameter estimation, equation (11) can be expressed as
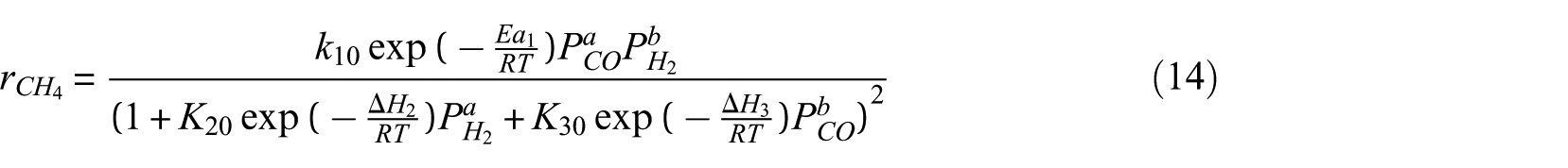
There are many methods of parameter estimation, such as the Gauss–Newton method, Powell method, damped least squares method, modified Gauss–Newton method, Runge–Kutta method, simplex method, and so on. The ideal parameter estimation method should have the characteristics of low initial value requirement, fast convergence speed, and high estimation accuracy. In this article, the range values of the parameters were searched first, and then the optimal parameters were obtained by the least squares method using MATLAB.
The intrinsic kinetic model of syngas methanation over Ni-0.5%Mo-SiO2 methanation catalyst was obtained by parameter estimation of the experimental data
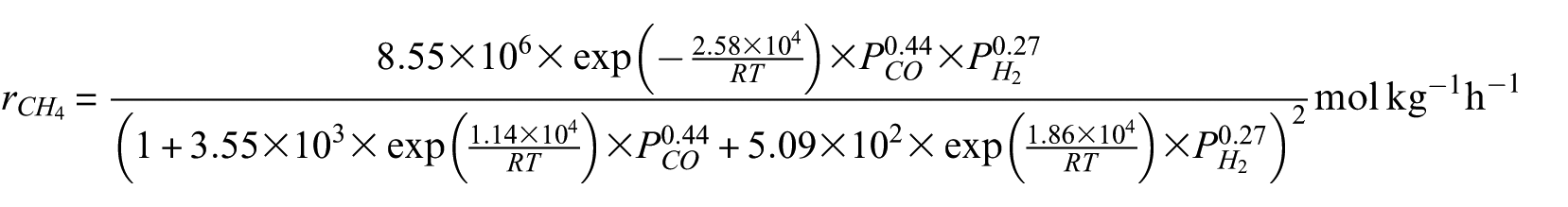
Model checking
Residual test
The comparison between the experimental values and the simulation values of the reaction rate of the outlet critical component CH4 is shown in Figure 3. It can be seen in Figure 3 that the residual distribution accorded with the normal distribution, and the relative error was less than 9.8%.
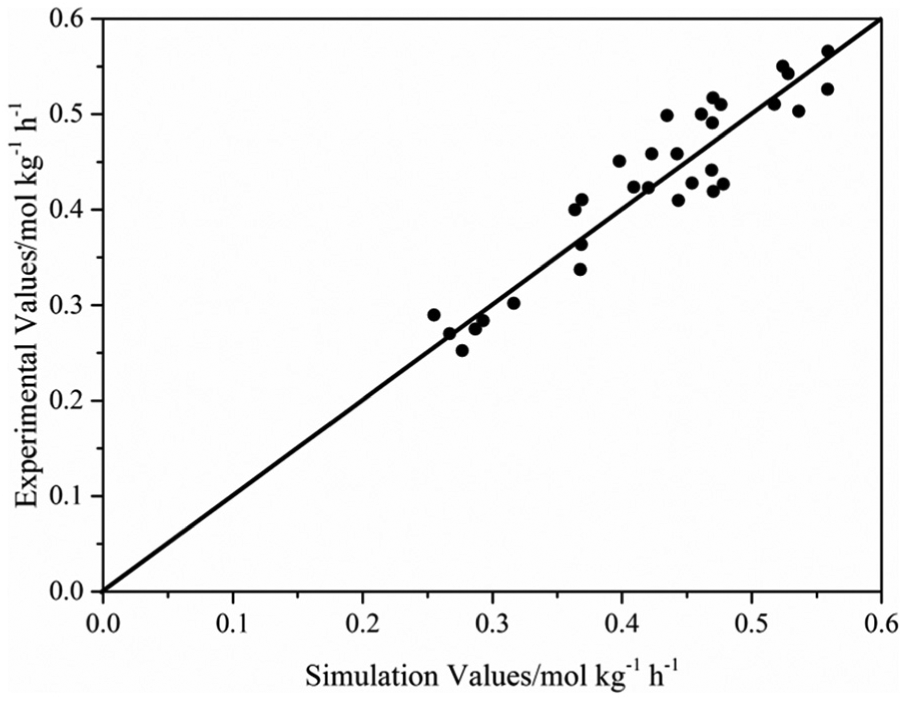
Comparison between experimental values and simulation values of CH4 reaction rate.
Checkout of ρ2F
According to the definition

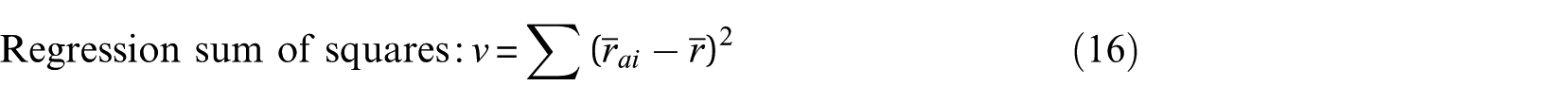



where ri is the experimental value, rai is the model simulation value,
If F > F1-α(P, N-P-1), it shows that the model is significant at level α. For nonlinear regression, it is generally believed that the F > 10 F1-α(P, N–P–1) model is highly significant. For the relevant index, when ρ2 > 0.9, the regression model is valid. The simulation values of the model statistic and the standard values under different confidence levels are shown in Table 7. Table 7 shows that ρ2 > 0.99 and Fsimulation ≫ 10Fstandard(0.05). Therefore, it is believed that the intrinsic kinetic model established in this article is credible in the range of temperature, pressure, and concentration studied. 18
The model statistics.

Arrhenius linear regression
The regression rate constant kCH4 was obtained at the isothermal experimental point, and the results are shown in Table 8. An Arrhenius plot of the data for four temperatures gave an excellent fit to a straight line, yielding an activation energy of 45.4 kJ mol−1 and the pre-exponential factor of methanation was 1.4836 × 10−5 mol−1 kg−1 h−1 calculated from the slope and intercept. The above results show that the estimated parameters are credible, and the estimated values of reaction order are accurate.
Reaction rate at different temperatures.

The above results also confirmed that the influence of mass transfer process in methanation was completely eliminated, and the reaction mechanism did not change in the range of temperatures tested.
Conclusion
In this article, the L–H kinetic model was adopted in the intrinsic kinetic study of methanation of synthetic gas in a fixed-bed reactor over Ni-0.5%Mo-SiO2 catalyst. The kinetic model and parameters based on the catalyst were obtained. The results of material balance showed that the experimental results were basically reliable. The pre-exponential factor of methanation was 1.4836 × 10−5 mol−1 kg−1 h−1 calculated from the slope and intercept and the activation energy, Ea = 45.4 kJ mol−1. According to the residual test, statistical test, and Arrhenius test, it could be concluded that the L–H kinetic model of syngas methanation over Ni-0.5%Mo-SiO2 catalyst fitted well with significant regression of the parameters and gave a reasonable residual distribution. The established model could truly reflect the reaction characteristics of syngas methanation over Ni-0.5%Mo-SiO2 catalyst, and was completely reasonable and reliable.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support from the Doctoral Foundation of Shijiazhuang University of Applied Technology (15YB001).
