Abstract
Ordered mesoporous silica, SBA-15 and MCM-41, and three-dimensionally ordered macroporous SiO2 were used as the supports of H4PMo11VO40 heteropolyacid for methacrolein oxidation. The dispersion and structural evolutions of the heteropolyacid along with thermal treatment were investigated. It was found that the heteropolyacid entered the one-dimensional mesoporous channels of SBA-15 and MCM-41, and the crystallization and growth were limited, leading to high dispersion of the heteropolyacid. However, the thermal stability was decreased under high dispersion. The migration of the heteropolyacid was observed to the end of the one-dimensional channels of SBA-15 and the outer surface of MCM-41 with calcination, accompanied by the decomposition of the heteropolyacid and the formation of MoO3. In comparison, the crystallization and growth of heteropolyacid were not limited in the open macropores of three-dimensionally ordered macroporous SiO2. Dispersed particles on the surface of the macropores with size of about 5 nm exhibited a higher thermal stability. The decomposition of the heteropolyacid in the SBA-15 and MCM-41 supported catalysts resulted in the loss of strong acid sites, causing low selectivity to methacrylic acid in methacrolein oxidation. High thermal stability with high exposure of the active sites in the three-dimensionally ordered macroporous SiO2 supported catalyst contributed to the enhancement in the catalytic performance.

Introduction
Due to the desirable acidity and oxidizing ability, heteropolyacids (HPAs) have been widely used as the catalysts in selective oxidation and acid-catalyzed reactions, such as the oxidation of methacrolein (MAL) to methacrylic acid (MAA), which is a key step in an industrial process for methyl methacrylate manufacturing via the so-called “C4 route.” 1
The surface areas of HPAs are usually low, which limit the exposure of the active sites and result in insufficient activity and low space time yield. Supporting HPAs on various supports with high surface areas has been investigated for the enhancement in catalytic activity. The most commonly used supports are various forms of SiO2, attributed to the tunable pore structure and abundant surface hydroxyl groups. Among which, ordered mesoporous silica materials, such as MCM-41 and SBA-15, have been used as the supports of HPAs catalysts in oxidative desulfurization,2–4 selective oxidation,5–7 esterification,8–10 dehydration of glycerol to acrolein,11,12 ethanol dehydration,13,14 acylation, 15 hydroxylation,16,17 and isomerization,18,19 and so on. Mesocellular foam silica, as a modified form of SBA-15 with three-dimensional pore structure and larger pore diameter, has been used as a support for Cs2.5H0.5PW12O40, and the obtained catalyst showed good performances in benzylation of phenol 20 and hydroxyalkylation of anisole with benzaldehyde. 21
It has been found that HPA can enter the mesopores of MCM-41 or SBA-15, and the resultant high dispersion enables more active sites, bringing about the improvement in catalytic activity. However, to date, there have been only a few reports on supported HPA employed as the catalysts for the oxidation of MAL to MAA, and the catalytic performances were far from satisfactory. Kanno and colleagues22,23 supported H4PMo11VO40 on SiO2 and further on ammonia-modified SiO2, giving higher catalytic activity than unsupported H4PMo11VO40, whereas with a selectivity to MAA below 80% when the MAL conversion was higher than 50%. Kim et al. 24 used SBA-15 to support H5PMo10V2O40, on which an MAL conversion of 74% with an MAA selectivity of 40% was obtained. The reaction temperature for MAL oxidation is usually up to about 330°C, the thermal stability of the supported HPAs may be a problem, especially when they are highly dispersed.
In our previous work, H4PMo11VO40 was supported on Cu-doped three-dimensionally ordered macroporous (3DOM) SiO2, and the effects of Cu doping amount on the physico-chemical properties of the 3DOM supports and the catalysts, as well as the catalytic property were investigated. 25 It was found that Cu doping in 3DOM SiO2 formed an efficient support for HPA, bringing about high catalytic performance for the oxidation of MAL to MAA. In this study, a simpler method was applied for the modification of 3DOM SiO2 with CuSO4, and enhanced performances of the as-synthesized catalyst were obtained. More importantly, the effects of supports pore structure on the dispersion and thermal stability of the HPA were explored using different silica-based supports, and the structural evolution of the HPA along with thermal treatment was investigated in detail. It was found that, under thermal treatment, highly dispersed HPA in the one-dimensional (1D) mesopores of SBA-15 or MCM-41 tended to decompose more easily than that supported on 3DOM SiO2, resulting in the loss of strong acid sites and the sharp decline of catalytic performance. In comparison, the crystallization and growth of HPA was not limited within the open macropores of 3DOM SiO2, rendering it a higher thermal stability. High catalytic performance in MAL oxidation was obtained on 3DOM SiO2-supported HPA catalyst because of the intact Keggin structure and the increased specific surface area.
Results and discussion
Structural features of the supported HPA
Bulk and the supported H4PMo11VO40 were analyzed by thermal gravimetric (TG) measurement, and the results are shown in Figure 1. There are two characteristic weight losses in the TG curve of bulk H4PMo11VO40. The first one, between 50°C and 250°C, is attributed to the loss of crystallization water, and the second one, between 250°C and 380°C, is ascribed to the loss of constitutional water. 26 The total weight loss is 8.26%, very close to the theoretical value (8.49%) for H4PMo11VO40 with seven H2O molecules. Two stages of weight loss are also seen in the curves of the supported samples. If the H4PMo11VO40 is present on the supports with seven water molecules per Keggin unit, the total weight loss would be 3.67% according to the loading amount of the HPA on the supports. The actual value obtained by the TG curve is 2.90% for HPA/3DOM SiO2, indicating the lower extent of hydration of the HPA when dispersed on 3DOM SiO2. In comparison, the TG curves of HPA/SBA-15 and HPA/MCM-41 are quite similar and the total weight loss reaches 10.93%, according to which the water content is calculated to be equivalent to 27 H2O molecules per Keggin unit. Thus, the relatively stronger adsorption properties for water of the mesoporous molecular sieves provide an environment making the HPA tend to be highly hydrated.

TG curves of H4PMo11VO40 and the supported samples before calcination.
The X-ray diffraction (XRD) patterns of the supported catalysts calcined at 350°C are shown in Figure 2. The supported catalysts uncalcined and calcined at 300°C, 325°C, and 400°C were also characterized by XRD analysis (Supplemental Figure S1), in order to explore the changes in phase composition with thermal treatment. The XRD patterns of the uncalcined HPA/3DOM SiO2 can be well indexed to the Keggin-type HPA with a triclinic structure (JCPDF No. 75-1588). Differently, there are almost no reflections in the patterns of uncalcined HPA/SBA-15 and HPA/MCM-41, indicating the supported HPA without good crystallization. It might be due to the dispersion of the HPA in the 1D mesoporous channels, which limited the aggregation of the HPA. With calcination at 300°C, all the three samples do not show obvious changes in XRD patterns compared with the uncalcined ones, indicating that the calcination mainly caused the loss of water as observed in the TG curves. With the calcination temperature rising to 325°C, monoclinic MoO3 (JCPDF No. 47-1081) is observed in the patterns of HPA/SBA-15 and HPA/MCM-41, with an increase in intensity at 350°C. There are no reflections of HPA. The intensity of reflections in HPA/MCM-41 is weaker than that observed in HPA/SBA-15, revealing that the relatively small pore size of MCM-41 limited the growth of the supported species. In comparison, HPA/3DOM SiO2 retained Keggin-type HPA till the calcination temperature reached 350°C. When the calcination temperature was increased to 400°C, monoclinic MoO3 decreased along with the increase of orthorhombic MoO3 (JCPDF No. 35-0609) in HPA/SBA-15 and HPA/MCM-41. HPA disappeared in the patterns of HPA/3DOM SiO2, with the formation of monoclinic MoO3 and orthorhombic MoO3.

XRD patterns of the supported catalysts calcined at 350°C.
The above results indicate that the pore structure of the supports have significant effects on the crystallization and thermal stability of the supported HPA. The crystallization of HPA occurred in the drying process, rather than limited in the open macropores of 3DOM SiO2. So reflections for HPA were found in the patterns of uncalcined HPA/3DOM SiO2. In contrast, the growth of the HPA was limited in the 1D mesopores of SBA-15 and MCM-41, leading to high dispersion of the HPA. Therefore, there were no reflections for HPA observed. When the calcination temperature was 350°C, decomposition of the HPA caused the formation of monoclinic MoO3, which tended to aggregate under thermal treatment. So it was observed in the XRD patterns of HPA/SBA-15 and HPA/MCM-41. Meanwhile, thermal treatment drove the transformation from monoclinic MoO3 to orthorhombic MoO3.
MoO3 and MoO3 hydrates are known to display a variety of structural types involving linked MoO6 octahedra, 27 among which the orthorhombic MoO3 has been studied more extensively. Distorted MoO6 octahedra share both edges and corners to form a unique thermodynamically stable structure in orthorhombic MoO3. While the monoclinic MoO3 involving corner sharing, distorted octahedra in a ReO3-type structure is a metastable phase. It has been reported 28 that monoclinic MoO3 can be reversely transformed into H3PMo12O40 with phosphorus oxide in the presence of water vapor. So the collapse of the HPA is limited. However, this reverse transformation did not take place on orthorhombic MoO3.
The Fourier-transform infrared (FTIR) spectra of the supported catalysts calcined at 350°C are shown in Figure 3. The absorption peaks in the FTIR spectra were generated from three aspects: the SiO2 supports, the HPA, and the possible decomposition products of the HPA. Six absorption peaks can be observed in the spectra of the uncalcined samples. The peaks at 1093, 795, and 467 cm−1 are attributed to the dissymmetry stretching, symmetry stretching, and bending vibrations of Si–O–Si, respectively. 29 The peaks at 1066, 963, and 869 cm−1 are related to the Keggin structure of the HPA, representing the P–O, Mo–Ot and Mo–Ob–Mo asymmetric stretching vibrations, respectively30,31 (Ot: terminal oxygen atoms, Ob: bridging oxygen atoms of the connecting O-bridges between the Keggin Mo3O13 triads). There should be an absorption peak at about 780 cm−1 corresponding to the Mo–Oc–Mo asymmetric stretching vibration of the HPA (Oc: bridging oxygen atoms of the internal O-bridges in the Mo3O13 triads), which has probably been overlapped with the symmetry stretching peak of Si–O–Si. The FTIR spectra of the supported catalysts uncalcined and calcined at 300°C, 325°C, and 400°C are shown in Supplemental Figure S2. The strong characteristic peaks of the Keggin structure indicate the existence of the HPA in HPA/SBA-15 and HPA/MCM-41, and it should be highly dispersed in the mesopores of the supports according to the XRD results. Both the FTIR and the XRD results show that the supported HPA can retain stable structure with calcination at 300°C. When the calcination temperature was increased to 350°C, the characteristic absorption peaks of Keggin structure decreased greatly in the spectra of HPA/SBA-15 and HPA/MCM-41, due to the decomposition of the HPA. And meanwhile obvious absorption peaks at about 620 cm−1 can be found, indicating the formation of MoO3. There are still strong characteristic peaks of Keggin structure in HPA/3DOM SiO2 calcined at 350°C, showing its higher thermal stability than the other two samples. When the calcination temperature was 400°C, the characteristic peaks of Keggin structure can hardly be seen for each sample, demonstrating the nearly complete decomposition of the HPA.
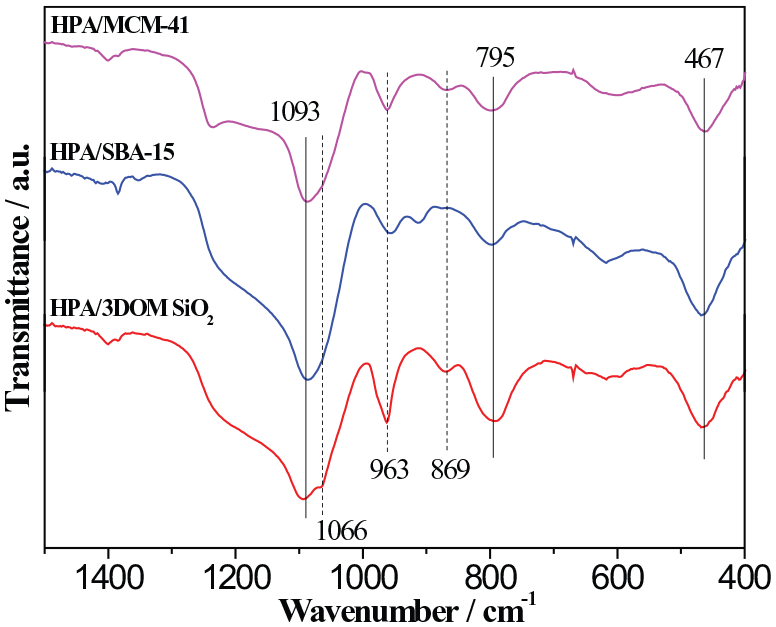
FTIR spectra of the supported catalysts calcined at 350°C.
Morphology of the supported catalysts
Scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDS) mapping measurements were carried out in order to observe the morphology of the catalysts and determine the element distributions, as shown in Supplemental Figure S3. The elemental mappings clearly show that Cu, P, Mo, and V exhibit uniform distributions in a large scale for all these three samples.
Transmission electron microscopy (TEM) images of the supported catalysts calcined at 350°C, and those uncalcined are shown in Figure 4. The pore morphology of the supports can be seen by the TEM images. There are interconnected macropores with size of about 250 nm in 3DOM SiO2, and there are 1D mesoporous channels in SBA-15 and MCM-41 with the pore diameter of 8.69 nm and 3.57 nm, respectively. HPA particles with size of about 5 nm can be observed in the macropores of the uncalcined HPA/3DOM SiO2 (Figure 4(a)). In comparison, no aggregated particles are found in the channels or on the outer surface of HPA/SBA-15 and HPA/MCM-41 (Figure 4(c) and (e)), indicating the high dispersion of the HPA, and it is consistent with the XRD results. After having been calcined at 350°C, the size of the supported HPA in HPA/3DOM SiO2 increased obviously as shown in Figure 4(b).

TEM images of the supported catalysts before ((a), (c), (e)) and after ((b), (d), (f)) calcination at 350°C. ((a), (b)) HPA/3DOM SiO2, ((c), (d)) HPA/SBA-15, and ((e), (f)) HPA/MCM-41.
As shown in Figure 4(d) and (f), there are also nanoparticles appearing in HPA/SBA-15 and HPA/MCM-41 after they have been calcined at 350°C. However, formation of these nanoparticles is not due to the growth of the HPA, as the situation in HPA/3DOM SiO2. Because as it has been found by XRD and FTIR measurements, most of the HPA decomposes to MoO3 after calcination at 350°C. Thus, it can be considered that the HPA will become more prone to decomposition if it was highly dispersed on the support. Moreover, these nanoparticles are found to be mainly distributed at the ends of the 1D channels of HPA/SBA-15 or on the outer surfaces of HPA/MCM-41, as shown in the high-magnification images (Figure 4(d) and (f)). It indicates that the HPA tends to migrate from the inside of the channel to the outside during calcination. The relative higher content of crystallization water, as found by TG measurement, probably contributes to such migration. Figure 4(d) shows that some channels of HPA/SBA-15 might be blocked by the aggregated particles. The pore diameter of HPA/MCM-41 is smaller than that of HPA/SBA-15. So the growth of the particles is more remarkably limited, leading to the migration to the outer surfaces of the support.
Textural properties of the supported catalysts
Supplemental Figure S4 shows the N2 adsorption–desorption isotherms and the pore size distribution curves of the supports and the catalysts. The experimental parameters of specific surface areas, porous volumes, and pore sizes are listed in Table 1. 3DOM SiO2 exhibits the Type-II isotherms, while SBA-15 and MCM-41 present the Type-IV isotherms. The specific surface areas of them were 189, 563, and 874 m2 g−1, respectively, which decrease significantly after the HPA has been supported. There are two reasons for that: one is that the supported HPA has occupied a certain proportion in mass, whereas with very low surface areas; and the other is that the pores in the supports might be blocked by the supported HPA. After calcination at 350°C, the increases in specific surface area are found for all the samples, which might be due to the loss of crystallization and constitutional water, and the newly formed pores of the aggregated particles. The clear inflection points at very low relative pressure (P/P0 < 0.05) in the isotherms of 3DOM SiO2 indicate the existence of micropores. Meanwhile, Supplemental Figure S4(b) shows that there are mesopores in the walls of 3DOM SiO2 framework with a wide distribution. After the HPA has been supported, the inflection points in the isotherms almost disappear, indicating the great decrease of the micropores, which might be blocked by the supported HPA, leading to the decrease of the specific surface areas and the increase of the average pore sizes. For SBA-15 and MCM-41, a fairly uniform pore size distribution can be seen in Supplemental Figure S4(d) and (f), representing the 1D mesoporous channels with a diameter of 8.7 and 3.6 nm, respectively. The obvious decrease in pore size after the support of HPA indicates that the HPA has entered the channels of SBA-15 and MCM-41.
Textural properties of the supports and the supported catalysts before/after calcination.

Catalytic performances of the supported catalysts
The performances of the supported catalysts calcined at 350°C for the vapor-phase oxidation of MAL are shown in Figure 5. It can be found that there are significant disparities in catalytic performances among the catalysts with different supports. On HPA/3DOM SiO2, the conversion of MAL and the selectivity to MAA at 300°C were, respectively, 45% and 89%. With the increase of the reaction temperature, the MAL conversion is greatly increased to 67% at 315°C and to 81% at 330°C, meanwhile the MAA selectivity is decreased slightly to 87% at 315°C and then to 67% at 330°C. With the reaction temperature increased from 300°C to 330°C, the MAL conversion increase from 58% to 66%, and to 78% and also from 59% to 61%, and to 65% on HPA/SBA-15 and HPA/MCM-41, respectively. There are no distinct differences in MAL conversion among the three catalysts. However, the MAA selectivities are only 21% and 8% on HPA/SBA-15 and HPA/MCM-41 at 300°C, much lower than that on HPA/3DOM SiO2. Besides MAA, the main by-products, acetic acid and COx (CO and CO2), were also detected in the reaction to determine the carbon balance. When the reaction temperature is 315°C, the selectivities to acetic acid and COx on HPA/3DOM SiO2 are 3.5% and 6.7%, respectively. It is found that the increase in the reaction temperature obviously increases the selectivity to COx and with little effect on the formation of acetic acid. Similarly, the low selectivities to MAA on HPA/SBA-15 and HPA/MCM-41 are corresponding to the high selectivities to COx, and the selectivities to acetic acid were all less than 12%. It has been shown that the oxidation of MAL to MAA proceeds in two steps.22,32 The first step reaction is catalyzed by Brønsted acid sites to form the intermediates having C–O–Mo bonds, and then they are oxidized by the lattice oxygen to form MAA via Mars and van Krevelen mechanism in the second step. Strong acid sites are indispensable in the first step. 22 The acidity of MoO3 is weaker than that of the HPA, and it cannot promote the first step of the reaction. MAL can be oxidized directly to acetic acid and COx besides the selective oxidation, and the formed MAA can also be successively oxidized to acetic acid and COx. So the decomposition of the HPA to MoO3 results in the decrease of the selectivity to MAA.

Catalytic performances of the supported catalysts for the oxidation of MAL. The values above the bar are reaction temperatures.
In order to explore the changes in acidity of the HPA after it had been supported, NH3-TPD measurement was carried out on the catalysts and the results are shown in Figure 6. In the profile of bulk H4PMo11VO40, two broad desorption peaks are observed in the temperature range of 50°C–300°C and 300°C–550°C with the Tmax value being 139°C and 463°C, representing the weak acid sites and the strong acid sites, respectively, as has been reported previously.30,33 The profile of HPA/3DOM SiO2 is similar to that of bulk H4PMo11VO40 except that the Tmax value of the desorption peak related to the strong acid sites shifts to a lower temperature (433°C), indicating the well-retained Keggin structure on the 3DOM SiO2. And the acid strength decreases due to the strong interaction between the HPA and the support. 34 The desorption peak related to the strong acid sites becomes much smaller in the profile of HPA/SBA-15 and almost disappears in the profile of HPA/MCM-41. And meanwhile, the desorption signals in the temperature range of 200°C–400°C increase, especially in the profile of HPA/MCM-41 with a newly emerged peak at 293°C, suggesting the occurrence of other acid sites probably related to the formation of MoO3.

NH3-TPD profiles of H4PMo11VO40 and the supported catalysts calcined at 350°C: (a) H4PMo11VO40, (b) HPA/3DOM SiO2, (c) HPA/SBA-15, and (d) HPA/MCM-41.
A long-term performance test was carried out to evaluate the durability of the HPA/3DOM SiO2 catalyst, and the result is shown in Supplemental Figure S5. In the XRD pattern of the used catalyst (Supplemental Figure S6), besides the main phase of HPA, there emerged reflections assigned to MoO3, which are probably responsible for the slight decrease in the catalytic performance. For comparison, catalytic performances of various supported catalysts employed for the oxidation of MAL to MAA are presented in Table 2. It can be seen that HPA/3DOM SiO2 exhibits much higher catalytic activity than the referenced catalysts, indicating the 3DOM SiO2 of a competitive and promising candidate as support for HPA catalyst with broad applications.
Comparison in catalytic performance of different supported catalysts.

MAL: methacrolein.
The value was the mass of the HPA in the catalysts.
mL min−1.
Conclusion
In consideration of the relatively high reaction temperature of MAL oxidation, the thermal stability of the HPAs catalysts is particularly concerned. When HPA is supported in the 1D mesoporous channels of SBA-15 and MCM-41 with high dispersion, it migrates toward outside under calcination, and it starts to decompose at a lower temperature. When supported on HPA/SBA-15 or HPA/MCM-41 and calcined at 350°C, the HPA is almost completely transformed to monoclinic MoO3, which is further changed to more stable orthorhombic MoO3 at a higher calcination temperature. For HPA/3DOM SiO2, the intact Keggin structure can be retained after calcination at 350°C. Because the crystallization and growth of HPA are not limited in the open macropores of 3DOM SiO2, dispersed particles with an average size of about 5 nm are formed on the surfaces of the macropores with a higher thermal stability, which guarantees the existence of the necessary strong acid sites for MAL oxidation. The MAL conversion and MAA selectivity are, respectively, 67% and 87% on HPA/3DOM SiO2 at a reaction temperature of 315°C, respectively.
Experimental
Preparation of supported HPA
SBA-15 and MCM-41 were synthesized according to the typical methods as described in literatures.39,40 3DOM SiO2 was synthesized using polystyrene (PS) colloidal template with tetraethyl orthosilicate (TEOS) as the precursor. PS colloidal template was prepared by centrifugation of the slurry-containing monodispersed PS microspheres. After being dried completely, the template was impregnated in a mixed solution with the mole ratio of ethanol: TEOS:HCl:H2O = 1:1:0.25:0.2 for 30 min, followed by drying for 2 h. The impregnation and drying were repeated for three times. The dried sample was calcined in an air flow with a temperature-rising rate of 2°C min−1. The calcination temperature was kept for 3 h at 300°C and 6 h at 500°C, respectively, then 3DOM SiO2 was obtained.
The supported catalysts were prepared by the following procedure. First, 1.0 g of support was added into 20 mL copper sulfate solution (1 mol L−1). The slurry was kept at 95°C under constant magnetic stirring for 1 h. Then it was separated by centrifugation, and the solid was washed with 300 mL deionized water. After being dried at 75°C, the solid was calcined at 500°C for 2 h to be used as the support. A small amount (0.38 g) of H4PMo11VO40 (with seven molecules of crystallization water as determined by TG measurement) was dissolved into deionized water, and then 0.5 g of support was added to the solution. Then the mixture was vibrated at 50°C for 12 h and dried at 75°C for 24 h. Finally, the sample was calcined at a desired temperature for 6 h in a tube furnace. The obtained catalysts were designated as HPA/3DOM SiO2, HPA/SBA-15, and HPA/MCM-41. The loading amount of H4PMo11VO40 in the catalysts was 41.5 wt%.
Characterization
XRD patterns were recorded on a MiniFlex600 Powder X-ray powder diffractometer (Rigaku, Japan). Elemental mapping images were obtained by EDS using a Merlin Compact field emission scanning electron microscope (ZEISS, Germany). TEM measurements were carried out on a JEM-2100PLUS high-resolution transmission electron microscope (JEOL, Japan). TG analysis was carried out on an STA 409C TG analyzer (Netzsch, Germany) in air with a heating rate of 10.0°C min−1. Nitrogen adsorption–desorption isotherms were measured using an SSA-4200 surface area and porosity analyzer (Builder Electronic Technology, China), and the specific surface areas were calculated using the Brunauer–Emmett–Teller (BET) equation. Pore size distributions were evaluated by the Barrett–Joyner–Halenda (BJH) method using the adsorption branch of the isotherms. FTIR spectra were recorded on a NEXUS-470 spectrophotometer (Nicolet, USA). NH3 temperature-programmed desorption (NH3-TPD) measurements were carried out on an Autochem II 2920 apparatus (Micromeritics, USA). In a typical NH3-TPD experiment, 100 mg of catalyst was pretreated in a helium flow at 200°C for 1 h. Then the sample was treated with a 10% NH3–He flow for 1 h at room temperature. After that, the sample was purging in a helium flow for 1 h until the baseline was stable. The desorption was carried out in flowing helium at a heating rate of 10°C min−1 to 750 °C.
Evaluation of catalytic activity
The oxidation of MAL was carried out in a fixed-bed reactor. In a typical experiment, MAL was delivered with a flow rate of 2.4 × 10−3 mol h−1 by an injection pump to a mixer kept at 150°C. Air, nitrogen, and steam were also continuously fed into the mixer. After mixing, the gaseous reactants with a mole ratio of MAL:O2: N2: H2O = 1:2.5:15:8 were fed into the fixed-bed reactor loaded with 0.5 g catalyst, and the reaction temperature was maintained at 315°C. After the catalytic reaction was carried out for 6 h, the products were gathered and analyzed by an SP-6890 gas chromatograph (Rainbow Chemical Instrument, China) equipped with a flame ionization detector (FID) detector using an FFAP capillary column. The gaseous by-products were detected by the GC system with TDX-01 packed column and a thermal conductivity detector (TCD).
Supplemental Material
Supplementary_file-R1_1 – Supplemental material for Comparison in thermal stability and catalytic performance of H4PMo11VO40 heteropolyacid supported on mesoporous and macroporous silica materials
Supplemental material, Supplementary_file-R1_1 for Comparison in thermal stability and catalytic performance of H4PMo11VO40 heteropolyacid supported on mesoporous and macroporous silica materials by Heng Zhang, Chunhao Yang, Shengying Zhao, Tingting Wang and Wancheng Zhu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the National Natural Science Foundation of China (grant no. 21206179) and the Natural Science Foundation of Shandong Province (grant no. ZR2018MB033).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
