Abstract
Introduction
Asthma is the most common non-communicable respiratory disease, affecting an estimated 339 million people worldwide.1,2 It is a heterogeneous disease that varies in severity, phenotype, and natural history. 3 Despite the availability of highly effective treatments, uncontrolled asthma affects ∼30% of patients. Poor asthma control is associated with decreased health-related quality of life along with increased morbidity, mortality, healthcare resource utilisation, and associated costs.4–6
For many patients, asthma remains poorly controlled because of poor inhaler technique, suboptimal adherence to inhaled therapies, or both.7,8 Failure to recognise and correct these common causes can lead to unnecessary treatment escalation, which increases the likelihood of treatment-related adverse effects, inappropriate use of high-cost therapies, and misclassification of asthma severity. 9 Studies have demonstrated that once adherence and inhaler technique are optimised, the true prevalence of severe asthma falls below 5%.7,10
In the 2014 National Review of Asthma Deaths, preventable factors, including the underuse of inhaled corticosteroids and overuse of short-acting beta agonists, lack of timely specialist referrals, and failure to follow important steps of the asthma review, were identified as key contributors to asthma mortality in the United Kingdom. 11 Despite this devastating report of preventable deaths, a more recent analysis from the United Kingdom revealed that many patients who have features consistent with severe asthma continue to be managed in primary care, even though secondary care assessment and treatment is advised. This scenario is likely due to multiple reasons, including poor understanding about what constitutes severe asthma, a failure to understand the harm associated with repeated courses of oral corticosteroids, as well as misperceptions about the benefits of referral to a specialist, which includes access to newer, more advanced treatments. 12 The phase 2, open-label, single-arm REcognise Asthma and LInk to Symptoms and Experience (REALISE) study surveyed asthma control across 11 European countries to assess asthma improvement via symptoms, exacerbations, and Global Initiative for Asthma (GINA)–defined control in real-life populations of patients with asthma and to provide insights on patient attitudes toward asthma. 13 Results from the study indicate that many patients consider their asthma to be controlled, despite ongoing symptoms and exacerbations, with high short-acting beta agonist use as a marker.13,14 Misperceptions about asthma control and specialist referral, in combination with failures to use up-to-date national and international treatment guidelines, infrequent provision of asthma reviews, and inadequate assessments of modifiable factors, all contribute to global and regional variations in asthma management; these realities highlight the significant unmet need to improve the quality of care and help standardise disease management for patients with asthma. 15 More robust, comprehensive strategies that allow for timely diagnosis and care are warranted.
Recent digital advances in healthcare, including smartphone apps and electronic monitors, offer clinicians new options to improve asthma care in both the clinic and the home setting. 15 For self-management, several programs have shown to be effective in improvement of asthma control. Among Australian First Nations people, the Asthma APP multilingual mobile application has been shown to improve health knowledge and understanding of asthma among those caring for children with asthma. 16 Another mobile application geared toward adolescents with asthma was found to improve their asthma self-management skills. 17 A study of a computerized decision support tool for asthma management (the Asthma Control Evaluation and Treatment [ACET] Program) found the tool facilitated standardized asthma assessment and treatment in multicentre randomized controlled trials. 18 Moreover, a study recruiting patients with asthma with limited health literacy found that another self-management mobile application had favourable adoption, adherence, and retention of use. 19 A meta-analysis of electronic health technologies in paediatric asthma care showed potential superiority of electronic monitoring of medication use, lung function, and digital environment compared to hospital-based monitoring. 20 Another self-management educational interventional tool, the AsmaDroid mobile application, showed efficacy in improving asthma education among young patients. 21 While all of these recent developments have shown promise in their ability to offer education and self-guided asthma care among particular patient populations, with the assistance of healthcare professionals, there remains a need for more robust, comprehensive digital tool that can eventually be adopted into everyday clinical practice focusing on the healthcare professional. ReferID+ is a novel digital tool currently at the prototype stage that was designed to ensure optimised asthma management at every step of the treatment paradigm. 22 Its aim is to guide structured consultations to cover all aspects of the asthma review, including timely referrals to specialist clinics in cases of potentially severe asthma, through guiding healthcare professionals from all levels of training. 22 ReferID+ was developed for use in the United Kingdom, but the content is universally appropriate for asthma care in any healthcare system. To increase accessibility, it is a website that can be accessed via an office computer or portable tablet. ReferID+ incorporates all the key factors that contribute to poor asthma control and exacerbation risk while simultaneously identifying patients with suspected severe asthma who may benefit from referral to a specialist. ReferID+ was adapted into the AsthmaOptimiser tool, expressly for use in the Netherlands, where nurses perform the majority of care in clinical practice. The AsthmaOptimiser consists of a similar panel of questions and topic areas for the asthma review and is similarly aimed to facilitate structured consultations between primary care nurses and patients. The ReferID+ and AsthmaOptimiser tools differ in regional adaptations (e.g., language, questionnaires used, and electronic medical record system integration); however, both tools support healthcare professionals in more effectively managing their patients’ asthma, with the aim of improving long-term outcomes. From a global perspective, the use of the ReferID+ and AsthmaOptimiser tools have the potential to improve healthcare professional knowledge and familiarity with each aspect of the asthma review, resulting in high-quality care for patients with asthma.
Here we describe the ReferID+ and AsthmaOptimiser tools, including key challenges in development, qualitative research insights on the key components for each tool, opportunities to enhance the utility of the tools, and two clinical trials that are currently underway to assess the effectiveness and implementation strategies for ReferID+ and AsthmaOptimiser. This assessment will help pave the way for validation of these tools for adoption into common clinical practice.
Methods
ReferID+ and AsthmaOptimiser development
ReferID+ was developed primarily in London, for use in the UK National Health Service primary care environment to identify uncontrolled and suboptimally treated patients, promote patient engagement through collaborative structured asthma consultations, and upskill healthcare professionals on best practice guidelines for asthma care. Further development of ReferID+ was accomplished through extensive collaboration with a diverse group of international asthma experts, which was facilitated through an industry-sponsored programme. The programme is a non-promotional global initiative that seeks to improve clinical care and long-term outcomes for patients with severe asthma. 23 As primary care asthma reviews in the United Kingdom are frequently undertaken by nonphysicians, additional feedback on content and ease of workflow was sought from a group of primary care nurses and pharmacists.
The ReferID+ prototype was developed in January 2019, and concepts were developed for international scaling between July and September 2019; implementation began in 2020. The workflow for ReferID+ was constructed using assessment guidelines as described in the Global Initiative for Asthma report 1 and was then tested and refined by asthma experts and clinicians who designed the tool.
Development of the ReferID+ tool focused on implementation in the United Kingdom. With the potential to be used in other healthcare settings, the Netherlands was chosen as an additional country to develop and adapt the ReferID+ as template for future scale-up. One of the first comments received in discussions with Dutch healthcare professionals was on the name. ReferID+ was seen as a tool to increase referrals, but the tool could be used to optimise treatment, preventing unnecessary referrals. Therefore, the name was changed to AsthmaOptimiser. In the Netherlands, because general practice nurses perform most of the routine asthma visits in primary care, it was necessary to adapt the AsthmaOptimiser tool for use by a wide range of healthcare professionals. In this healthcare setting, nurses often encounter difficulties in translating guidelines to a practical and systematic approach, which can lead to suboptimal disease control and reduced health-related quality of life.
24
Qualitative research during the development of the AsthmaOptimiser included interviews with nine healthcare professionals across the Netherlands and France (two nurses, two experts, and five general practitioners) with structured questions on needs, contents, and feedback on mock-ups; evaluations of multiple iterations of the tool; and assessments of key factors required for electronic medical record integration. Developers of the tool went to considerable lengths to ensure seamless integration between AsthmaOptimiser and eight different electronic medical record systems in the Netherlands (Figure 1). Several key insights related to practice and workflow were described in the healthcare professional interviews, including the need to highlight “how patients feel” about living with asthma and how their symptoms affect their day-to-day life. Healthcare professionals believed the asthma consultation should be structured to promote a conversation between the healthcare professional and the patient. Furthermore, because most practices in the Netherlands rely on nurses to perform lung function tests and assess inhaler technique, healthcare professionals suggested the AsthmaOptimiser should also support nurses who manage patients with other chronic diseases, and occasionally, patients with chronic asthma. Integrating ReferID+ or AsthmaOptimiser into electronic medical records. API: application programme interface; APP: application; DMDP: digital medical device platform; EDIFACT: electronic data interchange for administration, commerce, and transport; EMR: electronic medical record; GP: general practitioner; HCP: healthcare professional; mgmt: management; NL: Netherlands; PDF: portable data format; PHI: protected health information; UI: user interface.
Results
ReferID+ and AsthmaOptimiser content
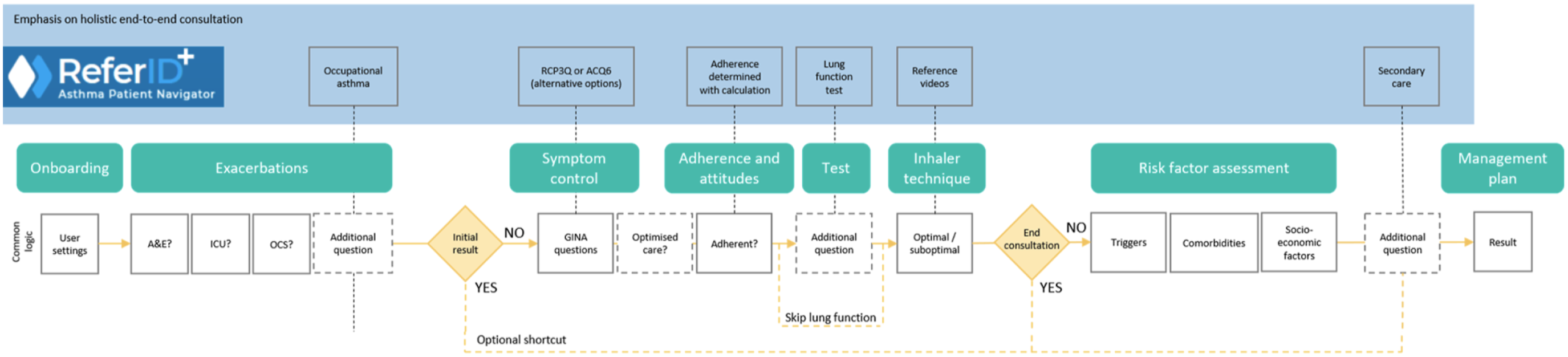
ReferID+ and AsthmaOptimiser workflow. A&E: accident and emergency; ACQ6: asthma control questionnaire; GINA: Global Initiative for Asthma; ICU: intensive care unit; OCS: oral corticosteroid; RCP3Q: Royal College of Physicians 3 questions. The solid contours indicate steps that will lead to specific suggestions based on the guidelines in the final report. The dashed lines indicate a step to collect more information, which is then included in the final report; however, the treatment algorithm is not dependent on the step.

“Red flag” questions to identify patients who may benefit from a specialist review. GINA: Global Initiative for Asthma; ICU: intensive care unit; OCS: oral corticosteroid; SABA: short-acting beta agonist.
After the questions regarding exacerbation history, the next steps in the workflow assess symptom control, inhaled corticosteroids adherence, and inhaler technique, as well as an option to input lung function test results. Healthcare professionals can choose to end the ReferID+ question workflow after step 5 or proceed with the risk factor assessment in step 6 to assess triggers, comorbidities, and socioeconomic factors that may contribute to symptoms or exacerbations. On the basis of healthcare professional responses to the assessment questions, ReferID+ generates a summary of the consultation with individualized recommendations for guideline-based strategies to optimise asthma management (e.g., improvement of inhalation technique, medication adherence, or change in treatment step, including step-down). In the context of suspected severe asthma or unexplained poorly controlled symptoms, ReferID+ can support healthcare professionals in referring the appropriate patients to a specialist (Figure 4(a) and (b)). (a) Actionable output generated by ReferID+ or AsthmaOptimiser; and (b) example referral letter for ReferID+ and AsthmaOptimiser.
In the Netherlands, AsthmaOptimiser promotes collaboration between primary and secondary care providers with an easy-to-use and transparent documentation system for the asthma review. According to qualitative research conducted during the development of the tool, the AsthmaOptimiser is most likely to be used by group practices that have familiarity with digital tools and specification around their practice setup (e.g., tech-savvy practices pushing for a better patient experience and allowing for more direct general practitioner–patient interactions [vs practices primarily using digital tools for electronic medical records] and relying heavily on nurses for assessments). Designed to support primary care nurses in conducting a proper asthma assessment, the AsthmaOptimiser facilitates structured conversations between nurses and patients and supports a standard assessment/work-up of asthma control, with a workflow and questions similar to those included in ReferID+.
The AsthmaOptimiser tool follows guidelines from Global Initiative for Asthma as well as from the Dutch College of General Practitioners.1,30 The 8 Dutch primary care electronic medical records use the coding table developed by the College of General Practitioners to store test results and laboratory values, and the AsthmaOptimiser results were mapped to these codes. 30 For full integration, an XML message is sent to one of the national communications providers in the Netherlands, translated to the EDIFACT (Electronic Data Interchange For Administration, Commerce and Transport) standard, and sent to the general practice systems. The AsthmaOptimiser results can then be imported into the electronic medical record system, in the same manner as for laboratory results, and can be used for reimbursement for structured care. The Asthma Control Questionnaire–6 test is routinely used to assess asthma control in the Netherlands.
For successful widespread adoption of these tools into routine clinical practice, seamless integration with existing clinical workflows and electronic medical record systems is necessary. This technical challenge and any information transfer between electronic medical record systems is currently the focus of ongoing work in the United Kingdom and Dutch healthcare systems. Direct data transfer from the tool to an electronic medical record system is being piloted in the Netherlands through the AsthmaOptimiser to simplify use and encourage healthcare professionals to adopt the tool. Additional training may be necessary for both tools to address technical challenges; for example, options within the tool allow nurses to demonstrate inhaler techniques via standardised instructional videos linked to the tools. Both tools could also be enhanced with an ability to add additional assessment components, such as additional lung function measurements or remote assessments, which would enable further considerations for remote patient care. Indeed, during the COVID-19 pandemic, the use of remote assessments has become critical to minimise potential exposure for patients with pre-existing respiratory conditions, such as asthma. Following the studies, the tool will be made available to the healthcare systems.
ReferID+ and AsthmaOptimiser validation and implementation
Effectiveness on patient outcomes when the ReferID+ is in use are currently being evaluated in the Optimisation of Asthma in Those With Uncontrolled Symptoms (OASIS) study in London, United Kingdom (NCT04941001). This prospective randomised study will compare outcomes of patients in primary care who receive care with ReferID+ via telephone or video consultations with outcomes of patients who continue to receive the standard of care without ReferID+. The 18-month pilot study aims to include 200 adult patients with asthma from primary care who have had one or more courses of oral corticosteroids in the previous 12 months. Participants will be identified using a patient filter to select those with at least one course of prednisolone for asthma in the previous year. The primary objective is to evaluate the clinical effectiveness of ReferID+ in primary care by comparing asthma exacerbation rates after 12 months between patients who received usual asthma care and patients who received an asthma review with ReferID+. Secondary objectives include change in Asthma Quality of Life Questionnaire score, Asthma Control Questionnaire–6 score, and salbutamol usage at 12 months, as well as an assessment of outcomes of patients referred to secondary care. Additional objectives are to determine the usability of ReferID+ for facilitating an asthma review via telephone or video consultation, including the healthcare professionals’ perspectives and their time to complete the review.
The AsthmaOptimiser is currently being evaluated in the CAPTURE study (NCT04456270), which will evaluate the feasibility and utility of the tool in primary care in the Netherlands. CAPTURE is a prospective, observational study that will enrol 30 primary care healthcare professionals. These healthcare professionals will use the AsthmaOptimiser in their daily clinical practice with 5 to 10 adult patients with asthma who have appointments with the practice nurse for regular monitoring visits. The primary objective is to evaluate the proportion of patients with inadequate asthma control measured with the Asthma Control Questionnaire–6 questionnaire. Secondary objectives are to examine the association between asthma control and patient demographics, clinical characteristics, work and productivity loss, and the proportions of patients whose asthma management can be optimised in primary care versus patients who need a referral to a specialist. Healthcare resource utilisation by patients who are referred to a specialist will also be evaluated in the 6 months after the study visit. In addition to the quantitative outcome measures, qualitative research will be performed by interviewing healthcare professionals about their expectations, facilitators, barriers, and experiences with using the tool. Healthcare professionals will be interviewed before they start using the AsthmaOptimiser and after they have used the tool in their daily clinical practice with 5 to 10 patients with asthma. Any feedback will be used to improve the tool before making it available outside the study setting.
ReferID+ was recently adapted into a simple, concise, four-question version known as the ReferID tool, which has been launched or implemented in 30 countries and 21 languages to date. 22 This simple and concise tool was also launched as the AsthmaID tool in Singapore in 2020. 31 ReferID can be used to quickly identify patients who may benefit from a referral to a specialist in countries with fewer resources for asthma reviews in primary care.
Discussion
The streamlined digital interface of the ReferID+ and AsthmaOptimiser tools and simple question format allow for ease of use and step-by-step guidance and could eventually be adapted for use in other chronic conditions or the combination of conditions in patients with comorbidities. Currently a version is in development for chronic obstructive pulmonary disease and we are seeking funding for the development for cardio-vascular and metabolic diseases. A limitation of this study is the implementation in select healthcare systems, which underscores the need to test the ReferID+ and AsthmaOptimiser tools in broader healthcare systems across different organizations. Another limitation is the use of Global Initiative for Asthma and Dutch College of General Practitioner guidelines, rather than local or national guidelines, for the AsthmaOptimiser tool; however, given that Global Initiative for Asthma recommendations are updated annually based on the most recent literature, the AsthmaOptimiser tool has the added value of being based on the latest research.
The long-term goal for these authors is to integrate multiple tools of this type, as part of the disease management process in patients with comorbid diseases. Eventual updates such as electronically measured compliance and medication adherence data would improve the practical application of these tools. Other areas for improvement include better integration with existing healthcare systems and enhanced healthcare professional training modules. Patient privacy data is a key consideration for adoption of new systems in healthcare. The current versions are developed with the privacy by design mindset, not collecting identifiable information and not storing identifiable results. The OASIS study will show effectiveness in a study setting, and the CAPTURE study will evaluate implementation, but long-term studies are needed that focus on real-world effectiveness, including cost-effectiveness analyses, as well as sustainability of these tools over time. The ultimate goals are to evaluate and adapt these tools in other countries by refining them for each specific healthcare environment and ensuring seamless integration into local electronic medical records.
Conclusions
The ReferID+ and AsthmaOptimiser tools aim to support high-quality, structured asthma consultations in a primary care setting by providing individualised recommendations for guideline-based strategies to optimise asthma management. These recommendations support healthcare professional decision-making regarding treatment optimisation and when to refer the patient to a specialist, which will help to minimise risks associated with inadequate disease control, as well as inappropriate and excessive corticosteroid use.
Footnotes
Acknowledgments
Medical writing support was provided by Jamie King, PhD, and Dan Jackson, PhD, C.M.P.P., from Citrus Scientific, a Citrus Health Group, Inc. (Chicago, IL), in accordance with Good Publication Practice (GPP3) guidelines, and funded by AstraZeneca (Gaithersburg, Maryland, USA). The authors would like to thank Hans Wouters (General Practitioners Research Institute, Groningen, The Netherlands) for his contributions in the development of the CAPTURE study protocol.
Author contributions
All authors contributed to the design of the study the analysis of the results and the development of the manuscript. All authors read and approved of the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AstraZeneca (Gaithersburg, Maryland) funded the development of the ReferID+ and AsthmaOptimiser tools.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David J. Jackson has received advisory board and speaker fees from AstraZeneca, Sanofi, Novartis, Chiesi, Boehringer Ingelheim, and GlaxoSmithKline. Julie Hales is an employee of AstraZeneca. Mona Al-Ahmad has received advisory board and speaker fees from AstraZeneca, Sanofi, Novartis, and GSK. Janwillem Kocks reports grants, personal fees, and non-financial support from AstraZeneca; grants, personal fees, and non-financial support from Boehringer Ingelheim; grants and personal fees from Chiesi; grants, personal fees, and non-financial support from GSK; non-financial support from Mundi Pharma; grants and personal fees from Teva; personal fees from MSD; personal fees from COVIS Pharma; and grants from Valneva outside the submitted work Janwillem Kocks holds <5% shares of Lothar Medtec GmbH and 72.5% of shares in the General Practitioners Research Institute. Hetal Dhruve, Bertine Flokstra-de Blok and Birgit Wijnsma report no conflicts of interest.

