Abstract
Keywords
Introduction
Immunization programs are a cornerstone of public health systems around the world,1,2 helping to prevent the spread and impact of communicable diseases such as polio, measles, mumps, diphtheria and pertussis.2–5 Accurate, real-time immunization data is integral to the success of immunization programs in both routine and outbreak scenarios.6–8 In order for this data to be used most effectively, data should be standardized, thereby allowing it to be aggregated, analyzed, and leveraged to determine whether vaccination coverage targets are being met, to conduct robust public health surveillance, and to inform strategic public health planning and prioritization. 8 Standardization is also a critical part of ensuring that immunization data can be shared within and between jurisdictions. The importance of standardization is evident in the context of Canadian immunization programs, where data comes from several sources in each province and territory.9,10
The need to improve Canada’s capacity to collect, share and use health data became particularly apparent during the COVID-19 pandemic, when existing gaps in these areas challenged public health systems’ ability to respond effectively to this global health emergency. 9 In 2022, the final report from the Pan-Canadian Health Data Expert Advisory Group identified the implementation of interoperable data standards as a key recommendation in Canada’s health data strategy. 11 However, the importance of immunization data interoperability pre-dates this recommendation by many years. 12
The Canadian Vaccine Catalogue (CVC), was created to support interoperable immunization systems in Canada.
10
The CVC was funded and maintained from 2016 to 2024 and represents the culmination of many years of work from individuals and organizations across the country. This paper summarizes the CVC’s efforts to support immunization interoperability in Canada, with an overarching goal of providing other jurisdictions with valuable lessons from the Canadian context. Our objectives are to: 1. Provide the history of the CVC’s development, in the context of the history of immunization standards and terminology development in Canada. 2. Describe the activities of the CVC and how it supported interoperability in Canadian immunization systems. 3. Provide lessons learned and recommendations for other jurisdictions on how to enhance standards and terminology development and adoption.
History of the CVC
Immunization standards and terminology are longstanding issues in the Canadian public health community. Indeed, the need for a robust Canadian Immunization Information System (IIS) was identified as early as the 1996 Canadian National Immunization Conference. 12 The 1998 Canadian Consensus Conference on a National Immunization Records System further highlighted the need for interoperability between provincial and territorial immunization registries across Canada. 13 The National Working Group for an Immunization Records Network (NWGIRN) was formed to implement these recommendations, and successfully published functional standards and minimum (core) data sets for a national immunization registry network and vaccine-associated adverse events. 14 Thereafter, Canada Health Infoway (CHI), a federally funded non-profit organization that supports the implementation of digital health initiatives, spearheaded the development of immunization terminology content to facilitate the collection of standardized data. As a result of these efforts, the National Immunization Data Dictionary (NIDD) was developed to provide a national standard for defining, collecting and sharing immunization data required for use in Canadian public health systems, electronic medical records (EMRs), pharmacy systems, hospital systems, and patient (consumer) applications.
The impetus for the CVC arose from two main needs among immunization stakeholders. First, it became clear that the Vaccine Identification Database System (VIDS), the database originally developed by the Public Health Agency of Canada (PHAC) that housed a portion of standardized immunization content, would not support the capture of all minimum national immunization data elements (NIDE) for immunization registries that were required for fulsome clinical documentation. Eventually, VIDS was decommissioned.
Second, CHI had developed Systemized Nomenclature of Medicine – Clinical Terms (SNOMED CT) immunization content and clinical subsets to aggregate immunization information, which were aligned with the NIDE. However, with the decommissioning of VIDS, there was no longer an accessible resource that would allow pan-Canadian immunization stakeholders to easily implement standardized immunization content.
To address the need for interoperable vaccine registries in Canada, the CVC was created in 2016 to provide a solution for hosting all vaccine clinical terminology, regulatory information, variable product data, as well as concept mappings to support forecasting in clinical systems into a single, downloadable dataset. Furthermore, it was designed to align with functional immunization registry standards outlined by PHAC and included additional subsets, effectively becoming a one-stop-shop for documenting immunization events in health information systems. The CVC served as an accessible resource for immunization stakeholders to reference in system implementation, and aimed to support stakeholders who were originally intending to leverage VIDS to access vaccine packaging information and lot data.
Importantly, when the CVC was initially conceptualized, it was primarily developed in partnership with system implementers of jurisdictional immunization registries. Eventually, some jurisdictions encouraged EMRs to use standardized terminology to capture vaccine data and facilitate interoperability between systems, as bidirectional data flow between immunization registries and source systems are crucial to achieve a complete picture of an individual’s immunization history.
About the CVC
Overview of Data Inputs to the CVC.

CVC Data Formats
The CVC was accessible as a web page, an Excel workbook, and a Health Level 7 (HL7) Fast Healthcare Interoperability Resources (FHIR) application programming interface (API). The FHIR API was developed to support system-to-system integrations between electronic health record software and the CVC. Figure 1 summarizes data inputs, formats, and use cases of the CVC. Overview of data inputs, data formats, and use cases of the CVC.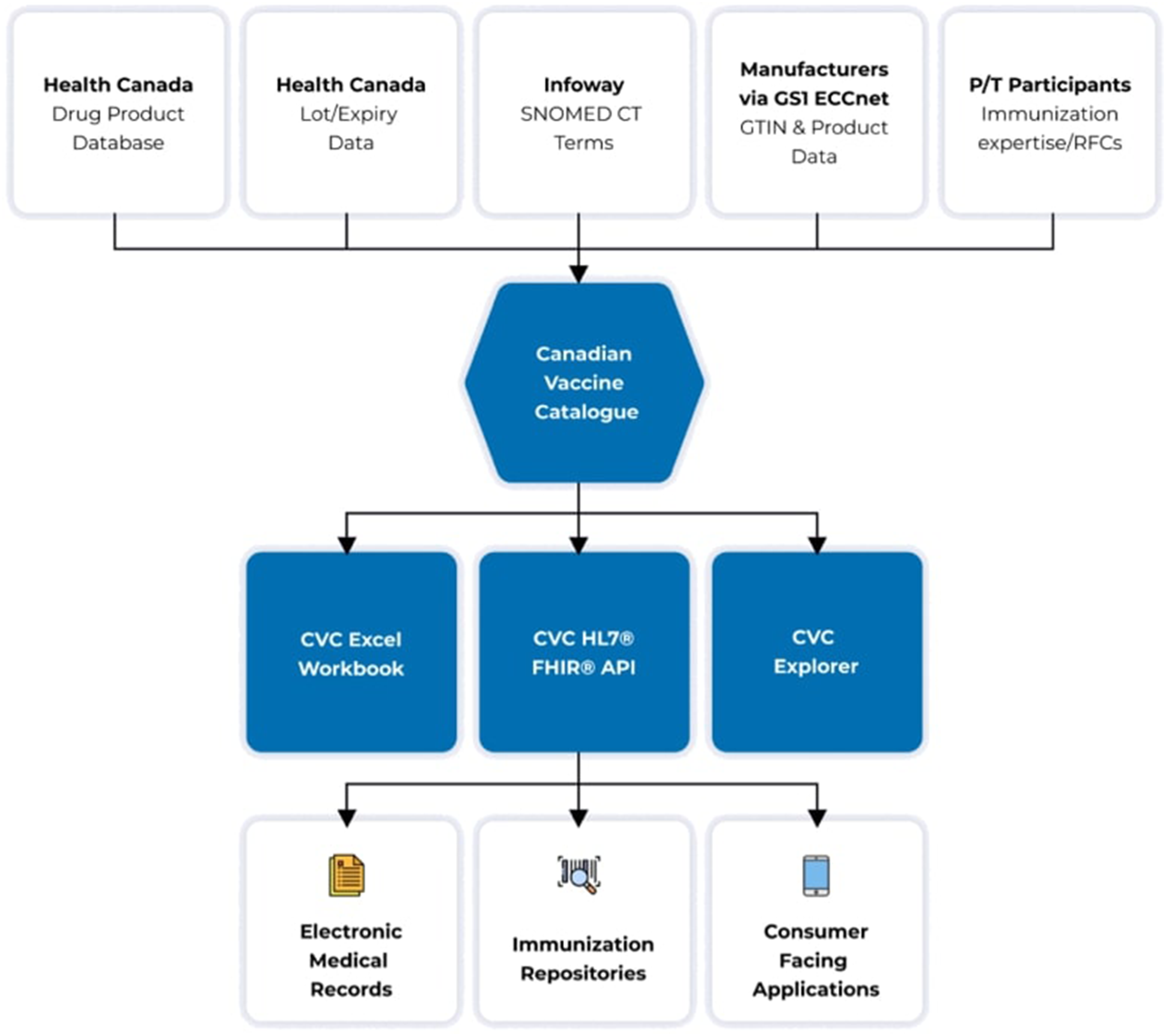
CVC use cases
The CVC aimed to enhance immunization interoperability within and between jurisdictions by: 1. Providing standardized picklists in EMR software The CVC could be used when configuring EMR software to ensure vaccine picklists were up-to-date, associated with standardized clinical identifiers (i.e. SNOMED CT), and used standardized display terminology to improve the accuracy of clinical documentation. The CVC permitted an immunization record to be added to an electronic health record by immunizing agent or tradename, lot number, or Global Trade Item Number (GTIN) (through barcode scanning). One example of the CVC being leveraged for this use case was its use by OntarioMD, a government-funded body that exists to support clinicians with the use of digital health technology. To facilitate the transmission of standardized immunization documentation to Ontario’s provincial immunization repository, OntarioMD encouraged EMR users to implement the CVC API and/or use the CVC to document immunizations. 2. Configuring immunization information systems The CVC was designed to be a key resource in the configuration of jurisdictional immunization information systems that would support a consistent approach to aggregating immunization data from various sources into their immunization repository, as well as supply chain management tracking. Furthermore, SNOMED CT concept mappings for the agent-antigen-disease relationship would support vaccine forecasting, helping immunization program administrators maintain optimal vaccine coverage and facilitate program planning. An illustration of this use case in Canada’s Panorama public health surveillance system is presented in Box 1.
Box 1. Implementation of Panorama Implementation of Panorama The global Severe Acute Respiratory Syndrome (SARS) outbreak in 2002 highlighted major gaps in Canada’s ability to respond to pandemic threats. This prompted a number of national committee recommendations to strengthen public health infrastructure, which led to the development of a public health surveillance system known as Panorama. This system was expected to achieve a number of benefits that would directly address the system failures that were identified after the SARS outbreak, including the ability to collect, share, and analyze communicable disease information in real-time. Standardized immunization terminology content (i.e., SNOMED CT) was developed to help support Panorama in achieving its intended outcomes and to enable the interoperability of immunization information. Throughout its rollout and in the years since then, system implementers of Panorama have leveraged the CVC as a reference resource to configure immunization information according to SNOMED CT standards. 3. Documenting immunizations in digital health applications The CVC was designed to be compatible with consumer-facing applications, which would eventually support patient-reported immunization records and integrate with jurisdictional systems to retrieve immunization records. An example of this use case is a pan-Canadian consumer-facing digital immunization record that leverages the CVC to populate its standardized vaccine picklists (Figure 2). A consumer-facing digital immunization record that consumes the CVC API, which is populated with standardized picklists of vaccines available in Canada. To date, the app has been used to document the vaccine records of over 400,000 Canadians.

CVC Working Group
Standards development is a collaborative exercise, and decisions to include specific content in the CVC have always leveraged the expertise of the Canadian public health community. The CVC Working Group was developed to serve as a forum to discuss new and upcoming vaccine products on the Canadian market, requests to develop or modify existing SNOMED CT terminology, as well as business and technical requirements of immunization terminology across jurisdictions and systems. The Working Group consisted primarily of jurisdictional partners, but also saw representation from Health Canada, vendors, and non-profit organizations.
Discussion
To function optimally, immunization registries should contain high quality, accurate data across the lifespan, starting at the point of vaccine administration. Public health organizations, clinicians and individuals should have simultaneous access to their immunization record, digitally and in real-time. Immunization records should also follow individuals within and between jurisdictions, facilitating the provision of safe and up-to-date vaccination, and enabling active public health surveillance.
15
The ability to meet this vision rests on a solid foundation of standardized immunization data and timely access to the information required to adhere to the standard. Figure 3 illustrates our vision for a patient-centric, interoperable IIS.
10
Data sources and components of a modern immunization information system. Abbreviations: AEFI, adverse event following immunization; AI, artificial intelligence; IIS, immunization information system.
Summary of recommendations.

Recommendation 1
Establish clear, sustainable governance processes in immunization standards and terminology development and maintenance
One of the major barriers to efficient standards development and implementation we encountered in Canada was related to an underdeveloped governance process, a weakness that became particularly evident during the COVID-19 pandemic. In its current state, standards and terminology development is a relatively siloed process. However, a well-defined and solidified governance structure of how data feeds these registries is imperative.
Recommendation 1a: Enhance processes for the development of standards and terminology at the ground level
Collaboration, subject matter expertise, and solicitation of feedback when developing immunization terminology are complex undertakings and require input from multiple stakeholders. The COVID-19 pandemic presented an unprecedented challenge in this regard in immunization terminology development. As COVID-19 vaccines were imminently approaching emergency authorization approval in Canada, it was unclear which organizational body was technically responsible for leading this important prerequisite to vaccine administration. In addition to critical product details, stakeholders required access to vaccine lot and expiry date information in near real-time to support their vaccine rollouts. It became clear that the CVC Working Group was the forum where this was being discussed, leading the Working Group to spearhead the development of SNOMED CT concepts for the Moderna and Pfizer-BioNTech vaccines that arrived in December 2020.
Further to a lack of clarity around roles and responsibilities in standards development, there were also many gaps in how to approach creating terminology for a novel vaccine technology platform (i.e., mRNA vaccines). In the absence of a formal body to advise on essential data elements from a clinical perspective, the CVC Working Group relied heavily on individual connections with public health subject matter experts for support. Canada’s regulator was also able to accommodate the need for variable product data by delivering ad-hoc reports to the CVC whenever new lots were released. The hard-working collaboration among all participants of the CVC Working Group to develop SNOMED CT concepts for both domestic and international vaccines was an impressive accomplishment, but also revealed a key area for improvement. To mitigate the challenge of gathering input from experts in the field of immunization, we recommend formalizing a more rigorous process with the regulator to provide variable product data, as well as assembling an Immunization Terminology Advisory Committee.
The purpose of the Immunization Terminology Advisory Committee would be to advise on terminology requirements from a clinical and vaccine safety perspective, and provide resolution when information is not consistent between respected sources. For example, the committee could advise on the level of granularity required for the documentation of vaccines in an IIS in order to ensure patient safety, assess protection from vaccine-preventable diseases at a population level, support vaccine safety surveillance, and conduct epidemiological investigations. Given that this committee would consist of senior-level professionals with many competing demands, it is also our recommendation that members be remunerated for their time to secure accountability and commitment to this work.
Recommendation 1b: Establish the national public health body as the accountable party to ensure the long-term sustainability of vaccine standards and terminology
An open-access resource with critical information on immunization data is a gap in Canada’s immunization strategy. It has become clear that a vaccine catalogue is essential for jurisdictions and vendors to adhere to a standard, and to achieve IIS interoperability. It is our recommendation that the national governing body for public health take ownership for both the resource hosting the data, and for the governance structures that underpin the creation of content within the resource. Furthermore, we recommend that national public health bodies commit to a long-term, sustainable funding and program support plan to support this critical resource. The lack of consistent and reliable funding for the CVC throughout its existence occasionally led to disruptions in our ability to maintain the CVC, despite reliance on it from stakeholders. Moreover, during the COVID-19 pandemic, when vaccine terminology was changing rapidly, resource limitations meant that we were unable to update the CVC more frequently than our usual bi-weekly schedule. Dedicated resources for IIS interoperability can help to ensure that the structures underpinning the system remain up-to-date, especially during times of crisis.
Recommendation 2
Bridge the gap between public health and information technology by engaging medical officers of health and chief technology officers in an immunization interoperability strategy
The implementation of standards requires balancing requirements from clinical, business, and IT perspectives; all of these sectors may have different use cases for standardized immunization data and often work in silos. During the initial development of the CVC, the most engaged end-users included those in the public health and clinical implementation domains. Of course, the participation of these users was essential for developing clinically accurate content, but in hindsight, it was equally important to engage and establish buy-in among the IT sector and digital transformation offices at the jurisdictional level. For example, had Canadian provincial and territorial public health authorities built a strong line of communication with IT teams while developing SNOMED CT immunization content, they could have requested (or required) that vendors use these standards when documenting and transmitting immunization data.
Through our experience developing the CVC and other immunization software products, we have learned first-hand the importance of fostering strong partnerships between public health experts and technology implementers to build solutions that optimize end-users’ workflows and achieve immunization program goals. To make progress towards national interoperability, we recommend developing a national immunization interoperability strategy. This strategy could serve to bridge the gap between public health and information technology stakeholders, and ensure jurisdictional partners stay accountable to the country’s immunization interoperability roadmap.
Recommendation 3
Implement strategies to enforce standards use among EMR vendors
A common barrier to a comprehensive immunization registry that is populated with immunization records across the lifespan is the difficulty of retrieving data from vaccine providers using EMR software. In the Canadian context, there are significant gaps in vaccine data housed in jurisdictional registries that are administered by family physicians, hospitals, and pharmacies. Barriers to EMR adoption of immunization standards can be multi-faceted: outdated, legacy software is often unable to support the implementation of new fields and pre-populated picklists to support standardized data capture; the sheer number of EMR vendors makes it nearly impossible from a resource perspective to ask every provider to align to a common standard; and primary care providers often use different instances of EMR software.
If jurisdictions are to have one interoperable immunization record for every individual, EMR vendors will need to support data collection workflows that align with Canada’s national standard, which can then be transmitted to immunization repositories. This may require implementation through a top-down approach by enforcing regulation of standardized vaccine data collection, as jurisdictions do not currently have the business case to support such a requirement from EMR vendors.
Alternatively, jurisdictional bodies could establish subsidiaries that have a mandate to support clinicians in the adoption of standards. For example, OntarioMD, funded by the Province of Ontario, exists to support family physicians, specialists, nurse practitioners and medical administrative staff with the use of digital health technology, connect them with provincial digital health tools, and optimize EMR workflows.
Recommendation 4
Provide comprehensive onboarding resources and support for immunization standards adopters
In a similar vein to enforcing standards adoption among EMR vendors, there is a clear need to provide comprehensive educational materials and onboarding resources to help system implementers with standards adoption. This includes the development of training and reference material on implementation and maintenance of vaccine terminology, including accessible content that would facilitate understanding by those not trained in health informatics. In addition to written material, recorded presentations that provide an overview of the terminology structure — all specific to the immunization use case — would be a valuable asset. These educational tools must be appropriate for both technical and non-technical audiences.
Educational materials should also include information about functional immunization registry standards and minimum (core) data elements for vaccine documentation. This includes documentation requirements at the time of vaccine administration, as well as for recording a previously administered vaccine. While this information does currently exist, it is siloed from other vaccine standards and terminology information; an information bank containing all material and tools needed to adopt immunization standards would be a welcome resource for system implementers.
Finally, for those who are not currently adopting immunization standards, standards adoption may (or may be perceived to) require significant administrative and technical resources, particularly in the initial stages. In order to support widespread adoption, integrating and utilizing standards needs to be simple, rapid, well-coordinated, and measurable. It will be important to offer technical support and subject matter expertise to facilitate the onboarding process for stakeholders, particularly for the adoption of API integrations.
Global perspectives for IIS implementation
While our experiences with the CVC are reflective of the unique needs and challenges faced in the Canadian context, many other countries have implemented electronic immunization systems that can offer important lessons. A 2023 article by Carnahan and colleagues 17 describes the experiences of implementing electronic immunization registries in Vietnam, Tanzania, and Zambia. Like our experiences with the CVC, across all three settings, interdisciplinary teams with representation from a wide range of stakeholders were essential to the success of the projects. However, in Vietnam, consistent with our experience in Canada (Recommendation 1a), a lack of clarity around roles and responsibilities created challenges and delays in implementation. 17 Similarly, in Zambia, difficulty coordinating decision-making between two different ministries complicated the design, development, and implementation of the system, 17 underscoring the need for a dedicated governing body that can take ownership of the IIS and ensure its sustainability and effectiveness (Recommendation 1b). This finding was also described in a 2023 study of Australia’s whole life immunization program, which emphasized the importance of clear governance and data access policies, which contribute to effective management. 18 Ensuring that the responsible governing body provides adequate and dedicated resources for ensuring the sustainability of an IIS was another important lesson taken from all of these settings, consistent with our experiences.17,18
In their evaluation of Australia’s immunization registry, Kpozehouen and colleagues 18 also noted that a key lesson learned concerned the need for high-quality, bidirectional data flow between the system and healthcare providers. Moreover, they argued that a successful IIS must integrate with existing health registries and IT systems. This finding aligns with our recommendation for a comprehensive IIS roadmap that includes both public health and information technology partners (Recommendation 2).
Evaluations of IIS interoperability in North Dakota 19 and New York 20 indicated that linkages between an IIS and EMR can improve data quality and completeness, reduce data errors, 19 help to increase on-time vaccination and series completion rates, and reduce rates of over-vaccination. 20 These benefits align with our findings that coordination with EMR vendors can substantially enhance the value and utility of an IIS (Recommendation 3).
The challenges posed by the fragmentation of the Canadian healthcare system are similar to those experienced in the United States (US), where IIS are managed at the state or local level, and no centralized vaccine data registry exists. 21 A 2023 assessment of registries in the US argued that minimum functional standards are essential for an IIS that can meet the needs of healthcare providers and other immunization stakeholders, but the majority of state laws do not require that these minimum data elements be provided, limiting interoperability. 21 Similar lessons can be gleaned from a 2017 study evaluating the roll-out of an electronic IIS in Kenya. 22 In line with our findings in Canada, many of the challenges experienced in Kenya stemmed from a lack of consistent standards and data elements, which limited the system’s utility, particularly regarding interoperability and data sharing. 22 By contrast, a 2024 analysis of Korea’s IIS notes that the country’s efforts to create a nation-wide, standardized dataset has led to a comprehensive vaccine registry that helps considerably with efforts to evaluate vaccination timeliness and coverage across the country. 23 These findings highlight the importance of minimum data standards and adequate training materials that can support system implementers with standards adoption and implementation (Recommendation 4). This finding was also consistent with a systematic review by McGinn and colleagues, 24 which found that healthcare professionals and patients both expressed hesitancy over electronic health records due to concerns over a lack of familiarity with the technology.
Conclusion
At its foundation, interoperability requires the standardized collection of data, allowing health systems to speak the same language and share critical information with each other. This is integral to promoting public health and ensuring patient safety in several ways. It helps patients stay up-to-date with their immunizations by supporting vaccine forecasting, allows for swift follow-up in the event of a vaccine recall, permits active vaccine safety surveillance, and can be cost-saving for the healthcare system by better managing vaccine inventory. On the heels of the COVID-19 pandemic, we have seen first-hand the importance of high-quality data to inform a data-driven public health response. The lessons we have learned from our work administering and maintaining the CVC can help to drive these public health efforts in the years ahead.
Footnotes
Acknowledgements
CANImmunize gratefully acknowledges the support of the Public Health Agency of Canada and Canada Health Infoway in the development and maintenance of the Canadian Vaccine Catalogue, as well as the Canadian Public Health Surveillance Community for their contributions over the years.
Ethical approval
This manuscript is a review and did not involve any participants or health information; therefore, ethics approval was not required.
Author Contribution
Rubens-Augustson, T completed research, analysis, and wrote the manuscript. Wilson, LA reviewed and edited the manuscript. Bell, C and Wilson, K provided subject matter expertise and informed the conceptual design of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would also like to acknowledge the Canadian Institutes of Health Research for funding to support the development of this article (CIHR Project Grant #492071).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CANImmunize Labs is the research arm of CANImmunize Inc., a for-profit company specializing in the development of immunization software and technology solutions.
