Abstract
Background: Although exercise benefits female cancer survivors, clinical decision-making regarding timing, frequency, duration, and intensity is lacking. Optimizing exercise interventions in this population is necessary. This study aimed to describe existing digital home-based exercises and to assess their effectiveness at improving physical health in female cancer survivors upon completion of therapy. Design: We conducted a systematic review using articles from Web of Science, Embase and Medline (Ovid). We included intervention studies examining the effects of digital home-based exercise programs on post-treatment recovery in female cancer survivors. Rob2 and ROBIN I were used to assess quality of studies. Quality-of-life, fatigue score, and physical performance were assessed using meta-analysis. Results: This study involved 1578 female cancer survivors in 21 interventions. Following guidelines and supervised exercise with coaches led to better outcomes than interventions without guidelines, programs without coaches, or lower intensity exercise. Exercise led to significant improvement in some physical performance outcomes. Significant improvements were seen in physical performance outcomes, including the 6-min walk test, metabolic equivalent task, and number of steps per day. Conclusion: Providing cancer survivors with standard guidelines for home-based, coach-supervised, vigorous exercise on digital platforms could improve their physical function, health, and quality-of-life.
Introduction
Cancer has caused a high burden of diseases among women worldwide 1 because not only the high mortality and incidence rate but also the high number of disability-adjusted life year (DALY) compared to men. 2 Female cancer survivors tend to have higher incidence rates 3 and live longer than male cancer survivors. 4 Females have a significantly higher incidence rate of cancer than males between the age of 18 and 60, according to an official report from the United Kingdom. 3 In 2012, there were an estimated 6.7 million new cases and 3.5 million deaths attributed to cancer among women globally. 5 Fortunately, average 5-year survival rates of almost all cancer types have increased significantly over the last three decades, particularly for breast cancer (74.8% in 1970 vs 89.7% in 2007). 6 This is most likely attributable, at least in part, to the progress that has been made in the diagnosis and treatment of cancer among women.
Indeed, since the widespread introduction of radiotherapy in cancer treatment in the 1990s, 60% of cancer patients have received therapeutic radiation alongside chemotherapy, immunotherapy, hormone therapy, and surgery to shrink tumors or alleviate pain in patients with metastases. 7 Worldwide, as a result of radiation therapy 3.5 million people may have a chance of cure, with a similar number receiving post-treatment palliative care. 8 Cancer treatment extends the lives of half of those diagnosed with the disease for 10 years or more. 3 While the benefits are evident, one must also acknowledge potential downsides. The quality-of-life can be adversely affected by side-effects of cancer treatment, including fatigue, sleeplessness, and cancer-related cognitive impairment. 9 Therefore, post-treatment care for cancer survivors is essential to ease the burden of treatment-related side-effects and hence to reduce the time to recovery.
Exercise interventions are increasingly used to help cancer survivors to improve their health and functionality following recovery.
10
Exercise therapies are safe, have the potential to minimize the risk of mortality, and to produce significant improvements in clinical, functional, and quality of health outcomes in cancer-surviving patients.
11
Exercises are also proven to improve in quality-of-life, muscular and aerobic fitness, and overall health
12
and improve management of post-therapy symptoms, especially fatigue and sleep disturbance.13,14 With cutting-edge digital tech, home-based exercise programs offer a promising post-treatment strategy. They efficiently manage interventions, motivate home-based physical activity, and maintain contact with healthcare providers after hospital discharge.15–17 A digital home-based program is an intervention that an individual can undertake from home for any duration of follow-up using a smartphone, tablet, laptop, or home computer.16,17 Hence, digital home-based exercise treatments should be developed and put into practice with a view to improving health outcomes of cancer patients. The purpose of this systematic review was to provide a synthesis of evidence regarding the characteristics, implementation approach, and effectiveness of digital home-based exercise treatments on health outcomes. Specifically, the following research questions are addressed: 1. What are the characteristics of digital home-based exercise interventions for female cancer survivors after completing cancer treatments? 2. What effects have these interventions had on health outcomes of interest for cancer-surviving women following cancer treatments?
Methods
This systematic review was conducted following the “The Preferred Reporting Items for Systematic Reviews and Meta-Analyses” (PRISMA) guideline. 18 The detailed protocol has been registered on PROSPERO (#CRD42022348049).
Eligibility criteria
Inclusion and exclusion criteria for literature selection were defined using the Population, Intervention, Control, Outcomes, Study Design (PICOS) method. 19
Types of population
The following inclusion criteria were all required to be met: (1) women (18 years of age or older) with a diagnosis of any cancer type; and (2) completed an episode of main and adjuvant cancer treatment, such as radiation alongside chemotherapy, immunotherapy, or hormone therapy. Following treatment, each woman could have hospital outpatient examinations according to their treatment schedule. Exclusions were for: (1) patients receiving acute or inpatient hospital care; and/or (2) metastatic cancer patients (more than 80% of participants).
Types of intervention
Digital home-based exercise interventions were considered for: (1) implementation at home; (2) patients who had completed cancer treatment; and (3) when exercise interventions needed to include at least one type of physical exercise, such as aerobic exercise (AE), resistance exercise (RE), Yoga, or tai chi 11 ; and (4) when the participant possessed a mobile device, handheld tablet, or laptop computer to offer the intervention via smartphone apps, online platforms (websites, Zoom, Skype, WhatsApp, social media channels, etc.), video gaming, and DVD videos. 20 The review excluded hospital/clinic-based or home exercise interventions like cooking, cleaning, and gardening without supervision or intensity.
Types of control and study design
Intervention studies included randomized controlled trials (RCTs), quasi‐experimental studies, and multiple intervention arms trials with no control groups. Non-experimental studies such as case reports, cross-sectional and observational studies were excluded. Before/after studies with no comparison groups were also deemed as ineligible for inclusion.
Types of outcomes
This review focused on self-reported quality-of-life (QoL) outcomes and selected physical outcomes: - Fatigue: the outcome could be assessed or self-reported using validated questionnaires, such as Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F). - Health-related quality-of-life (cancer-specific or, cancer site-specific): using validated questionnaires, such as Functional Assessment of Cancer Therapy-General (FACT-G), European Organization for Research and Treatment of Cancer Quality-of-life Questionnaire-Core 36 (EORTC QLQ-C30), and Functional Assessment of Cancer Therapy-Breast (FACT-B). - Physical activity: maximum rate of oxygen consumption during physical exertion (VO2 max), distance walked per unit time (MET – hour/week), 6-min walk test (6MWT), and metabolic equivalent task in minutes per week (MET – min/week).
Language of publication
No restrictions were set on the language of publication, although all search terms used were in English.
Search methods
On 25 July 2022 a search was performed for published interventions in four different bibliographic databases: Web of Science; Embase; Medline; The Cochrane Central Register of Controlled Trials. The search strategy was presented in Appendix 1, Table S1. Our search included a combination of Medical Subject Headings (MeSH) and key words describing the population and intervention. An initial limited search of Medline (Ovid) was undertaken to identify articles on the topic and key terms used to describe the articles. Those key terms were then matched to search for MeSH terms and key words were used to search for titles, abstracts and keywords in Medline.
Study selection
Search results were imported into EndNote 20, where duplicates were eliminated before a title screening. The two-part study selection process involved a title and abstract review, followed by a full-text review. Two reviewers (KHN and LB) independently assessed titles/abstracts, reaching a consensus on full texts. Potentially eligible articles were assessed by the same reviewers, with a third reviewer (HHN) available for consultation if needed.
Data extraction
Data were extracted from selected articles, encompassing general information (country, authors, publication details), participant details, intervention specifics, data analysis methodology, outcome measures, and results. Continuous outcomes, such as physical fitness, fatigue, and quality-of-life scores, were extracted with means, standard deviations, and participant numbers assessed. Two researchers (KHN and LB) independently extracted data, verified by a third researcher (HHN) for accuracy. Disagreements could be resolved by consulting a fourth researcher (HDT).
Assessment of risk of bias
Two reviewers independently conducted a risk of bias assessment. According to a recommendation from the National Institutes of Health (NIH), 21 the Cochrane Handbook for Systematic Reviews of Interventions (Risk of Bias 2; Rob 2) tool and the Risk of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool were used.22,23
Data synthesis
The meta-analyses were conducted using R software (version 4.2.0) using meta package. Given methodological heterogeneity in target population, intervention implementation, and outcome measurement, the random-effects model was employed to address variations across studies. 24 Statistical heterogeneity was assessed using the I2 statistic, which quantifies the inconsistency between separate studies. An I2 value of up to 25% was considered as predictive of low heterogeneity, an I2 value of around 50% as moderate heterogeneity, and an I2 value of 75% or more as considerable heterogeneity. 25
Results
Study selection
Figure 1 depicts the flow diagram of this study’s selection process. The bibliometric search found 5406 studies. After removing 1881 duplicates, 3525 articles underwent title and abstract screening. Of these, 3476 were excluded for not meeting criteria, leaving 49 for full-text screening. Twenty-six studies were excluded for not meeting eligibility criteria. In the end, the review included 23 experimental studies reporting outcomes from 21 interventions. There were two pairs of articles reporting the same interventions.7,26–30 Flow diagram of this study’s selection process.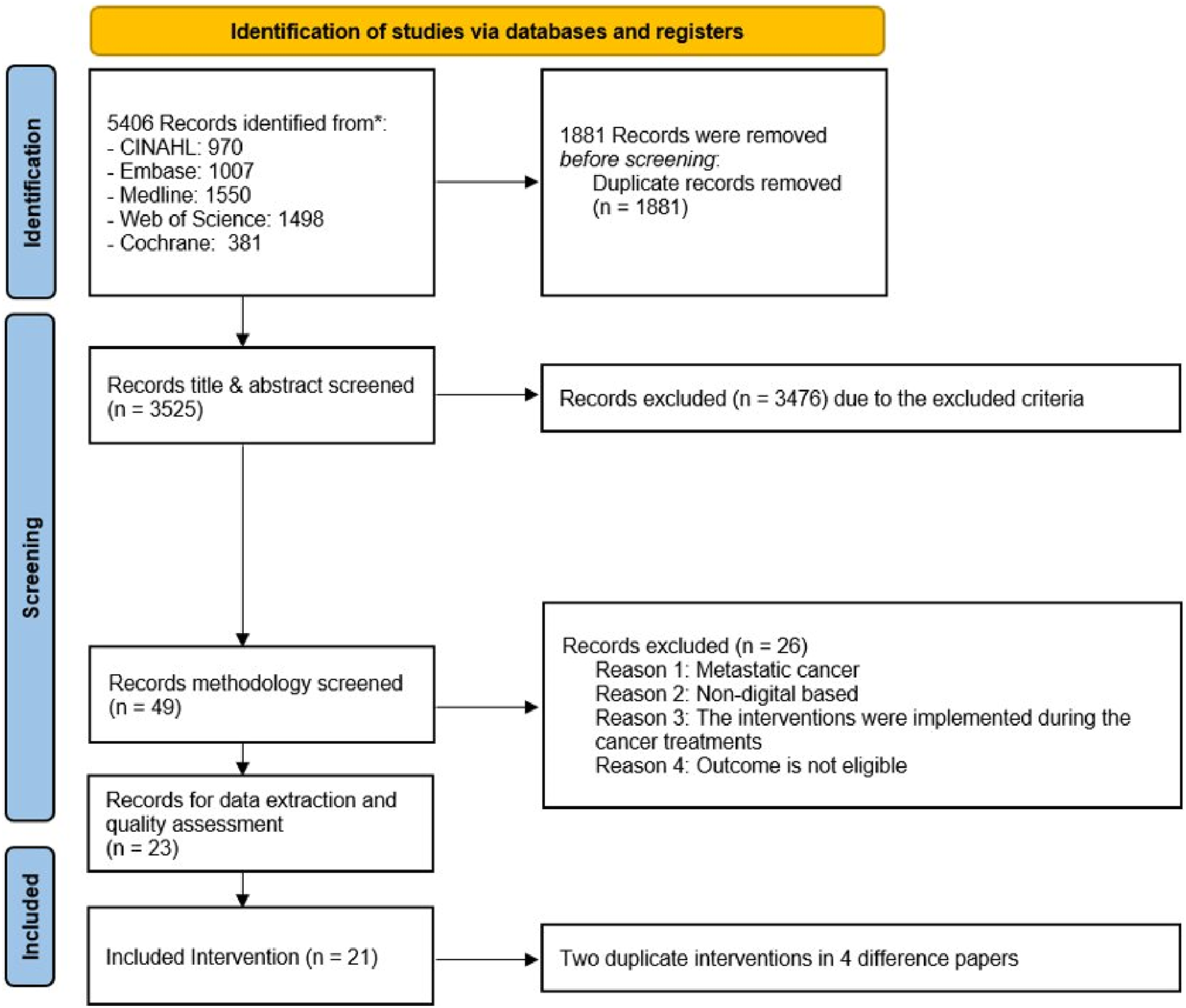
Characteristics of participants
Characteristics of participants among 21 selected interventions.
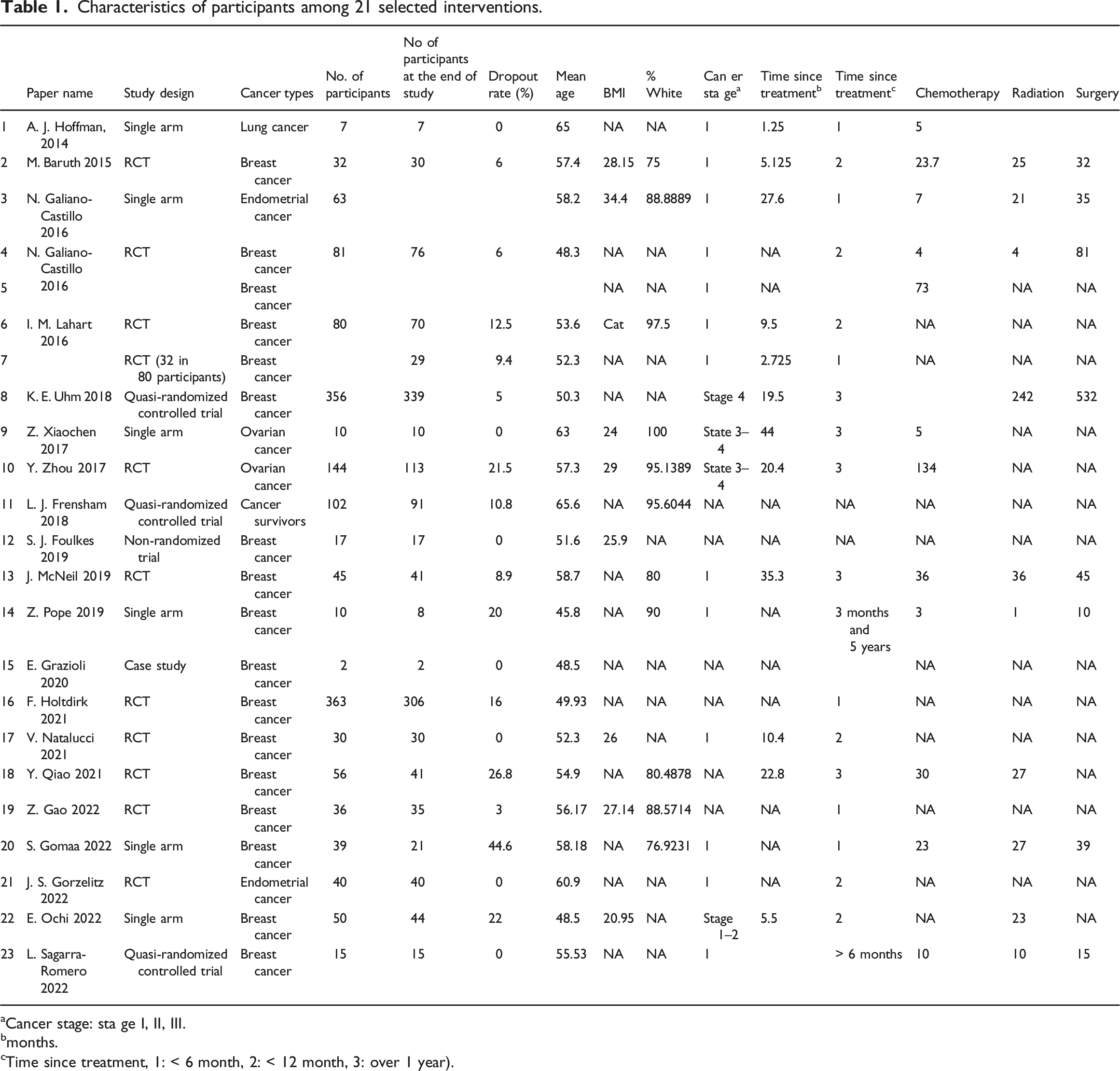
aCancer stage: sta ge I, II, III.
bmonths.
cTime since treatment, 1: < 6 month, 2: < 12 month, 3: over 1 year).
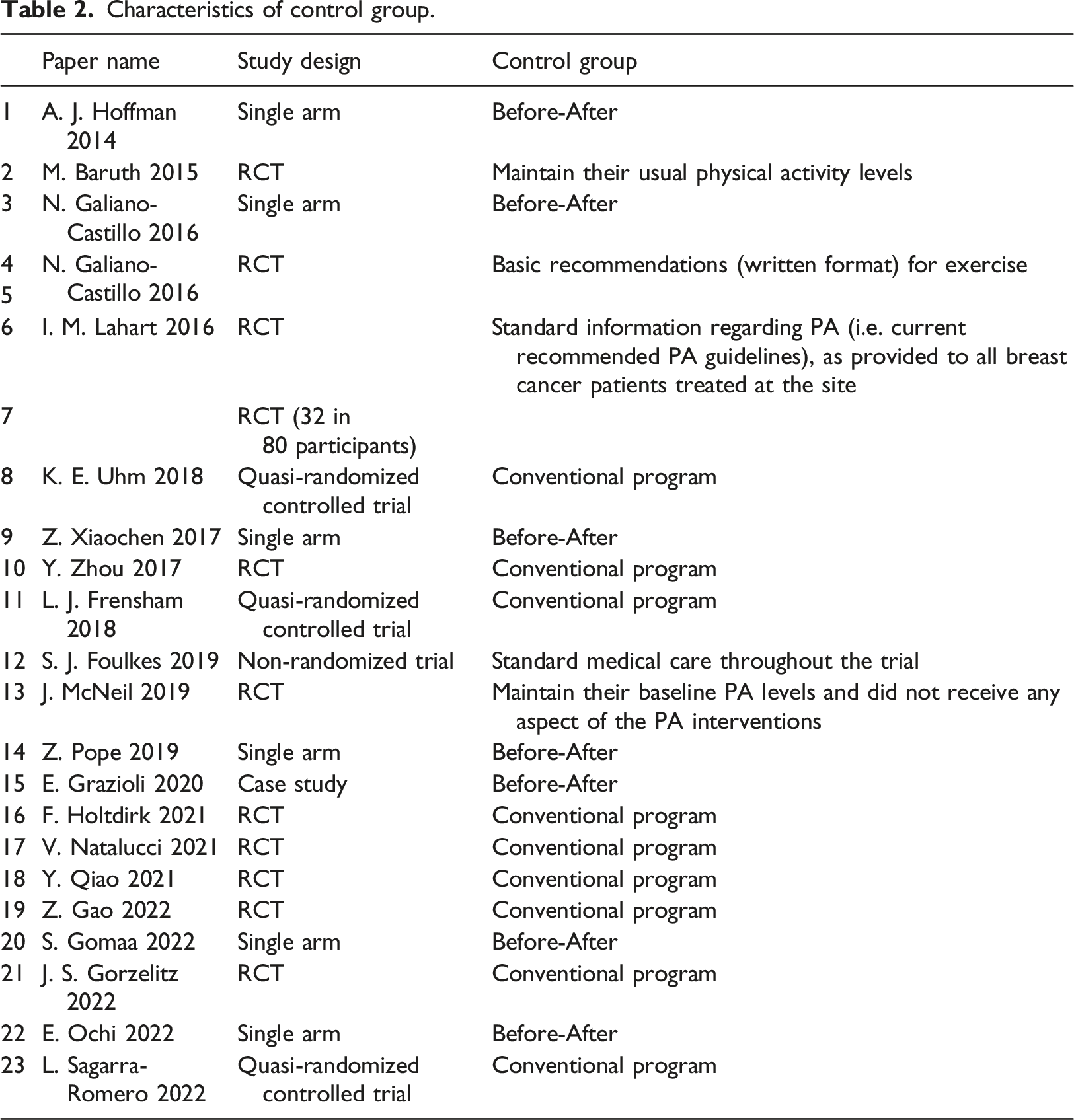
Characteristics of control group.
Characteristics of interventions
Characteristics of 21 selected interventions.

Guidelines
Six interventions26,28,33,36,39,44 used the American College of Sports Medicine recommendations. 50 In which, participants were first evaluated pre-exercise, which comprised taking their medical history, performing a physical examination and fitness testing. Based on an individual’s pre-exercise evaluation results, an appropriate exercise program was prepared to include components (warm-up, stimulus or conditioning phase, recreational activities, and cool-down) and exercise prescription (mode, duration and intensity of exercise). An intervention for lung cancer survivors applied World Health Organization guidelines, 51 which provide details for the intensity of physical activities among different age groups and specific population groups. 31
Supervision
Exercise classes were delivered by a coach to participants and were conducted either through in-person home visits or using online meeting platforms such as Zoom or Skype.46,49 In contrast, for other interventions,26,28,31,33,34,37,40–42,45,46,48 the coach only guided exercise at the baseline appointment, then participants did it at home without program staff monitoring. Smartphone apps and websites were employed in this case. These networks offered peer support, health education, and weekly class recordings for at-home performance.33,37,40,41,45,46,48 Other digital supplementary materials such as video games or DVD videos were provided for participants to support them to practice exercises at home.26,28,31,34,42 For example, the Nintendo Wii Fit Plus video game was used to provide a variety of video exercise routines such as yoga, strength training, aerobics, and balance in a mini games format, which participants could practice at home. 31
Interventions using standard guidelines and programs supervised by trained fitness coaches improved participant results more than those without. The study of Galiano et al.26,27 (using the American College of Sport Medicine for cancer survivor guidelines) reported a two-fold increase in quality-of-life score following intervention compared to a study that did not apply any standard physical exercise guidelines. 32 Furthermore, the mean difference in 6MWT results between treatment and control groups in a supervised intervention 26 was higher than that for either of two comparable but unsupervised interventions37,48 (Figure 4).
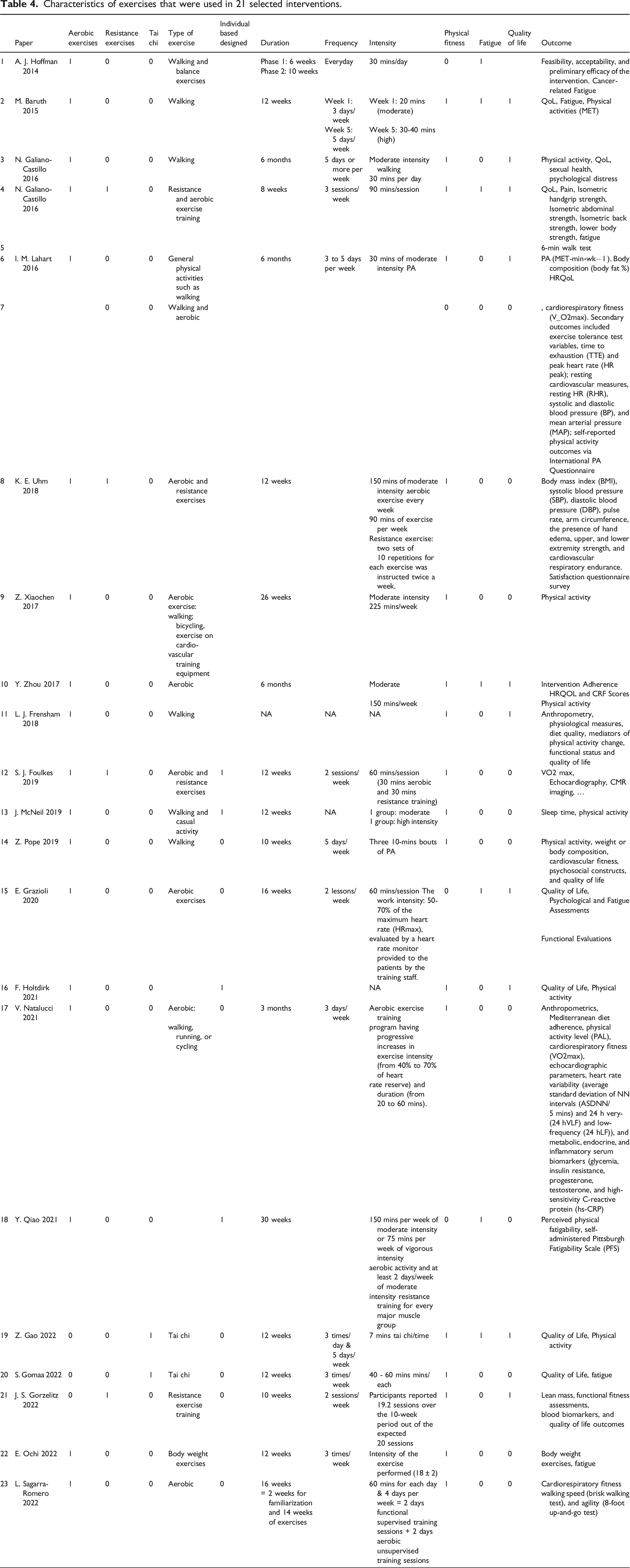
Characteristics of exercises
Table 3 summarizes exercise programs that were included in this study. In terms of physical exercise type, programs can be of a single type or a combination of aerobic exercises, resistance exercises, and tai chi. The majority of included studies combined exercise types, with a mix of aerobic and resistance exercises being the most common format.26,34,38,47 Only 4 studies involved walking26,32,37,40 and 2 interventions offered tai chi exercise as a single type of exercise.45,46 As an intervention, tai chi appears to achieve less significant improvement to physical functioning of female cancer survivors than do either aerobic exercise or resistance training. Pope et al. 40 (aerobic intervention) reported greater impact on physical functioning among the intervention group compared to the result of Gao et al. 45 (tai chi intervention), wherein both studies used the same questionnaire (Patient Reported Outcome Measurement Information System).
Duration, frequency, and intensity
Characteristics of exercises that were used in 21 selected interventions.
Risk of bias assessment
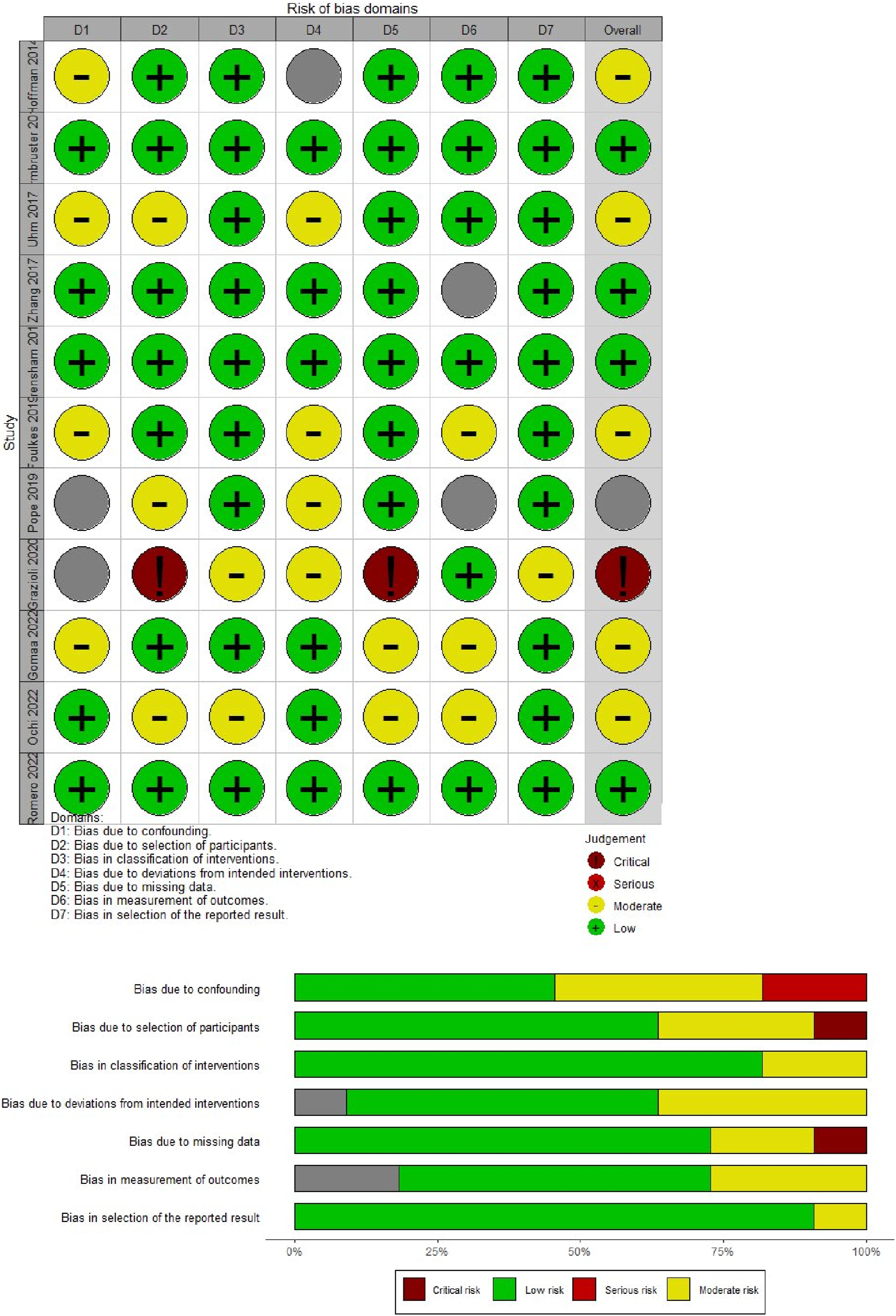
“Risk of bias” assessments for RCTs and non-RCTs are depicted in separate Figures 2 and 3, respectively. Baruth et al.’s and Lahart et al.’s RCTs showed a high risk of bias due to selective reporting and incomplete outcome, respectively.28,32 Lack of allocation concealment, random sequence generation, and participant and staff blinding raised problems in seven research.36,39,42–45,47 ROBINS-I showed that Grazioli et al’s study had a high risk of bias due to recruiting issues and potential intervention variations.
41
Pope et al. had a serious risk of bias due to important concerns regarding confounding.
40
The remaining nine studies were rated as having moderate risk of bias.31,33–35,37,38,46,48,49 Risk of bias assessment for RCTs. Risk of bias assessment for non RCTs.
Effects of interventions
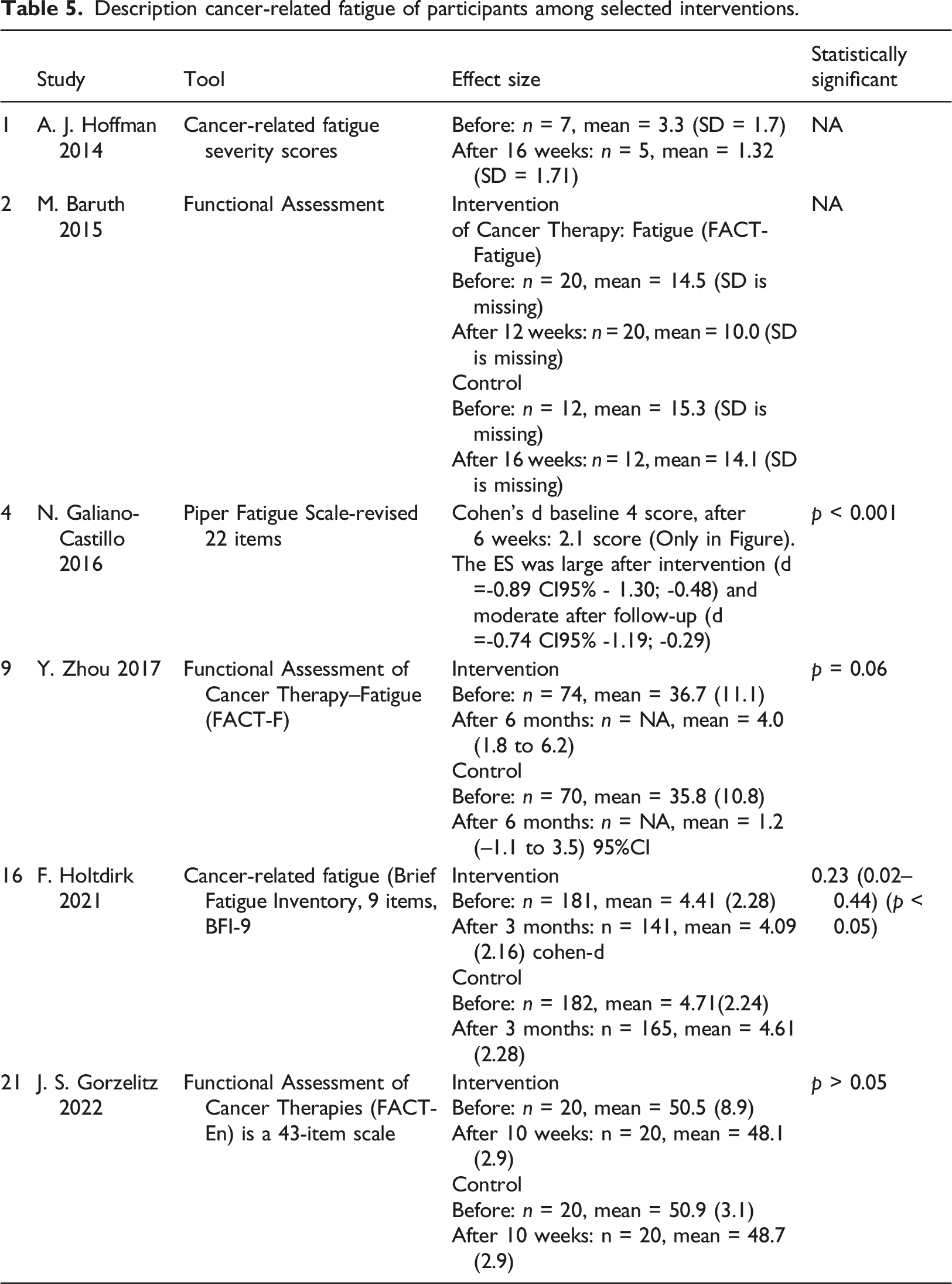
Fatigue
Description cancer-related fatigue of participants among selected interventions.
Quality-of-life
Description Quality of life of participants among selected interventions.

Physical activity
Description Physical activity of participants among selected interventions.
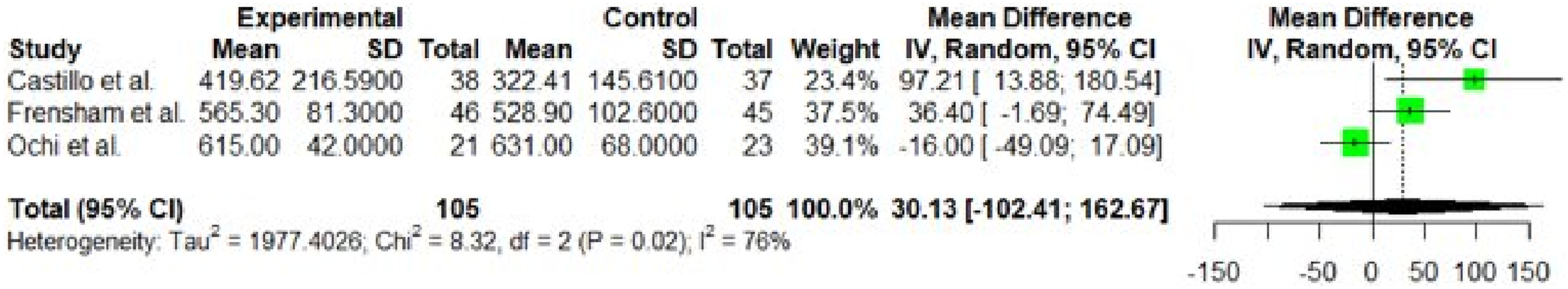
Meta-analysis results for the outcome of 6-min walk test.
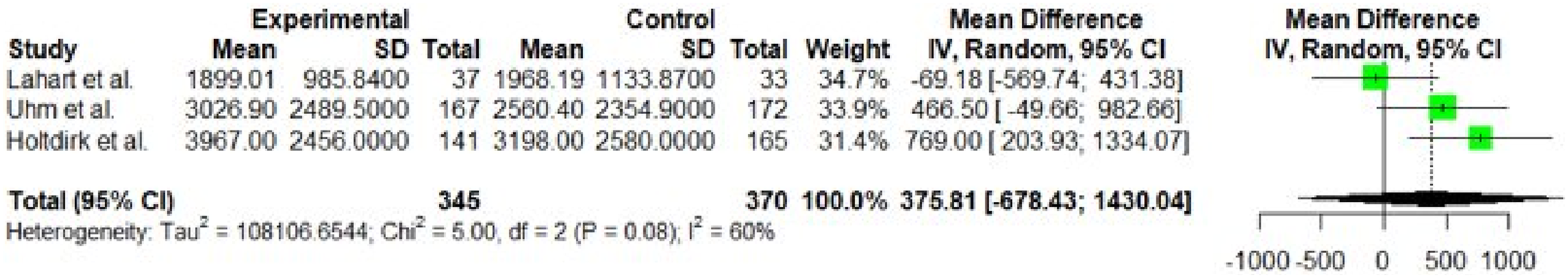
Meta-analysis results for the outcome of MET minutes per week.
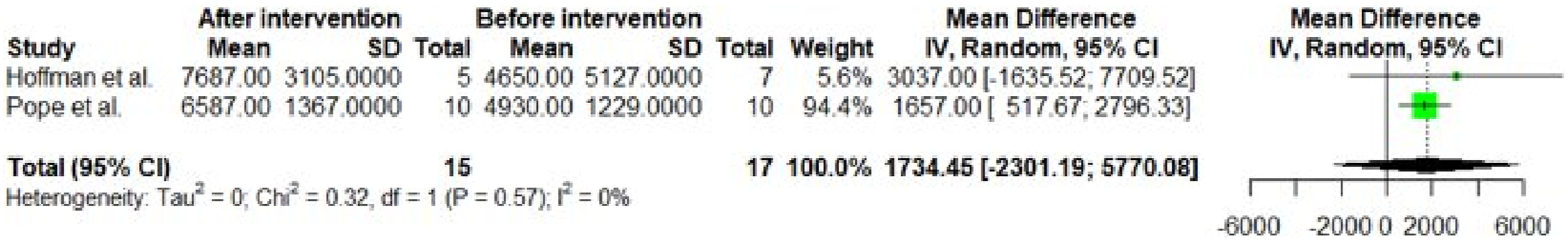
Meta-analysis results for the outcome of Steps walked per week.
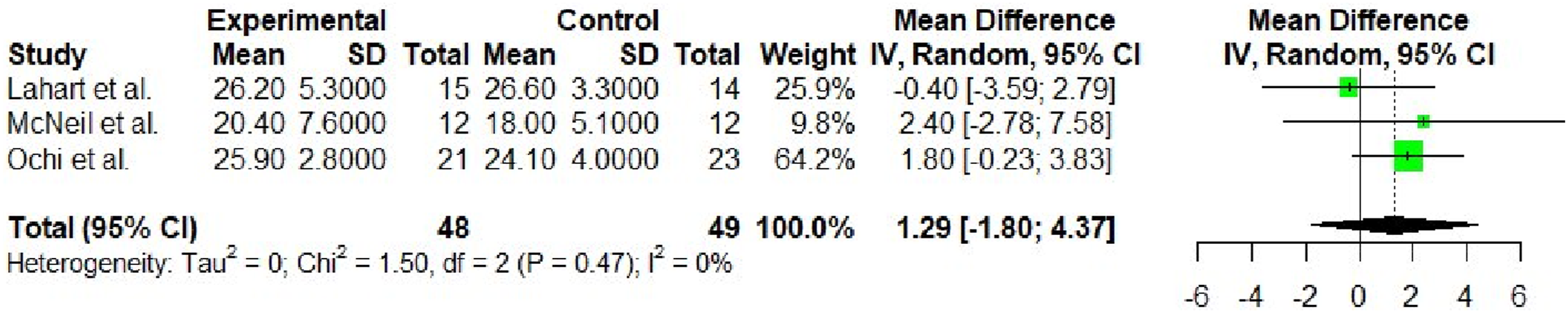
Meta-analysis results for the outcome of VO2 max.
Discussion
Our review included 21 digital home-based exercise interventions among a combined total of 1578 participants. This review found that digital home-based exercise interventions improve quality-of-life (7 studies), physical function (20 studies), and cancer-related fatigue (6 studies) in female cancer survivors post treatment. Interventions involving standard guidelines, programs monitored by qualified exercise coaches, and intense exercise improved participant outcomes more than those without standard guidelines, programs without exercise coaches, and lower intensity exercise.
Our study suggested that following standard recommendations with trained coaches and engaging in intensive activity improved participant outcomes compared to programs without guidelines, without coach supervision, or lower-intensity workouts. This finding corroborates observations from previous studies.55,56 Corres et al. found that a 16-week hypocaloric diet intervention improved cardiometabolic profiles, moving people from Metabolically Unhealthy Obesity (MUO) to MHO. Unfortunately, after 6 months without observation, participants’ biochemical profiles returned to MUO. 55 The importance of guidelines and coaches was previously shown by Gunderson et al. and Church et al.57,58 Following standard guidelines with trained coaches provides better structured, safer workouts that enhance participant compliance and reduce confusion. This expert guidance to vigorous exercise stimulates greater physiological adaptations, fostering improved outcomes compared to when following less structured or lower-intensity programs.
The current study included three types of exercise intervention, namely aerobic exercise, resistance training and tai chi. Tai chi appears to improve physical functioning of female cancer survivors less than aerobic exercise or resistance training. The relative intensity of these two forms of exercise may help to explain this difference. While aerobic exercise and training often demand moderate to rigorous intensity, in the study of Gao et al. 45 tai chi required only minor intensity (7 min of balance exercises). Due to the marked heterogeneity of exercise interventions, the difference in impact on quality-of-life and fatigue were not comparable between the 21 reviewed interventions. Hence, there is a pressing need for further research to focus on creating exercise guidelines that support recommendations at different stages of the cancer care state. This should consider both current and anticipated side-effects of treatment, as well as the health status of an individual.
All 21 programs in this review were in high-income countries. While residents of low- and middle-income countries (LMIC) account for > 70% of all cancer-related deaths, there is a scarcity of supportive care interventions as well as a general lack of research to evaluate their efficacy in these resource-poor settings. 59 Therefore, more exercise interventions are required to be implemented in LMIC, which should also consider targeting women living with types of cancer other than breast cancer.
Limitations
This study has some inherent limitations. Many included papers were excluded from the meta-analysis due to outcome measurement and reporting heterogeneity. However, the interventions’ findings were narratively presented and compared. In addition, none of the RCTs included in this review had high-quality evidence due to a poor blinding process. The risk of bias for allocation concealment and blinding of participants and personnel is high in intervention groups because both patient and coach know about the intervention. Finally, interventions were often conducted in small trials. A consequence of this is that statistical power was compromised in several studies and also negatively impacted on the generalizability of the study.
Conclusion
Digital home-based exercise interventions improved side-effects of cancer treatment. Standard guidelines, exercise coaches, and intense exercise should be considered when designing exercise interventions for cancer survivors.
Supplemental Material
Supplemental Material - Digital home-based post-treatment exercise interventions for female cancer survivors: A systematic review and meta-analysis
Supplemental Material for Digital home-based post-treatment exercise interventions for female cancer survivors: A systematic review and meta-analysis in Huyen Thi Hoa Nguyen, Nguyen Thi Khanh Huyen, Linh Khanh Bui, Ha Thi Thuy Dinh, Andrew W Taylor-Robinson in Health Informatics Journal
Footnotes
Acknowledgements
This study received financial support from the VinUniversity 2022 Fast Track Funding Scheme. We are grateful to the College of Health Sciences, VinUniversity, Hanoi, Vietnam, for providing in-kind support.
Author contributions
HT, LK, TT, and AWT-R generated study aims and collaborated to conduct the study. HN designed the study, organized the search methodology, and data analysis. HT and HN performed data screening and data extracting. HT wrote the first draft of the manuscript. HN wrote the methodology sections of the manuscript. All authors contributed to the manuscript revision, and read, and approved the submitted version. AWT-Rrevised the writing style for the final version of the manuscript and HN is responsible to submit the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by Vin University 2022 Fast Track Funding No. 22-0011-P0001.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
