Abstract
Patients desire greater control over sharing their digital health data. Consent2Share (C2S) is an open-source consent tool offered by SAMHSA and the VA to support granular data sharing (GDS) options that align with patient preferences and data privacy regulations. The need to validate this tool exists. We pilot tested C2S with 199 English and Spanish-speaking patients with behavioral health conditions (BHCs) and patient guardians. Data were analyzed using mixed methodology. All participants desired granular control over the sharing of their health data. Most participants (87%) were highly interested in using a tool that offered granular options for executing data sharing decisions, with over half (55%) indicated that being able to specify the data type, data recipient, and data use purpose made them more willing to share their medical records. Majority (83%) indicated that the supported data type sharing categories satisfied their data-sharing privacy preferences. Majority (87%) also reported that knowing the purpose of data use made them more comfortable in sharing. Some participants (28%) accessed the education materials provided on data type sharing options. Patients want granular choices when sharing medical records. C2S and its supported data type sharing categories are adequate to capture patients’ data sharing preferences. Further development is needed before deployment in clinical environments.
Keywords
Introduction
Granular information sharing refers to “a detailed choice an individual makes to share specific types of health data…[enabling] the capture and exchange of patients’ preferences to advance coordination of care in multiple settings for treatment, payment, healthcare operations, and research.” 1 The Office of the National Coordinator for Health Information Technology (ONC) promotes granular information sharing: “Patients should have a greater degree of choice to determine, at a granular level, which personal health information should be shared with whom, and for what purpose.” 2
Honoring granular data sharing (GDS) requires the development of technologies offering meaningful consent options that align with patient preferences and applicable federal and state data privacy regulations. While there has been a movement toward GDS research in the last decade, the main focus has been on developing data sharing solutions for research,3–7 while tools to support patient-driven GDS for care have been rare.
Consent2Share (C2S) is a consent tool developed by the Substance Abuse and Mental Health Services Administration (SAMHSA) and the Veteran Administration (VA) to support granular segmentation of protected health data. 8 This tool allows patients to share their health information through an online consent process, choosing which sensitive information they wish to share from a list of common sensitive data categories, with whom (individuals or health care organizations) and for what purposes (treatment, payment, or research). 9
C2S is a stand-alone, web-based tool that can also be integrated with existing electronic health record (EHR) and health information exchange (HIE) systems. C2S is compliant with HIPAA and 42 CFR Part 2, the federal regulation that controls the sharing of substance use medical records. 8 The tool supports the segmentation of HL7 CCD or CCDA clinical documents and HL7 FHIR resources and the filtering of structured medical record information codified in standard terminologies (such as ICD-10 diagnosis). 10 C2S does not support the sharing of sensitive unstructured data elements (such as psychiatric clinical notes). C2S relies on terminology-based data sharing models (i.e. taxonomies) for specifying meaningful categories of medical records (e.g. Mental Health Information) and segments data from the patient’s EHR into those categories (e.g. the ICD-10 concept ‘Depression’ is classified as Mental Health Information).
C2S allows customization of data sources, data recipients, sensitive data types and data use purposes. The two most commonly used data type taxonomies in data sharing research were created by the National Committee on Vital and Health Statistics (NCVHS) and SAMHSA.11,12 In a card sorting exercise, 42% (N = 25) of patients using the SAMHSA taxonomy recommended a modification, e.g. substituting the term Drug Abuse for Drug Use or merging categories like Communicable Diseases with Sexual Health and Family History with Genetic Data.13,14 The NCVHS categorization has appeared more frequently in the GDS literature15–20 and has been evaluated using an online consent tool.15,18 The NCVHS categories have not yet been used to assess patients’ preferences on GDS for care.16,17,19,20
The accuracy of C2S when adopting the SAMHSA taxonomy to segment structured data elements has been assessed by Grando et al. 21 The study found significant differences between sensitivity classification by C2S and health providers (χ2 (2, N = 584) = 114.74, p = < .0001). There were 56.0% agreements in sensitivity determinations, 31.2% disagreements, and 12.8% partial agreements. C2S has also been pilot tested using EHR data, but it has not been field validated with individuals at the point of care. 22 There is a need to validate this tool with key stakeholders, namely patients, guardians, and health providers, on its capacity to reflect GDS desires of individuals as well as its usability in a variety of clinical environments.23–26
Literature shows that patients overwhelmingly want choices when sharing their medical records,14,16,27,28 but there are very few studies that focus on data sharing preferences of patients with sensitive information.15,26,29–32 This is surprising because GDS is purported to protect sensitive data.14,24,26,27,33–36 Studies focusing on understanding GDS preferences of individuals with behavioral health conditions (BHCs), i.e. conditions that impact the mental, emotional well-being and/or actions that affect wellness,33,37 are scare.15,26,29,30,38 Understanding the sharing preferences of people with sensitive information is critical in the design of consent tools that support granular segmentation of such information.
Objectives
This study aims to pilot test the C2S tool with patients with BHCs and assess their GDS choices.
Materials and methods
Clinical settings and study consent
The Arizona State University Institutional Review Board approved the recruitment of adult (18 years or older) patients diagnosed with BHCs or legal representatives (guardians) of patients from two integrated, community-based care facilities that together provide care for more than 34,000 patients with BHCs.
Recruitment was performed in collaboration with the facilities using electronic flyers sent via email to eligible individuals (Supplementary Appendix 1). Participants could choose to complete the study using the C2S tool or with a researcher by phone. Participants could choose to take the study in English or Spanish. An electronic consent was administered to patients at the beginning of the study. During the study, participants were informed that their responses would have no effect on the sharing of their actual health records. After the study, participants received a $30 gift card as compensation.
Study design
After obtaining participants’ consent, the University of California, San Diego Brief Assessment of Capacity to Consent (UBACC) test was administered online to access consent comprehension and decision-making capacity of participants (Supplementary Appendix 2). 39 Patients with scores below the established threshold of the test engaged and received compensation, but these responses were not included in the analysis.
A questionnaire was administered electronically to collect participant demographics and care histories within and outside of the integrated facility.
C2S users are required to sign on through email communications and to verify their identify. C2S does not authorize patient representatives or guardians to complete the registration process or to make GDS decisions on behalf of patients. For our study, participants (patients or guardians of patients) were manually registered into C2S to allow those without an active email to participate. We enrolled all participants in the tool as patients and kept track of their participants ID. C2S supports the customization of data sources, data recipients, data types, and data use purposes. We chose behavioral health providers (BHP) within participant’s facility as C2S data source (patients’ choices grantors). We selected (1) primary care providers (PCPs) within participants’ facility, (2) PCPs outside the participants’ facility, and (3) BHP outside of participants’ facility as C2S data recipients (patients’ choices grantees). We adopted the NCVHS data categories (Domestic Violence, Genetic Information, Mental Health Information, Reproductive Health, and Substance Use) to model C2S sensitive data types. We selected research and treatment as C2S data use purposes.
After reviewing available materials and finding none to be satisfactory, we designed custom patient educational material to explain each of the NCVHS sensitive data types. This sixth-grade level, ‘on-demand’ information in English and Spanish was designed using national pedagogical guidelines and Information Systems research.
40
The educational material was embedded into the C2S tool via info buttons (Figure 1). Screenshot of on-demand educational material for mental health information in English and Spanish.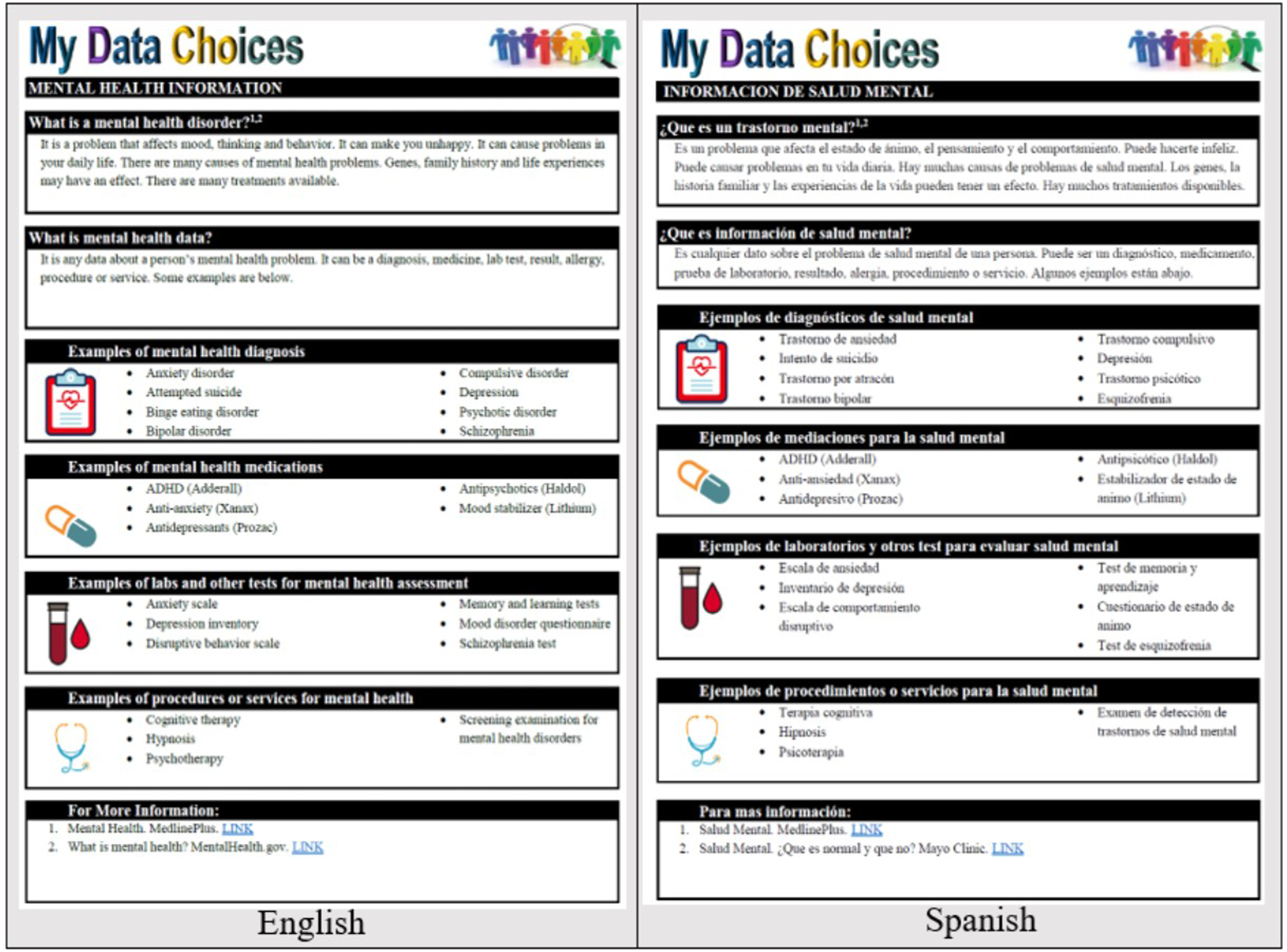
Study participants used the C2S tool to make GDS choices (Figure 2) and provided feedback electronically on their experience using the tool and their views regarding the choices offered by the tool. The study materials are summarized in Supplementary Appendix 3. Screenshot of C2S user interface. Info button activation provides an on-demand explanation for each data type.
Data analysis
Overview of the data sharing options model in C2S and the mixed methodology used for data analysis.
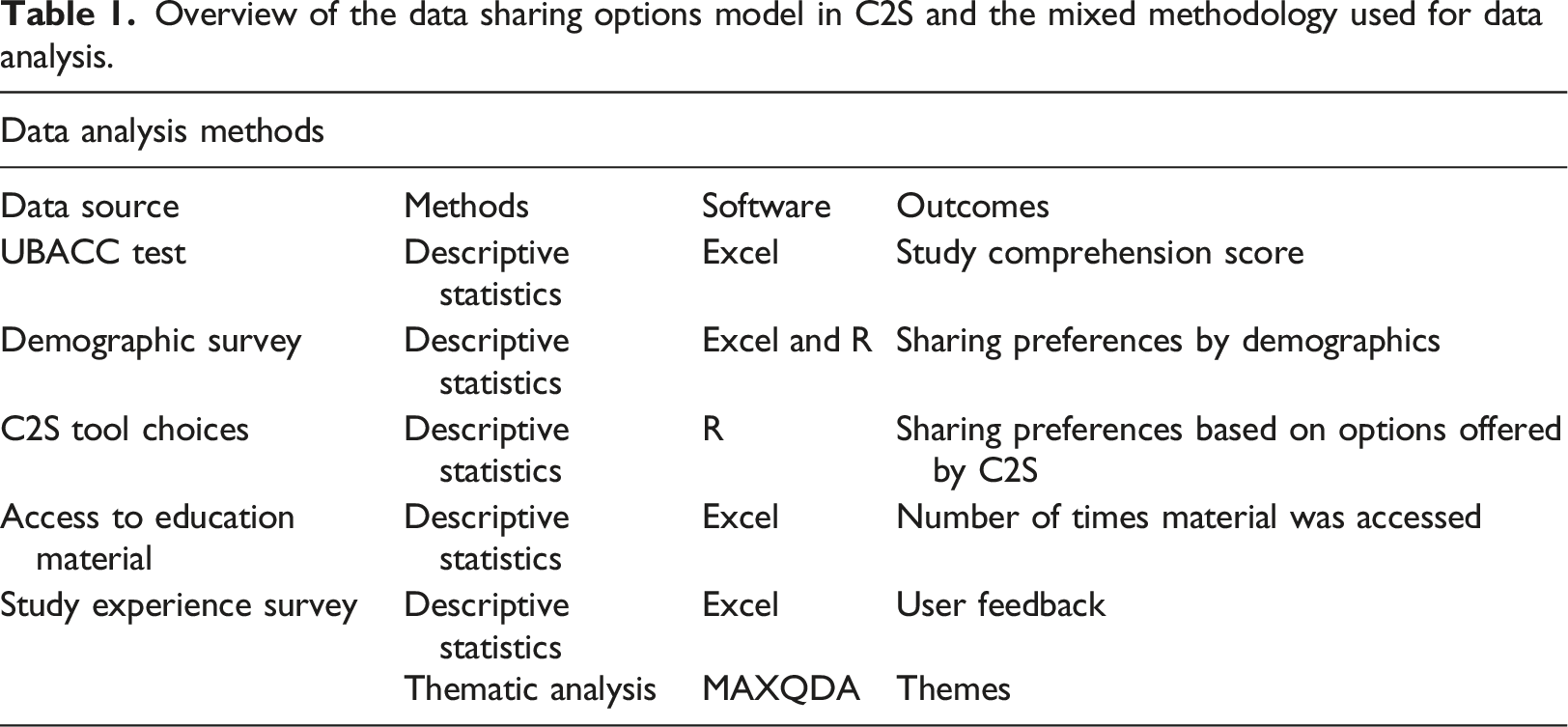
Participants’ references to the educational materials and the impact of education material on sharing preferences was analyzed using descriptive statistic. Descriptive statistic and qualitative content analysis were used to analyze users’ feedback provided in a multiple choice and free response format on the tool.
Results
Demographics
Demographics of participants completing the study (N = 199). Percentage may not total 100 due to rounding.

Access to education materials
Fifty-five participants (28%, N = 199) activated the info button to access educational materials across the three data sharing options 86 times. Across the five supported data types, educational material for Genetic Information was accessed the most (36 times) followed by Sexual and Reproductive Health (26 times), Substance Use (15 times) and Domestic Violence (7 times). Mental Health Information was accessed the least (2 times).
On average, participants who accessed the education material were more willing to share data (86%) than participants who did not (69%). However, sharing all health data was selected fewer times by those who had accessed material (58%) than for those who did not (74%). A majority of those who accessed the education materials were more likely to restrict some health data (40%) than those that did not access the materials (24%). Overall, there was no difference in participant perspectives about the adequacy of the NCVHS data types to capture data sharing needs between participants who accessed the educational materials and those who did not.
User experience survey feedback
Majority of the participants (83%, N = 199) indicated that the provided NCVHS data types captured their data privacy needs (Figure 3(a)). Over half (55%) reported that being able to specify data recipient, data type, and data use purpose made them more willing to share their medical data (Figure 3(b)). Most respondents (87%) indicated that knowing how their data is being used (for care or research) made them feel more comfortable sharing their data. (Figure 3(c)). Most (82%) of the participants wanted to be notified each time someone used their medical data for care or research (Figure 3(d)). Results of user experience survey.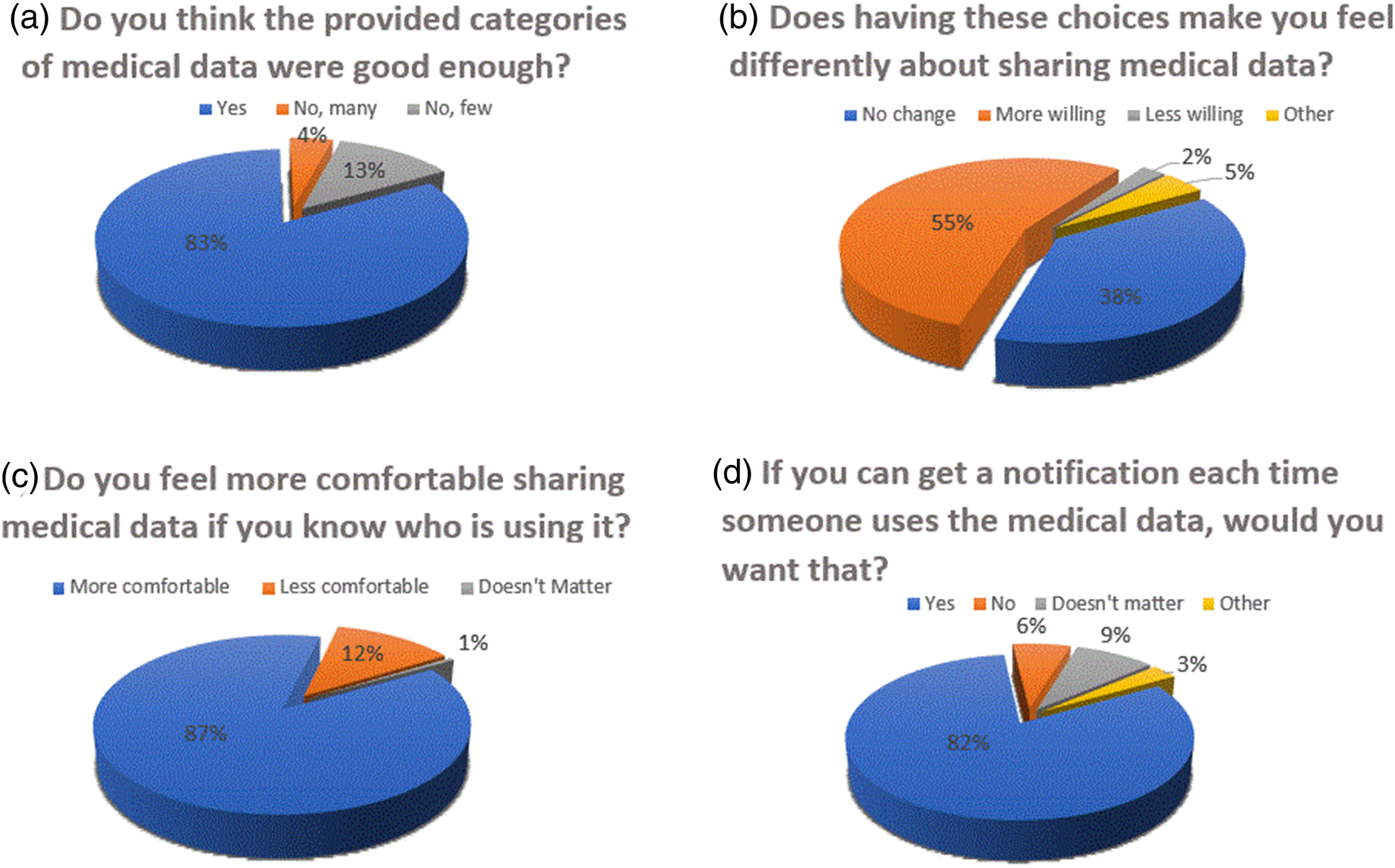
Of the 199 participants, 91 (46%) provided free text responses. Thematic analysis was performed yielding 173 codes that were designated positive (131 codes), mixed (33 codes), or concerned (9 codes). Mixed responses encompassed four key themes: (1) sharing notification (2) changes to categories, (3) additional options in sharing, and (4) more details needed (Figure 4). Thematic analysis results of 173 comments from 91 participants with example commentary.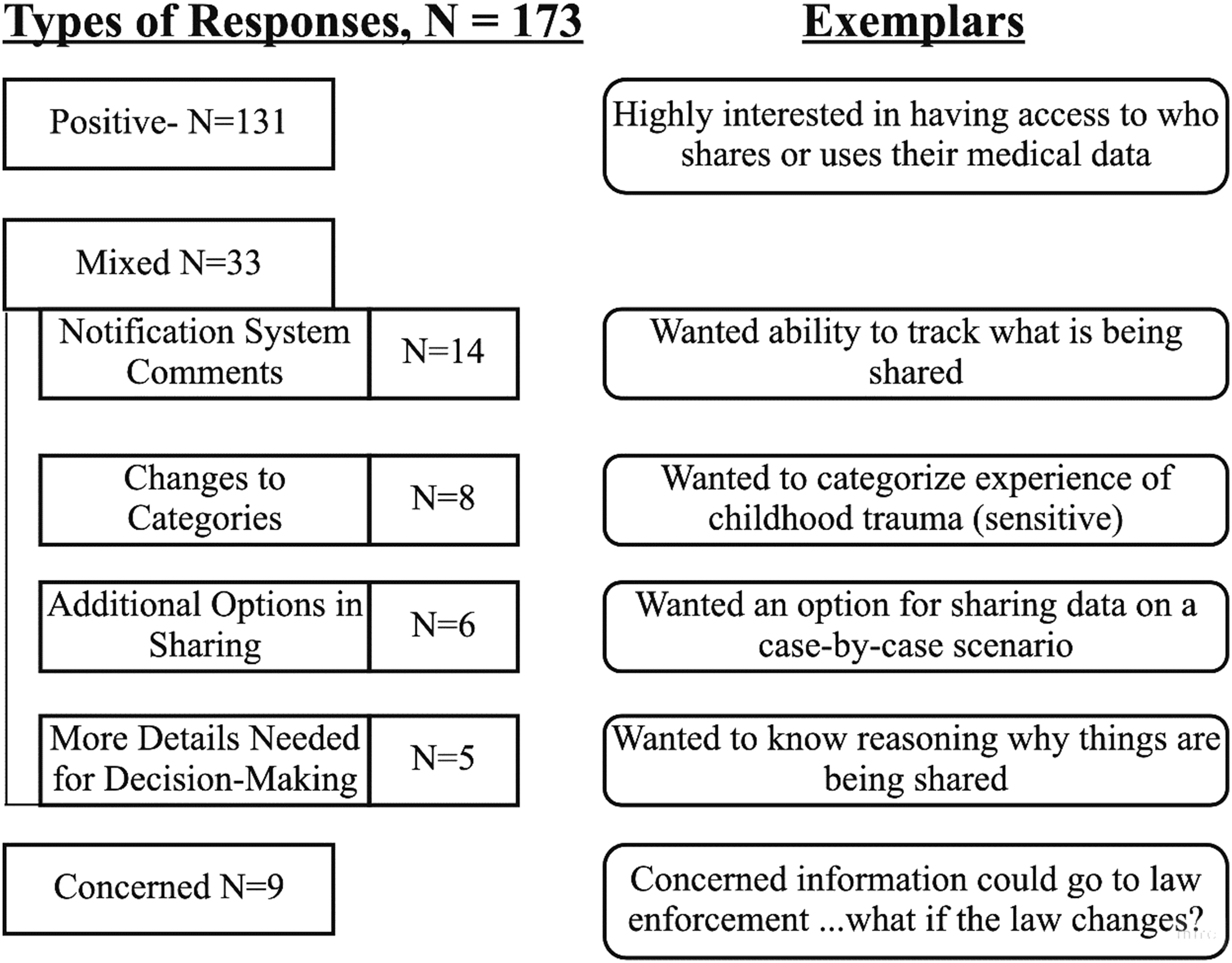
Discussion
This is the first study to pilot test the CS2 tool with patients. Main lessons learned from this experiment are summarized below:
Patients with BHCs want granular control over the sharing of their health data
No participant wanted to share all their data with all data recipients, though most indicated that having granular choices would make sharing more appealing. Most participants wanted to know how their data was to be used (for care or research) and reported that such control made them feel more comfortable sharing. This is consistent with previous studies that focused on patients with BHCs.13,26,30 Our results suggest that the prevalent “share all” or “share none” approach does not accommodate medical record sharing preferences for patients with BHCs. Tools and processes that offer GDS options are needed to increase patient convenience, lessen privacy concerns, and activate patients in cross-organizational health data sharing.
Data type, data recipient, and data use purpose impact patient willingness to share health data
In a complementary study, Karway et al. performed a deeper dive into the sharing preferences of these participants. They reported a significant (p < .001) impact of the data sharing variables supported by C2S, i.e. data recipient, data type and data use purpose, on willingness to share health data. 41 Previous studies showed that sharing was impacted by patients’ contextualization and comprehension of the type of data being shared; by their level of trust in the data recipients and how the data would be used, among others.13,25 Other variables16,19,42–44 such as duration of data access by year and opt-in versus opt-out policies may also impact GDS and the design of user-facing tools like C2S.
Access to information about sensitive data variables impacted sharing choices
Though educational material was accessed by a limited number of participants (28%) there was frequent access to data categories like Genetic Information, Sexual and Reproductive Health and Substance Use. Participants who accessed the education materials were more willing to share their health records, less likely to share all records, and more likely to restrict access to some data. Limited willingness of patients with BHC to consult education materials on sensitive information has been reported previously by Soni et al. 13 Future studies might focus on identifying patient educational needs and receptivity and best format to deliver on-demand information on GDS and sensitive data types.
A simple, customizable user interface using sensitive data type options met GDS needs of patients
Most participants (87%) were highly satisfied with the GDS options supported by C2S and the NCVHS sensitive data types. While these encouraging results open the door to future consent preference studies on a wider range of patient populations, the tool is not ready for production. Improvements are needed in the following: • • • •
Study limitations
Our study mode was modified from in-person to online and phone due to the COVID-19 pandemic. Despite our efforts to recruit a diverse, representative sample, COVID-19 restrictions made this challenging, necessitating the combination of race and age for analysis. The resulting homogeneity of participants in this study may not reflect that of the broader population. We manually registered participants in C2S to simplify the process and allow those without an active email to participate. This approach may not be practical in live clinical environments.
Although our study completion rate was very high, nonresponse bias due to the use of digital devices and network is possible. Participants who did not have access to a phone may have declined to participate. However, our study participation times were flexible, including weekends, and did not require transportation to a physical location. Moreover, the physical absence of a recruiter may have given participants more autonomy when making data sharing decisions.
We did not assess participants’ health status. Although the literature suggests that health status is not a significant predictor of sharing attitude 45 less healthy individuals may have different views regarding data sensitivity and their willingness to share.
Like previous studies on GDS,15,18 this study was hypothetical. Participants’ willingness to share their actual health data may be different from hypothetical willingness. Previous studies on genomic data have shown that factual willingness was actually greater than hypothetical willingness.46,47 Additionally, the COVID-19 pandemic could have increased participants’ willingness to share data.46,47
Finally, we were unable to explore the variations in motivations behind information sharing in detail. Following IRB advice, we limited the duration of the study to minimize patients’ risks related to anxiety or stress. Future studies focusing on exploring the desires and motivations behind medical record sharing preferences of patients are needed to better understand these factors.
Future work
This pilot study evaluated the C2S patient interface. Future studies are encouraged to evaluate the provider, staff, and master interfaces supported by C2S. Also, this study assessed C2S as a stand-alone tool. Additional studies are needed to evaluate the applicability of this tool when integrated into other electronic systems, such as patient portals and EHRs. Additional functionalities of the tool, including patient identification and consent segmentation mechanism, remain unexplored.
Conclusions
The study showed that patients with BHCs wanted granular control over the sharing of their sensitive medical records. When supplemented with educational materials, the C2S tool and the NCVHS data types offered data sharing choices that honor GDS desire of patients. Future work is needed to fully assess C2S in clinical environments.
Supplemental Material
Supplemental Material - My Data Choices: Pilot evaluation of patient-controlled medical record sharing technology
Supplemental Material for My Data Choices: Pilot evaluation of patient-controlled medical record sharing technology by George Karway, Julia Ivanova, Tina Kaing, Michael Todd, Darwyn Chern, Anita Murcko, Kazi Syed, Mirtha Garcia, Michael Franczak, Mary J Whitfield, and Maria A Grando in Health Informatics Journal
Footnotes
Acknowledgements
The authors would like to thank the My Data Choices Community Advisory Board for supporting this work. They want to thank Michaela Statt, Theresa Pinky Marie Guerrero, Diane Palacios, Deborah Freeman, Robert Ouitmette, Rochelle Ryan, Adalesa Meek and Byron Hoston for helping with recruitment. They would also like to thank the patients, administrators, and clinical staff at the clinical sites for their cooperation.
Author contributions
All authors made substantial contributions to manuscript revisions and approved the final version. GK, MAG, and AM contributed to the design of the study. GK, MAG, JI, TK, KS, MGR, HD, MJW, DC, MF were involved in data collection. GK, JI, and MT were involved in data analysis. MAG and AM supervised the conception, design, and revision of the manuscript. All authors also agree to be accountable for the accuracy and integrity of the work presented here.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Mental Health through My Data Choices, evaluation of effective consent strategies for patients with behavioral health conditions (R01 MH108992) grant.
Ethical approval
The Arizona State University Institutional Review Board approved this study.
Article Note
The following updates were made to this article:
• ‘SAMHA’ has been changed to ‘SAMHSA’ on page 2 of this article.
• Some occurrences of ‘Consent2Share’ has been changed to ‘C2S’.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
