Abstract
Atrial fibrillation (AF) is the most common arrhythmia. myAlgos is an mHealth disease management system consisting of physician-oriented platform and patient-oriented smartphone app. Our purpose was to assess the usability of myAlgos by physicians and patients and the effect of myAlgos on the quality of life (QoL) in patients with paroxysmal AF (PAF). Physicians rated the platform with the Post-Study System Usability Questionnaire (PSSUQ). Patients rated the app with the mHealth App Usability Questionnaire (MAUQ). The e-medicine Platform for Optimizing the Workflow in hEaRt Diseases (emPOWERD-AF) study investigated the effect of myAlgos in PAF patients randomized to full/control version. QoL was measured by the Atrial Fibrillation Effect on QualiTy-of-life (AFEQT) and 5-level EQ-5D (EQ-5D-5L) questionnaires. myAlgos got a PSSUQ score of 2.52 ± 0.36 by five physicians and a MAUQ score of 79.9% by 33 patients. In emPOWERD-AF, 80 patients were randomized 1:1 (58.1 ± 8.7 years, 66% male). The median AFEQT change at 6 months was +2.63% in full version users and −1.63% in controls (p < .001). The myAlgos platform and app were easy-to-use and improved QoL in patients.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia, affecting more than 30 million people globally. 1 Aging, male sex, and cardiovascular comorbidities are the main risk factors for AF. 2 In paroxysmal AF (PAF), AF episodes last less than 7 days before patients revert to sinus rhythm with or without medical intervention. AF in general conveys a nonhomogeneous risk of severe complications, most commonly due to thromboembolic events, which may affect patients’ health status and quality of life and in some cases prove fatal. 1 However, AF need not incite life-threatening complications to severely impact patients’ quality of life – in most patients the arrhythmia itself elicits burdensome symptoms such as palpitations, exertional dyspnea, fatigue and lightheadedness. 3
The rapid rise in hospitalizations due to AF or AF complications indicate that there is significant room for early identification of the arrhythmia and improvement in the treatment of the disease. 4 Notably, a high proportion of non-AF related hospitalizations can be observed in study populations (2196 out of 2614 hospitalizations observed in the 14171 patients in ROCKET-AF were not AF or bleeding related). This highlights the significance of enhancing structured and multidisciplinary care pathways in AF patients. Indeed, the integrated, patient-centered chronic care model put forward by Wagner et al. has been tested successfully in AF.5,6 All available evidence shows that such patient-centered approaches are powerful tools in improving outcomes in AF. 7
mHealth interventions in AF are equally promising since they enable a high degree of patient involvement in AF management and personalization depending on individual needs.8–10 The 2020 European Society of Cardiology guidelines for the management of AF acknowledged the utility of mHealth projects that enhance patient education and improve and streamline physician-patient communication. 11 The ESC also launched “myAF”, 12 a smartphone application that enhances patient education and self-engagement, while optimizing communication between physicians and patients. In terms of detecting AF, both Apple and Huawei have investigated the capability of their wearable devices to detect the arrhythmia.13,14
myAlgos combines patient care with an mHealth component. It is an integrated, cloud-based, cooperative chronic disease management system which offers clinicians, patients, and caregivers an electronic environment where automated procedures address their own different perspectives for remote monitoring, supported self-management, personalized treatment plan, and enhanced communication as well as collaboration. The system’s philosophy of operation is based on precision medicine. myAlgos features patient education, in-app medical contact via messages, medication reminders and heart rate log. Any improvement of medication adherence is expected to benefit patients, as declining adherence in anticoagulation medication in this patient group has been associated with increased risk of thromboembolic events.
15
In terms of the AF Better Care pathway,
11
the app supports patients and physicians in every step – aiding physicians optimize stroke prevention by highlighting high-risk patients, providing real-time feedback on symptom management efficacy, and in-app monitoring of risk factors for AF-related complications. Real depictions of the myAlgos platform for physicians and the myAlgos smartphone app for patients are presented in Figure 1, while a schematic representation of main functionalities is present in Figure 2. Presentation of the myAlgos web platform patient list (a) (left) and patient overview (a) (right) and the myAlgos mHealth Application (b) – main menu, pain map, medication reminder, app adherence statistics and care team messaging. Schematic representation of the myAlgos platform main features and functionality. AF: atrial fibrillation.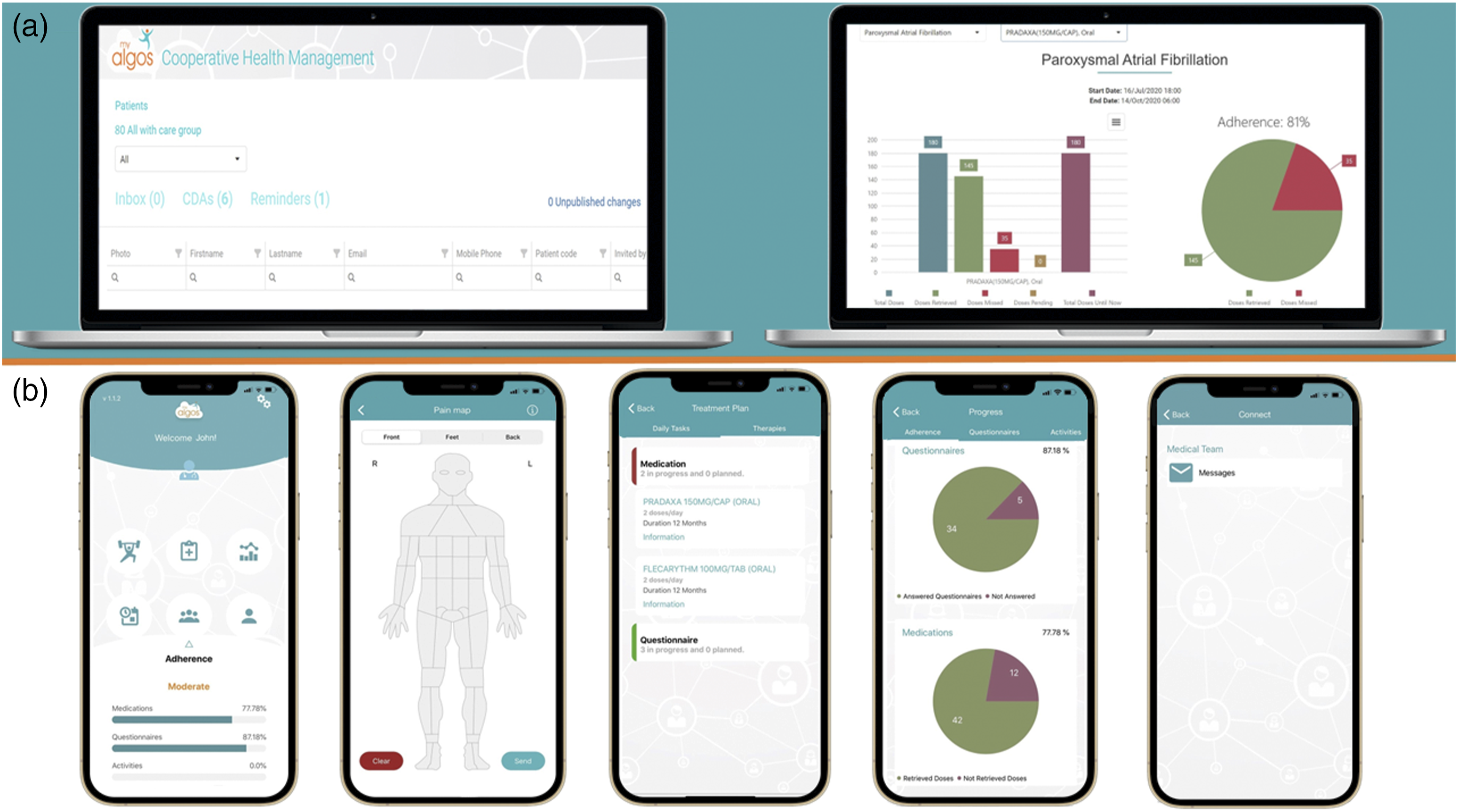
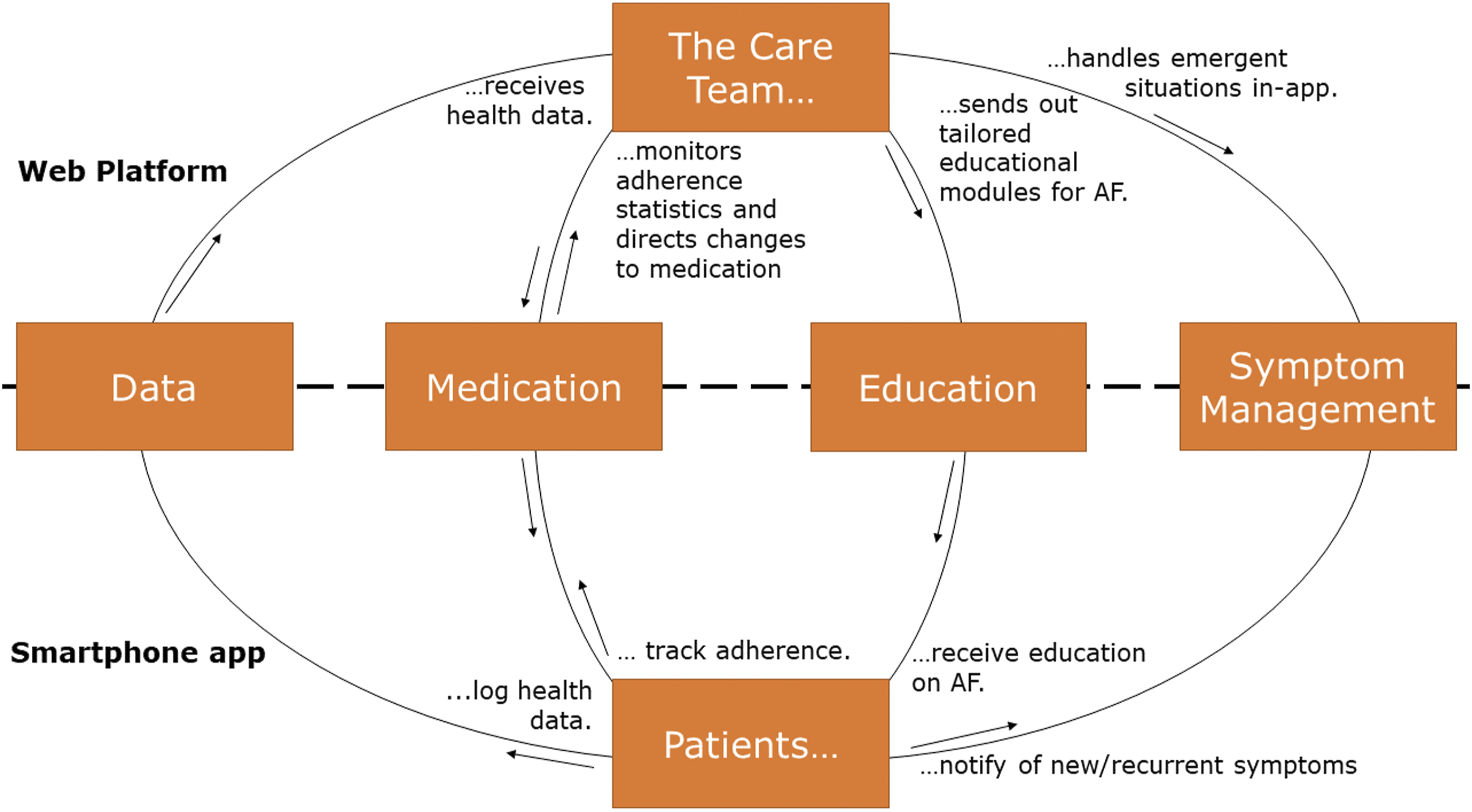
The physician-side of the platform is a website (hence often referred to as the myAlgos web platform) with a layout optimized for use via any internet browser. Each physician has unique credentials that enable access to the files and data of patients they treat. Upon accessing the web platform, physicians are first notified of high- and mid-level alerts automatically generated by the platform upon detection of abnormal patient inputs (e.g., high heart rate, palpitations, high blood pressure etc.). Regarding patient files, a thorough but AF-focused patient history is always available, as is all clinical data from the previous patient visit and an overview of the reported medication adherence. Physicians also have access to a summary and detailed log of patient inputs on the app (arterial pressure, heart rate, palpitations, anxiety) as well as any messages the patient may have sent to the management team. Most importantly, the physician is able to direct treatment changes through the app or even instruct the patient to contact emergency services, upon which the patient is immediately notified. One of the main goals of the myAlgos physician platform was to streamline the workflow of PAF patients follow-up by giving treating physicians the ability to review a patient’s file, medications, recent disease course and potential urgent situations as well as give tailored medical advice in a safe and user-friendly digital environment.
The patient-side of the myAlgos platform is a smartphone app installed in patients’ smartphones. Inside the app, the patients have a detailed list of prescribed medications, including dosage and frequency, along with an extensive patient empowerment module that contains information about atrial fibrillation and relevant medications and interventions. Most of this information has been prepared by physicians and targeted for laypeople, although it is supplemented with physician-approved articles from allied health professionals and/or medical journalists. Medication reminders are also present and serve dual purpose as tools for monitoring medication adherence, since patients are kindly notified to confirm they took their prescribed doses. Patients can also inform the care team that they are experiencing an episode of palpitations or send unstructured messages. It should be pointed out that in the interest of patient safety, users are always reminded that the app does not offer urgent/emergency health services and should not be used in an emergency situation.
Our aim was to firstly investigate the usability of the physician-centered myAlgos platform and the patient-oriented myAlgos smartphone app through patient and physician usability studies. Subsequently, we sought to measure the effect of the use of the myAlgos app by patients with PAF on their quality of life through the e-medicine Platform for Optimizing the Workflow in hEaRt Disease (emPOWERD) study.
Methods
Usability study of the myAlgos web platform
Five physicians from our cardiology department, belonging to a Greek tertiary hospital, were asked to use the myAlgos platform to monitor patients and direct treatment. The treatment plan consisted of medications typically prescribed in atrial fibrillation, self-care behavior recommendations (e.g., feeling the heart rhythm for irregularity, blood pressure measurement) and patient education material. After a tutorial session with technical personnel, users were free to use the platform.
After 4 weeks of using the platform, the participating physicians were asked to complete the Post-Study System Usability Questionnaire (PSSUQ). 16 The PSSUQ is a 16-item questionnaire that measures users’ perceived satisfaction of a system. PSSUQ scores can range from 1 to 7, lower being better. Its questions can be divided in three subdomains: system usefulness, information quality and interface quality.
Usability study of the myAlgos mHealth app
Patients visiting our department’s outpatient arrhythmia clinic (belonging to a Greek tertiary hospital) were invited to participate in the usability study. Participants downloaded the myAlgos app on their smartphone, while researchers assumed the role of the care team, transposing patients’ treatment plan to the myAlgos platform. After an in-app tutorial, users were free to use the app at will. To simulate real-life conditions, patients were discouraged from contacting study personnel and physicians outside the app during the duration of the study, although tech support was available during working hours and patient appointments at the AF clinic continued as normal. After 4 weeks of using the app, participants were asked to complete the mHealth App Usability Questionnaire (MAUQ), specifically the version for interactive mHealth apps used by patients. 17
The MAUQ was created exclusively for usability testing of mHealth apps. Its performance and consistency in assessing mHealth apps during usability testing has been validated by comparing it to the PSSUQ and the System Usability Scale (SUS). 17 The version for interactive mHealth apps used by patients consists of 21 questions divided into three categories: ease of use and satisfaction, system information arrangement, and usefulness. Each item is a Likert scale ranging from 1 to 7, higher the better.
emPOWERD-AF study design
The open, randomized, non-interventional, single-center cohort study recruited patients with PAF from June 2020 to November 2020. Patients visiting our department’s arrhythmia clinic (belonging to a Greek tertiary hospital) that fulfilled inclusion criteria were eligible to participate. Follow-up lasted 6 months. Inclusion criteria were age greater than 18 years old, thorough understanding of the Greek language, a history of PAF as well as owning and regularly operating a smartphone. Visual acuity lower than 40%, cognitive impairment defined as Montreal Cognitive Assessment test score <20, stroke within the previous 12 months and uncontrolled mental disorders comprised the study’s exclusion criteria. The study was designed and performed in accordance with the World Medical Association Declaration of Helsinki regarding ethical conduct of research involving human subjects and received approval from the institutional review board of our hospital. Written informed consent was obtained from all participants.
Patients were randomly divided into two groups using block randomization (researchers blinded to block size). The full version group received the full version of the myAlgos app, while the control group received a simplified version of the app. Patients randomized to the full version were signed on the myAlgos platform and received the full myAlgos smartphone app. The patient was encouraged to organize every aspect of self-care through the app. Physicians received all data that the patients logged in the app, including vital signs and other physiological parameters (blood pressure, heart rate, body weight, blood glucose levels, oxygen saturation) and medication adherence statistics, obtained by patients’ response rate to medication adherence notifications. They could also instruct changes to patient medication. Patients could send messages to the care team at any point, although it was made clear that the app was not meant to replace contact with emergency health services. All communication between patients and members of the care team were asynchronous and text based. The control app version consisted of a heart pulse log only, which the patients and physicians could view at any time.
In total, three visits were performed at baseline, three and 6 months later. Patients used the app throughout this 6-month period. The baseline and 6-month visits demanded physical presence of the patient, whereas the 3-month visit was performed via phone call. Basic demographic characteristics, complete medical and AF-related history were collected at the baseline visit. Quality of life (QoL) was the study’s primary outcome, assessed with the Atrial Fibrillation Effect on QualiTy-of-life (AFEQT) Questionnaire at every visit and with the 5-level EQ-5D (EQ-5D-5L) health assessment tool at the baseline and the last 6-month visit.
The EQ-5D-5L questionnaire consists of five short questions and a visual analogue scale (VAS). Each question provides a 5-point scaled answer regarding five health subdomains including mobility, self-care, usual activities, discomfort/pain, and depression/anxiety. The VAS ranges from 0 to 100 (100 being best possible health). 18 The AFEQT questionnaire quantifies AF-related QoL. AF related symptoms, daily activities, treatment concern and treatment satisfaction comprise the four sub-domains of the questionnaire, which consists of 20 items, with a 7-point Likert-type response scale. Each item and subdomain are transposed to a 0–100 scale with 100 describing the least severe condition. Domain-specific and total scores are calculated and assessed. 19 Both questionnaires have been validated for the Greek population.20,21
To estimate the optimal sample size, we performed power analysis with an objective of to adequately recognize an increase in AFEQT score of 5 points or more as significant. This has been recently defined as the minimal clinically important change in AF. 22 To his effect, AFEQT position and dispersion data were extracted from a study on European Heart Rhythm Association (EHRA) that reported AFEQT scores, 23 while serial score correlation was extracted from the AFEQT Greek validation study. 21 A Monte Carlo model simulating a 5-point increase with a paired sample correlation coefficient of 0.8 was designed to acquire a reliable estimate of the expected effect size. With an alpha of 0.05 and 80% power, the minimum sample size to reliably detect a 5-point increase in AFEQT between the full app group and the control app group with a Mann-Whitney U test 24 was calculated at 100 (i.e., 50 for each group).
Statistical analysis
Power analysis was performed in Python (using modules from the numpy library) 25 and R (using power analysis from the wmpow package). 26 Data were tested regarding their normality of distribution via the Kolmogorov–Smirnov test. Normally and not normally distributed variables are presented as Mean ± SD and Median (IQR, Interquartile range), respectively. Categorical variables’ frequency distribution was compared with the Pearson Chi-square test, while normally distributed variables with the t-test and non-normally distributed parameters with the Mann-Whitney U test. p-values lower than 0.05 were considered as statistically significant. All statistical analyses were performed using IBM SPSS Statistics 25.0 (IBM Corp, Armonk, NY).
Results
Usability study of the myAlgos web platform
The myAlgos platform received a mean overall PSSUQ score of 2.52 ± 0.36 (Figure 3(a)). The highest score was noted in the system usefulness subdomain (2.27 ± 0.7). The platform was rated at 2.35 ± 0.51 in regard to interface quality and at 2.8 ± 0.4 for information quality. When it came to user satisfaction, the platform received a mean 75% satisfaction score by physicians. Bar charts of (a) PSSUQ overall score and subdomains in the myAlgos web platform usability study and (b) of MAUQ overall score and subdomains in the myAlgos smartphone app usability study. PSSUQ: Post-System Survey Usability Questionnaire; MAUQ: mHealth App Usability Questionnaire. *The presented results are inverted for ease of comprehension.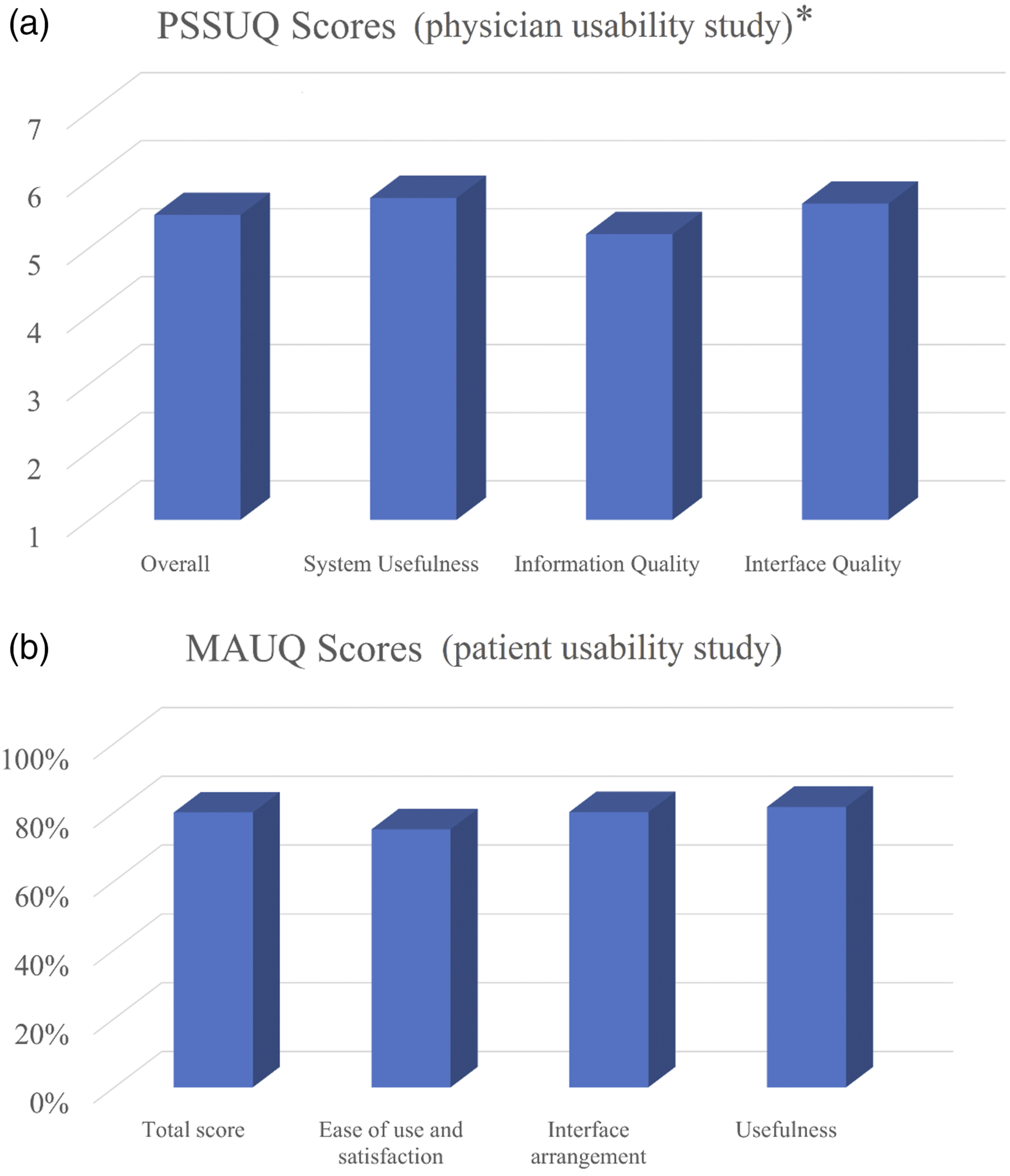
Usability study of the myAlgos mHealth app
Thirty-three patients aged 57.1 ± 36 years (63% male) participated in the usability study. They gave the myAlgos app an average overall MAUQ score of 117 ± 17.7 out of a perfect 147 (79.9%, Figure 3(b)). In the subdomain of ease of use and satisfaction, the app received an average score of 16 ± 5.2 out of 21 (75% of perfect score). Regarding the arrangement of the app’s interface, users rated myAlgos at 56 ± 8.1 out of 70 (80%). Users rated the app’s predicted usefulness in the self-management of AF at 45 ± 6.3 out of 56 (81.5%). The aforementioned scores are graphically presented in Figure 3(b).
The emPOWERD-AF study
Patient demographics at baseline.
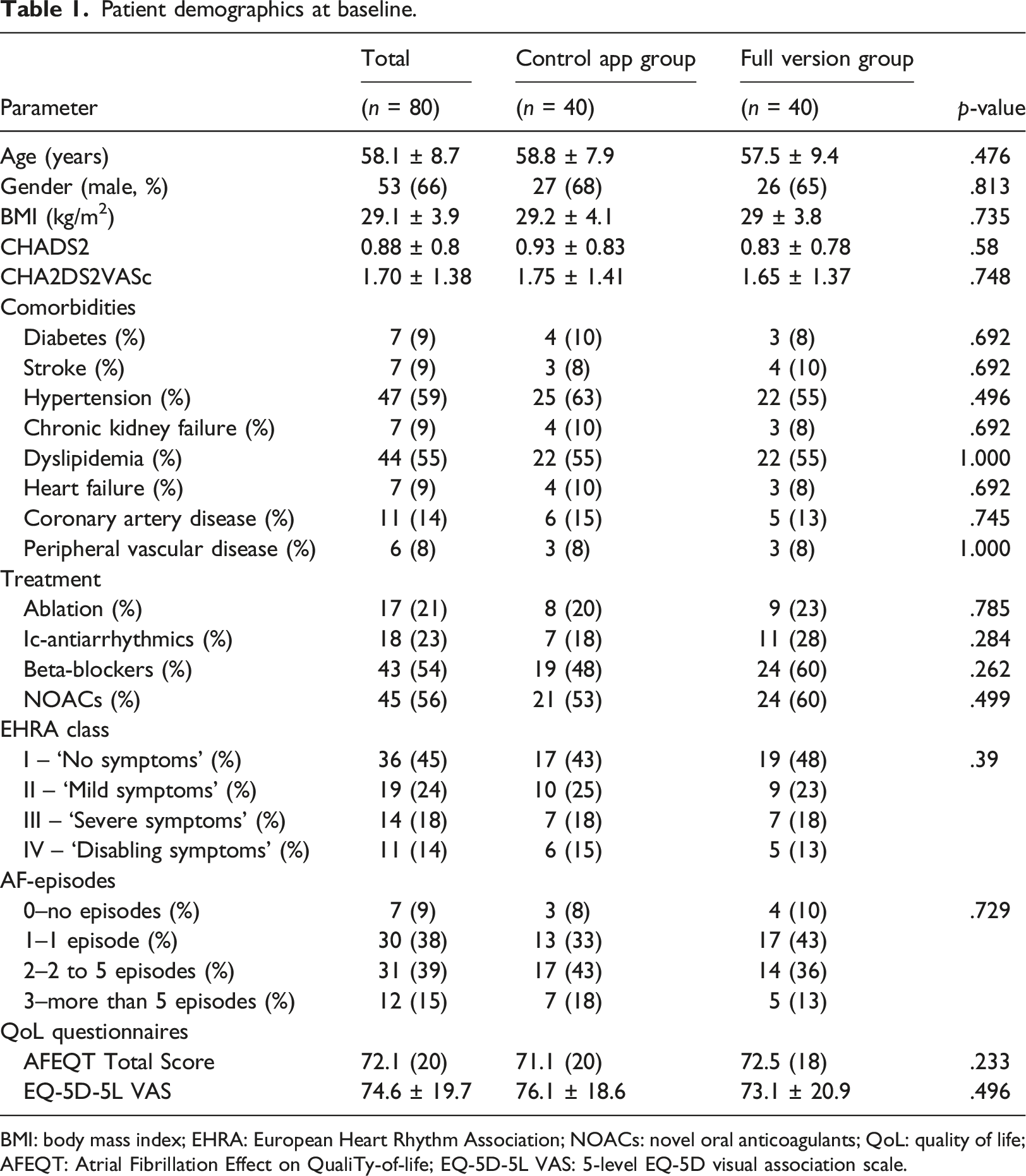
BMI: body mass index; EHRA: European Heart Rhythm Association; NOACs: novel oral anticoagulants; QoL: quality of life; AFEQT: Atrial Fibrillation Effect on QualiTy-of-life; EQ-5D-5L VAS: 5-level EQ-5D visual association scale.
Over a follow-up of 6 months, no major cardiovascular events or deaths occurred in the study population. The control and full app version group experienced a similar number of AF-related hospitalizations (2 and 3 respectively, p = .644) Patients experienced a median of 1 (IQR = 2) paroxysmal AF episode over the follow-up period. The median AFEQT score increase was 2.63
5
in the full version group and −1.63
4
in the control group (p < .001, Figure 4). Regarding AFEQT sub-domains, the difference between groups was highest in treatment satisfaction, which had a median increase of 7%
13
in the full version group and a median decrease of 3%
5
in the control group (p < .001), followed by the treatment concern subdomain, which saw a median 5.5%
11
increase in the full version group and simultaneous 1%
7
drop in the control group over the follow-up period (p < .001). The symptom burden and daily activities subdomains remained relatively stable in both groups (p = .46 and p = .118 for between-groups change, respectively). At the same time, mean EQ-5D-5L VAS results remained stable in the control group (+0.23 ± 7.4%) while exhibiting a minor increase in the full version group (+3.5 ± 9%, between-groups change p = .078). Bar chart of median AFEQT total scores for the control and full version group during follow-up. Note that the difference between medians (which can be inferred from this figure) rarely equals to the median difference in paired data (which is presented in-text).
32
AFEQT: Atrial Fibrillation Effect on QualiTy-of-life.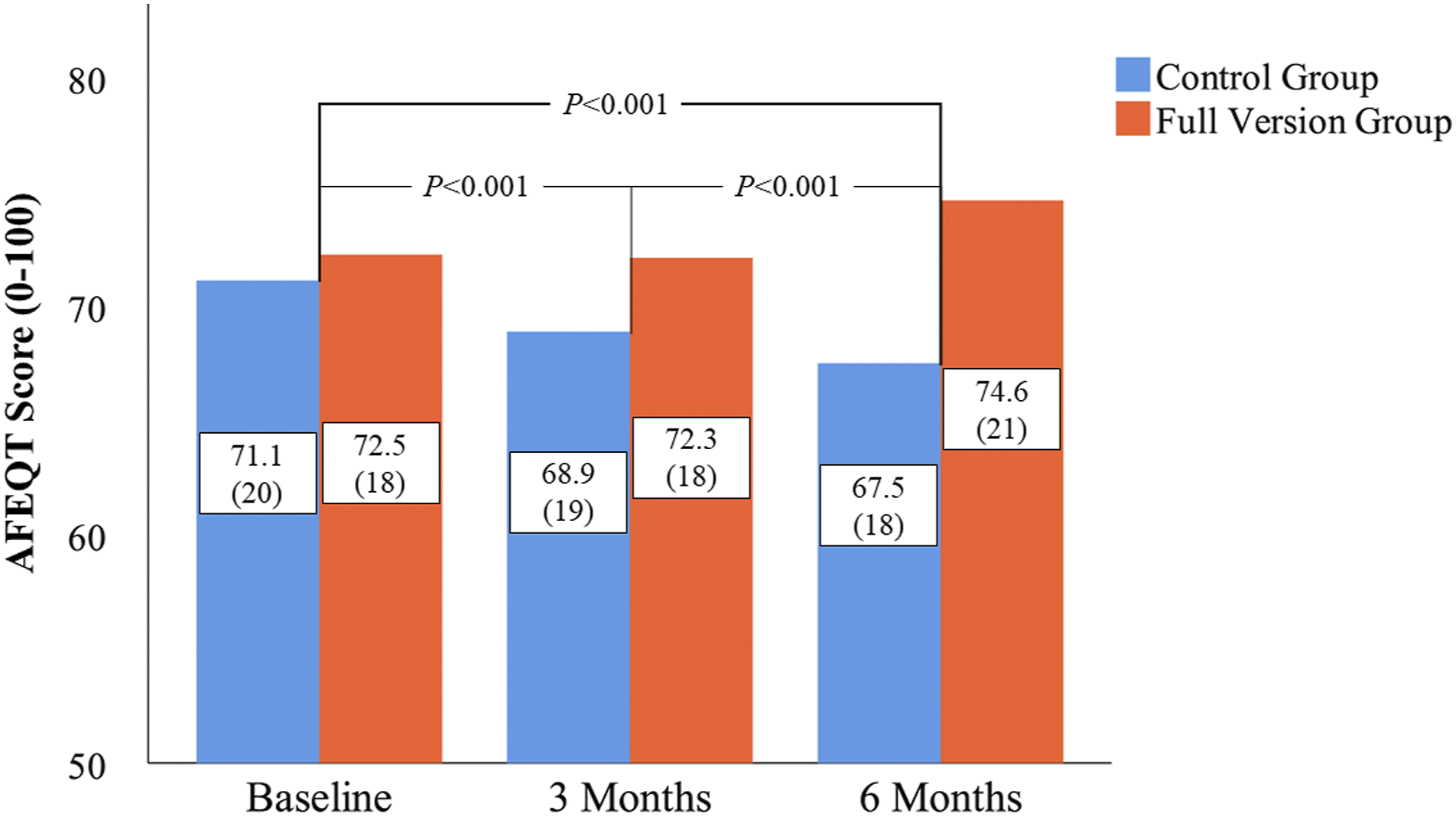
Overall, 16 patients (40%) in the full version group were classified as responders, having experienced an increase in AFEQT score of 5 points or more, recently defined as the clinically important change in AF. 22 Responders did not significantly differ with non-responders in age nor sexual distribution (p = .405 and p = .599 respectively). In fact, among all recorded variables, responders differed significantly in that they interacted more with the app (73.9 ± 14.3% vs 40.5 ± 26.8% for non-responders, p < .001) and that they handled all their AF episodes via the app, using the pill-in-the pocket treatment approach (100% vs 60% in non-responders, p = .045). Responders were also receiving more types of medication (2 vs 4, p = .044).
Usage statistics were available for patients that used the full version of the app through app analytics coded in-app. Overall, patients fulfilled 53.9 ± 28% of all possible self-care interactions prescribed to them by physicians over a 6-month period. Compliance with the app decreased in the first 3 months before eventually stabilizing (Figure 5). Interestingly, patients using the full version used myAlgos to notify medical personnel of AF episodes in 85% of all episodes (such a feature was unavailable in the control group). App adherence (defined as percentage of actual over possible interactions per time period) for responders and non-responders.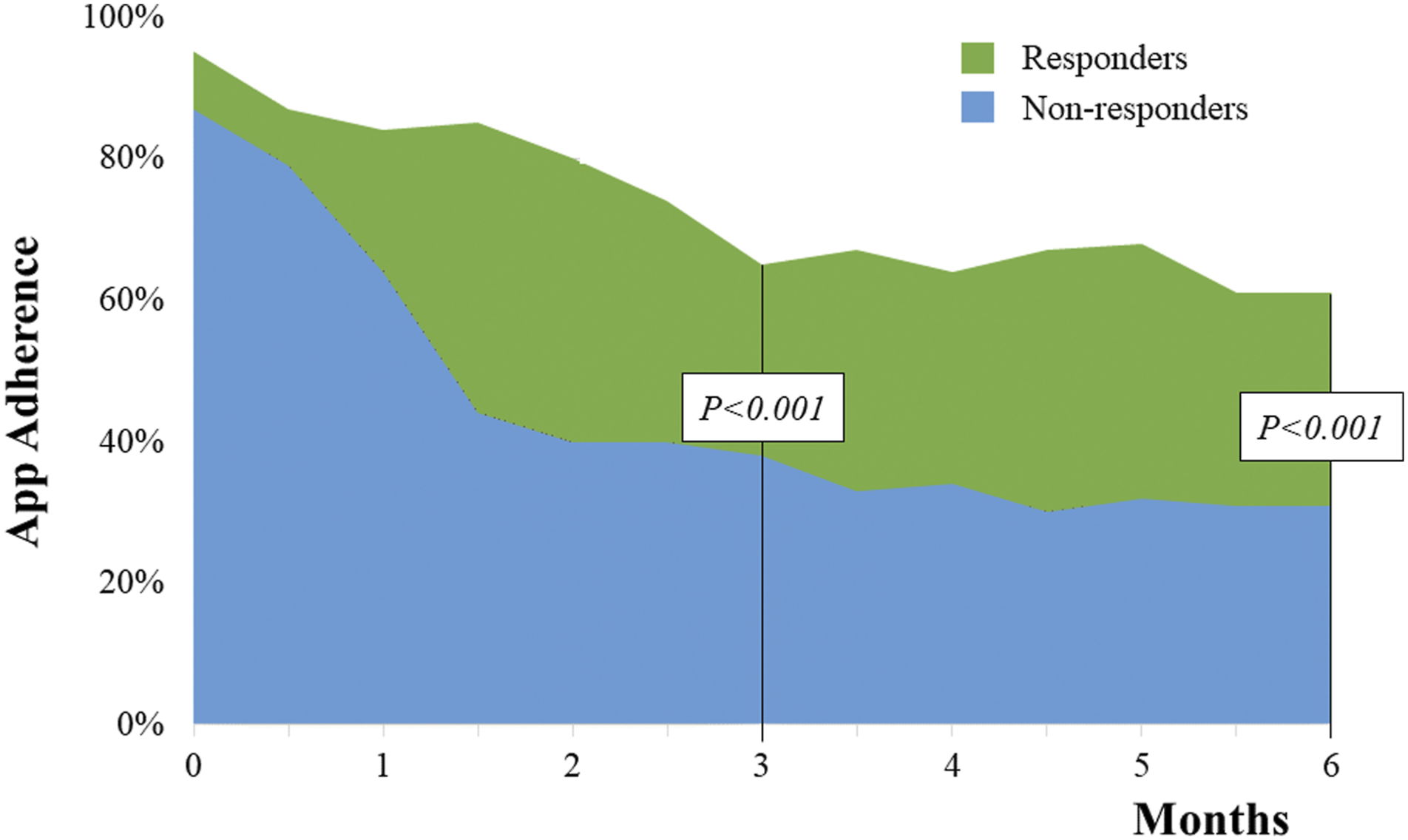
Discussion
The main finding of the myAlgos physician-oriented web platform and patient-oriented smartphone app usability studies was that physicians and patients alike found them useful and easy to use. The platform’s interface assisted physicians in tracking their patients’ medication, heart rhythm and blood pressure, while patient-testers had little difficult in navigating the menus necessary to fully access the app’s features.
Regarding the emPOWERD-AF study, the features of the app’s full version appear to have contributed significantly to conserving and even improving patients’ quality of life. Treatment-related QoL subdomains displayed the most prominent increase in the full version group, likely driven by the myAlgos’ medication tracking and ease of contact with the care team.
Indeed, 40% of patients using the full app version experienced a clinically significant increase in quality of life over the follow-up period. Based on app usage statistics, these patients appear to have made more frequent app use, and interestingly, handled all their PAF episodes through the app in the 6-month period of the emPOWERD-AF study. This fact indicates that although the myAlgos platform contains features that patients benefited from, helping them stay committed to their digital treatment plan is the primary challenge. It is probably no coincidence that the app deployed in the mAFA-II trial long-term extension, one of very few mHealth interventions in AF to achieve a hard composite endpoint (in this case lowering stroke, death and hospitalization risk), boasted a 91.7% adherence. 27 Other self-care apps in cardiovascular disorders report similar app adherence rates to our 53.9% over a 6-month period, 28 which shows that maximizing patient compliance remains an industry-wide concern.
Compared to apps like myAF, which was developed by the ESC, 12 myAlgos has the main advantage of serving as a platform for telemedicine in addition to providing features such as medication reminders and educational articles about atrial fibrillation. Such innovations will be necessary as the state of the art regarding mHealth apps for AF patients evolves.
The COVID-19 pandemic may have affected the study’s results in two further ways. Firstly, a trend towards a decrease in quality of life was detected in the control version group, one that was averted in the full version group. This could be attributed to AF patients feeling a particular need to perform social distancing due to the disease, as well as the attested fact that care quality for non-COVID diseases diminished significantly during peak months of the pandemic. 29 Secondly, relatively few AF hospitalizations were observed in both treatment arms, suggesting that patients – and physicians – may have opted for a more conservative treatment approach to minimize patient exposure to the virus.
Regarding COVID-19 in general, this unprecedented crisis may accelerate the use of telehealth platforms such as myAlgos. In response to the pandemic, specialist societies now encourage telehealth provision in treating arrhythmias whenever possible. 30 TeleCheck-AF, one of the largest integrated telehealth projects in AF, has the explicit purpose of enabling teleconsultations during the COVID-19 pandemic. 31
Our study had some limitations. First of all, emPOWERD-AF was a single-center, open-label study, which included patients that were already smartphone users and relatively free of visual/cognitive impairment. Thus, larger, multi-center studies with more expansive inclusion criteria will be needed to definitively assess the effect of mHealth apps on the quality of life of patients with PAF. The relatively young mean age of our sample indicates that our findings cannot be necessarily extrapolated to older AF patients, who may struggle with using mHealth applications. Additionally, the relatively low proportion of patients receiving NOAC therapy could suggest that patients in real-life AF cohorts may deal with more complex pharmacotherapy, with unsure effects of the efficacy of mHealth applications. The proportion of asymptomatic patients with a priori high QoL could have attenuated the beneficial effect of the myAlgos platform on patients’ QoL. Although the study was marginally underpowered to detect an increase in AFEQT-measured QoL, a significant result was nonetheless observed. This may be in part due to the smaller within-groups variation observed in the emPOWERD-AF study. The smaller variation is likely attributable to the fact that the relatively stringent inclusion led to an arguably more homogeneous sample (all had PAF, smartphone owners, no visual/cognitive impairment etc.). Finally, there was no patient group that received no intervention except for optimal treatment; this decision was made in order to eliminate any potential placebo effect derived by factors related to app installation and use.
Conclusions
The myAlgos platform was rated highly by physicians and patients alike regarding ease-of-use and presentation of available information. The use of the myAlgos app by patients with PAF significantly increased their disease-related quality of life, which was especially pronounced in patients that displayed adherence to the app’s integrative treatment approach. As such, a potential future objective lies in motivating patients to continue mHealth app use over sustained time periods. Furthermore, prolonged investigation of a larger population using the myAlgos app could yield further insights as to its effect on hard endpoints such as hospitalization risk, thromboembolic events, and mortality.
Footnotes
Acknowledgements
We would like to thank all patients that were a constant source of inspiration, support, and patience during the app’s usability testing and the emPOWERD-AF study, especially in the setting of the COVID-19 pandemic.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval protocol number: 820/04-11-2019
