Abstract
Facility based retrospective study was conducted in three regions in southern Ethiopia to assess quality of medical records. A total of 2,145 medical records were reviewed from 73 public health facilities. Minimum standards of medical records were considered to assess completeness and legibility of records. The completeness of medical records were judged systematically according to national HMIS formats. From total of 2,145 medical cards reviewed, only 394 (18.4%) records had all complete and readable data. Gaps observed include 29.0% missed at least one of identification data, 14.3% lack chief compliant, 20.1% lack diagnosis, 12.5% lack medication and 60.3% records had no date and/or signature. Moreover, 9.5% cards had at least one non-readable component. Records at health centers were 56.8% less likely to be quality record as compared to records in hospitals. Even though completeness of every single record is must, only less than one fifth of records met quality of national medical record standard. Ministry of health should consider rules and regulation to maintain data quality and switching to electronic record, and finally progress in data quality should be monitored routinely.
What is already known?
• In most low-income countries like sub-Saharan Africa, medical records are not accessible or lack significant information required for quality healthcare.
What does this paper add?
• This study was first to present quality of medical records in southern Ethiopia considering multiple zones.
Background
Health system generates huge volumes of data every day that may cost up to 30% of the total health budget. 1 Data delivered through health management information system (HMIS) come from service delivery and administrative records kept as part of routine transactions at health facilities and management offices. 2
Medical record is “a collection of facts about a patient’s health history, including past and present illness(es) and treatment(s) written up by the health care professional treating the patient.” 3 The goals of recording information in medical records are to support the delivery of good care, clinical decision-making, communication between healthcare workers, continuity of care, scientific research, quality assurance and transparency of the delivered care. 4 It is also important for measuring and improving the quality and coverage of health services and policy directions and promote equity, to detect and control emerging and endemic health problems and for empowering individuals and communities with timely and understandable information.3,5–7
To be useful, data in a medical registry must be of good quality, meaning it should be complete, legible, reliable, accessible, timely, and so on. 3 However, in practice, quite frequently incorrect patients data are registered or data items can be inaccurately recorded or not recorded at all. 8 And in most low-income countries like sub-Saharan Africa, such information is either lacking or not accessible and overall quality of medical records is poor.9–12
Limitations related to data quality also include missing values, bias, measurement error, and human errors in data entry and computation. 13 Medical records are of no value to medical science or health care management if they are not accurate, reliable and accessible. For these reasons, data utilized must be monitored for its quality. 3
Ethiopia, including the study regions, has been implementing HMIS at all levels of health system and standardized indicators, data collection and reporting forms, and procedures.14,15 All facility level recordings in the study settings are paper based. History and cares given are written in individual files and are summarized in registers at service delivery points. Each service delivery point then tally services given and prepare reports and overall facility reports are compiled by HMIS focal persons. Implementation of these activities including format duplication have been supported by partners.
To have confidence in the health data presented and to ensure proper implementation of HMIS, the routine data quality should be monitored qualitatively and/or quantitatively.3,13 In addition to these facts, there was no regional level study addressing medical record quality in public health facilities. So, this study was designed to address this information gap and support the system improvement.
Methods
Facility based retrospective study was conducted done in April 2017 in Southern Nations Nationalities and People’s Region (SNNPR), Ethiopia. SNNPR was the third largest administrative region of Ethiopia representing about 20% of the country’s population. From 2007 census, the regional population is estimated to be 19,170,005 in 2016. It was the most diverse region in the country in terms of language, culture and ethnic background. Administratively, the region was divided into 14 zones, 1 city administration and 4 special woredas. Woreda, equivalent to district, is administrative structure in study setting with approximate total population of 100,000 and is sub-divided in to smallest administrative structures called kebele. In 2016, there were 57 hospitals of all types and 736 health centers reporting data through eHMIS. Currently, the region is administratively divided in to three regions: Sidama, Southwest and SNNP regions.
A total of 73 institutions (65 health centers and eight hospitals) were included in the study. The number of facilities were limited by considering existing logistics and time.16,17
According to the World Health Organization (WHO) standard for developing countries, all medical records should be assembled within 24 h of patient discharge with 100% accuracy. 3
The same guideline states that a qualitative analysis of medical records should be performed on a random sample of 30 patient records or 5% of records of discharged patients. 3 Considering these assumptions, we included the first 30 client cards seen (for any reason) and discharged the day before data collection. The day before data collection day was considered to ensure completeness of records after 24 h of clients discharge. For facilities with low patient flow (below 30 patients discharge), we included 2 days data in the review.
In the context of a medical records, data quality can be defined as “the totality of features and characteristics of a data set that bear on its ability to satisfy the needs that result from the intended use of the data.” 8 As dimensions for measuring data quality, completeness and legibility were considered. Completeness indicates that all required data are present in records while legibility shows data are readable for users. 3
Completeness of patient care data on cards were evaluated considering minimum national medical record standards. These were data on patient identification (name, sex, age and address), presentation (reason for the visit), main diagnosis (or service), treatment/s (or service) and date and signature. The completeness of medical records were judged systematically according to national recording formats.
BSc holder nurses or health officers who have experience in HMIS were used to review documents. Considering the study context where majority of cares, especially at primary healthcare levels, were provided and/or supervised by BSc holder nurses or health officers, records were coded “legible” if these people could read data. Records of each variables were categorized to “clearly written,” “not written,” and “not readable/legible.” Records with “not applicable” fields based on recording formats were excluded for specific variable. Records with complete information about these all variables based on recording formats were considered “complete” and missing of any of variables was considered “incomplete.”
Training of data collectors (with pre-test) was given for 3 days. Prior experience on HMIS and data collection was also considered in selection of data collectors and supervisors. Daily supervision was done by principals and supervisors. All collected data were examined for the completeness of data by principal investigators.
Data was entered, cleaned and analyzed using SPSS for Windows version 20. Descriptive statistics was used to describe data quality. Binary logistic regression was used to test quality of records between levels of health facilities.
Results
Sixty five health centers and eight hospitals, a total of 73 public health facilities were included in the study. From these facilities, 2,145 medical records were reviewed. The minimum data that medical records should comprise were assessed and presented as follows.
Patient identification data
Quality of medical records in southern Ethiopia, 2017.

*Date and signature was not applicable for 20 cards (of family planning services).
Chief complaint (reason for visit)
Chief complaint like clinical signs and symptoms for those seeking medical care, diagnostic service and/or services (in case of maternal service utilization like antenatal care, family planning etc.) were reviewed and more than eight out of ten (81.3%) had clearly written data while 14.3% and 4.4% lack the data and were not legible respectively (Table 1).
Diagnosis
Diagnosis, a condition to be chiefly responsible for occasioning the patient to the health facility for care, was assessed considering classifying disease condition and/or services provided. In case of maternal health service utilization, main services like antenatal care, family planning etc. were accepted.
In this way, about three fourths (76.4%) records had clearly written diagnosis. In addition, one fifth (20.1%) lacked this data while 3.5% were not legible (Table 1).
Treatment (medication)
To assess records of treatment or medication, curative medication/s and/or other medical supplies like contraceptive methods to address reasons for visit and diagnosis were reviewed. Accordingly, more than eight out of ten (84.7%) records had clearly written records while 12.5% and 2.8% had no records and not readable respectively.
Date and signature
In addition to supporting continuity of care, medical records are used to maintain accountability among healthcare workers, which is usually addressed by signature (including name of health worker) and date. From 2,125 records with space for signing and writing date, only less than four out of 10 (38.2%) had clearly written data.
Summary
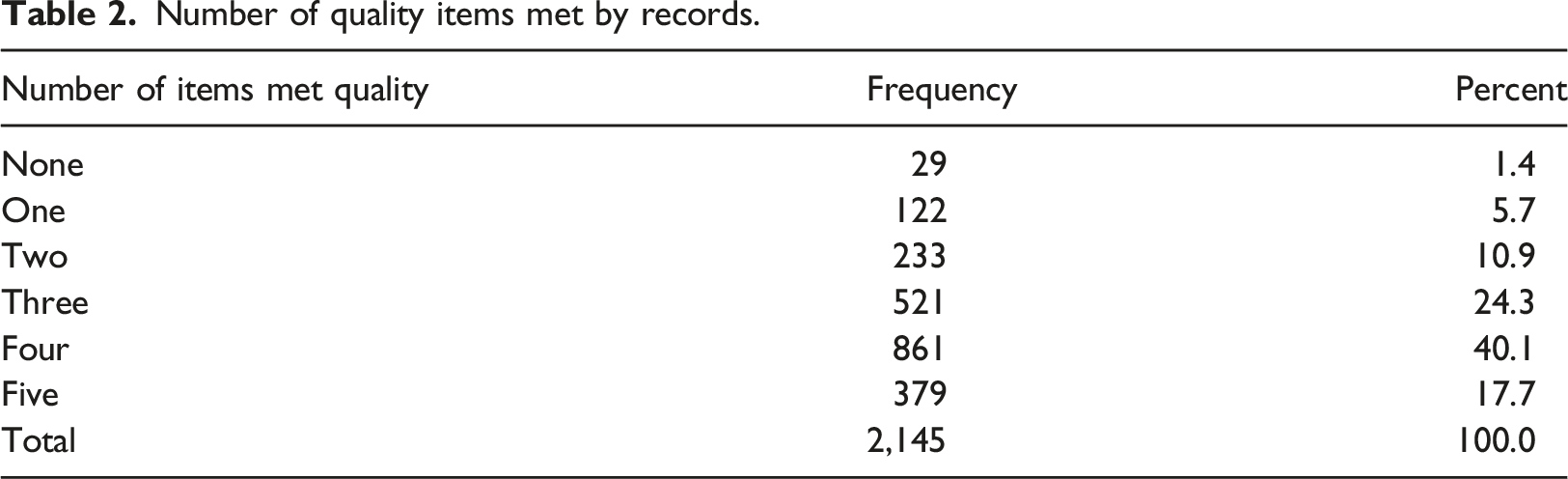
Number of quality items met by records.
Cross tabulation of facility type and quality of medical record.
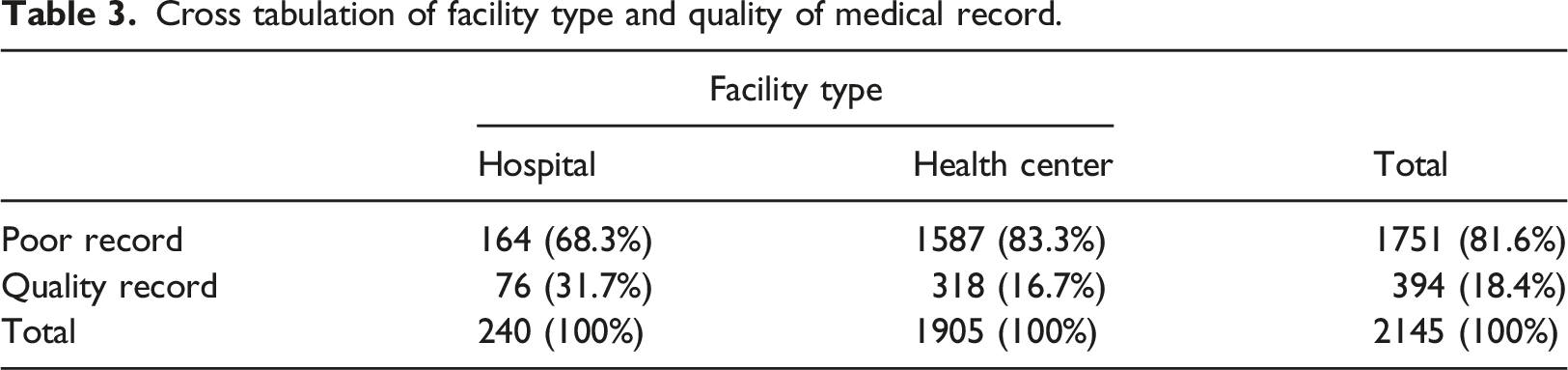
The binary logistic regression showed that records at health centers were 56.8% less likely to be quality record as compared to records in hospitals with 95% CI crude odds ratio of 41.8–67.9%, p < 0.001.
Discussion
This study was conducted with the objective of assessing medical records data quality in terms of completeness and legibility. Key findings show that there are notable gaps in completeness and legibility of patient profile, complaint, diagnosis, treatment and date and signature.
As a written collection of information about patient’s health and treatment, medical records are used essentially for the present and continuing care of the patient. In addition, medical records are used in the management and planning of health care facilities and services, for medical research and the production of health care statistics. 18 The records must be compiled in a timely manner and contain sufficient data to identify the patient, support the diagnosis or reasons for the healthcare encounter, justify the treatment, and accurately document the results. They are the visible evidence of the hospital’s clinical activities and accomplishments.
In this study, however, only 69.0% had clearly written demographic data while about one-third (31%) of cards reviewed had no or unreadable demographic which can challenge management and follow-up of patients. In addition, 84.7% medical records had clearly written treatment while the rest 15.3% had no or unreadable records. Moreover, only 38.2% of cards with space for signs and signature had sign and signature and only less than one fifth (18.4%) records had all five items assessed indicating significant proportions of medical cards lack important data. Such inadequacy of clinical documentation was also reported by study done in Nigeria. 19 Inadequate record can challenge provision of healthcare and also can challenge detection of adverse effects. 4 It has been reported that poor quality of the information in patient records may be a cause or a consequence of poor quality of care. Clinicians recording more data are supposed to expose themselves to detection of adverse events. 4
Records at health centers were 56.8% less likely to be quality record as compared to records in hospitals (95% CI: 41.8–67.9%), p < 0.001. A study from the same country also reported that hospitals are more likely to report good data as compared to health centers. 20 This could be due to the setup of facilities. Hospitals are more equipped and staffed than health centers.
Despite the computer revolution and data digitalization, clinical records especially in study setting continue to be handwritten and difficult to read. A clinician writing the record can understand what has been written, but when another clinician at the same or referral facility gets it, difficulties can arise and may lead to prescription errors. In addition to challenging provision of healthcare, poor medical records challenges auditors, researchers, and other clinicians. So, the legibility dimension of records quality should also be monitored to ensure that data is useable.
Although we reviewed quality of medical records for completeness and legibility in multiple sites, limitations of this study include that other dimensions of medical records quality like security (confidentiality) and archiving were not addressed. In addition, though we used experts in HMIS as data collector, interpersonal variation in reading might have affected legibility. Moreover, significant factors that are related to medical record completion like staffing, training of staff; perceived value of medical record etc. were not addressed. Finally, we used structured checklist with options described in results and data were categorized during data collection at field levels, and detail information on some of items were missed. For example, which of demographic data (name, sex, age and/or address) was missing was not addressed.
Conclusions
Even though completeness of every single record is must, only less than one fifth of records met quality of national medical record standard indicating the need to strengthen data quality assessment. Ministry of health should consider rules and regulation to maintain data quality and switching to electronic record with ‘must enter’ security patterns not to skip key components. Progress in data quality should be monitored routinely against the data quality parameters quantitatively and qualitatively.
Footnotes
Acknowledgements
We would like to thank SNNPR health bureau for funding the study. We would also like to thank USAID Transform PHCU and L10 K 2020 for providing training on operational research as part of system support to enhanced program learning to impact policy and elimination of preventable child and maternal deaths, and also thank Agegnehu Gebru, Amare Assesfa and Firew Solomon for their continuous follow-up and encouragement to analyze and present secondary data for evidence-based decision-making.
Author contributions
ME managed data, prepared report and manuscript. AK and AA facilitated funding and managed overall study. FL validated result. All authors contributed to conceptualization, designing, data collection, read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the SNNPR Regional Health Bureau.
Data availability statement
All relevant data are within the manuscript. The datasets analyzed during the current study available from the corresponding author on reasonable request.
Ethics approval
Ethical approval was obtained from Ethical Review Committee of SNNPR Regional Health Bureau (Ref. £’6-19-2762). After getting ethical approval, official letter was written to study facilities. The information obtained kept confidential.
