Abstract
Mobile health (mHealth)—hand-held technologies to address health priorities—has significant potential to answer the growing need for patient chronic illness self-care interventions. Previous reviews examined mHealth effect on patient outcomes. None have a detailed examination and mapping of specific technology features to targeted health outcomes. Examine recent chronic illness mHealth self-care interventions; map the study descriptors, mHealth technology features, and study outcomes. (1) Information extracted from PubMed, CINAHL, and Web of Science databases for clinical outcomes studies published 2010–January 2020; and (2) realist synthesis techniques for within and across case analysis. From 652 records, 32 studies were examined. Median study duration was 19.5 weeks. Median sample size was 62 participants. About 47% of interventions used solely patient input versus digital input; 50% sent tailored messages versus generic messages; 22% augmented the intervention with human interaction. Studies with positive clinical outcomes had higher use of digital input. Software descriptions were lacking. Most studies built interventions: only two incorporated target audience participation in development. We recommend researchers provide sufficient system description detail. Future research includes: data input characteristics; impact of augmentation with human interaction on outcomes; and development decisions.
Keywords
Introduction
Chronic disease, with its high prevalence, increased mortality, and associated health costs, is a public health burden internationally. 1 For example, in the United States, the chronic illness cost burden was estimated at 78% of total health care spending. 2 People living with chronic conditions have economic challenges including medication related costs and reduced ability to work. 3 A technology category with significant potential to address this need is mobile health (mHealth)—application of mobile (e.g. hand-held) technologies to address health priorities. 4 Accordingly, mHealth studies have been published, among which are behavioral interventions intended to improve patient chronic illness management.5–7 This management encompasses tasks that persons need to take on so as to live well with chronic condition(s) 8 such as following a specific diet, daily monitoring for physiologic changes (e.g. blood glucose), and response to those changes.
Lately, reviews have examined mHealth chronic illness self-care interventions’ impact on clinical outcomes,6,7 whereas previous reviews were limited to reports on mHealth feasibility, acceptance, and usability5,9 and impact on behavior change9,10 for specific chronic conditions. The two recent articles focused on clinical outcomes are relatively small systematic reviews (in regard to number of studies) of mHealth randomized control trials (RCT) published between 2005 and 2016. Lee et al. included multiple common chronic diseases. Ten of 12 studies reported statistically significant improvement in health and clinical outcomes. Identified mHealth functionality characteristics were data input devices and automated text reminders. 6 Whitehead et al. focused on apps for four chronic conditions (i.e. diabetes types 1 and 2, cardiovascular diseases, chronic lung diseases). Six of nine studies reported statistically significant improvement in clinical outcomes. Identified mHealth functionality were apps which tended to include data input, data transmission, and sending text messages; and receiving automated text reminders. 7
However, mHealth interventions targeting self-care of chronic conditions have not had a detailed examination and mapping of the specific mHealth technology features (e.g. data input, messaging) to the health outcomes they target. This mapping would enable investigation of whether specific technology characteristics result in better patient outcomes.
Objectives
This review updates and expands previous reviews to examine mHealth features, viewed through the scope of self-care. Our objective was to: (1) identify empiric (i.e. observational or experimental) mHealth interventions studies explicitly designed for self-care of community-living adults with one or more chronic conditions which assessed the impact on patient outcomes, and among the retained studies (2) categorize and map the study descriptors, mHealth technology features, and study outcomes. We targeted studies sufficiently developed to include clinical outcomes, in contrast to evaluation limited to process outcomes such as feasibility or usability.
Methods
The authors were a health informatician with expertise in home health care and mHealth (PS), a nurse expert in geriatric patient self-care (HB), and a nurse with expertise in chronic conditions and software development for clinician chronic condition management (ES).
Study design
An integrative review methodology was used to synthesize the research results using a narrative analysis 11 for literature published from 2010 to January 20, 2020. The methodology was to: (1) identify applicable studies; (2) select for review studies that met criteria; (3) organize data from the retained studies; and (4) summarize and report the results. This paper is one of two papers addressing the topic area mHealth in chronic illness management as part of a larger examination of the mHealth human/technology interface. One paper addresses the human aspect or self-care behavior issues in mHealth studies. 12 This second paper is designed to address (1) the technology aspect or mHealth intervention technology features themselves (i.e. patient or digital device input, general or tailored patient feedback, software features) and (2) their relation to health outcomes and mHealth usage and satisfaction outcomes. Dissemination via this two-pronged approach was designed to support the advancement of the science.
Identify studies
Two authors (PS, ES) developed a search strategy with the assistance of a research librarian (see Acknowledgments). The authors identified a model article, 5 which was a scoping review of studies focused on the design, development, and evaluation of self-care mHealth for older adults with chronic conditions living at home. From this article the authors gleaned keywords which were used in a PubMed search. This search returned mostly literature reviews, the reference lists of which were hand searched for studies.
Referring to the model article, the authors consulted the research librarian to develop a definitive list of key keywords (i.e. MeSH terms and text words which would also search in the MeSH field) and subject headings in order to capture a comprehensive list of potential sources. They identified and combined keywords to address four research question components: (1) mobile or electronic devices, (2) technology-based health care delivery, (3) chronic condition, and (4) an adult population. The research librarian structured and ran a search query using Boolean operators for each database of peer-reviewed scientific journals searched: PubMed, CINAHL, Web of Science. These databases were available at the librarian’s institution and chosen for their salience to finding mHealth intervention studies. Delimiters in the query were that the articles were published in English, after 2010. Literature reviews related to patient-facing (e.g. for use by patients) mHealth for chronic conditions were also reviewed to retrieve studies to be considered for inclusion.
Selection for review studies that meet criteria
The two authors (PS, ES) established inclusion criteria for article retention: observational or experimental research studies explicitly focused on self-care and with clinical outcomes, which included mHealth technologies designed for use by community-residing adults living with at least one physiological chronic condition. The mHealth technology could be developed by the researchers, and/or publicly available.
They performed the article selection from each database sequentially, as follows. They independently identified articles for review by searching titles and abstracts and resolving conflicts through consensus. Exclusion criteria were: (1) solely mental health or behavioral health conditions; (2) children (younger than 18 years); (3) study designs which lacked clinical outcomes such as feasibility, pilot studies, protocols, software description; (4) mHealth designed solely for provider use; (5) technologies out of scope: telehealth, social networking, telephonic and telecounseling, telerehabilitation; (6) non-chronic conditions such as rehabilitation (e.g. stroke; cancer), risk factors (e.g. smoking), or health promotion; and (7) absence of clinical outcome.
Articles that met inclusion and exclusion criteria through abstract review were independently reviewed in full by two reviewers (PS, ES). The reviewers held regular virtual meetings to discuss their decisions regarding inclusion/exclusion criteria of articles. A result of these meetings was the revision of both inclusion and exclusion criteria as the search progressed so as to better address the research question. The adjustments were to include only physiological conditions (e.g. exclude behavioral conditions), include studies with adults of any age (e.g. not limited to older adults); include studies of mHealth sufficiently developed so as to be studies in an observational or experimental trial with measurable clinical outcomes (e.g. exclude studies limited to user acceptance); and exclude studies which did not electronically generate messages to patients (instead only people such as clinicians or coaches sent messages).
Organize, summarize, and report the review results
The reviewers (PS, HB, ES) developed a data charting form, implemented as an Excel spreadsheet, to record selected data from the retained articles. The spreadsheet was revised as the review progressed. 12 The following information from articles identified through the review process described above was entered into the spreadsheet:
Publication information (Author, title, journal, date)
Condition(s)
Sample size
Study duration
Research setting country
Participant age range
mHealth user interface characteristics (e.g. patient input, electronic device input, personal data used, patient feedback—general or tailored messages)
Human interaction to augment technology
Software characteristics: algorithm; gamification
Outcome type (clinical, satisfaction, etc.)
Outcome measures
Main study findings
The two reviewers (PS, ES) abstracted data from the retained articles using this standardized form by charting half the articles and cross-checking the other author’s charted articles. They discussed charting differences until disagreements were resolved. A third author, HB, reviewed the resulting form for completeness and confirmation of technology elements.
The reviewers (PS, ES) synthesized the data using realist synthesis techniques13,14: Analyzing data elements (i.e. technology and patient outcomes) across studies to determine the nature and relationships among and between these data elements to deduce higher order abstractions. This included examining interface characteristics, software characteristics, and human augmentation in light of study outcomes; and investigating patterns across studies. Study outcomes were classified into three types: clinical; performance of care processes (i.e. adherence, health care, or lifestyle changes measures); and usefulness, usage, and satisfaction. Studies were categorized as having positive, mixed, or null (no/null) outcomes based primarily on the reported clinical outcomes. Studies categorized as positive had statistical significance in the primary outcome measure. Studies that did not report clinical outcomes were classified by their reported clinical performance of care processes.
Results
The query used in the three databases, PubMed, CINAHL, and Web of Science, is shown in Figure 1. The search of articles published between 2010 and 2020, which also included those in relevant literature reviews, returned 652 unique studies, of which 32 studies were retained as shown in Figure 2.

Search query used against the three databases, PubMed, CINAHL, and Web of Science.
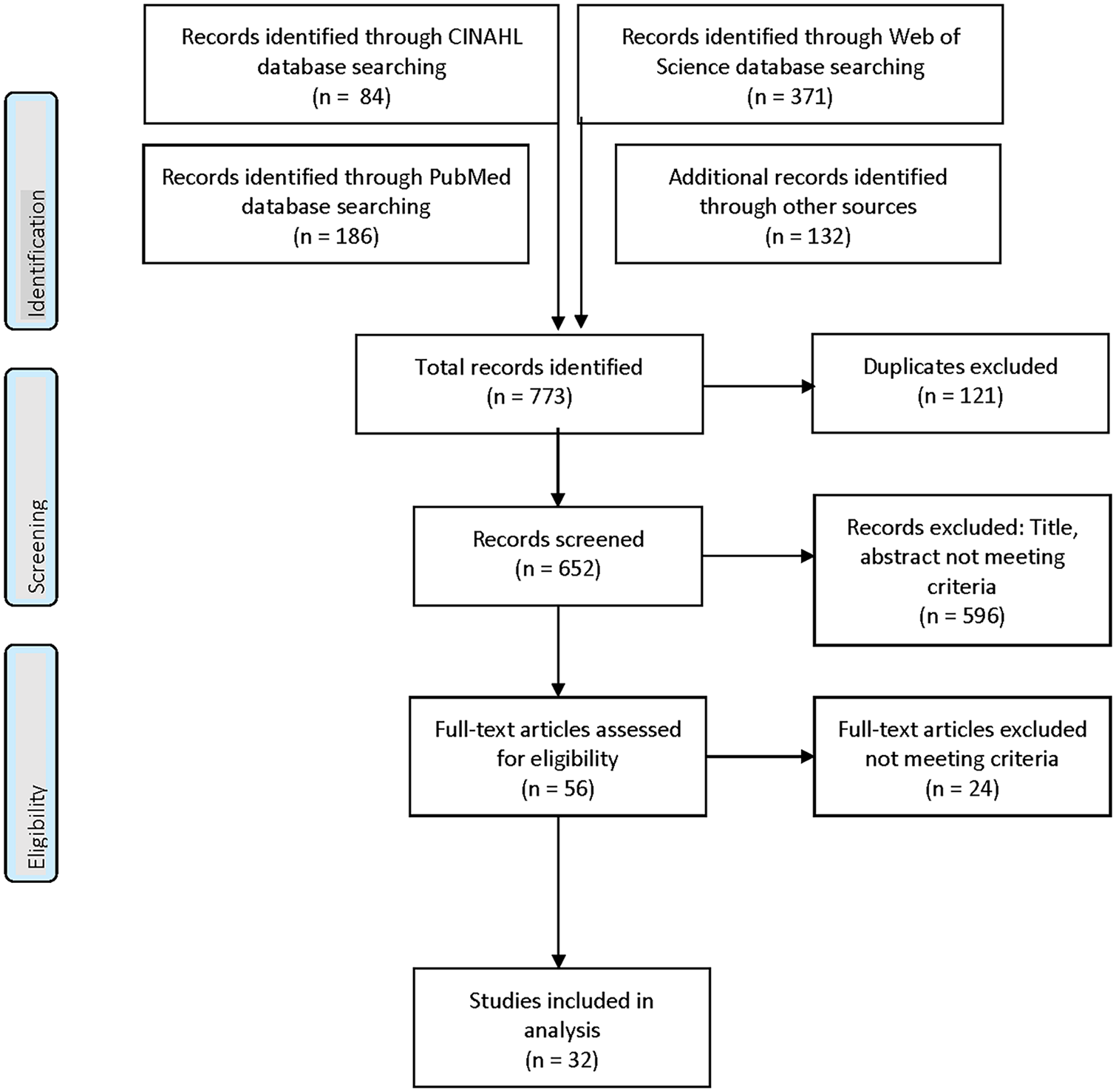
PRISMA flow study diagram.
Study descriptors
The location of most studies was North America (n = 17 studies (53%)) as shown in Table 1. The remaining studies were conducted in Asia (n = 6 (19%)), Europe and Scandinavia (n = 4 (13%)), Australia (n = 3 (9%)), and the Indian sub-continent (n = 2 (6%)). The study design of the majority of studies was randomized control trial (n = 20 (63%)). However, a few of these studies omitted the power analysis,15–17 or appeared to be under-powered (for all 18 or some 19 measures) suggesting possible threats to the study findings validity. Additional study designs were single-armed trials (n = 9 (28%)), a (3%) three-armed trial, 20 a two-armed case-control trial, 21 and an observational study. 22 A few single-armed trials omitted the power analysis, 23 or appeared to be under-powered for all24,25 or some 26 measures. The median of the studies’ duration was 19.5 weeks with a range of 427,28 to 52 29 weeks. The median of the studies’ sample size was 62 participants (range 6, 27 710 30 ). Two studies focused on multiple chronic conditions,31,32 one of which was in a population over 65 years old. 33 The remainder of the studies focused on 1 of 12 conditions, with diabetes (n = 10 studies (31%)) and a group of cardiovascular conditions (n = 8 (25%)) being the most frequent. The median age of the studies’ participants was 52 years of age, with a range of 29 21 to 72 32 years of age.
Descriptors of retained studies: location, study design, sample size, participant age, study duration, and chronic condition.
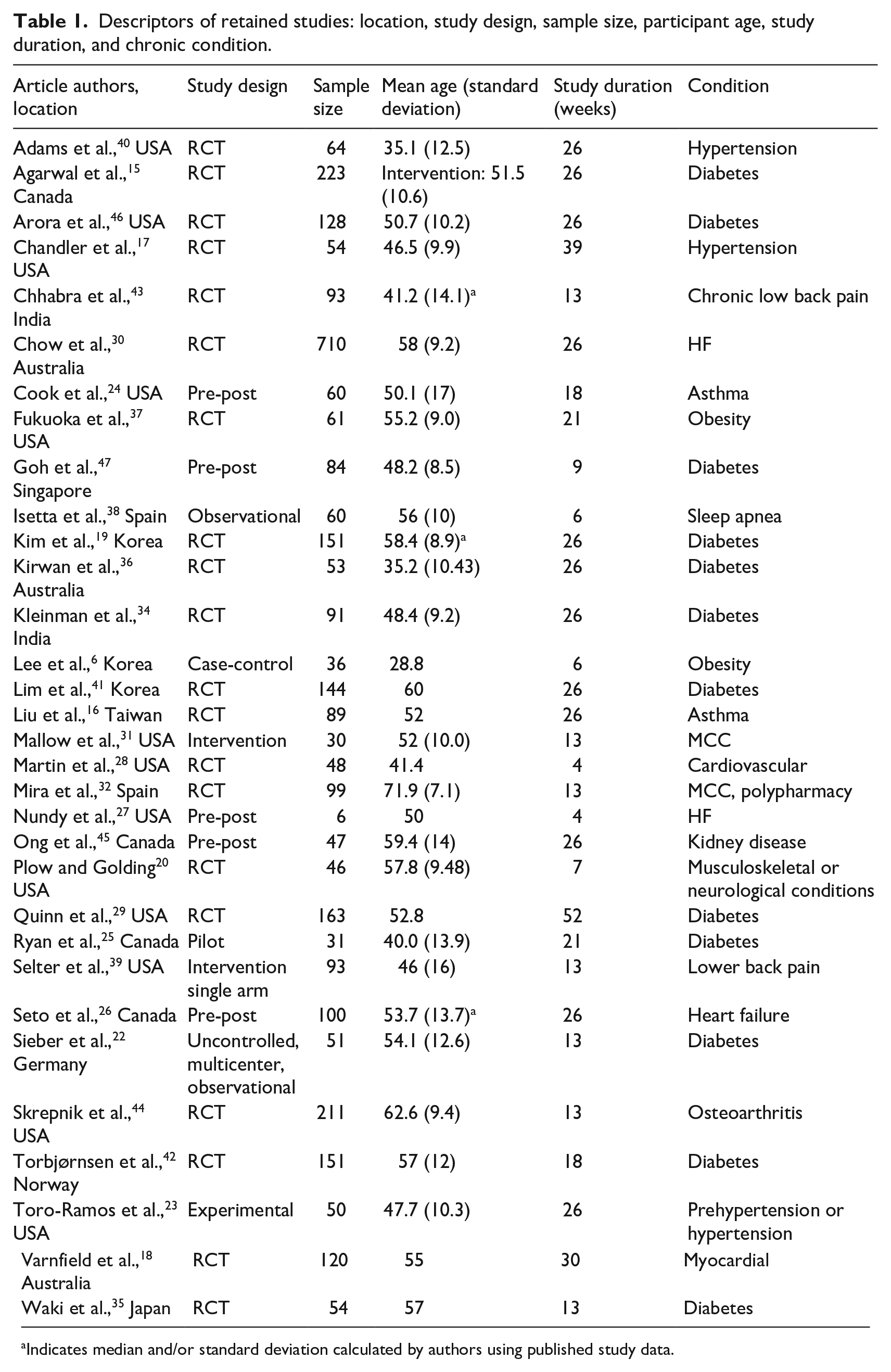
Indicates median and/or standard deviation calculated by authors using published study data.
mHealth characteristics
mHealth interventions mentioned in the retained studies had characteristics which included the user interface, whether the intervention was stand-alone, software features, and development approach. Two user interface characteristics and whether the mHealth intervention was augmented with human interaction are shown in Table 2.
Retained studies’ characteristics of user interface and human augmentation, with study outcome categorization.
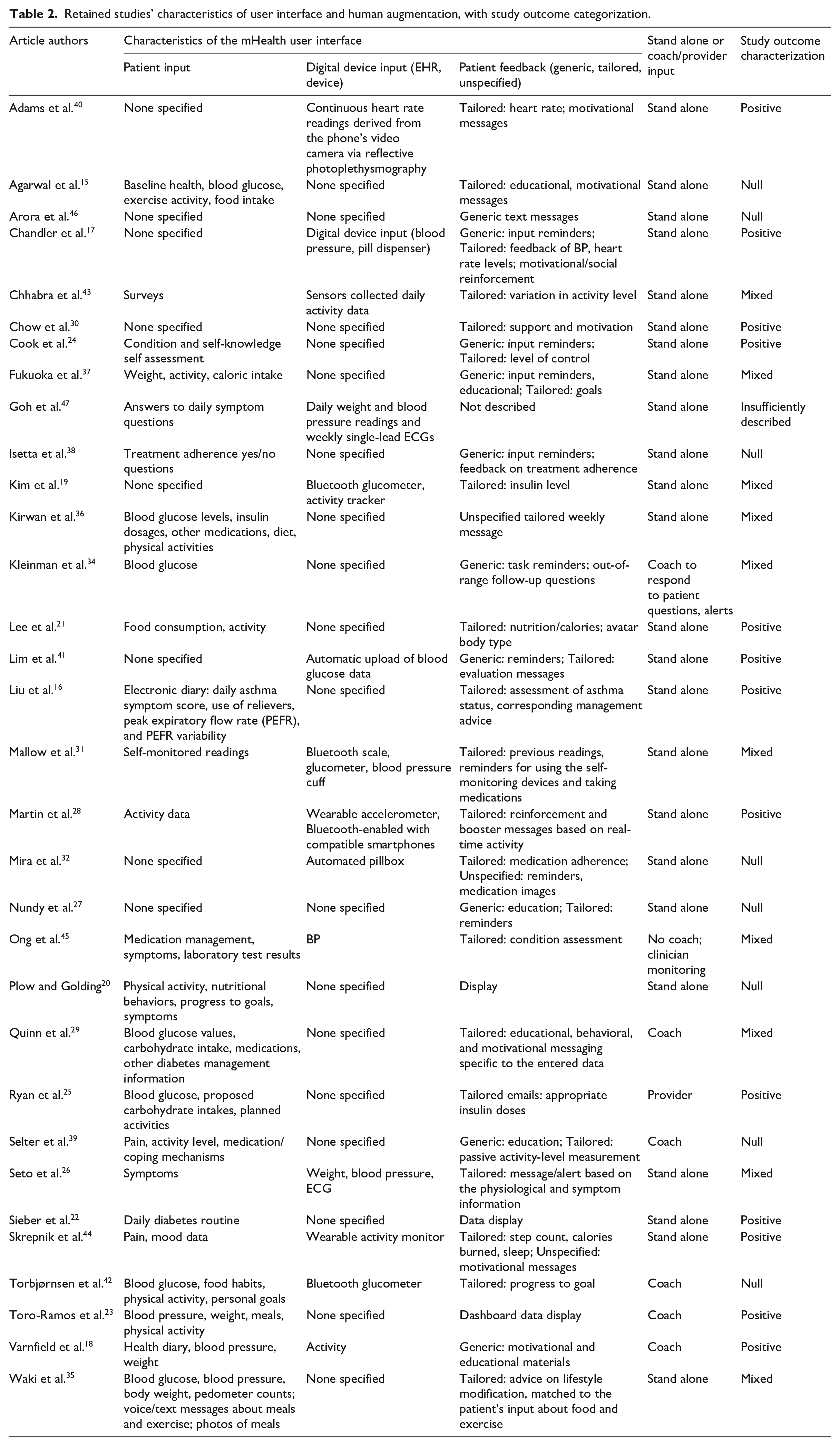
Aspects of the user interface were data input and patient messaging. Data input was characterized as patient input or via a digital device. About 15 studies (47%) described solely patient input, for example: blood glucose readings,22,25,29,34–36 food/caloric intake,15,20–23,25,29,35–38 exercise activity,15,19–23,25,35–39 body weight,23,35,38 and symptom monitoring.16,20,24 One of these studies enabled voice, text, and photo input. 35 Five studies17,19,32,40,41 (16%) described only digital input such as wireless transmission of blood pressure,17,31 glycemic level,19,41 weight, activity, 19 or heart rate 40 readings, or medication administration. 32 Nine studies (28%) described both patient and digital input.18,26–28,31,42–45 Examples of digital input included devices,17,19,31,32 and sensors in18,40 or not in cell phones.43,44 Three studies (9%) did not provide a description of the data input.27,30,46
Patient messaging was characterized as a generic message or display, a personalized or tailored message or display, or as unspecified as to either characteristic. About 16 studies (50%) described tailored messages solely. Examples include customized messages based on evidence or clinical targets15,16,19,21,26,29; and social reinforcement and motivation based on recently input adherence levels,31,40 and personal characteristics related to the condition (e.g. smoking). 30 Four studies (13%) described generic messages solely, such as reminders about the intervention.34,38 Five studies described use of both tailored and generic messages.17,24,27,37,39 One study (3%) described tailored and also unspecified messages. 44 One study sent tailored emails, not mHealth messages. 25 Four studies did not mention messages: Three studies reported displays of user data,20,22,23 one of which also sent tailored messages. 32 One study did not describe messages. 47
Human interaction that augmented the mHealth intervention was mentioned in seven studies (22%). Clinicians, or trained coaches or mentors provided the interactions which included monitoring,23,29,39 assessment, 25 support,23,39,42 counseling,18,23 and responding to patient questions and alerts.18,34,39,42 One study limited coaching to responding to alerts and questions. 34
Description of the mHealth software was infrequent and lacked detail. Although most studies generated tailored messages, algorithms (e.g. a rules engine which generated tailored messages based on the patient data) were mentioned or described in nine studies (28%)16,17,19,27,29,30,35,41 including machine learning. 43 One study also mentioned gamification (i.e. rewards points). 43 One study used natural language processing of patient-entered text. 35
Researchers in five studies incorporated publicly available apps.15,20,22,23,36 One app included a commercial curriculum and human coach intervention platform. 23
Among studies that mentioned the mHealth development process, most content (logic and messages) was developed by the research team. Two studies described target audience participation.17,32
Study outcomes
Categorization of study outcomes are shown in Table 3. One study 47 did not sufficiently describe any of the three outcome types which the study authors ascribed to attrition. Across outcome categories, more than half the studies were RCTs.
Characterization of retained studies’ outcomes: clinical, performance of care processes, usefulness/usage/satisfaction.
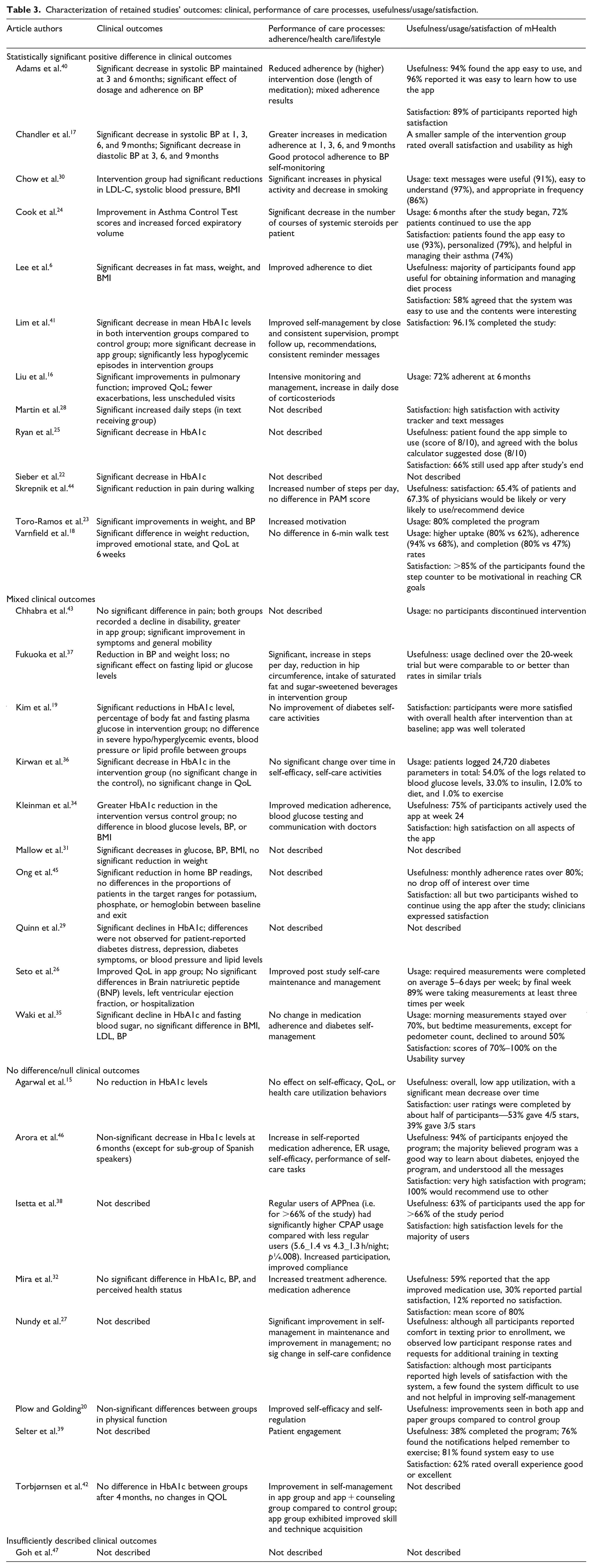
About 13 studies (41% of all studies) reported statistically significant positive outcomes, for example in weight,18,21,23,30 BP,17,30,40 HbA1c,22,25,41 LDL, 30 asthma control, 24 pain, 44 and pulmonary function. 16 Ten studies reported performance of care processes: eight reported improved performance (e.g. Intensive monitoring and management, increase in corticosteroid daily dose 16 ). One study reported no effect on care processes, 18 and one study reported mixed results. 40 Of the 12 studies which reported user experience, usage, usefulness, and/or satisfaction was described as high.
Ten studies (31%) reported mixed study outcomes. For example, Fukuoka et al. 37 reported reduction in BP and weight loss, yet no significant effect on fasting lipid or glucose levels. 37 Among the six studies that reported performance of care processes, assessments were mixed. Three studies indicated no effect19,35,36 (e.g. no significant change over time in self-efficacy, self-care activities 36 ) and three studies reported positive effects26,34,37 (e.g. improved medication adherence, blood glucose testing, and communication with doctors 34 ). Seven of the eight studies that reported comparative usefulness/usage/satisfaction reported generally affirmative effects.19,25,26,34,35,43,45
Eight studies (25%) reported null study outcomes.15,20,27,32,38,39,42,46 Five studies reported null clinical outcomes.15,20,32,42,46 For example, Torbjørnsen et al. 42 reported no difference in HbA1c between groups after 4 months, and no changes in QOL. Three studies did not report clinical outcomes.16,37,39 Six of the eight studies that reported performance of care processes described affirmative effects20,27,32,38,42,46 (e.g. increase in self-reported medication adherence and self-efficacy, performance of self-care tasks 46 ). Of the eight studies that reported usage and user satisfaction, two had affirmative effects.38,46
Integration of study descriptors, mHealth technology features by study outcomes
In each study outcome category (positive, mixed, or null), the most often occurring clinical outcomes were blood glucose (14 studies) and blood pressure (11 studies) followed by weight (5 studies) reflecting the large number of diabetes and cardiovascular disease studies. Across outcome categories, studies reported clinical care processes such as performance of care or usefulness/usage/satisfaction as generally affirmative. Careful analysis of study features and the technology by outcome reveals several interesting findings.
Of the 13 studies with positive study outcomes the study descriptor, median study duration, was 21 weeks, slightly above the median for all retained studies (19.5). Study sample size was 60 participants, slightly below the median (62) of retained studies. Median study participant age was 50 years, slightly less than the median for retained studies. 51 This analysis suggests that slightly longer but smaller sample size studies with more rigorous design in well-studied chronic illnesses in younger patients were more likely to report positive clinical outcomes.
Regarding mHealth technology features, 6 of the 13 studies used digital input: either alone or with patient input (three studies each). About 10 studies used tailored messages. Two studies used display output (and patient input). Of the three studies that used coaching, two included assessment and counseling for weight and blood pressure outcomes. Two studies used publicly available apps. One study described user participation in the mHealth development process. This synthesis suggests that digital input and tailored messages are the more successful user interfaces for supporting chronic illness management sufficiently to change clinical outcomes.
Of the 10 studies with mixed study outcomes, median study duration was 26 weeks, 6.5 weeks longer than the median for all retained studies. Study sample size of 57 participants was less than the median (62) of all studies. Median participant age for the 10 studies was 53 years, slightly above the median of retained studies. Of the studies that reported performance of care, the measure was mixed. This examination indicates studies with a design mostly similar to that of studies with positive study outcomes, with the exception of having a longer duration, were more likely to report mixed study outcomes.
Concerning mHealth features, nine studies used patient input: either alone (five studies) or with digital input (four studies). Nine studies used tailored messages. No study used display output. Two studies included coaching consisting of monitoring or replying to patients; neither included counseling. One study used a publicly available app. One study mentioned gamification with incentives for outcomes which included pain. The analysis suggests that studies that used patient input and tailored messages were more likely to reported mixed clinical outcomes.
Of the eight studies with null reported difference in study outcomes, median study duration of 13 weeks was much shorter than the median duration of all retained studies. Study sample size was 96 participants, much higher than the median (62) of retained studies. Study participant median age was 53.5, slightly above the median age of all retained studies. This exploration suggests studies of shorter duration with larger sample sizes with more rigorous design in a range of chronic illnesses in younger patients were more likely to report null outcomes.
As for mHealth technology, among the six studies which described data input, five used patient input. Five studies used tailored messages and four studies used generic messages (two studies used both). One study used a display output. Two studies included coaching for monitoring and support, and did not include counseling. Three studies used a publicly available app: one study outcome was physical function; another study did not report a clinical outcome. One study described user participation in development. This investigation suggests that studies that used patient input were more likely to report null outcomes.
Discussion
We conducted an integrative review of clinical studies of mHealth self-care applications for patients living with chronic conditions to examine the study descriptors, mHealth technology features, and study outcomes. We identified similarities and variations among the 32 retained studies, and offer suggestions as shown in Table 4. The resulting information enables researchers to identify gaps in knowledge and future avenues of inquiry as shown in Table 5.
Study results and related suggestions.
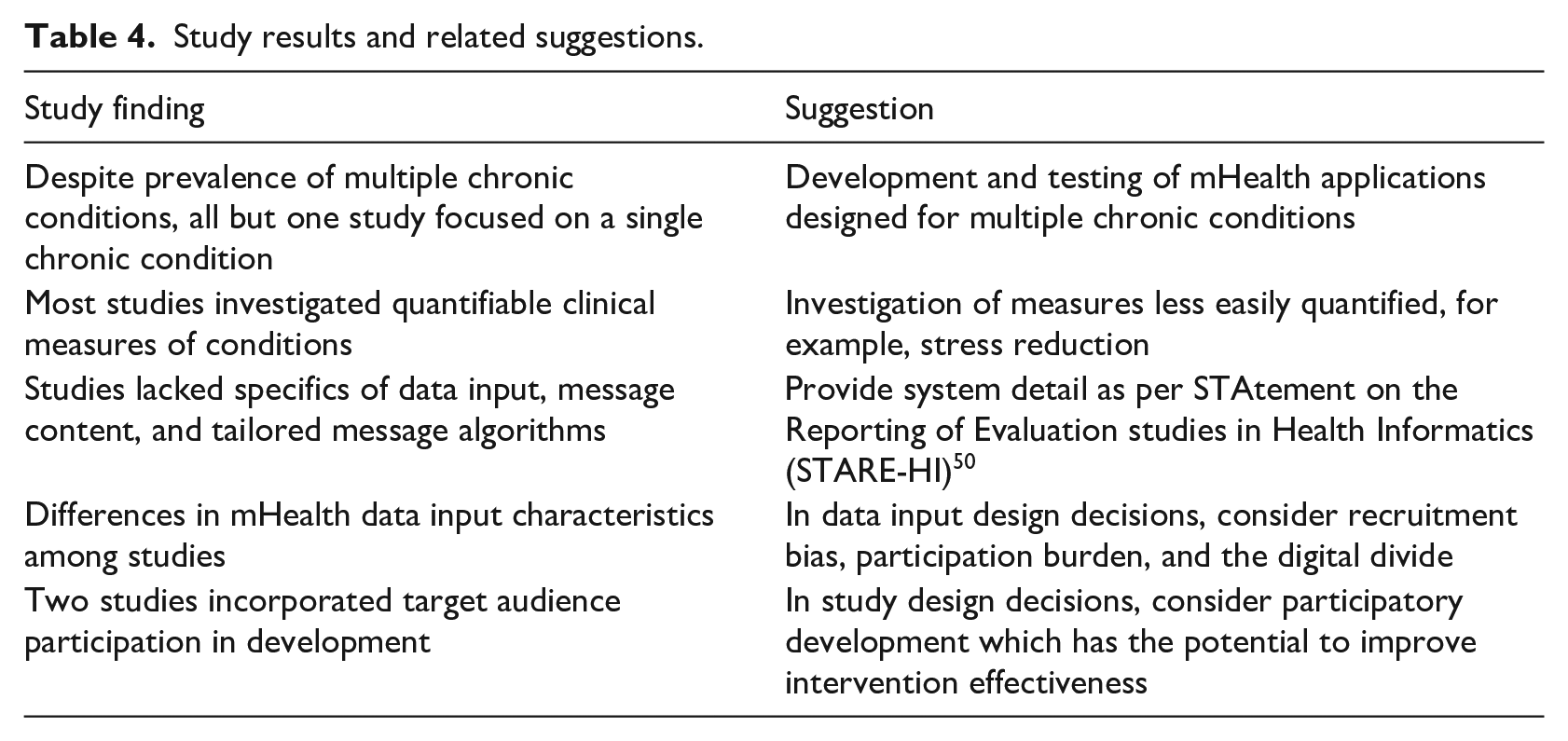
Recommendations for future research.
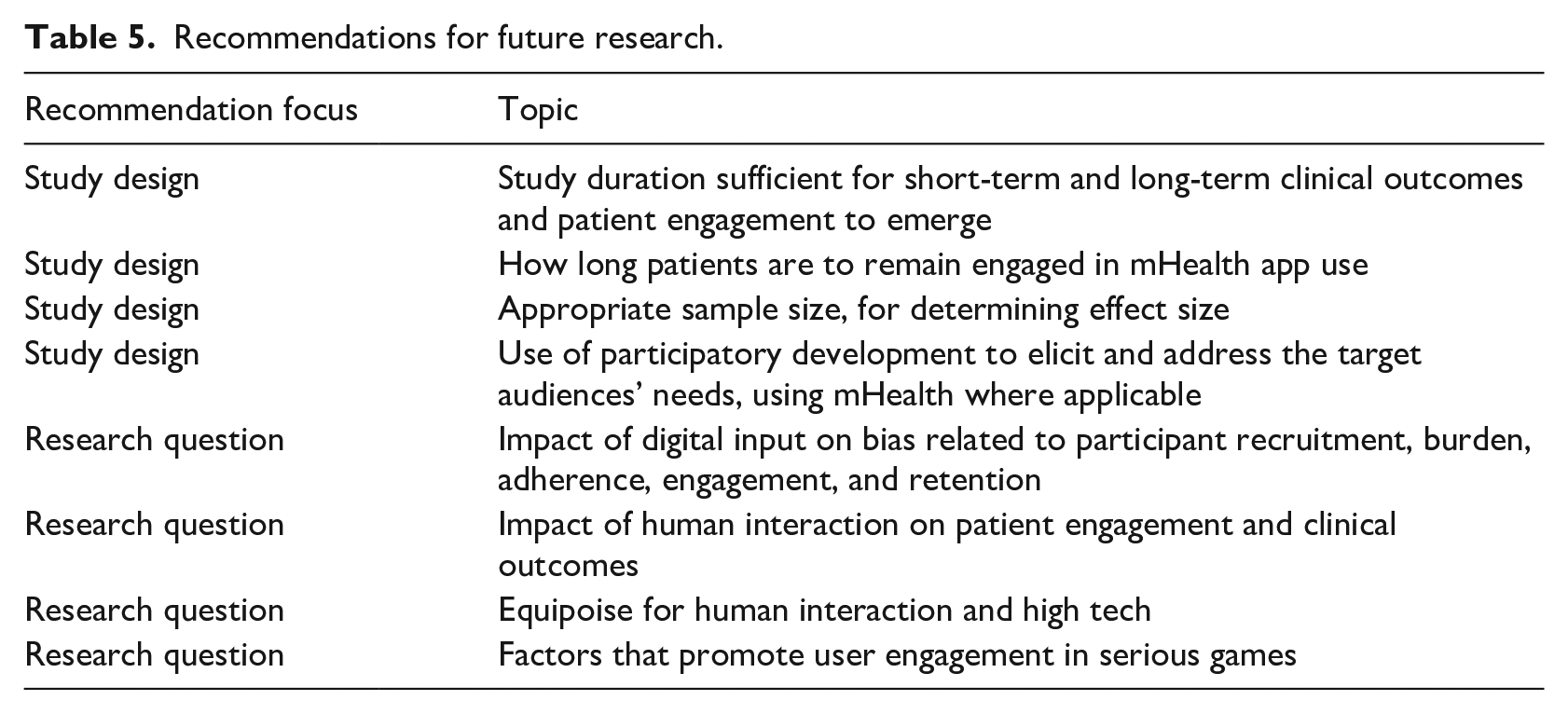
Study descriptors and mHealth characteristics similarities
All but one study focused on a single chronic condition in an adult population younger than 65 years. However, multiple chronic conditions is the most common chronic condition 51 internationally.48,49 Development and testing of mHealth self-care applications designed for multiple chronic conditions is warranted.
Among the dozen conditions targeted in the retained studies, the most common had quantifiable clinical measures such as blood glucose, blood pressure, weight, food intake, and exercise activity. Notable was an under examination of measures less easily quantified, such as stress reduction, although a few studies assessed symptom monitoring.
Description of mHealth characteristics was variable. Several studies lacked specifics of data input or messages. While most studies sent tailored messages based on data input, only some studies mentioned the software logic to determine which message to send when, and few studies described the logic. We recommend researchers provide a sufficient level of detail of the system description to permit the reader to understand how the system works, as per the STAtement on the Reporting of Evaluation studies in Health Informatics (STARE-HI). 50
Study descriptors and mHealth characteristics related to study outcomes
Among studies that reported measures, performance of care, and usefulness/usage/satisfaction outcomes were generally affirmative. These outcomes were not indicative of study outcome categorization.
Studies with positive or mixed outcomes tended to have a 20–26 week duration, an approximately 60 participant sample size, and use digital input. Whereas studies with null outcomes tended to have a shorter duration, larger sample size, and rely on patient input.
The study design differences, coupled with the wide range in study duration and sample size, highlight opportunities for future research. Questions include what is a sufficient duration for short-term and long-term clinical outcomes and patient engagement to emerge; and for how long do patients remained engaged in mHealth app use? For example, hemoglobin A1c, weight, and low-density lipoproteins change slowly: Measurement within months will not show large differences. Another question is an appropriate sample size, especially in regards to determining effect size.
Differences in mHealth data input characteristics among studies point to areas for further research. Dependence on patient input may introduce recruitment bias among older people uncomfortable with the technology. Furthermore, the participation burden of patient data input 24 coupled with the new hurdle of unfamiliar technology use may impact participant adherence, engagement, and retention. Digital input use, such as sensors or voice-activated data input 51 may address these challenges: another avenue of future research. However, use of digital input technologies which require capabilities that may be unavailable to vulnerable populations 27 or people living in under-resourced communities 46 may introduce recruitment bias.
Some studies augmented technology with human interaction such as coaching (“high-touch”). Studies with positive outcomes that included coaching offered counseling, a feature not included in studies with mixed or null study outcomes. Questions of interest are whether high-touch leads to better patient engagement 15 and clinical outcomes; and what is the high-touch/high tech equipoise? Prevalent use of tailored messages and infrequent use of coaching among studies, regardless of clinical outcome categorization, suggests these questions warrant further research.
mHealth development characteristics included build-or-buy approaches and participatory development. While most studies built mHealth interventions, only two incorporated target audience participation in development, with differing study outcomes. Participatory development has the potential to improve intervention effectiveness. 52 Its infrequent use among the studies raises the question whether researchers are implementing in mHealth existing approaches while expecting better outcomes, rather than eliciting and addressing the target audiences’ needs and using mHealth where applicable.
The few studies that used the buy-approach, incorporating commercially available apps, had diverse study outcomes. This finding may suggest a lack of app effectiveness, indicative of the lack of formal assessment, regulatory review, 53 and testing outside of a small, relatively homogeneous population. 15
An mHealth design feature mentioned in one study with mixed outcomes was gamification, a method that may address the challenge of initiating and maintaining patient engagement. DeSmet et al.’s 54 meta-analysis of serious games for healthy lifestyle promotion found small positive effects on healthy lifestyles, their determinants, and clinical outcomes. However, most games in the meta-analysis were designed for children and young adults, a younger population compared to those living with chronic conditions. Edwards et al.’s 53 systematic review of publicly available games for healthy behavior change found no relation between game content and user ratings. This finding indicates that the question of what promotes user engagement remains unanswered among gaming scientists, and is an opportunity for future research.
Practical implications
Those who use or intend to use mHealth self-care applications designed for patients living with multiple chronic conditions should keep in mind that these applications could benefit from further development and testing. Researchers should be aware that these applications often are inadequately described in the studies, that appropriate sample size for effect size estimation remains undetermined, and recruitment bias may not be accounted for in completed studies.
Limitations
Characteristic of literature reviews, this review does not claim to be comprehensive nor to fully implement PRISMA guidelines given the more targeted scope of the paper. Instead, it summarizes the research on mHealth for patient chronic illness self-management based on the search terms used, the databases included, and the review time period. That we used a medical librarian with extensive literature search experience and transparently reported the data acquisition process provides evidence for the dataset’s limitations and scope. Also, we acknowledge that positive results publication bias may have limited the studies available in the databases searched. We did not exclude studies that omitted power analysis or appeared underpowered, nor change authors’ study design classification to reflect sample size. As this was an integrative review, we did not report quality assessment which is required only in systematic reviews and meta-analyses.
Conclusions
We investigated recent chronic condition mHealth interventions experimental studies with a focus on technology features related to clinical outcomes. We offer suggestions about study duration and sample size, and data input and messaging design decisions. We provide these suggestions to advance mHealth science and to future technology developers and researchers for their consideration.
Footnotes
Acknowledgements
The research librarian, Janice Masud-Paul, for assistance with development of the search strategy and retrieval of articles from databases.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
