Abstract
A failure modes, effects and criticality analysis was supported by an observational medication error rate study to analyze the impact of Phocus Rx®, a new image-based workflow software system, on chemotherapy compounding error rates. Residual risks that should be a target for additional action were identified and prioritized and pharmacy staff satisfaction with the new system was evaluated. In total, 16 potential failure modes were recognized in the pre-implementation phase and 21 after Phocus Rx® implementation. The total reduction of the criticality index was 67 percent, with a reduction of 46 percent in material preparation, 76 percent in drug production and 48 percent in quality control subprocesses. The relative risk reduction of compounding error rate was 63 percent after the implementation of Phocus Rx®, from 0.045 to 0.017 percent. The high-priority recommendations defined were identification of the product with batch and expiration date from scanned bidimensional barcodes on drug vials and process improvements in image-based quality control. Overall satisfaction index was 8.30 (SD 1.06) for technicians and 8.56 (SD 1.42) for pharmacists (p = 0.655). The introduction of a new workflow management software system was an effective approach to increasing safety in the compounding procedures in the pharmacy department, according to the failure modes, effects and criticality analysis method.
Keywords
Introduction
Chemotherapy drugs used in the treatment of cancer are considered high-alert medications, meaning that they bear a heightened risk of causing significant patient harm when they are used in error. 1 Chemotherapy errors can occur at all steps of the medication process (prescription, preparation and administration) and include all types of medication errors from wrong patient or drug to wrong dose, schedule, infusion rate or compounding errors.2–4 Therefore, risk reduction in the antineoplastic drug use process should be a major objective for all relevant institutions.
The prevalence of errors in the chemotherapy prescription and administration stages is described in the literature,5–7 and their risk assessments have been published elsewhere.8–11 However, the risks in the chemotherapy compounding process remains uncertain, mostly because of the studies’ heterogeneity and the lack of observational studies specifically designed to detect chemotherapy compounding errors. Nevertheless, the compounding process is often considered a critical step, as many of the possible errors at this stage cannot be detected downward in the process. For example, the nurse who administers the treatment could not detect a dose error in an bag solution.
The implementation of technology-assisted quality control processes for the preparation of antineoplastic medications is recommended by several organizations to reduce the number of medication errors.12–14 These technologies include barcode identification of starting products, an electronic interface to guide the technicians during the compounding process and a dose measure control, usually gravimetric or volumetric.
However, a careful evaluation of their benefits and risks is needed, 13 as they change both the processes and their performance. In this context, the failure modes, effects and criticality analysis (FMECA) method is a risk-assessment tool that has been widely used to identify and address safety concerns in complex processes. It may be of great value for evaluating potential errors (failure modes) and their causes and prioritizes them according to the risk they pose. FMECA has been applied to different stages of the medication use process such as prescription, distribution and administration.15,16 It has also been considered an excellent tool for the evaluation of patient safety after the implementation of information technologies (ITs) like computerized physician order entry (CPOE) and smart pumps.17,18 However, given the scant evidence about the reliability and validity of FMECA, 19 it would not be appropriate to recommend its use alone as a tool for preventing patient harm. To overcome this limitation, we combined risk analysis using FMECA methodology with the objective measures of a traditional cross-sectional medication error rate study.
The primary objective of this study was to perform a comparative risk analysis of antineoplastic production before and after the implementation of an image-based compounding workflow software system and to quantify its impact on reducing medication errors. The secondary objectives were to identify any major residual risks that should be a target for additional action in the computerized system and to evaluate pharmacy technicians’ and pharmacists’ satisfaction with the new system.
Methods
Setting
The study was conducted in the chemotherapy compounding unit of the pharmacy department (PD) in a 1300-bed tertiary teaching hospital that prepares an average of 220 parenteral antineoplastic doses daily. It serves 39 infusion chairs for ambulatory care patients (32 for adults and 7 for pediatrics) and all inpatient chemotherapy doses, including pediatric and bone marrow transplantation units.
CPOE software (Farhos®; Visual Limes, Valencia, Spain), integrated with the electronic health record system is used for medication ordering, compounding and administration. Standard doses are automatically calculated based on standard protocols and body surface area (calculated from the patient’s height and weight) or creatinine clearance (computed from the serum creatinine concentration according to the Cockroft–Gault formula). Once the oncology pharmacists have verified the prescriptions in the CPOE system, the compounding protocols and medication labels are automatically available for printing in the chemotherapy compounding unit. Pharmacy technicians gather drug vials and supplies as required and compound the medications according to the compounding order and the unit’s standard operating procedures.
Quality control of the compounded medications was done by a different pharmacy technician from the one who had prepared the medication and was supported by a pharmacist. In the pre-implementation phase, technicians visually verified selected starting products, measured doses according to vials used, the final product appearance and label accuracy. In the post-implementation phase, quality control was performed by image checking assisted by the Phocus Rx® system.
Implementation of the compounding workflow management software system
A compounding workflow software system (Phocus Rx®; Grifols, Barcelona, Spain) was pilot tested between November 2016 and February 2017 in the chemotherapy compounding unit of the PD. After a validation and educational period Phocus Rx® was implemented in May 2017. This system consists of a series of hardware components, including a touchscreen computer installed next to the biological safety cabinet (BSC), an ultra-compact camera located in the clean room ceiling and a barcode scanner. The software components interface with the CPOE system to receive the information for the compounding order: drug vials, consumables, amount of drug and fluids, final volume and specific preparation instructions. All this information is visible on the touchscreen once the preparation has been started.
Pharmacy technicians in the BSC scan the compounding order to initiate preparation. Then, Phocus Rx® requires that products in the compounding order (drugs, fluids and consumables) also be scanned. This is an additional control step in products containing a barcode on primary packages, nowadays a minority. 20 If the barcodes match the prescribed products, workflow can proceed. The system guides the technicians through the compounding process taking pictures throughout the defined steps, during the preparation, without adding any steps to the process. Each step performed by the technician is thereby documented in real time and made available for quality control of the preparations.
In the post-implementation phase, a pharmacy technician reviewed the images associated with each dose and approved or rejected the preparations. If any discrepancy between the image and the procedure was detected, a pharmacist was responsible for giving final approval to or rejecting the preparation. Final approval involved verification of patient and drug identity, drug commercial presentation, reconstitution fluid and amount required, volume of drug required to achieve the dose, fluid type and volume, consumables, preparation procedures and labeling.
Study design
The FMECA procedure was performed to analyze the chemotherapy compounding process before and after implementing Phocus Rx®. The FMECA was completed with a before-and-after cross-sectional medication error study to measure the impact of Phocus Rx® implementation in compounding error rates.
The FMECA was performed in two phases: a pre-implementation phase (between October and November 2016) and a post-implementation phase (between October and November 2017), each of them consisting of six meetings lasting approximately 2 h, including the time taken to write the report. An introductory session of 1 h was held to explain the features of FMECA.
The pre-implementation observational error study was conducted from May 2016 to April 2017 and the post-implementation error study from June 2017 to May 2018. Between these two periods, experience of the pharmacy technicians, compounding procedures and number of preparations compounded in the unit were maintained.
An anonymous satisfaction survey specifically designed for this project was given to pharmacy technicians and pharmacists 1 year after Phocus Rx® implementation.
FMECA procedure
In accordance with FMECA guidelines, a multidisciplinary team of pharmacists and technicians was assembled. 21 Five specialist pharmacists from different areas of expertise were involved: oncohematology-chemotherapy protocols, sterile compounding, patient safety, design and safety of workflow management systems technology and quality assurance. In addition, two pharmacy technicians specialized in chemotherapy compounding and the supervisor of the PD joined the team. All members were asked to represent their colleagues’ opinions and to consult them if necessary during the analysis.
The process of centralized preparation of chemotherapy drugs in the PD before and after implementing Phocus Rx® was analyzed using FMECA. The team first developed a flow diagram of the process and the main subprocesses involved (from the moment the preparation order is received until dispensing). A brainstorming session was then organized to determine the ways in which the process could fail at each step. The main points of the discussion were summarized and the failure modes were established.
The severity of the potential patient outcome, the likelihood of occurrence and the likelihood of detection for each failure were graded on a scale of 1–10. The evaluation was based on the literature, the results of our previous observational study, 22 local context and workload (Table 1). Estimations were obtained for all failure modes by consensual discussions between team members. The riskiness of each element was expressed as a risk priority number (RPN), which was calculated as the product of the severity, occurrence and detectability scores (RPN = S × O × D). The RPN identified those elements that are the most likely contributors to medically serious failures. The criticality indices were compared for each mode of failure before and after the Phocus Rx® system implementation. The sum of all individual criticality indices was calculated for each subprocesses and compared to determine overall safety improvement and potential impact on patient outcome.
Rating scales for severity, frequency and likelihood of detection.

Medication error study
A compounding error was defined as any discrepancy between the compounding process and the requirements of the compounding order that was not detected and corrected in the quality control step of the process. Before starting the study, definitions and practical examples of each possible type of error were presented and discussed by the multidisciplinary team. Two pharmacists of this team were trained to evaluate the quality of the preparations compounded, before and after the technology was implemented, and they performed a daily audit in close collaboration with pharmacy technicians and nurses who administer the chemotherapy. Errors were detected at the time of administration by nursing staff or by the patients themselves or during treatment follow-up by the physician or pharmacist.
The multidisciplinary team classified and evaluated the detected errors. The type of error was classified according to the Ruiz Jarabo 2008 classification, which is an adaptation of the National Coordinating Council for Medication Error Reporting and Prevention (NCC MERP) taxonomy in the Spanish health system. 23
The hospital’s institutional review board did not require informed consent from the patient as the study was focused on the technicians’ work. The PD technicians’ involvement in the study was entirely voluntary.
The number of observations needed to adequately power this study was based on published data in cytotoxic drug preparation errors. Assuming a baseline error rate of 0.1 percent and an α of 0.05 and β of 0.20 (80%) power, 47,100 preparations would have to be analyzed in each period to demonstrate a reduction to 0.05 percent in the compounding error rate.
Statistical analysis was performed with SPSS, 21.0 and p-value <0.05 was considered statistically significant.
Acceptability and further actions
For each failure mode, the criticality index after Phocus Rx® implementation was analyzed by the team and the acceptability of the risk or the need for further recommendations was discussed. The proposed actions were divided into two categories: Phocus Rx® recommendations and process improvements. A priority classification was established by taking into consideration the value of the criticality index in the compounding process, the extent of the expected reduction in criticality and the volume of work and expenditure needed to develop the proposal.
Staff satisfaction analysis
A cross-sectional, anonymous questionnaire was carried out. The questionnaire, which included items on 10 influencing factors, was sent to 10 pharmacy technicians and 9 pharmacists who were working in the PD before and after the implementation of Phocus Rx® and were familiar with compounding and quality control processes. Specifically, satisfaction with the ease of use of the technology, the impact on pharmacy technician workload (added time) and perception of safety and quality improvement were evaluated by the pharmacy staff. All employees were asked if they would prefer the use of Phocus Rx® to manual quality control and if they would recommend their implementation to other PD.
The results (0–10 points) were expressed in mean ± standard deviation. Comparison between staff category was made using Mann–Whitney test.
Results
Failure modes definition
The sterile parenteral antineoplastic compounding process was divided into three subprocesses: (1) starting products’ preparation, (2) drug compounding in the BSC and (3) quality control of the compounded drug. The phases of the process before and after Phocus Rx® implementation are detailed in Figure 1.
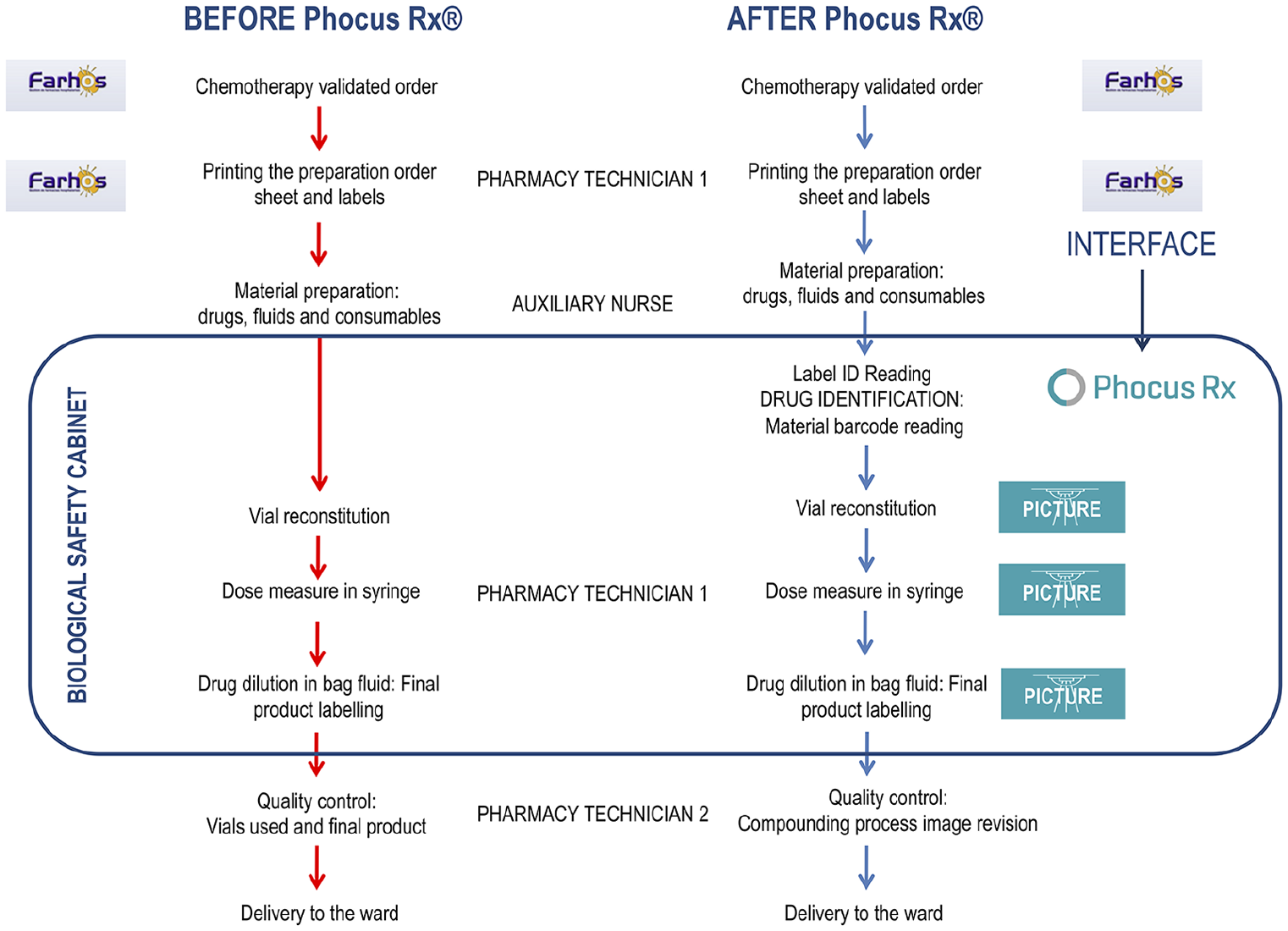
Flow diagram of the compounding process in the before and after implementation phases.
Criticality analysis
We recognized 16 potential failure modes in the pre-implementation phase and 21 in the post-implementation phase, and the sum of criticality indexes was 1999 in the pre-phase and 668 in the post-phase. Phocus Rx® implementation led to a criticality index reduction of 67 percent, with drug production in the BSC as the subprocess with the highest reduction (−76%; Figure 2).
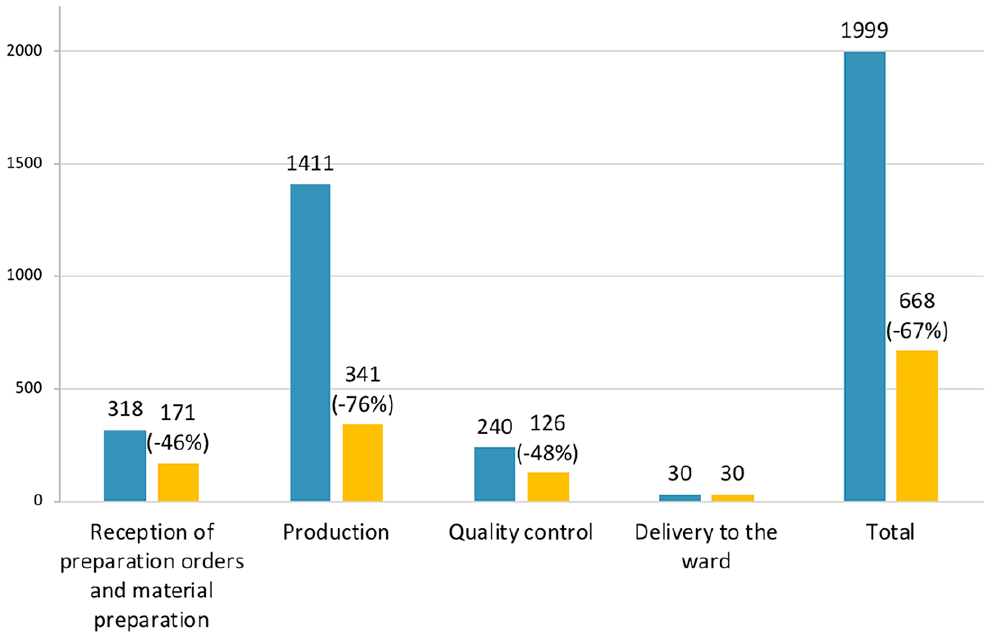
Total criticality indices before and after Phocus Rx® implementation.
For 13 of 22 failure modes (59%), the criticality index decreased after implementation, whereas the risk remained unchanged for four. Five new modes of failure appeared in the after phase, but they were associated with very low criticalities. The number of indexes classified as high risk (RPN > 100) diminished from 9 (56%) in the before phase to 0 in the after phase. The criticality indexes calculated from the frequency, severity and detection scores defined for each of the failure modes are illustrated in Figures 3 to 5, for each major step of the process.
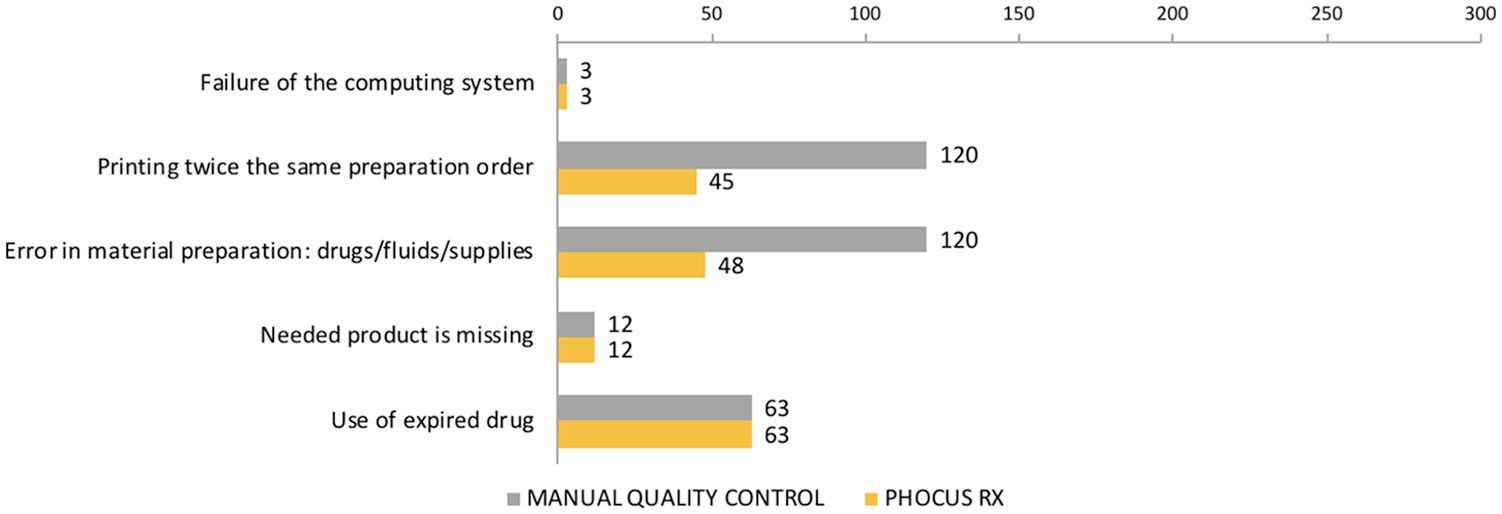
Reception of compounding orders and material preparation: failure modes and criticality indices.
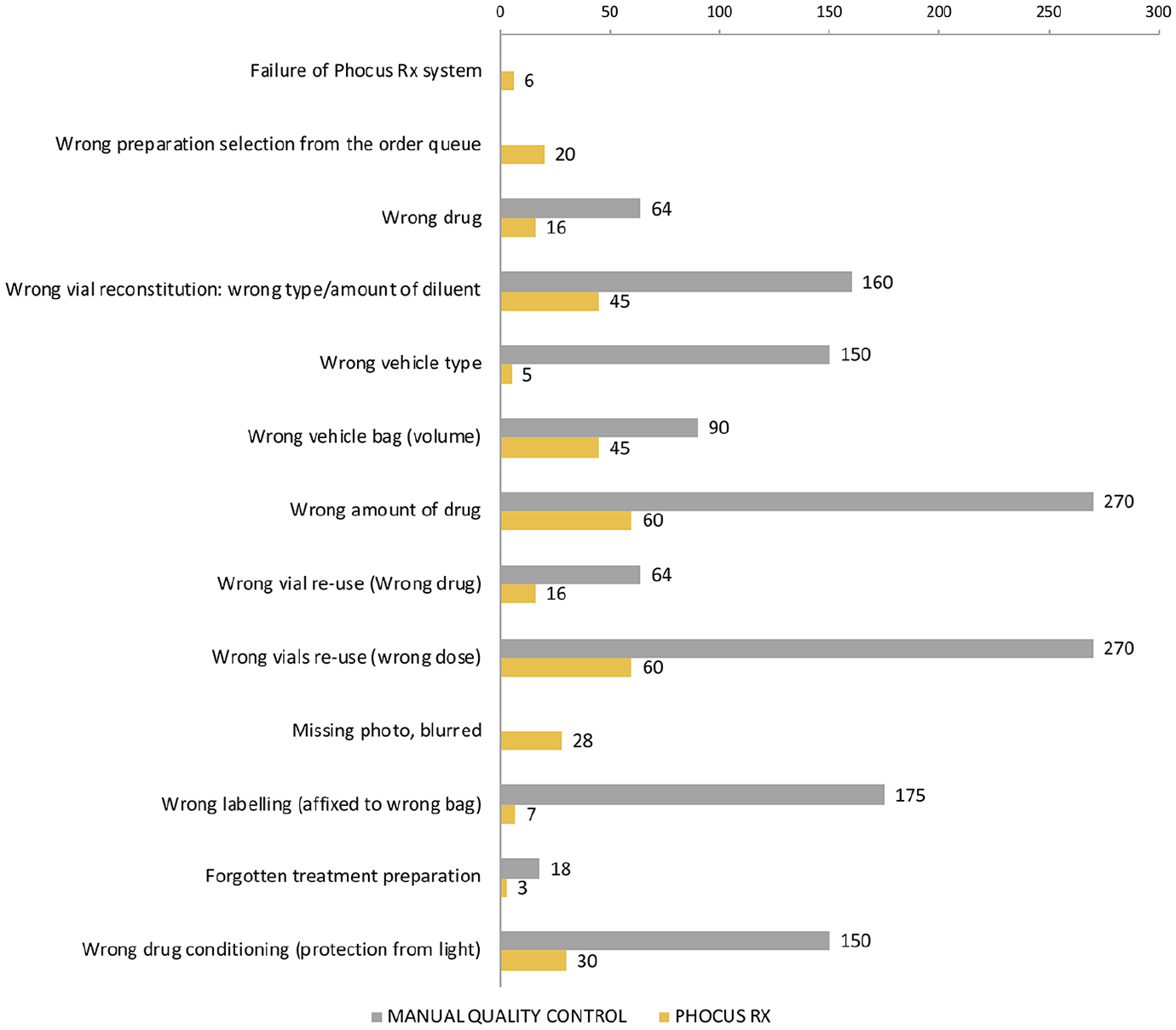
Antineoplastic drug compounding: failure modes and criticality indices.
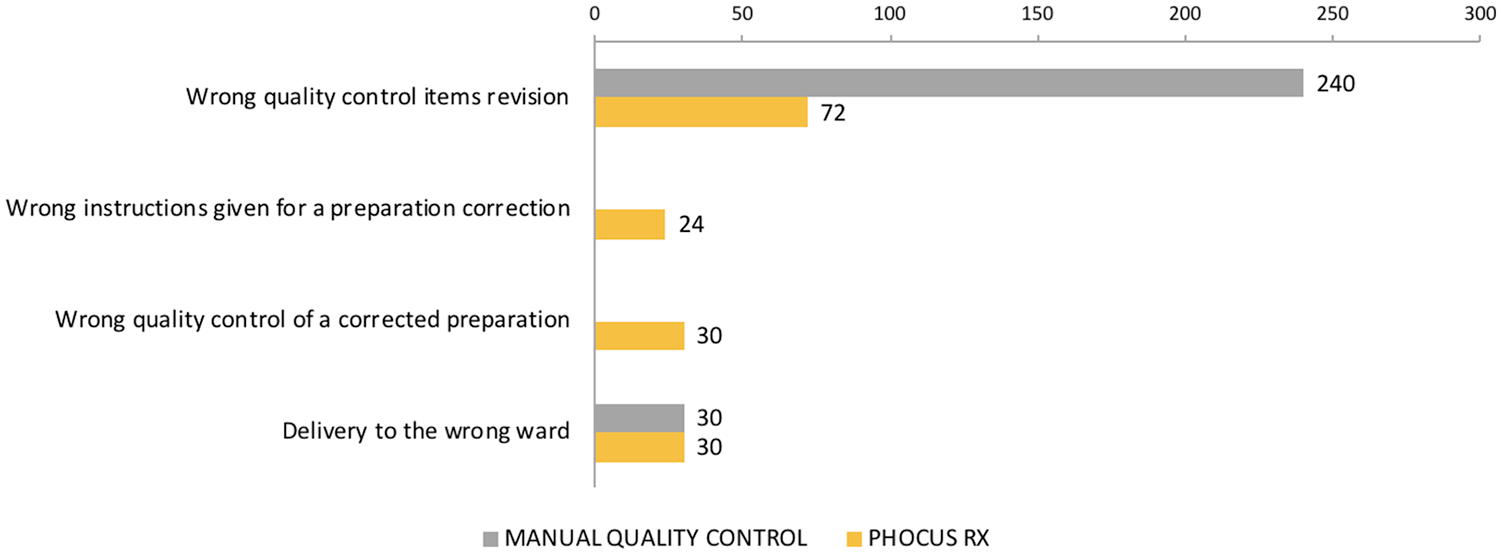
Quality control of antineoplastic drugs: failure modes and criticality indices.
In the pre-implementation phase, the subprocess that accumulated the highest value for total RPN was the drug compounding in the BSC (total RPN = 1411). The failure modes with the highest RPNs were as follows: incorrect quality control, incorrect dose measure, incorrect dose calculation when reusing vials, incorrect label (misapplication to another bag of the same patient), incorrect reconstitution fluid, wrong vehicle type and incorrect drug packaging.
With implementation of the compounding workflow management system, the failure modes with the highest criticality reduction were: wrong vehicle type (−96.7%), incorrect label (−96%), forgotten bag preparation (−83.3%), incorrect drug packaging (−80%), incorrect drug measure or incorrect measured dose with drug vials optimization to prevent waste (−77.8%) and incorrect drug (−75%).
Medication error study
During the observational study, 51,589 antineoplastic drugs (9.8% clinical trial preparations) were compounded in the pre-implementation period and 54,430 (12.1% clinical trial) in the post-implementation period. The number of errors that were not detected in the PD during the quality control was reduced after the implementation of Phocus Rx® (0.045% vs 0.017%, RRR = 62.9%). The error type is detailed in Table 2.
Compounding medication error rate before and after implementation of Phocus Rx® system.
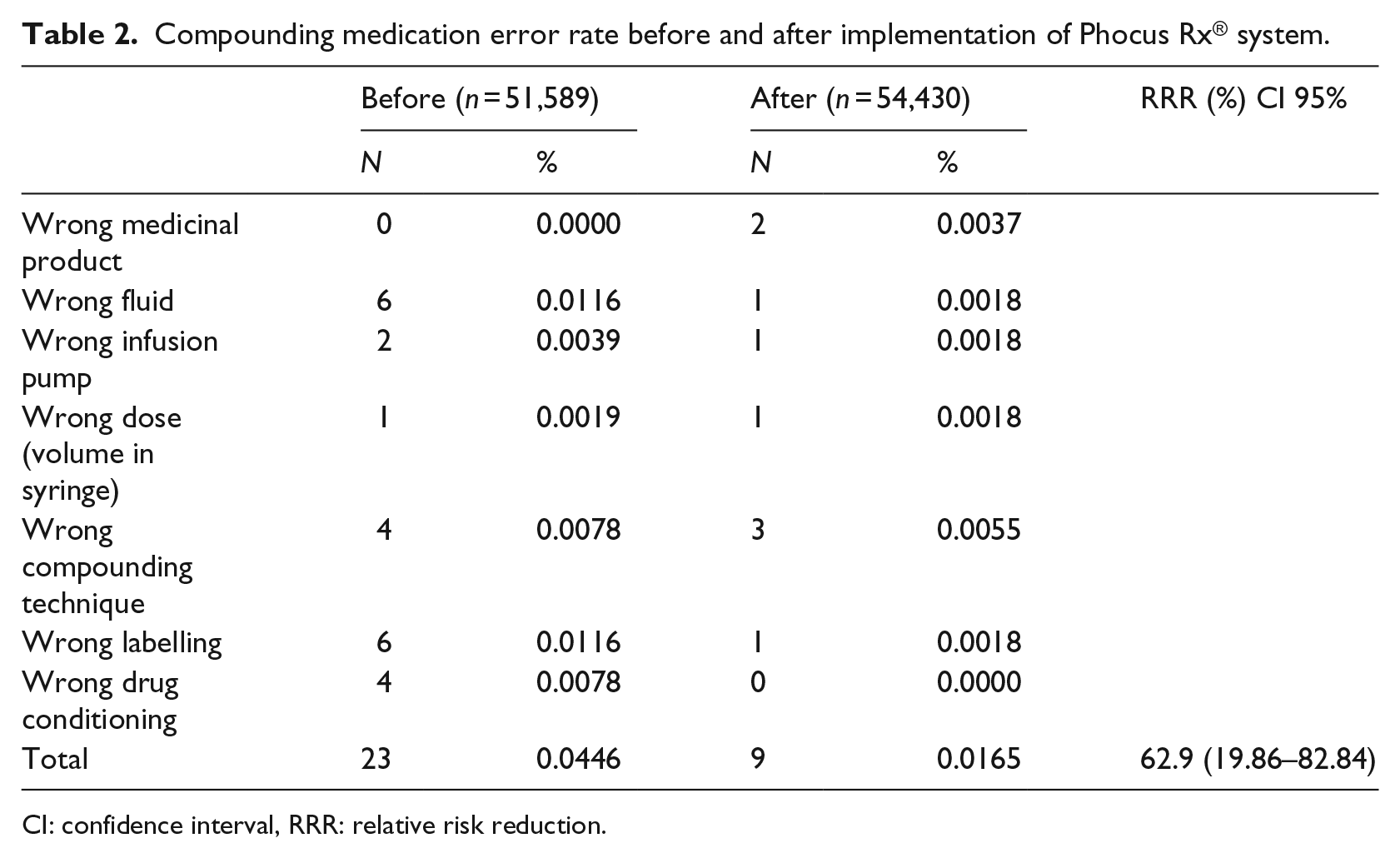
CI: confidence interval, RRR: relative risk reduction.
Most types of compounding errors were reduced after Phocus Rx® implementation. The two wrong medicinal product errors occurred because a different commercial presentation with a different concentration was selected for the treatment, in the context of a shortage. They are the two supposed under-dosing errors, which were not detected before administration to the patient. The vials lacked a barcode and technicians in charge of image verification did not identify the mistaken commercial presentation, accepting the treatment as correct.
Acceptability and further actions
The residual risks after the implementation were analyzed to evaluate their acceptability. Although the majority of them were not considered high risk (under 100 points), additional safety improvements were considered. To reduce the risk associated with each failure mode, recommendations regarding Phocus Rx® development as well as process improvements were described (Table 3).
Ranking for further system actions.
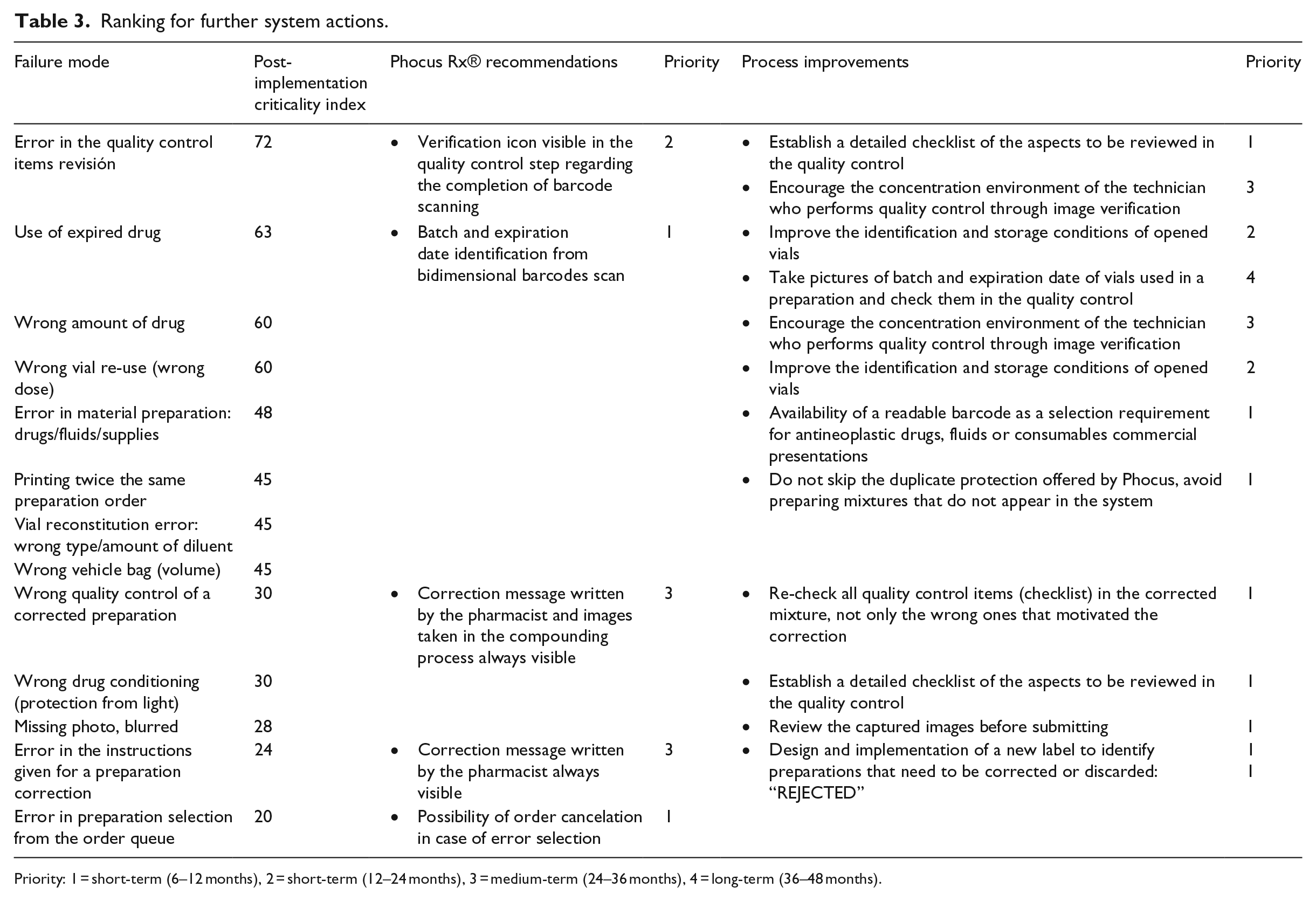
Priority: 1 = short-term (6–12 months), 2 = short-term (12–24 months), 3 = medium-term (24–36 months), 4 = long-term (36–48 months).
The priority initiatives categorized as short-term process improvements were implemented between January and May 2018. To ensure the achievement of recommendations, multidisciplinary sessions were held biweekly in the chemotherapy compounding unit, comprising pharmacists and technicians. In these sessions, the standard operating procedure was reviewed and updated taking into account the new initiatives, like the check-list of detailed aspects to be controlled in the image verification step, or the labels designed to identify the rejected preparations. Incidents related to these instructions were reported. Once solved and clarified, all the pharmacy technicians adopted the resultant recommendations. Moreover, this procedure was added to the welcome plan for new technician staff and a training plan was established to evaluate technicians’ competency before chemotherapy compounding. Simultaneously, a specific Phocus Rx® session was incorporated into the Compounding Technician Course organized in the PD every year.
To enhance the concentration environment of the technicians who perform quality control through image verification, a new organization of technicians’ activities was implemented. This new schedule guaranteed 30-min breaks every two compounding hours and alternated compounding with quality control tasks.
Staff satisfaction
The survey response rate was 100 percent. Overall satisfaction index was 8.30 (SD 1.06) for technicians and 8.56 (SD 1.42) for pharmacists (p = 0.655). Satisfaction was higher for pharmacists than technicians in all items evaluated, except contribution to safety. Specific satisfaction scores are detailed in Figure 6. Overall, some of the most valued items by the staff were the ease of quality control by image verification and the contribution of the system to safety, compounded drug quality and traceability of the process. The item with a lowest score was the compounding speed, with a score for technicians of 4.5 of 10. Although all employees would like the speed of compounding to be increased, all of them confirmed that they would prefer the use of the system to manual quality control and would recommend their implementation to other PD.
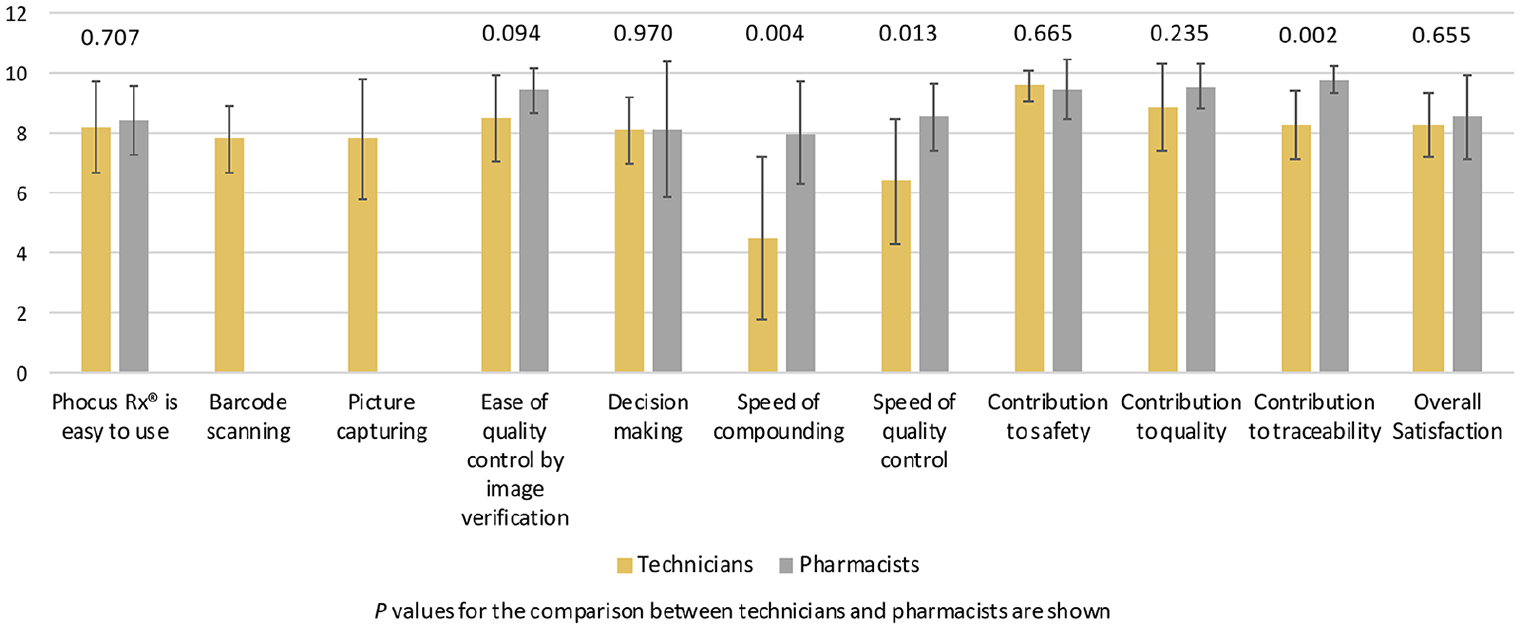
Staff satisfaction with the compounding workflow software system.
Discussion
The introduction of a new workflow management software system was an effective approach to increasing safety in the chemotherapy compounding process in the PD, in accordance with the FMECA method. The total reduction of the criticality index was 67 percent, with a significant reduction of the failure modes with higher criticality (RPN > 100). The relative risk reduction of error rate was 63 percent after implementation of the system, from 0.045 to 0.017 percent, and staff satisfaction was high.
FMECA procedure
In the before-implementation period, the priority order established for the failure modes seems reasonable. The failure modes with the highest RPNs (>100) involve the drug compounding in the BSC subprocess, where the severity of effects is higher and the probability of detection lower. After the implementation of Phocus Rx®, the highest risk reduction was observed in this subprocess (−76%), which corroborates the suitability of the system to improve the most critical points of the antineoplastic drugs compounding process.
In the material preparation subprocess, Phocus Rx® implementation reduced the criticality index in 46%. Although the system does not introduce any new action in this phase, the probability of detecting a material error was higher due to the drug identification step in the software, before starting the compounding. Nonetheless, low implementation of barcodes in primary packaging of commercial presentations represent a limitation in vial identification, 20 which accounted for the two medicinal product errors identified in the post-implementation phase. In this subprocess, the probability of compounding the same drug twice was also reduced, due to the double protection in the system.
In the compounding subprocess, the system’s drug identification step improved fluid selection for reconstitution or dilution and the re-use of opened vials. The detailed image documentation of every step in the compounding process improved the failure modes related to the reconstitution of vials, dose measure and bag labeling.
The criticality index in the quality control subprocess was also reduced 48%, due to the guidance the system offers to check every compounding step through the images. However, the implementation of Phocus Rx® added two new failure modes in this phase, related to instructions given to correct a preparation and quality control of this corrected admixture, but they were classified as low risk (RPN < 100).
Different technologies applied to antineoplastic drug compounding can improve the safety of the process. However, the potential failure modes and their criticality differ from one system to another so their implementation requires a detailed analysis of the risks associated with each one. 24 To our knowledge, this is the first study that analyzes, through the FMECA methodology, the impact of implementing image-based volumetric quality control software on the antineoplastic production process.
Bonnabry et al. 25 analyzed the failure modes and the criticality of all the chemotherapy use process, from prescription to administration, and their changes across five phases of development (from 1999 to 2006). Our study is comparable to their change between phases 3 (CPOE and BSC compounding without quality control) and 4 (compounding guided by CATO® software, based on gravimetric control). The overall criticality index decreased from 2385 to 2081, a 13% reduction. This reduction was based on two failure modes: wrong drug or dose (−81%: 54 vs 288) and incorrect labeling (−63%: 42 vs 122). The higher reduction in the criticality index observed in our study could be due to the analysis of a higher number of failure modes and a different perception of severity associated to them. Criticality reduction on wrong dose or drug is similar in our study (−78%) because gravimetric and image-based technologies can both detect this type of errors. However, wrong labeling was highly reduced in our experience, probably due to the established image-check of the label by a second technician before dispensing the drug in Phocus Rx® system (−96%), which increases detectability.
Medication error study
The existence of medication errors after the implementation of a quality control software like Phocus Rx®, highlights the need of validation of implemented ITs and continuous improvement of the processes.
The overall error rate decreased significantly between pre- and post-implementation periods, and all types of errors, except wrong medicinal product, had lower frequency after Phocus Rx® implementation. The system’s starting product identification step improved fluid and consumables selection, thus decreasing wrong fluid and wrong infusion pump errors. Quality control through image checking was responsible for the decrease in the rest of errors (dose measuring, compounding technique, labeling and conditioning).
However, this technology also has limitations. The lack of barcodes in commercial presentations accounted for the two medicinal product errors identified in the post-implementation phase. The remaining errors after Phocus Rx® implementation could have been detected during the quality control step of the process, emphasizing the importance of implementing process improvements in this phase and working under the stablished procedures. However, the ability to review the image history in the post-implementation phase allowed the pharmacist to detect the errors once the compounded drug had been approved as correct. It gave the healthcare professionals the opportunity to mitigate the errors’ consequences for the patients.
The adoption of such technologies is still accepted only by a few hospital pharmacies, probably because of the high cost. 26 In addition, time management needed for quality control can also be a limitation for medium to high volume hospital PD. Nevertheless, acquisition of this technology can be clearly cost-effective. The cost of the technology and its implementation was estimated at €106,995 the first year and €27,600 for subsequent years, including hiring a pharmacy technician for quality control performance. Compounding errors could reach and cause adverse events to the patients, which were estimated to have an average cost of €5584 according to a 2008 report by the Spanish Ministry of Health and Consumer Affairs. 27 Taking into account the prevention of a higher number of preventable adverse events due to reduction in compounding error rate after Phocus Rx® implementation, this technology prevents compounding errors from reaching a patient as well as it saves costs associated to intercepted errors.
Further actions
Although the implementation of Phocus Rx® has proven a benefit to patient safety, it is important to continue monitoring its use and to identify potential new recommendations and residual risks.
The highest priority was given to the identification of batch and expiration date from barcode reading in the system’s drug identification step, which has already been implemented in new versions of Phocus Rx®. The American Society of Health-System Pharmacists (ASHP) encourages hospital and health-system pharmacies to incorporate barcode scanning into inventory management, compounding and dispensing of medications in order to enhance patient safety and the quality of care. 28 This is crucial in achieving complete traceability of the compounding process and to improving safety in the antineoplastic drug use process. The capture of an image with the batch and expiration date of the vials might seem to be a solution for the quality control of the preparation, but this information would not be available in the system to be reviewed for pharmacovigilance purposes. Given the high number of preparations, this solution would not be operative in any case. However, we should push the pharmaceutical industry to include standard barcodes in the primary packaging, as cardboard boxes should not enter the clean room according to the USP 797. 29
The permanent display of pharmacist correction messages during the compounding process would improve technicians’ information, increasing safety. Moreover, the quality control step could be improved with some icons reflecting the completion of barcode scanning and with the development of a checklist to be completed by the technician, avoiding the omission of any of the items due to a lapse of concentration.
Process improvements should be complementary to the development of the tool and these safeguards can usually be implemented more easily. Thus, developing a detailed work procedure and continuing to train of health professionals who use the tool is essential. Multidisciplinary meetings greatly aid the adoption of improvement measures and enhance a safety culture in the organization, allowing high quality standards to be attained.
Staff satisfaction
This study identified a high rate of staff satisfaction with the workflow software system, although we identified a slightly lower degree of satisfaction in the group of technicians compared with pharmacists (8.30 ± 1.06 vs 8.56 ± 1.42). The item with a lowest score was the compounding speed. However, we should consider that Phocus Rx® has not been compared with other quality control systems, which may well be even slower as they usually incorporate additional control steps during the compounding process, like gravimetric control.
Limitations of the study
The major limitation of FMECA is the subjectivity in the selection of failure modes and calculation of criticality indexes. 19 This bias is minimized by the multidisciplinary composition of the team. In our case, the team formed was well balanced and included different pharmacist profiles involved in oncohematological processes and patient safety, as well as expert technicians in chemotherapy drug compounding. Moreover, frequency, severity and ability to detect a failure mode were determined not only based on other experiences but also taking into consideration the results from our previous observational study conducted in the PD, thus limiting variability. However, it is important to note that an RPN score that designates a failure is not the most important factor, as the main goal is to stratify risks and to determine orders of magnitude among possible failures.
The results of our FMECA cannot be easily extrapolated to other institutions, given the differences in workflows or personnel involved. However, most of the failure modes defined could be applicable to other chemotherapy compounding software systems, depending on the similarity of the process with ours.
Regarding the medication error study, we found the inherent limitation of conducting an uncontrolled before-and-after observational study, in which results may be biased because they lack a control group. However, this methodology is often used when there are practical and ethical barriers to conducting randomized controlled trials.15,30 Specifically, in this study, we would have incurred serious ethical problems if after the Phocus Rx® system implementation in the PD we had not used it to detect compounding errors in the control group, considering the serious consequences that these errors would have had for patients. However, Phocus Rx® system has the limitation of not having a report analysis which could help us to show the error data over time as a time series. The development of these reports would be useful to show the improvement of safety in the compounding procedures over time.
Conclusion
In conclusion, the introduction of a new workflow management software system was an effective approach to increasing safety in the compounding procedures in the PD, according to the FMECA method. It was confirmed with the decrease of compounding error rate in the post-implementation observational study.
Footnotes
Acknowledgements
Grifols is acknowledged for its support to this study. The authors thank pharmacy technicians, Mar Fresno, Raquel Culebras, Ana Canalejas, Estela López, Sofía Parra y Alejandra González, for their help in the risk’s evaluation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
