Abstract
Handover between physicians is a high-risk event for communication errors. Using electronic handover platforms has potential to improve the quality of informational transfer and therefore minimise this risk. This systematic review sought to compare the effectiveness of electronic handover methods on patient outcomes. Articles were identified by searching MEDLINE, EMbase, Scopus and CINAHL databases. Studies involving electronic handover between two healthcare personnel or teams, and which described patientspecific outcomes, were included. This search yielded 390 articles, with a total of nine publications included in the analysis. Outcomes reported in studies included length of stay, adverse event rates, time to procedure and handover completeness. This review suggests that e-handover may improve the handover completeness; however, it is unclear at this time if that translates to an improvement in patient care. The lack of reliable evidence highlights the need for further research exploring the effect of e-handovers on patient care.
Introduction
Healthcare is interdisciplinary, and patients are cared for by a range of health care providers (HCPs) across primary, secondary and tertiary care. 1 Accurate transfer of information between HCPs is crucial to maintaining patient safety. 2 The World Health Organization has recognised that this transfer of information – generally referred to as a ‘handover’ or ‘handoff’ – is a high-risk event for patient safety, and miscommunications during handover are often responsible for patient harms. 3 Despite handover between HCPs being recognised as a high-risk event, improving handover quality has remained challenging with up to 80 per cent of medical errors involving miscommunications.2,4
Instituting a systematic and uniform approach to communication processes reduces the opportunity for error; however, problems arise when these systems are too rigid to allow for the range of complexities encountered by HCPs. 5 The predominant barrier to initiating a universal solution is the multitude of circumstances and settings in which handovers take place. 3 Health information technologies such as electronic handover (e-handover) platforms are being increasingly utilised to overcome the logistical challenges of healthcare.6,7
E-handovers allow greater flexibility for HCPs to handover their patient using a structured platform that can be customised to meet the needs of user groups, without the need for HCPs to be simultaneously present during the exchange. E-handover software can be integrated with a patient’s full electronic health record to pre-populate fields and ensure all HCPs involved with the patient’s care have access to the information provided at each transition of care point, improving efficiency for users.8,9 Once created, e-handover information fields can then be adjusted as part of quality improvement cycles without the need for widespread educational interventions which are often tedious to implement and hinder systematic improvements. 10
No systematic reviews could be identified from existing literature specifically examining the effect of conducting e-handovers on patient outcomes. This systematic review aims to compare the effectiveness of e-handover methods on patient outcomes.
Methods
This systematic review was conducted in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 11
Data sources and search strategy
The electronic databases searched were MEDLINE via Ovid, EMbase, Scopus and CINAHL. The literature search was conducted in March, 2018, with no time limits for publication applied, as the use of e-handovers has only recently become prevalent and all research published has been recent and remains relevant.
The Population–Intervention–Comparison–Outcome (PICO) framework was used to define the search terms. 12 The search terms used were (P) patient OR healthcare AND (I) electronic hando* OR e-referral OR ereferral OR electronic referral AND (O) Continuity of care OR Quality of care OR Quality of documentation OR safe* OR efficien* OR accura*. The following search terms were also originally trialled however returned irrelevant or duplicate results only: (I) EHS, Health data network, EHR, Electronic health record, digital referral, digital hando*, e-handover, (O) time, length of stay, readmission, continuum of care, compliance, admission, quality of communica*.
All articles were then loaded into Covidence©, 13 a web-based referencing and data extraction software tool, where duplicates were removed. The titles and abstracts of all studies were then screened by two independent reviewers for eligible articles, with conflicts resolved via phone discussion between the two reviewers. Remaining articles underwent a full text review against the inclusion and exclusion criteria, performed independently by two reviewers. Any conflicts were resolved via a discussion between the two reviewers. The reference lists of all included articles were then hand searched for relevant title and abstracts, with no further articles located. The remaining articles proceeded to data extraction.
Inclusion and exclusion criteria
The following inclusion criteria were utilised: involves e-handover between two or more healthcare personnel or teams; and includes quantitative data in regards to patient outcome (e.g. time to diagnosis, time to treatment, length of stay, readmission rates, adverse event occurrences, morbidity, mortality or handover accuracy). The criteria for exclusion from the review were as follows: not a peer reviewed publication; or not available in English; or no full text available such as conference abstracts; or health-economics-based research; or e-consults; or e-booking data; or implementation protocols without data; or survey design as the sole methodology used in the study; or qualitative papers.
Study quality assessment
All included study designs were pre–post interventional studies, so the 12-point Quality Assessment Tool for Before-After (Pre-Post) Studies with No Control Group published by the National Heart, Lung & Blood Foundation was chosen as a quality assessment tool. 14 Each individual article was assessed independently by two reviewers, and conflicts were then discussed between the two reviewers. The body of evidence was then assessed by the primary reviewer as per the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group guidelines. 15
Synthesis of findings
Data were extracted by the primary reviewer using the following fields: (a) study setting, (b) number of participants, (c) intervention performed, (d) comparison prior to intervention, (e) outcomes measured and (f) risk of bias. Due to the heterogenous nature of the outcome measures, a meta-analysis was not appropriate and a narrative synthesis of data was used. Articles were grouped broadly into (a) length of stay, (b) adverse events, (c) time to procedure and (d) handover completeness for discussion purposes and comparison of individual study results.
Results
Search results
The search strategy returned 390 articles. There were 130 duplicates which were excluded. Of the 260 articles screened at the title and abstract level for relevance, 217 studies were deemed irrelevant and excluded. The reviewers screened 43 articles at a full text level and excluded 34 articles. Full text was unavailable in 21 of the studies, so they were excluded as were three studies which were not peer reviewed. E-consults were used in two articles as the intervention, and were therefore excluded. Implementation protocols without outcomes were reported in two studies, which were excluded. A further six articles were excluded due to their survey design as it was considered by the reviewers to be an unreliable measure of patient outcomes. After this exclusion stage, nine studies proceeded to data extraction. No new studies were identified through hand searching (Figure 1).
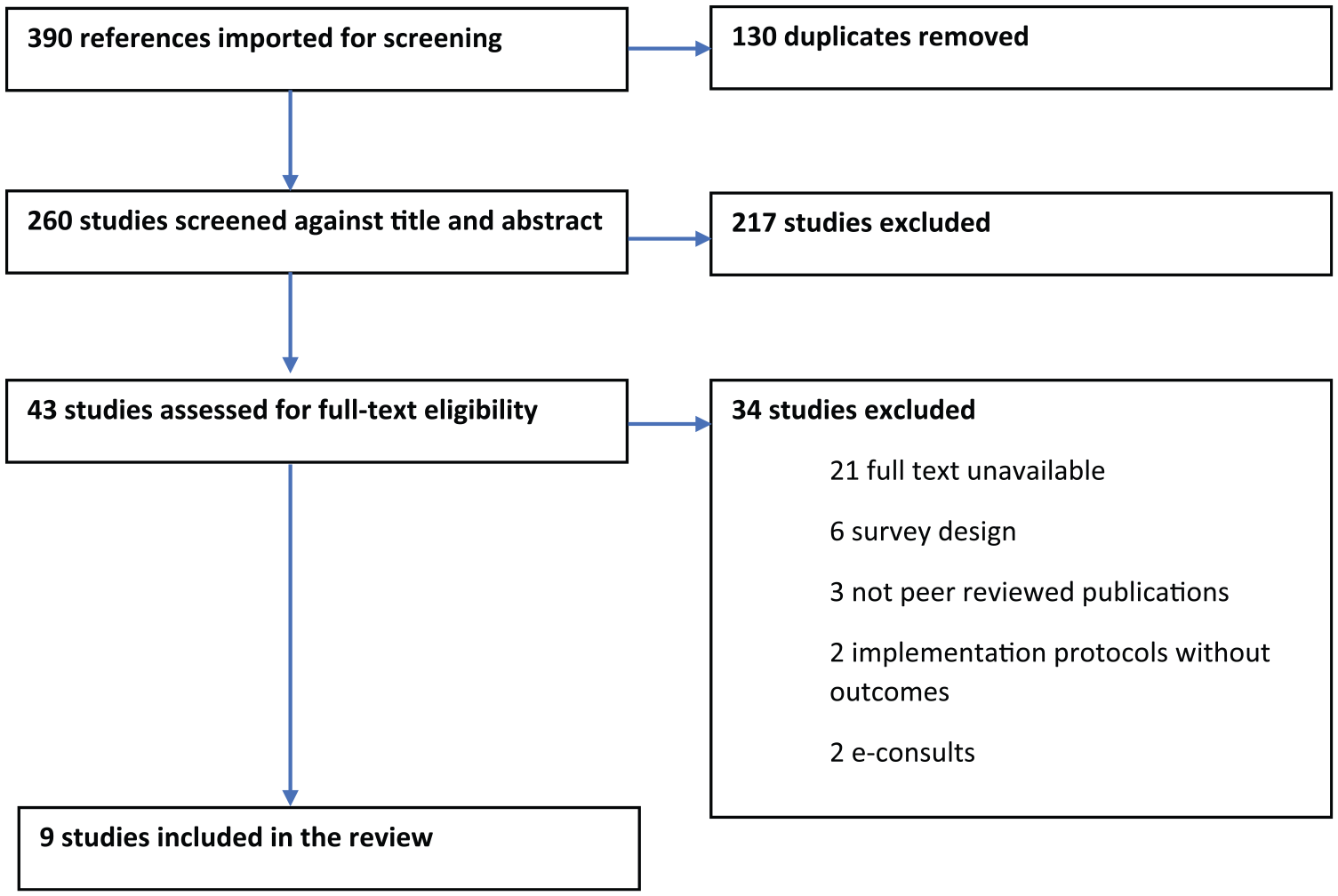
PRISMA flow chart.
Result characteristics
Publication dates ranged from 2009 to 2018. Studies originated from the United Kingdom (n = 4), the United States (n = 3), Taiwan (n = 1) and Ireland (n = 1). Study settings varied considerably and included handovers between residents on a ward (n = 3), from emergency department (ED) to a ward (n = 2), from general practice physicians to an outpatient urology clinic (n = 1), interhospital transfer (n = 1), from hospital wards to community pharmacists (n = 1) and from a hospital ward to an integrated hospital discharge team (n = 1). Number of handovers studied in each article also varied considerably. A third of the included articles studied ⩽100 handovers, a third studied between 101 and 1000 handovers and a third of the studies contained more than 1000 handovers, with the mean number of handovers being 828. All studies were of a pre–post interventional design (Table 1).
Characteristics of studies included in the analysis.

GP: general practitioner; STEMI: ST-elevation myocardial infarction; ED: emergency department.
Quality assessment
Risk of bias
As per the National Heart, Lung & Blood foundations’ Quality Assessment Tool for Before-After (Pre-Post) Studies with No Control Group, all studies were poor methodological quality. No included studies used blinding or an interrupted time-series design. Only four of the nine studies clearly described and prespecified the eligibility criteria for the study population, and five included all eligible participants. Outcome measures were not clearly defined or applied consistently in two of the studies. Nazar et al. 19 failed to clearly describe their aims for their study and also lost 69 per cent of their participants to follow up, well over the 20 per cent threshold for high bias. Singleton et al. 24 failed to utilise a large enough sample size to provide confidence in the findings. All studies provided a clear description of their e-handover and used the same e-handover format throughout their individual study. All studies used statistical methods to assess changes in outcome measures, taking individual measures into account and presenting p values. Individually, all articles therefore had multiple methodological flaws and were of poor quality.
GRADE assessment
As per the GRADE recommendations, the body of evidence starts as ‘low’ quality, due to the observational nature of the individual studies. 15 The significant inconsistency of outcome measures across the studies, combined with the high risk of bias and overall low quality of individual articles, further decreases certainty that the estimated effect of e-handovers aligns with the true effect of implementing the intervention. Therefore, the overall body of evidence is ‘very low’ quality.
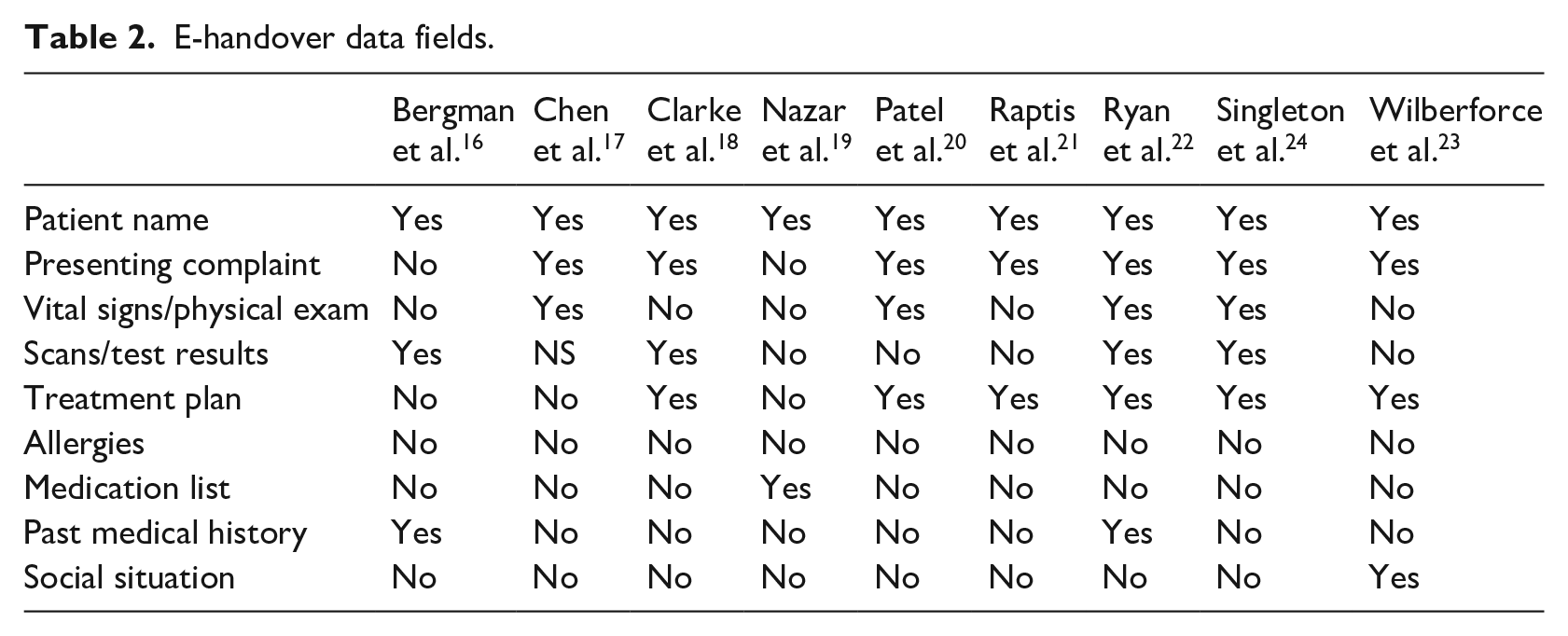
Pre-intervention studies reported handover occurring via handwritten paper referrals or pro formas (n = 5), via phone call (n = 1) or no formalised handover process in place (n = 3). The intervention for all studies was the implementation of structured e-handover platforms; however, the data fields within each handover protocol differed. For the purposes of this review, the studies included fields were compared against the IMISTAMBO (Identification, Mechanism/Medical Complaint, Injuries, Signs, Treatment, Allergies, Medications, Background Information, Other information) mnemonic which is an accepted handover format in the hospital and prehospital environments. 25 All e-handover platforms included patient identification. Presenting complaint or diagnosis was recorded in seven studies. Only four of the studies reported recording patient’s vital signs or physical examination results. Patient investigation scans or results were attached in four of the studies. Expected treatment or management plan was handed over in six of the studies. Only one study reported mandatory recording of patient medication lists, while no studies had a mandatory field for patient allergies. Past medical history was recorded in two studies, with patients’ social history being recorded in one study (Table 2).
E-handover data fields.
Narrative synthesis
Patient outcomes is a broad term that encompasses a range of non-specific measurements. This study aimed to include as many measures of patient wellbeing as possible, as well as the proxy outcome of handover completeness, given that handover completeness has been shown to be directly affect patient care in other studies.26,27 A variety of outcome measures were reported, which will be compared under the following subheadings: length of stay, adverse events, time to procedure and handover completeness (see Table 3).
Results synthesis.
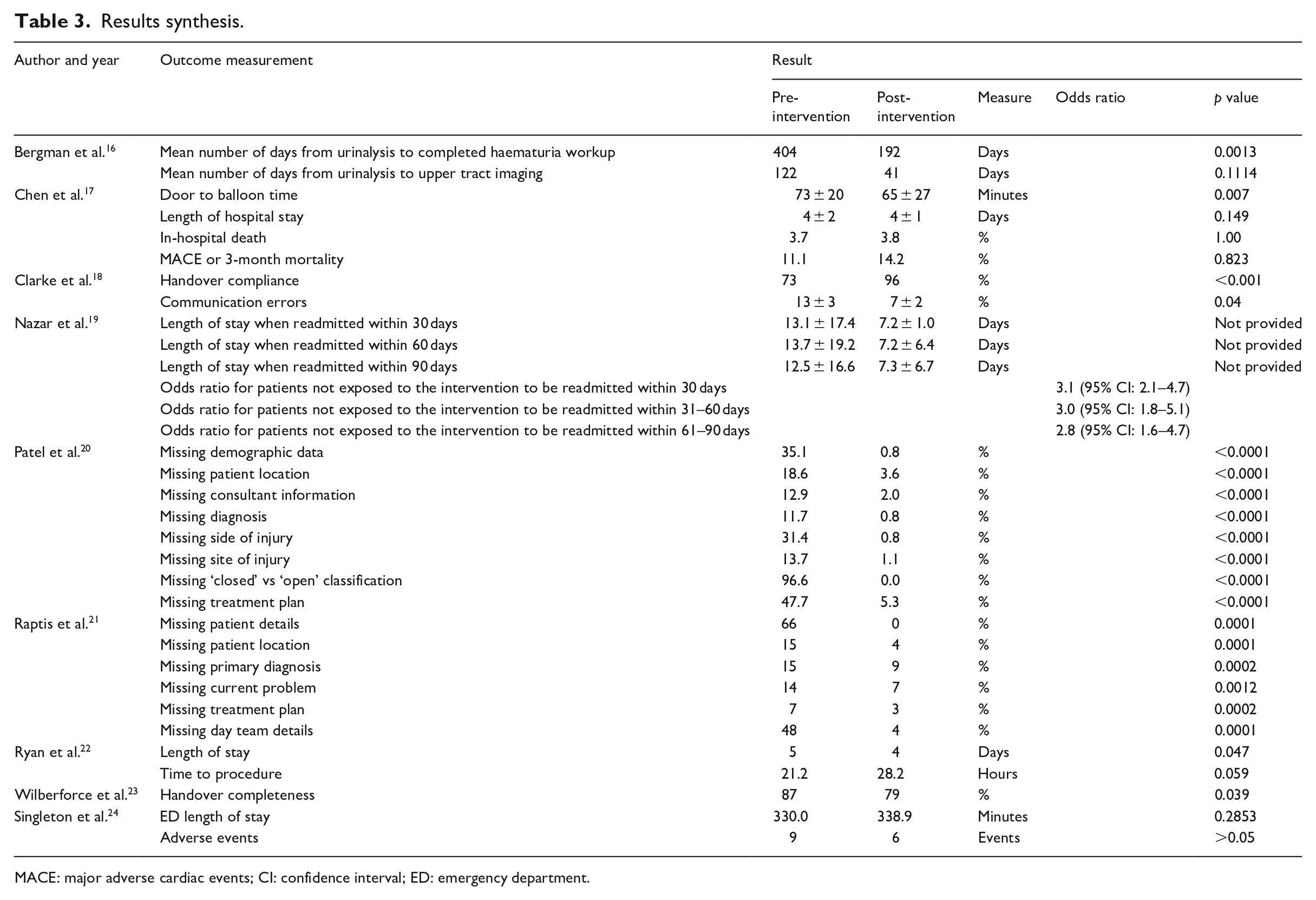
MACE: major adverse cardiac events; CI: confidence interval; ED: emergency department.
Length of stay
Length of stay was reported in three studies, with conflicting results. Nazar et al. 19 found that where patients were unexpectedly readmitted within 30, 60 or 90 days, their length of stay was reduced by 5.8, 6.5 or 5.2 days if they were exposed to the intervention. Ryan et al. 22 also reported a reduced median length of stay from 5 to 4 days (p = 0.047). Singleton et al. 24 and Chen et al. 17 however showed no statistically significant change in length of stay following implementation of e-handovers.
Adverse events
Frequency of adverse events was reported in three studies; however, each study defined adverse events differently, so the results are expectedly inconsistent. Chen et al. 17 and Singleton et al. 24 report no statistically significant difference in adverse events before and after implementation of e-handovers. However, Nazar et al. 19 found patients were 3.1, 3.0 or 2.8 times less likely to be readmitted to hospital within 30, 60 or 90 days, respectively, post-intervention.
Time to procedure
Time to procedure was included as an outcome in three studies. Bergman et al. 16 reported a reduction in time for lower urinary tract imaging from 404 days before e-handover and 192 days after implementation (p = 0.0013). Upper tract imaging was reduced from 122 days before e-handover, and 41 days after (p = 0.1114). Chen et al. 17 also reported a statistically significant decrease in time from ED arrival to percutaneous coronary intervention (PCI) lab from 73 ± 20 min to 65 ± 27 min (p = 0.007). Ryan et al. 22 however found no statistically significant change in time to procedure after e-handover implementation.
Handover completeness
Handover completeness was assessed in four studies. Three of the four studies reported increases in the amount of detail recorded during handover following intervention implementation. Each study defined handover completeness differently however, making the results non-comparable. Clarke et al. 18 showed an increase in trainee handoff compliance from 73 per cent to 96 per cent (p < 0.001) and decreased errors in communication by 50 per cent (p = 0.044). Raptis et al. 21 found patient details inclusion increased from 34 per cent to 100 per cent (p = 0.0001), patient location from 86 per cent to 96 per cent (p = 0.0001), primary diagnosis from 85 per cent to 91 per cent (p = 0.0002), current problem from 86 per cent to 93 per cent (p = 0.0001), plan of action from 93 per cent to 97 per cent (p = 0.0002) and day team details from 52 per cent to 96 per cent (p = 0.0001). 21 Similarly, Patel et al. 20 reported that following the introduction of e-handover, patients with missing demographic data reduced from 35.1 per cent to 0.8 per cent (p < 0.0001), missing patient location from 18.6 per cent to 3.6 per cent (p < 0.0001), missing consultant information dropped from 12.9 per cent to 2.0 per cent (p < 0.0001), missing diagnosis from 11.7 per cent to 0.8 per cent (p < 0.0001), missing information regarding side and anatomical site of the injury was reduced from 31.4 per cent to 0.8 per cent (p < 0.0001) and from 13.7 per cent to 1.1 per cent (p < 0.0001), respectively. Recording of ‘closed versus open’ injury pattern increased from 3.4 per cent in paper ad hoc handovers to 100 per cent in the e-handover group (p < 0.0001). A treatment plan was included only in 52.3 per cent of paper handovers compared to 94.7 per cent (p < 0.0001) of e-handovers. Contrastingly, Wilberforce et al. 23 reported that 21 per cent of e-handover documents contained incomplete fields versus 13 per cent of paper documents (p = 0.039). 23
Discussion
The aim of this systematic review was to understand how conducting patient handover electronically affects patient outcomes. Only nine studies were found for this review, and a lack of high-quality, consistent evidence in this field was identified. Due to the incongruent measures of patient outcomes, a meta-analysis was not possible, and terminology throughout the studies was highly inconsistent. Future research in this area should focus on protocol transparency and reproducibility.
Length of stay
There are several explanations for why the studies had inconsistent results regarding patient length of stay. Nazar et al. 19 reported a reduction of length of stay when e-handovers were utilised; however, the 95% confidence intervals were excessively wide and crossed zero, rendering the results statistically insignificant. They also failed to state how patients were chosen for recruitment into their study, so it is possible that selection bias is responsible for their substantial reduction of length of stay compared to the other studies.
Nazar et al. 19 implemented a new protocol where no handover process was in place prior, so their study is a comparison of e-handover to no handover, whereas the other three studies compared e-handovers to other handover formats, offering a possible explanation for why the other studies produced less drastic reductions. It is plausible that e-handover offers a way to implement beneficial informational transfer where other handover formats (such as face-to-face) are impractical.
Theoretically, using an e-handover platform to increase handover completeness should minimise length of stay by reducing unnecessary test repetition and adverse event rates. 26 Future research to determine if increasing handover completeness decreases unnecessary test repetition would be helpful to explore how e-handovers impacts on patient length of stay.
Adverse events
The definition of an ‘adverse event’ is specific to each study, and there is no agreed upon definition between the included studies. Chen et al. 17 defines adverse events as in-hospital death, ‘major adverse cardiac events’ (MACE) and all cause 3-month mortality rates. MACE itself however is a problematic term in that it is inconsistently defined among the literature and ranges from a patient requiring stent thrombosis to cardiac death. 28
Singleton et al. 24 do not list their entire definition of adverse events. They state that quality assurance (QA) events are automatically triggered by adverse events such as ED-to-ICU (intensive care unit) transfer, or death within 24 h. Non-automated QA events include physician complaints for communication errors and diagnostic error. This suggests that other automatic triggers are used other than the two listed, therefore decreasing transparency in their methodology. Furthermore, non-automated QA events are unreliable. Physician complaints against their colleagues for errors are likely to underestimate the amount of communication and diagnostic errors, as a multitude of factors including gender, position and previous experience with disclosure factor into the decision-making process of physicians when reporting errors.29–31
The complexity of measuring adverse events has been noted in research for many years, and no gold standard for defining and measuring adverse events has been universally endorsed despite multiple studies examining the way adverse events are reported.32–36 More evidence reporting on specific, well-defined outcomes is needed to effectively examine the way that e-handovers relate to adverse patient events.
Time to procedure
The three studies that measured time to procedure as an outcome produced varied results. The two studies demonstrating a reduction in time to procedure used an appropriate intervention likely to benefit the patient, whereas it is not clear if the other study chose an appropriate intervention to measure as an outcome.
Bergman et al. 16 and Chen et al. 17 both listed time to procedure as the primary outcome being investigated in their study and showed a significant reduction in time to procedure when e-handover is utilised to facilitate information sharing between physicians. Bergman et al. 16 measured time to completed urology workup, the primary reason for patients being handed over the specialists in a urology clinic. Chen et al. 17 measured time from presentation (‘door’) to PCI (‘balloon’), considered the gold standard for ST-elevation myocardial infarction (STEMI) patients, the cohort being studied. 37 Therefore, the intervention measured can be considered an appropriate variable.
In comparison, Ryan et al. 22 found no correlation between e-handover and time to procedure; however, a reason for this conflicting result may be that the intervention measured for all patients was computed tomography (CT) scan. It is not clear if all patients required a CT scan, or if it was relevant to their diagnosis or management. This lack of transparency decreases the likelihood that their result is an accurate representation of the true effect of e-handovers on time to procedure.
While further evidence is needed to determine if e-handover does reduce the time to procedure, this initial evidence does suggest that there may be a link that should be further explored. Decreasing time to procedure effectively reduces mortality and morbidity in patients suffering from a range of life-threatening conditions such as sepsis, acute myocardial infarction and cerebral vascular accidents.38–40 Therefore, this is an important outcome that should be further explored to meaningfully improve patient wellbeing.
Handover completeness
Handover completeness showed the most consistency between individual study results of the discussed outcomes. Implementation of e-handover considerably improved handover completeness in three of the four studies.
Wilberforce et al., 23 the only study to demonstrate a reduction in handover completeness post-implementation failed to describe if their e-handover platform had the same number of fields as the paper documentation or how they selected handovers to audit. It is therefore unclear if their findings are a result of selection bias or a genuine reduction in completed handovers. Wilberforce et al. 23 note that the limitations of their study include that their particular e-handover platform (‘Sharecare’) was tedious and not user-friendly, which could also explain their lack of success with improving handover completeness.
It is impossible to accurately compare the four studies against each other when the e-handover systems all differ in the information recorded, and the method of inputting the data differs between each platform. See Table 2 for included data fields. In 2009, a systematic review found no conclusive evidence to suggest superiority of any mnemonics for structured handover. 41 Since the publication of that review, no further systematic reviews have been performed to identify a gold standard structured handover. Additional studies comparing different e-handover platforms against one another are needed to establish a gold standard of e-handover field inclusions and format. This could minimise inconsistencies between studies and overcome the limitations of excessively heterogenous data that prevent clear comparison in this review.
Strengths and limitations
Optimal handover format is likely to be specific to individual specialties, settings and professions as there is a difference in perceived effectiveness of different handover types between healthcare professionals such as nurses, physicians and allied health professionals. 42 The diverse participant settings which included both in- and out-patients being seen by various specialists, decreases confidence in the findings of the review. When a larger body of available evidence allows, examination of e-handover in individual settings is likely to produce more meaningful results about which patient cohorts benefit the most from e-handover.
As highlighted throughout the discussion, the predominant limitation to this review is the significant heterogeneity of both terms used within the literature, and the conflicting results produced. The small sample of included articles further reduces confidence in the findings. A total of 21 articles were excluded as full text was unavailable, most of which were conference abstracts only. The results of these studies may have been highly relevant and applicable, clarifying whether e-handover improves patient care; however, without publishing the research for peer review and methodological appraisal, these studies’ lack the detail required to ensure they meet the review criteria. They also lack details in the results to allow an accurate interpretation of their findings.
This review was also focused on directly measurable patient outcomes or handover completeness. It is likely that there is flow on effects to patients from other outcomes that e-handover effects such as improved workflow freeing up physicians’ time for other clinical tasks and decreasing waiting list times.9,43,44 A more comprehensive analysis of the effects of e-handover on all stakeholders would be helpful for organisations deciding if implementation of e-handover systems would be beneficial to their stakeholders.
Conclusion
This systematic review highlights the lack of consistent or high-quality evidence evaluating the effectiveness of utilising e-handovers on improving patient outcomes. While the results suggest that handover completeness is likely to be enhanced by the implementation of a user-friendly electronic platform, it is unclear if that translates to an improvement in patient care. The diversity of results may indicate that e-handover is more effective within particular settings or specialties; however, due to the lack of robust and high-quality studies, this is currently unclear. If more organisations using e-handovers were to report on the effect of the intervention, it may clarify which patient cohorts are most likely to benefit from the implementation of e-handovers. In conclusion, the results of this review show a clear need for further research in the field of e-handovers to improve the available body of evidence.
Footnotes
Acknowledgements
The authors are grateful to Ric (Frederic) Bouvier and his family, for the support and inspiration in completing this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
