Abstract
For the treatment choice of localized prostate cancer, effective patient decision aids have been developed. The implementation of decision aids in routine care, however, lags behind. Main known barriers are lack of confidence in the tool, lack of training on its use, lack of resources and lack of time. A new implementation strategy addresses these barriers. Using this implementation strategy, the implementation rate of a decision aid was measured in eight hospitals and questionnaires were filled out by 24 care providers and 255 patients. The average implementation rate was 60 per cent (range 31%–100%). Hardly any barriers remained for care providers. Patients who did not use the decision aid appeared to be more unwilling than unable to use the decision aid. By addressing known barriers, that is, informing care providers on the effectiveness of the decision aid, providing instructions on its use, embedding it in the existing workflow and making it available free of charge, a successful implementation of a prostate cancer decision aid was reached.
Keywords
Background
For primary localized prostate cancer, different treatment modalities are available: active surveillance, surgery, interstitial radiotherapy (brachytherapy) and external beam radiotherapy. For most patients with localized tumours, current guidelines do not indicate one ‘best’ option.1,2 The treatments are comparable for survival, but differ with respect to side effects. This implies that patient values and preferences should play a key role. A useful tool to involve patients in the choice of their treatment is a decision aid (DA).
A DA is a tool (booklet or website) with two goals: (1) to inform patients about the risks and benefits of their treatment options and (2) to stimulate patients to consider which risks or benefits are most important for them. According to the International Patient Decision Aid Standards (IPDAS), DAs should at least contain the following main elements: explaining the fact that there is a decision to be made, describing the treatment options with their pros and cons and helping patients to make a personal trade-off by presenting the pros and cons in a structured and balanced way.
DAs appear to have effects on patients, as shown in Lin et al. 3 and Stacey et al. 4 DAs lead to more knowledge about treatments, more realistic expectations and more shared decision-making without increasing anxiety or regret. With DA, patients are more certain about the choice, the choice reflects what matters to the patient and fewer patients want intensive treatment.3,4 Although some patients prefer to leave the decision to the physician, the vast majority of patients consider it a good idea to be offered the possibility to choose, even of patients who decline to make their own treatment choice. 5
Given the fact that DAs have been shown to be effective, more effort needs to be made to implement this evidence 6 as the implementation of DAs in routine care lags behind. 7 To date, many patients who are eligible for more than one treatment option, do not receive a DA. A systematic implementation strategy aims to improve this. According to the model by Grol, 8 for successful implementation, different players in health care should be involved, interventions should be adapted to local needs and barriers to change should be identified. Literature reviews have identified barriers for implementation of DAs; main barriers reported by physicians were lack of confidence in the content, lack of training on how to use decision support, fear of disruption of the workflow, time constraints, low accessibility and high cost.7,9–11 For example, a recent study concluded that implementation would be facilitated by informing professionals about the positive effects of the DA and by embedding the DAs in the existing workflow. 12
We designed an implementation strategy for DAs, in which we incorporated the implementation lessons from earlier studies.7,9–11 The implementation strategy described in this study applies to both web-based and printed DAs. The strategy focuses on removing identified barriers. Respectively, lack of confidence, lack of skills, fear of workflow disruption, time concerns, accessibility and cost are addressed (see the ‘Methods’ section for more details). This implementation strategy was used to implement a recently developed DA booklet for localized prostate cancer.13,14 DAs have been shown to be equally effective in web-based and in paper format.15,16
In short, we developed and applied a strategy to achieve implementation of a DA, aiming to provide DAs to as many eligible patients as possible. Eligible patients are all patients who have more than one treatment option to choose from. Aim of our study was to measure the implementation rate of this tool, when using the implementation strategy. To date, studies lack information on the implementation rate that was actually achieved. 7 Second, as the usage of DAs is determined both by professionals providing DAs and by patients using those DAs, we aim to identify remaining barriers and facilitators for use of the DA as perceived by both health care professionals and patients.
Methods
The implementation rates of a DA for prostate cancer were calculated per hospital, and barriers for both care providers and patients were assessed by questionnaire.
Implementation strategy
The implementation strategy is presented in Table 1. Barriers that were identified in the literature were taken into account. (1) Lack of confidence in the content was addressed, in each participating hospital, by explaining the content of the DA to the care providers, together with the results of a randomized trial13,14 showing its effectiveness. (2) To reduce chance of disruption of workflow, local health care professionals were involved in the logistic decisions on how to best embed the DA in the existing care path in their own hospital. (3) To address accessibility and cost barriers, the DAs were provided to the hospitals free of charge. (4) To address the lack of training on how to use decision support, the following instructions for the use of the DA were incorporated in the above presentations: (a) make clear there is a treatment choice to be made, (b) identify the treatment options for this individual and mark these in the DA booklet, (c) discuss pros and cons of each option, (d) stimulate the patient to think about which pros and cons are most important to him, (e) give the DA with this information to take home, allow the patient some time to consider, (f) discuss, and reach a shared decision. These instructions were also handed out to the health care professionals on plasticized cards at the start of the implementation phase. As shown in Table 1, our implementation strategy also included several monitoring and feedback steps (step 5 through step 8) and a survey of other potential barriers and facilitators (step 9).
Implementation strategy.

Procedure
We studied the implementation of a DA for localized prostate cancer. This DA has been shown to improve patients’ knowledge and participation in decision-making 13 without inducing anxiety or regret. 14 It consists of a booklet to be used during the consultation and to take home for further consideration. The booklet describes the treatments, it compares outcome information, that is, the likelihood of cure and of the side effects associated with each treatment, and a table listing the pros and cons of each treatment. The outcome information is presented for two risk groups: (A) Gleason ⩽ 7 and prostate-specific antigen (PSA) ⩽ 20 and T1T2 or (B) Gleason > 7 and/or PSA > 20 and/or T3. On a dedicated first page of the DA booklet, the care provider indicated which treatment options are open to this individual patient and which risk information applied (A or B). The DA booklets were distributed to the hospitals. Care providers were asked to hand out and go through the DA with each eligible patient, according to the instructions described above as item 4 of the implementation strategy. All patients who have more than one treatment option are eligible. The only exclusion criterion is insufficient Dutch language proficiency. Depending on the local care path, this could be done by a urologist or a specialized nurse. In addition, the care provider asked patients whether a researcher could contact them about the study. The care provider documented whether or not the patient agreed with the telephone call and gave this information to the researcher. The researcher (J.J.v.T.-G.) explained the aim of the study to the patients by telephone and, after verbal informed consent, sent a written consent form, a questionnaire and a reply-paid envelope.
Questionnaires
Health care professionals filled out an online questionnaire after using the DA for a period of 6 to 8 months. This questionnaire was based on the MIDI tool, 17 which identifies barriers and facilitators at the level of the innovation (DA), the user (health care professional) and the organization (hospital).
Patients filled out a questionnaire 2 weeks after they received the DA. It contained questions on the presentation and content of the DA, whether they had used the DA and possible barriers and facilitators for the use of the DA.
Analyses
Our primary outcome measure was the implementation rate per hospital per year. The implementation rate per hospital was defined as the number of patients who received the DA divided by the number of eligible patients as estimated from a registry. More precisely, for the numerator, patients were counted who received the DA and who were also enrolled in the study. For the denominator (number of eligible patients), hospital-specific data were extracted from the National Cancer Registry of the Netherlands (IKNL). The number of patients with prostate cancer, excluding clinical T4, N+ and M+, were counted per hospital over the preceding 6 years and corrected for the duration of inclusion.
Barriers for care providers who used the DA 10 times or less were compared to colleagues who used the DA more often. For each barrier of facilitator, the percentages of care providers that endorsed it were analysed using chi-square tests. Patients who did or did not use the DA were also compared using chi-square tests. To correct for the number of barriers and facilitators tested, a Bonferroni correction was applied by dividing the significance level α by the number of barriers/facilitators.
Results
Patients and health care professionals
Ten hospitals in the region were asked to participate in the study, of which eight hospitals agreed. Two declined because they already participated in a study on DAs. Between spring of 2013 and summer of 2014, 30 health care professionals presented the DA to 316 patients. Of these patients, 11 were not eligible for the study, because additional diagnostics showed they had tumour growth outside of the prostate (N = 8) or had other health issues (N = 3) which meant they did not have a choice of treatment options. Of the remaining 305 patients, 294 (96%) gave informed consent and 255 (84%) returned the questionnaire. Of the 30 health care professionals, 24 filled out their questionnaire, representing a response rate of 80 per cent. The characteristics of the participating patients and care providers are listed in Table 2.
Characteristics of the professionals and patients who filled out the questionnaires.
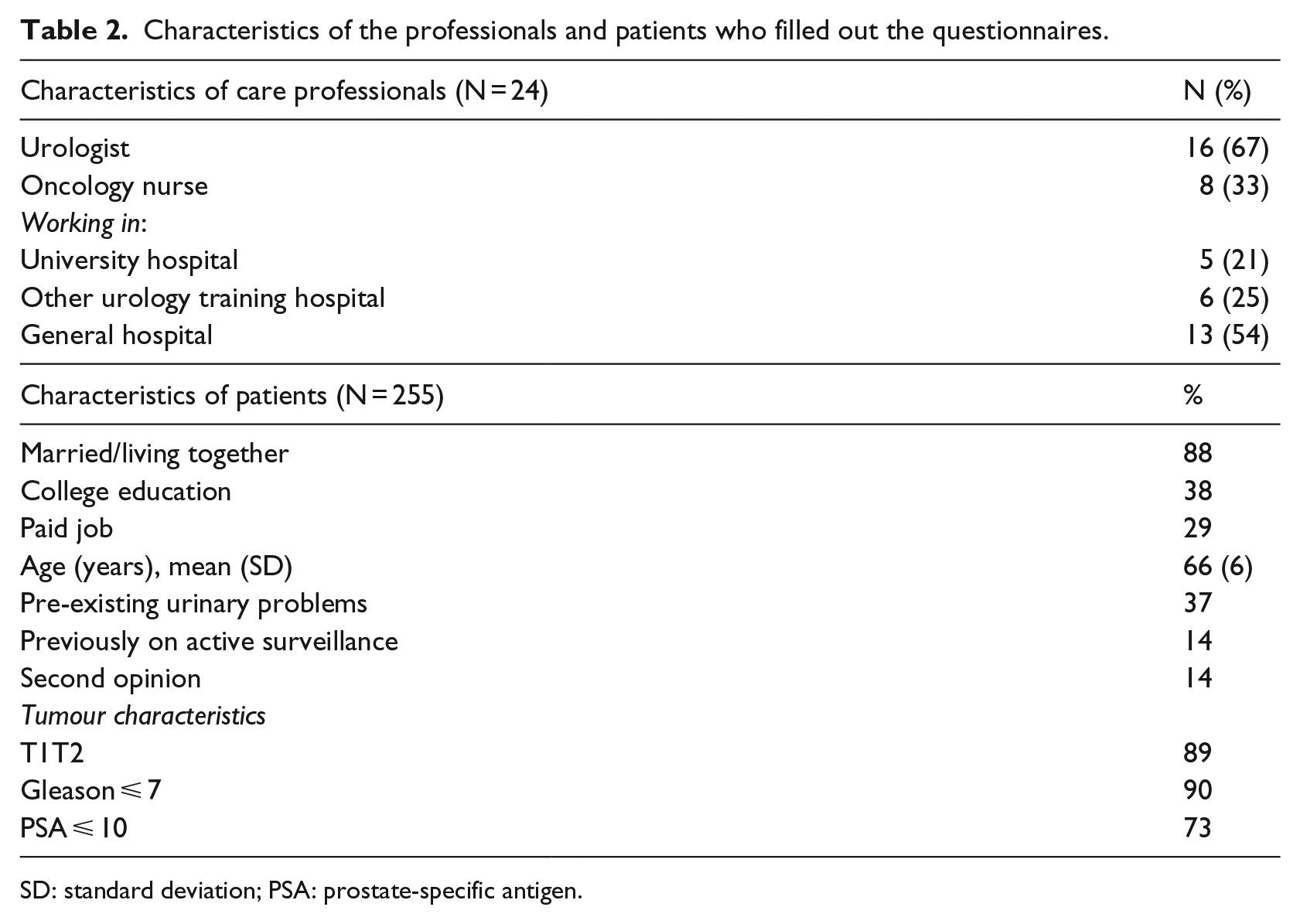
SD: standard deviation; PSA: prostate-specific antigen.
Implementation rate
Table 3 presents the estimated implementation rate of the DA. The number of patients who received the DA and were enrolled in the study in each hospital (A) was divided by the estimated number of eligible patients in each hospital (B). Since one hospital did not give permission to obtain registry data, the results are presented for seven of the eight hospitals. Overall, the average implementation rate was 60 per cent, ranging from 31 to 100 per cent.
Estimated minimal implementation rate.
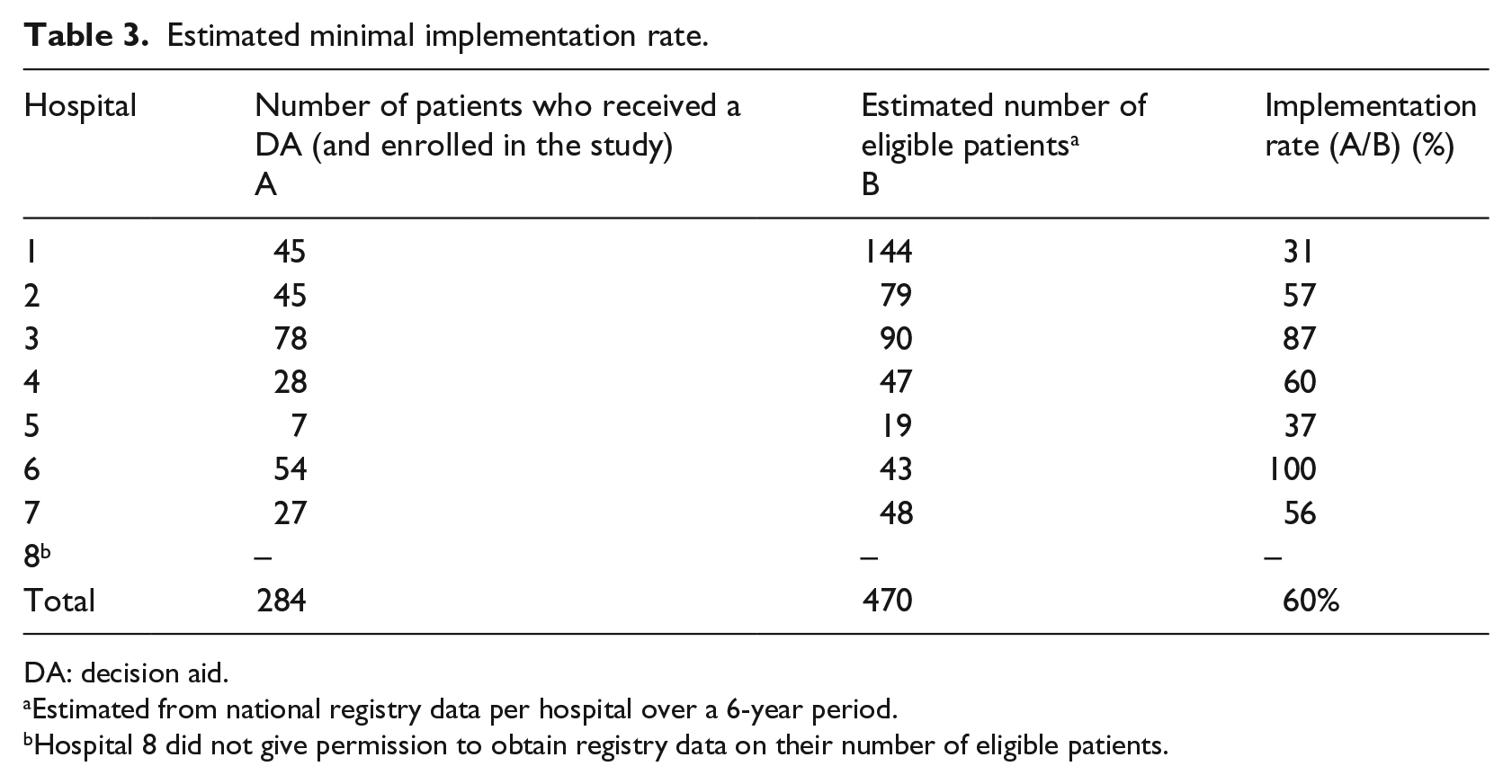
DA: decision aid.
Estimated from national registry data per hospital over a 6-year period.
Hospital 8 did not give permission to obtain registry data on their number of eligible patients.
Evaluation by health care professionals
The reception of the DA by the health care professionals was positive. They rated its value 8.1 (range = 6–9) on a scale from 1 to 10.
Presenting the DA
The vast majority of care providers reported that, when presenting the DA, they explained to patients that there were different treatment options, indicated which options were open for the patient and stimulated the patient to consider the pros and cons and make a personal trade-off (96%–100%). Most providers (88%) reported having asked their patients for their preference after the patients had gone over the DA.
Barriers and facilitators
The majority of health care professionals (92%) stated that they had offered the DA to all eligible patients. Those who had not always offered the DA endorsed various reasons which were related to logistics (somebody else offered the DA), physician-related (I forgot) or patient-related (patient nervous, already decided, unwilling). Time constraints did not appear to be an important factor; some care providers indicated that the DA required extra time (17%), but most answered neutrally (71%) or agreed with the statement that the DA saved time (13%). Moreover, none of the care professional indicated that they ‘lacked the time’ to offer the DA. An open question yielded no additional barriers.
Table 4 shows the percentages of care providers who endorsed potential barriers and facilitators. Most professionals disagreed with the negative statements (suggested barriers) and agreed with the positive statements (suggested facilitators). The facilitator that was endorsed by all care providers (100%) was ‘Offering the DA fits well with the existing guidelines and procedures’. After Bonferroni correction, no significant difference was found between care professionals who presented the DA to more than 10 patients versus those who presented the DA to less patients.
Agreement with facilitators and barriers for professionals who were high DA providers (to more than 10 patients) versus low DA providers (to 10 patients or less).
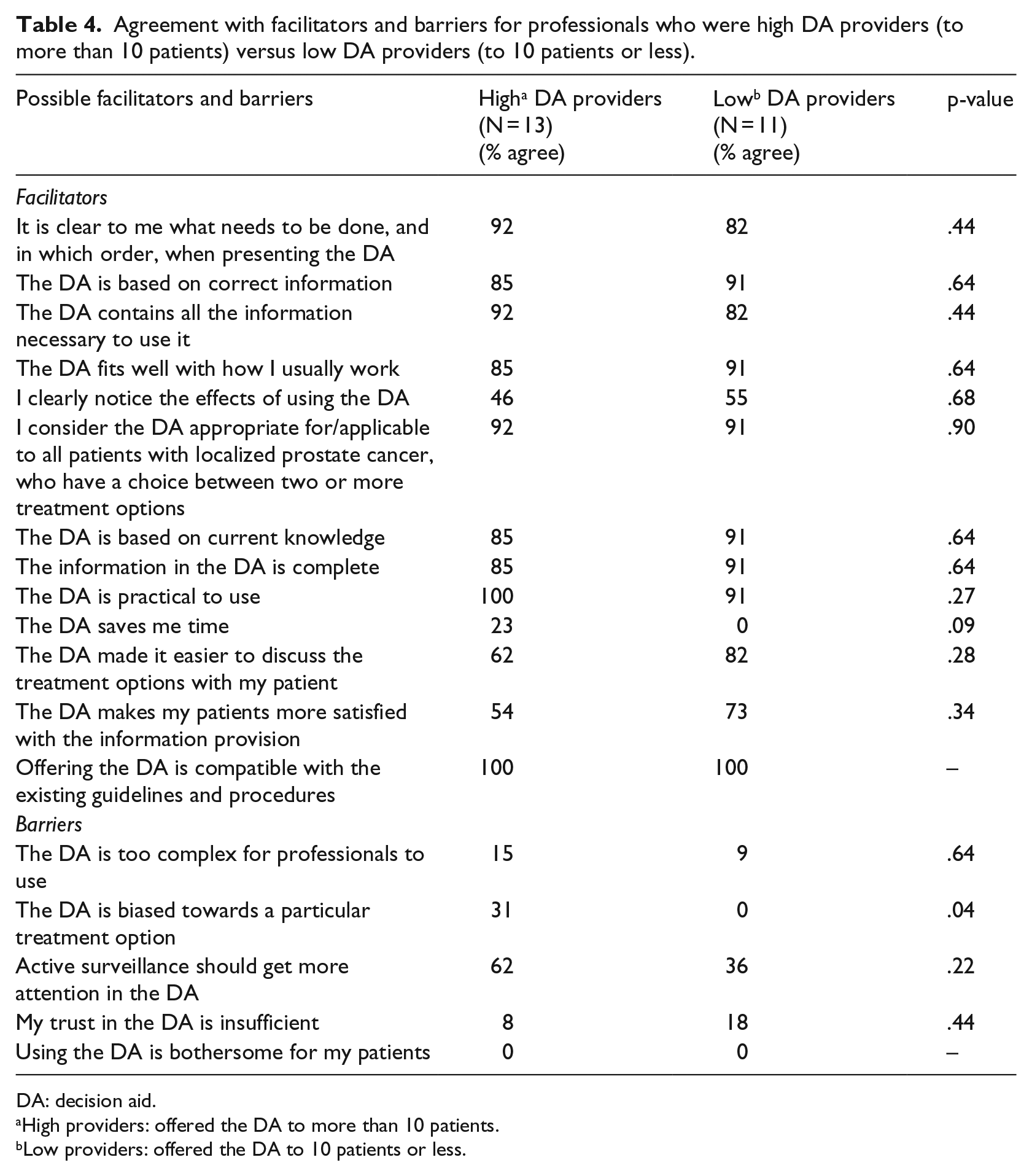
DA: decision aid.
High providers: offered the DA to more than 10 patients.
Low providers: offered the DA to 10 patients or less.
Evaluation by patients
Use of the DA
Of the 255 patients who received the DA, 88 per cent indicated that they had used the DA (N = 225) and 12 per cent that they had not used it (N = 30). Users were comparable to non-users for demographic characteristics, such as age (66.1 ± 6 vs 65.1 ± 6, p = .36), marital status (12% vs 10%, single, p = .73) and tumour characteristics (90% vs 90%, Gleason score ⩽ 7, p = .95 and 70% vs 77%, PSA ⩽ 10, p = .69). A difference was found for education; more users had a college degree than non-users (41% vs 20%, p = .028).
Barriers and facilitators
The barriers and facilitators that users and non-users endorsed are listed in Table 5. Clearly, non-users experienced more barriers and less facilitators compared to users. After Bonferroni correction, 11 barriers/facilitators still differed significantly between users and non-users (Table 5).
Agreement with facilitators and barriers for patients who used the DA versus those who did not use the DA, after receiving it.
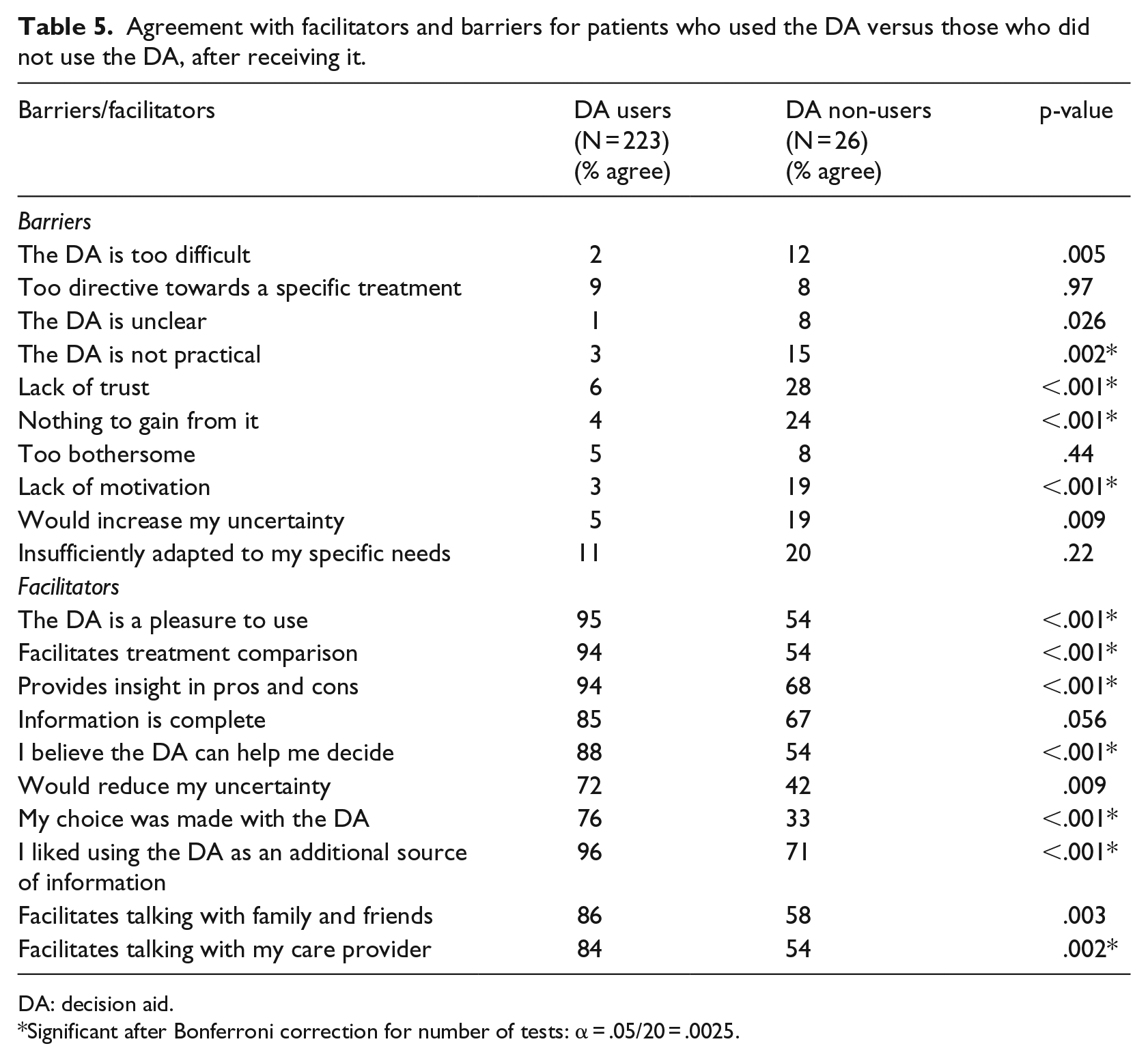
DA: decision aid.
Significant after Bonferroni correction for number of tests: α = .05/20 = .0025.
Of the users, 83 per cent said that the DA had ‘much’ or ‘very much’ added value compared to other information they had received, and all (100%) would recommend the DA to others. The evaluation of the decision-making process was also positive. Most patients found it meaningful (97%) and desirable (89%) to be involved in the treatment choice. It was easy for most patients to reach a decision (83%), and most patients felt no pressure during the decision-making process (98%).
Content of the DA
Patients were positive about the content of the DA. The information was clear (98%), complete (84%) and easy to use (96%). Most patients (95%) indicated that the information about the different treatment options was balanced. To the question how often words were unknown, the content was too complicated or they needed someone else to help them understand the text, almost all patients (96%) responded that this was rarely or never the case.
Presenting the DA (by whom and when)
Most patients were satisfied with the way the DA was applied by the health care professionals. Table 6 shows that the DA was presented most frequently by the urologist (79%, N = 181) and less often by the oncology nurse (21%, N = 47). The urologist was considered the most appropriate person to do so, according to the vast majority (97%) of patients who received it from the urologist. However, of the patients who received the DA from the nurse, a majority (64%) considered the nurse to be the most appropriate person.
Most appropriate professional to present DA according to patients.
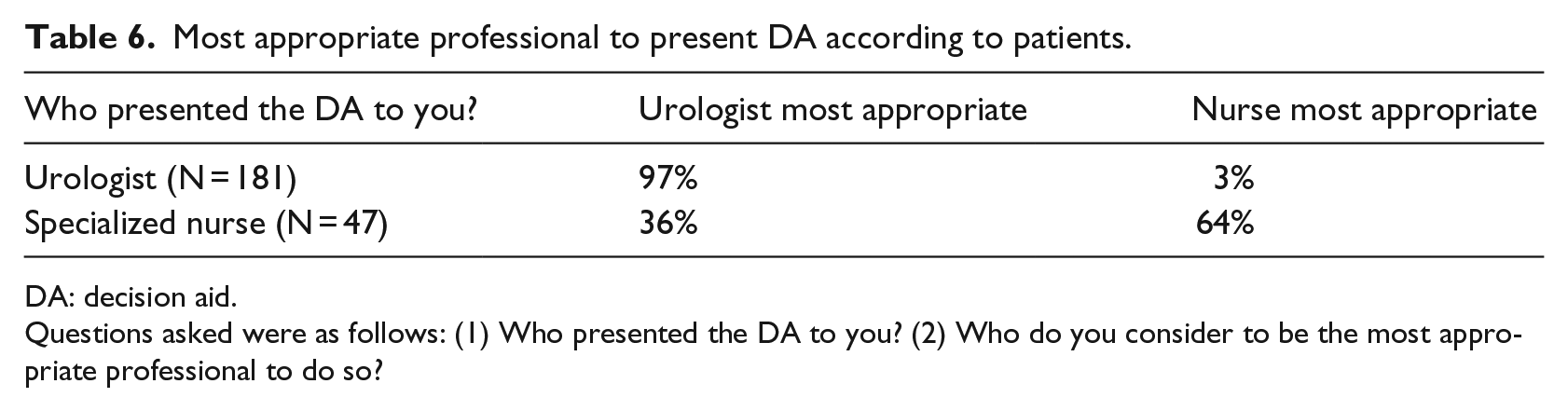
DA: decision aid.
Questions asked were as follows: (1) Who presented the DA to you? (2) Who do you consider to be the most appropriate professional to do so?
Most patients (59%) received the DA on the same day as their diagnosis of prostate cancer. Other patients received the DA either 1 day to 1 week after diagnosis (10%), 1 to 2 weeks after diagnosis (12%) or more than 2 weeks after diagnosis (19%). Whether patients felt that they had received the DA at the appropriate moment is shown in Figure 1. Of the patients who received the DA on the day of diagnosis, 1 per cent would have preferred to receive it later. Of the patients who received it 1 day to 1 week after diagnosis, all considered this the appropriate time to receive the DA. After more than 1 week, some patients would have preferred to receive the DA earlier.

Timing and satisfaction with timing of the decision aid.
Most patients (89%) indicated that the care provider had asked for their treatment preference. Almost all indicated that they had felt sufficient opportunity to discuss their preference with the professional (95%) and to get their questions answered (92%).
Discussion
The implementation strategy, used in this study, yielded an implementation rate of 60 per cent on average over eight hospitals. The strategy reduced previously identified barriers for implementation. No additional barriers were identified from the care providers’ perspective or from the patients’ perspective. The DA was well received by patients and care professionals. Patients considered both physicians and nurses to be appropriate professionals to hand out the DA. Receiving the DA on the same day as the diagnosis was acceptable to almost all patients.
Below, we will discuss the results regarding the implementation rates, the barriers and facilitators, first from the physicians’ point of view and second from the patients’ point of view. Finally, we will discuss the incorporation of DAs in the care pathway.
Successful implementation
The estimated implementation rate was higher than expected, that is, 60 per cent. Still, this may be a conservative estimate. It was calculated using the number of patients who received the DA and who were also enrolled in the study. This may have resulted in an underestimation of the actual implementation rate, since not all patients receiving the DA may have been enrolled in the study. Indeed, roughly twice as many DAs were used from stock by the participating hospitals as there were patients included in the study.
It is hard to compare the average implementation rate of 60 per cent with implementation rates achieved in other settings. Surveys in the Unites States on implementation of DAs showed that about one-third of physicians reported using a DA.18,19 These studies, however, do not report an implementation rate, that is, an estimate on how many of the eligible patients received a DA. One study on the use of a DA for cystic fibrosis patients reported an exceptional 95 per cent implementation rate, but this was self-reported by care providers and could not be verified in patients or otherwise. 20 A systematic review of 17 studies on the implementation of patient decision support reported a lack of specific information about the implementation rates that were achieved. 7
This study measured the implementation rate that was attained and, as such, adds to the existing literature in which such data are lacking. It is important to measure the attained implementation rate, as a considerable gap has been reported between physician’s intention to adopt DAs and their actual behaviour. 18 This intention–behaviour gap may underlie the poor implementation in some hospitals.
In this study, a large variation in implementation rates was found between hospitals. No obvious reasons for this variation were found. The type of hospital, university or regional, large or small, was not related to implementation rates. After 2 years, the DAs were reportedly still used by all eight participating hospitals. In one hospital, the use of the DA was reduced in comparison with the study period. Two other hospitals reported an increase in implementation rate, as their care pathway was reorganized and the DAs were now handed out by a dedicated nurse who saw all patients with prostate cancer. Underlying reasons for the variation in implementation rates will be further explored by interviews.
Barriers and facilitators
Some care providers indicated that the DA required extra time, but most answered neutral or even agreed that the DA saved time. In other settings, 20 to 32 per cent of the physicians also agreed that DAs will save time. 18 The main facilitating factor for the care providers in this study turned out to be its agreement with the guidelines. Guidelines do urge the use of shared decision-making.2,21 However, it would be even more helpful if, in addition, they would support the use of DAs more explicitly, as this might ensure greater adoption of DAs. 19
A recent survey in urologists and radiation oncologist showed that most were familiar with DAs. Still, only a minority used them in daily care. An important barrier for professionals who did not use DAs was the assumption that patients could not process information from DAs. 19 This assumption was not supported by the data of the patients in our study, since 96 per cent indicated that the content of the DA was not too complicated and that they did not need someone else to help them understand.
For patients, other barriers have been reported in literature. To participate in shared decision-making, patients do not only need knowledge but also need to be empowered to participate. 22 DAs that solely focus on the provision of information fail to address the acceptability and need for patient involvement. A review of 44 studies revealed that patient barriers include a lack of perception that there is a preference-sensitive decision to be made, incorrect timing of receiving the DA and physicians who do not inquire about patients’ preferences or needs. 23 In our study, the DA and the instructions for care providers on when and how to engage patients appear to have reduced such barriers. The patients overwhelmingly agreed with the suggested facilitators and hardly agreed with the suggested barriers.
The differences in endorsed barriers between patients that were users versus non-users do not suggest that non-users were unable to use the DA; barriers such as ‘too difficult/unclear/insufficiently adapted to my needs/information is (not) complete’ were not endorsed more by non-users. Instead, non-users appear to be unwilling or unmotivated to use the DA; significantly more often, they endorsed barriers such as lack of trust/lack of motivation/(not) believe the DA can help me decide. This suggests that the use of DAs could be increased by explaining the potential benefits more clearly to patients. Here, the care providers could potentially play a role in motivating their patients.
In all, most barriers were removed by the implementation strategy, for both care providers and patients. This confirms the notion that barriers are modifiable rather than fixed. 23 This is important because in physicians treating prostate cancer, a strong relation was found between their endorsement of barriers and their actual application of DAs. 19 Our study suggests that using an implementation strategy that takes known barriers into account may result in adoption of DAs for prostate cancer.
Implementation strategy
Designing implementation strategies is challenging. Successful implementation requires active targeting of the users, that is, health care providers and patients. 24 It is known that successful implementation requires tailored interventions based on identified barriers. 25 The implementation strategy, used in this study, involved reducing previously identified barriers for implementation. Our study did not reveal any additional barriers or facilitators that could be incorporated in adapting or optimizing our strategy. The strategy resulted in a satisfactory implementation rate. Since the introduction of the DA was well received by both patients and care professionals, this strategy may be useful for other clinical contexts using DAs as well.
How and when to implement the DA in the care pathway
The first week after diagnosis appears to be the preferred moment for patients to receive a DA. Even the day of diagnosis was considered an appropriate time by almost all patients who had received the DA on the day of diagnosis.
The experience with delivery of the DA by a nurse appears to make patients more appreciative of delivery by the nurse. This fits well in a recent development; a growing number of hospitals in the Netherlands allot a role to specialized nurses in informing patients about their treatment options.
Limitations
To calculate the implementation rate, the estimated number of eligible patients was derived from historic data (i.e. the mean number of eligible patients per year in the participating hospitals in the preceding 6 years). Although no trend was found over those years, the number of eligible patients may have changed.
All data on decision-making were self-reported by patients and physicians. There were no video or audio recordings of the consultations to confirm whether care providers followed all instructions.
Another limitation may be the selection bias professionals and patients who are not interested in using DAs may have been less willing to fill out the questionnaire and may have had a different opinion on the barriers. 26 This is a common bias in studies on health care innovations. Please note that the implementation rates in our study are not based on the questionnaires and are calculated for the entire urology departments, including the patients of possibly reluctant care providers.
Conclusion
A successful implementation of a DA on prostate cancer can be reached, by addressing known barriers. By informing caregivers on the effectiveness of the DA, by providing instructions on its use, by embedding it in the existing workflow and by making it available free of charge, implementation barriers were strongly reduced. This article may help to facilitate the implementation of DAs, both paper-based and online versions, in daily practice.
Footnotes
Acknowledgements
This study is part of a larger research project on the implementation of DAs for prostate cancer in three regions in the Netherlands (the JIPPA study), co-initiated and funded by CZ, a not-for-profit Health Insurance Company. The authors thank Dr Linda van Mierlo from CZ for her efforts in bringing the three research groups together. In addition, the authors would like to thank Astellas BV for providing the DAs free of charge without conditions regarding the content.
Author contributions
J.J.v.T.-G., C.F.v.U.-K., M.d.V. and P.F.M.S. contributed critically to the design of the study. J.J.v.T.-G., I.M.v.O., D.M.S, C.J.W. and A.G. contributed substantially to the acquisition and interpretation of data. All authors read and revised the manuscript critically and gave final approval for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
The need for approval was waived by the Medical Ethics Committee of the Radboud University Medical Centre (registration no. 2012/518).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Financial support for this study was provided by a grant from CZ (No. 201300071), a not-for-profit Health Insurance Company. The funding agreement ensured the authors’ independence in designing the study; in collecting, analysing and interpreting the data; and in writing the manuscript.
Trial registration
The study was registered at the Dutch Trial Register NTR 6245.
