Abstract
Implementation of patient’s decision aids in routine clinical care is generally low. This study evaluated uptake and usage of a novel Dutch web-based prostate cancer treatment decision aid within the Prostate Cancer Patient Centered Care trial. From an estimated total patient sample of 1006 patients, 351 received a decision aid (35% implementation rate; hospital ranges 16%–84%). After receipt of the decision aid, most patients accessed the decision aid, utilized most functions, although not completely, and discussed the decision aid summary in a subsequent consultation with their care provider. Including nurses for dissemination of decision aids seemed to positively affect decision aid uptake. Once received, patients seemed able to use the decision aid and engage in shared decision-making as intended; however, decision aid uptake and complete usage of all decision aid components should be further improved. Prior to the diagnosis consultation, handing out of the decision aid should be prepared.
Background
When diagnosed with localized prostate cancer (Pca), patients can face a choice between multiple surgical and radiotherapy options or decide not to be treated immediately by following an active surveillance (AS) protocol. In the absence of a generally superior treatment option, patient preferences should guide this treatment selection process.1–3 When such a preference-sensitive health decision has to be made, a collaborative approach from both the patient and care provider is preferred to select the best suiting treatment. 4 In this process of shared decision-making (SDM), a doctor provides all relevant information about the disease, treatments and consequences, and the patient shares his preferences and concerns. 5
SDM can be initiated or enhanced with help from decision support tools, such as decision aids (DAs). DAs come in multiple formats (e.g. booklet, web-based), but all provide structured and balanced information about the disease, available treatments and the risks and benefits associated with these treatments.4,6 Often, DAs also include values clarification exercises (VCEs) to elicit patient preferences. 7 Previous studies have shown that patients, after using a DA, have more knowledge, lower decisional conflict and more accurate risk perceptions. 8 If VCEs are included, patients are also more likely to select a treatment that is consistent with their values. 8
While many trials have reported beneficial patient outcomes after the use of decision support tools, routine use of such tools in clinical care still is low. 9 Only a few examples are known of sustained DA use after their initial evaluation in a clinical trial.10,11 Studies that have evaluated DA implementation from a care providers’ perspective identified important barriers, such as limited confidence in the (content of the) tools, time pressure and concerns about disrupting work procedures.9,12 A common patient-reported barrier is the feeling of being unable to engage in SDM or to use a DA, rather than being unwilling to do so. 13
Recently, policy makers in the Netherlands adopted SDM and DAs in their effort to improve quality of care. 14 Funding programs are encouraging SDM and usage of DAs. As such, patient groups, professional bodies and healthcare insurers and hospitals are stimulated to implement DAs for multiple medical conditions, and DA developers are stimulated to develop new DAs. 15 Although much is known about attitudes, barriers and facilitators toward SDM and DAs, there is limited data available about actual achieved degrees of implementation and the precise proportion of patients using DAs once distributed. Often this is because the tool or patient population does not allow for detailed registration of the exact number of eligible patients, number of tools distributed or monitoring of actual usage. For example, leaflet DAs could be distributed in waiting room areas, and in such circumstances, it is unknown who takes them. For an online DA that is publicly available, it is often unknown which visitors are actually patients facing a treatment decision at that particular moment. Moreover, if a DA is used in multiple hospitals, and detailed information on actual uptake and usage is lacking, less can be learned from local best practices.
A novel web-based Dutch Pca treatment DA, which was investigated in a cluster-randomized controlled trial (RCT), allowed for structural monitoring of DA uptake and usage in a quantifiable patient population. 16 Therefore, this study aimed to improve understanding of the implementation results by comparing the DA uptake across hospitals and the actual usage of the DA and its elements (e.g. VCEs) by patients.
Methods
Study sites
A total of 18 Dutch hospitals agreed to participate in the trial, and after randomization, the DA was implemented in 9 Dutch hospitals. The other nine hospitals formed the control arm and delivered care as usual. 17 One academic medical center participated in the trial, which was randomized into the control arm. All other hospitals were teaching hospitals. Two hospitals that were involved in DA development (before randomization) were randomized by pure chance into the intervention arm. Randomization was performed by an independent statistician, blind to hospital names. 17 The hospitals involved offered either one (AS) or two treatments (AS and surgery or radiotherapy) at their own location. Referral to another hospital for specific treatments is common in the Netherlands. Implementation started in August 2014 and data collection ended in January 2016. The need for ethical approval was waived by the regional medical ethics committee (reference: NW2014-03).
Implementation/delivery method
All urology departments of the hospitals enrolled in the trial’s DA group were visited by the researchers and received an explanation of the purpose of the RCT. The DA was presented to the medical staff and the proposed method of delivery was explained. The explanation included that the DA would be introduced to patients in addition to the presentation of standard information. Depending on local work routines, the DA would usually be introduced by the urologist at diagnosis or by an oncology nurse during consultation following diagnosis. Next to the DA, patients received all information and materials (e.g. hospital brochures) that would also have been provided otherwise. Patients accessed the DA with an individual access code that was provided to them on a card by their care provider. On that card, the care provider also indicated which of the treatments covered by the DA (AS, radical prostatectomy, brachytherapy and external beam radiotherapy) were eligible for the patient. For gathering DA user data, receiving the post-questionnaire and combining DA user data and questionnaire data, patients signed informed consent. The DA could be used by patients regardless of trial participation and consent, and DA usage was then still monitored, as this was anonymized data not linked to individual patients.
DAs
The DA is in Dutch language and available online only after login (http://prostaat.keuzehulp.nl). After accessing the DA, patients are presented with general information about Pca and treatments first. Based on the treatments available to the patient, detailed information is then provided about all treatments. Information about treatments is broken up into a section about AS versus treatment and a section about surgery versus radiotherapy. Per section, VCEs are included to elicit patient preferences for treatment. These VCEs are presented as trade-offs between treatment attributes. The DA ends with a summary of all indicated preferences, including a treatment preference. The DA summary can be printed and brought to the following consultation with their clinician, or re-accessed online, to enable a shared patient–doctor conversation about aspects relevant to the patient. The development process and DA content (including examples of the VCEs and DA summary page) have been described in more detail before. 16
Data collection and patient questionnaires
For this study, data were gathered on the number of DAs provided by healthcare providers and the usage of the DA by patients. As the DA was provided as part of an RCT, both healthcare providers and patients signed informed consent. Study numbers on the informed consent forms were hospital specific and linked to the DA access codes. An informed consent signed by a healthcare provider represented a DA being provided to a patient; actual patient use of the DA was monitored by means of log files. Data from the questionnaires were linked to DA user data based on study number. Patient characteristics from informed consent were saved separately from the DA user data to ensure anonymity.
To determine the degree of implementation, information on the total number of eligible Pca patients per hospital was obtained from the Netherlands Cancer Registry (NCR) for a 5-year period (2009–2013) prior to the start of DA implementation. All participating hospitals allowed the NCR to provide us with data about their number of patients diagnosed, except for one. From these registry data, an estimation was made on the expected number of eligible Pca patients during the trial period. Eligibility was defined as being diagnosed with low or intermediate risk Pca (prostate specific antigen (PSA) level below 20 and a maximum Gleason score of 7). 18 In addition, patients were required to have access to the Internet and to be able to read and understand Dutch language.
An online questionnaire to evaluate DA usage by patients was sent (paper version on request) to patients after the treatment decision was made. The questionnaire included items about when the DA was received and from who (urologist or nurse), whether the DA was sufficiently explained and whether the DA summary was discussed during a subsequent consultation.
Data analysis
The degree of implementation was calculated by dividing the number of DAs provided to patients (based on access cards distributed by care providers) by the estimated total number of eligible Pca patients (based on national registry data) per hospital. Descriptive statistics were presented as means (and SDs) for continuous variables and frequencies (and percentages) for categorical variables. Group comparisons between DA users and non-DA users and between questionnaire responders and non-responders were made with t-tests for continuous variables and chi-square tests for categorical data. Statistical analyses were conducted using SPSS 22.0 (Statistical Package for Social Sciences, Chicago, IL, USA). Tests were considered statistically significant if p < .05.
Results
From national registry data, it was estimated that during the trial period, 1006 patients were diagnosed with localized Pca in participating hospitals. With 351 DAs distributed to patients, the average achieved degree of implementation across all study locations was 35 percent, varying between 16 and 84 percent across hospitals. Highest implementation levels (84% and 79%) were achieved in hospitals who were also involved in DA development. Implementation did not succeed in one hospital due to a lack of organizational support (also no registry data were obtained for this hospital), and this hospital was therefore excluded from further analyses. Detailed implementation results across all participating hospitals are presented in Table 1.
DA uptake per study site.
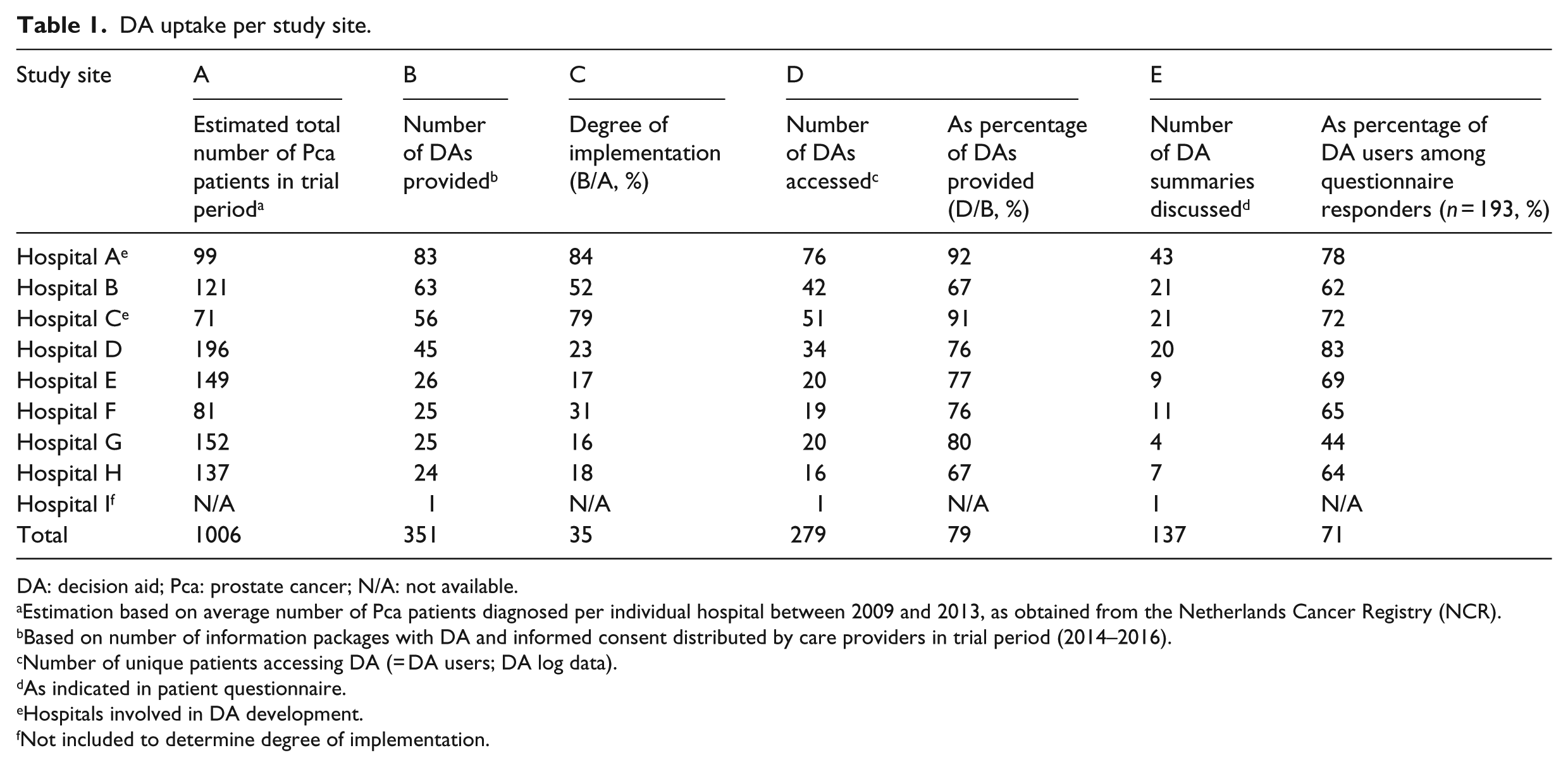
DA: decision aid; Pca: prostate cancer; N/A: not available.
Estimation based on average number of Pca patients diagnosed per individual hospital between 2009 and 2013, as obtained from the Netherlands Cancer Registry (NCR).
Based on number of information packages with DA and informed consent distributed by care providers in trial period (2014–2016).
Number of unique patients accessing DA (= DA users; DA log data).
As indicated in patient questionnaire.
Hospitals involved in DA development.
Not included to determine degree of implementation.
From all patients (N = 351) receiving a DA, 277 patients accessed the DA (79%; Table 1). Age did not significantly differ between DA users versus non-DA users (65.2 vs 65.6; p = .60). A larger proportion of non-DA users were less educated, but differences in education level between DA users and non-users did not reach statistical significance. The DA was used more often by patients with a Gleason grade 6 tumor (usage rate 92%), compared to patients with a Gleason grade 7 tumor (usage rate 83%, χ 2 (1, N = 220) = 5.18, p < .02; Table 2).
Self-reported sociodemographic and clinical characteristics of sample.
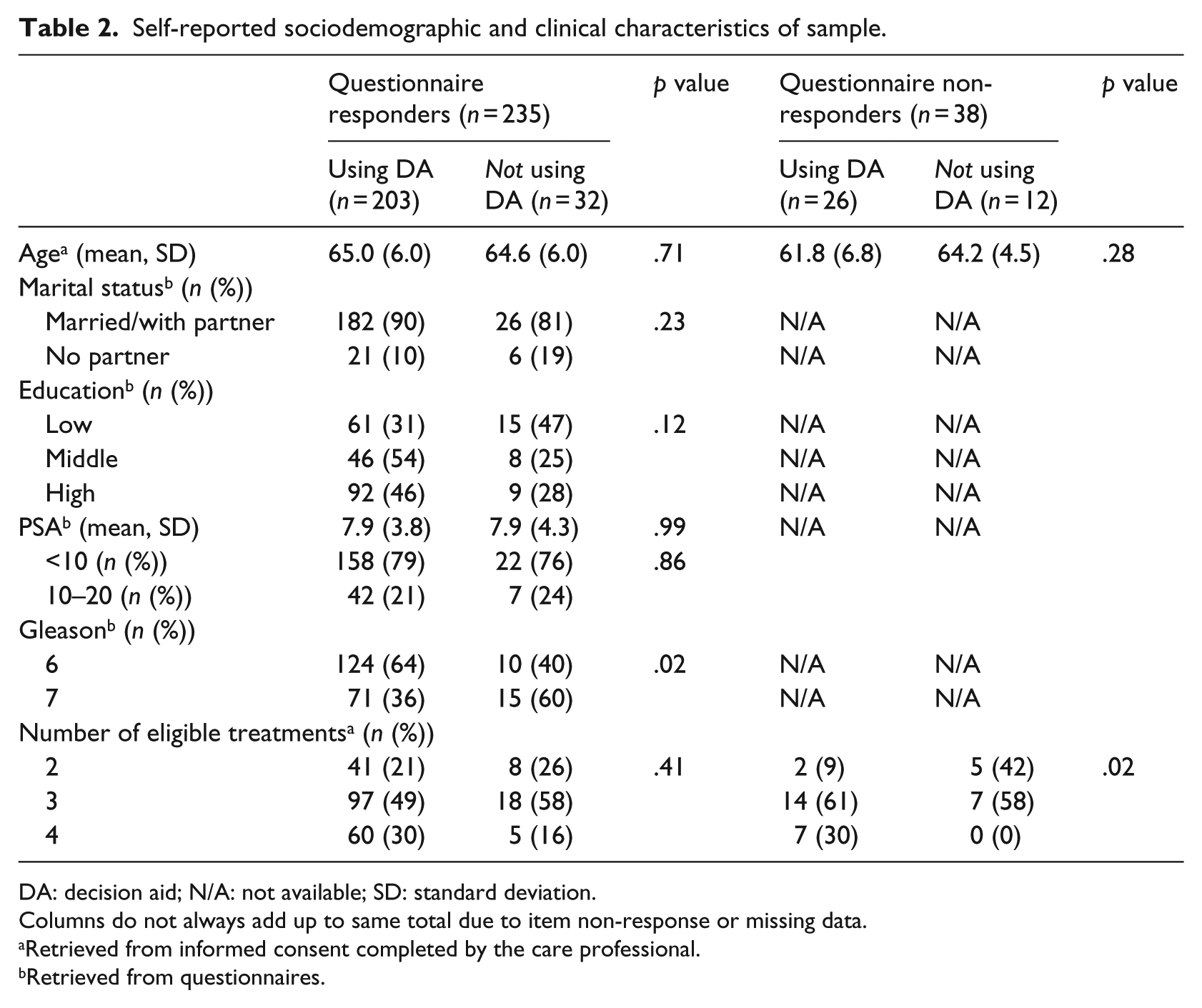
DA: decision aid; N/A: not available; SD: standard deviation.
Columns do not always add up to same total due to item non-response or missing data.
Retrieved from informed consent completed by the care professional.
Retrieved from questionnaires.
DA log file data showed that of 106 patients eligible for AS, 69 patients (65%) read all information about the comparison between AS and treatments, and 163/277 (59%) completed the section about surgery and radiotherapy (all DA users were eligible for at least one of these treatments). The VCEs after the first step (AS versus treatment) were fully completed by 67 of 106 eligible DA users (63%) and after the second step (surgery versus radiotherapy) by 66/277 (24%). A treatment preference was indicated by 197 DA users (71% of all DA users). Usage of the DA elements was consistent among patients from different hospitals (Table 3).
DA reception and completion.
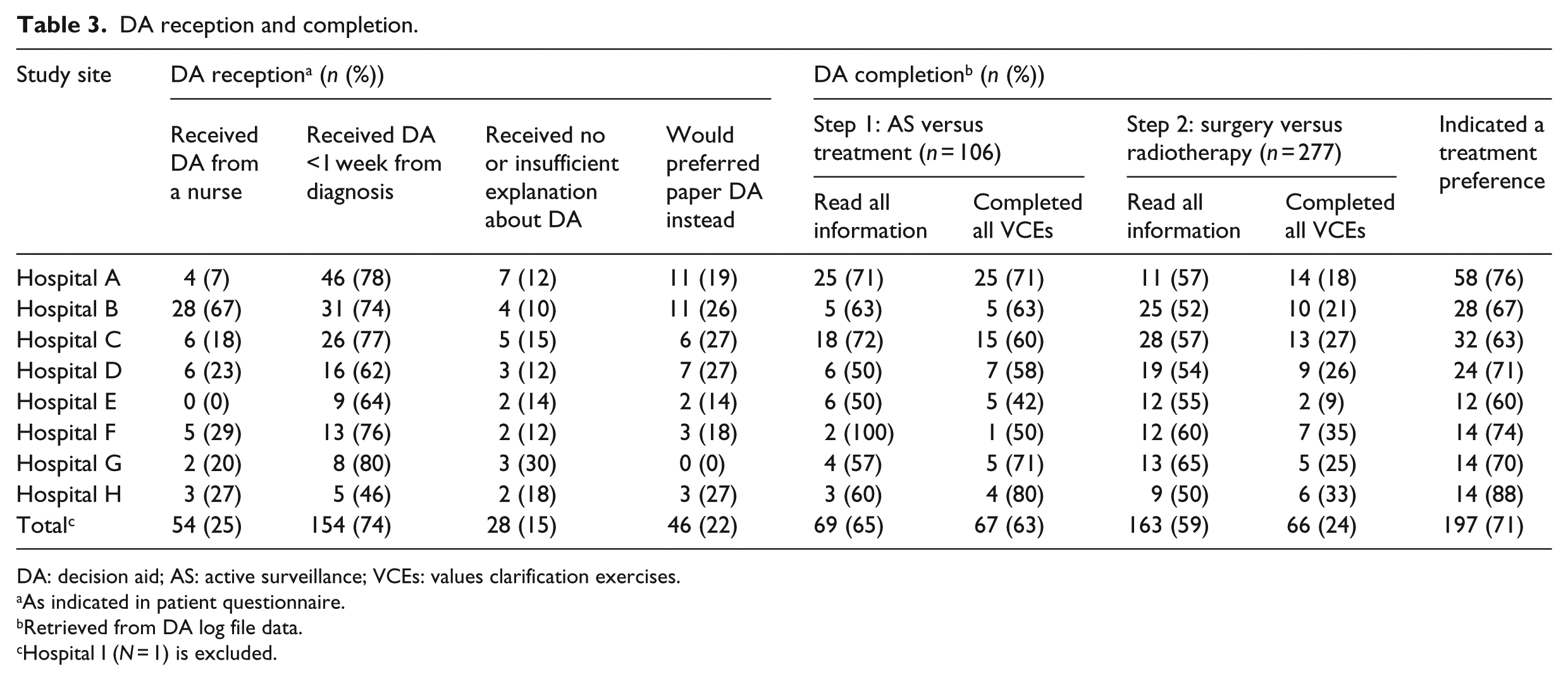
DA: decision aid; AS: active surveillance; VCEs: values clarification exercises.
As indicated in patient questionnaire.
Retrieved from DA log file data.
Hospital I (N = 1) is excluded.
The post-decision-making questionnaire was sent to 273 patients who gave informed consent (consent rate 78%) and was filled out by 235 respondents (response rate 86%). Questionnaire responders were more likely to have also used the DA (usage rate 86% vs 68%, χ 2 (1, N = 273) = 7.81, p < .01) and were slightly older (62.5 vs 64.9, t(271) =−2.29, p = .02) compared to non-responders. Marital status, educational level, PSA level and Gleason score were collected with the questionnaire and therefore not available for non-responders (Table 2). The flow diagram of patients included in this study is presented in Figure 1.
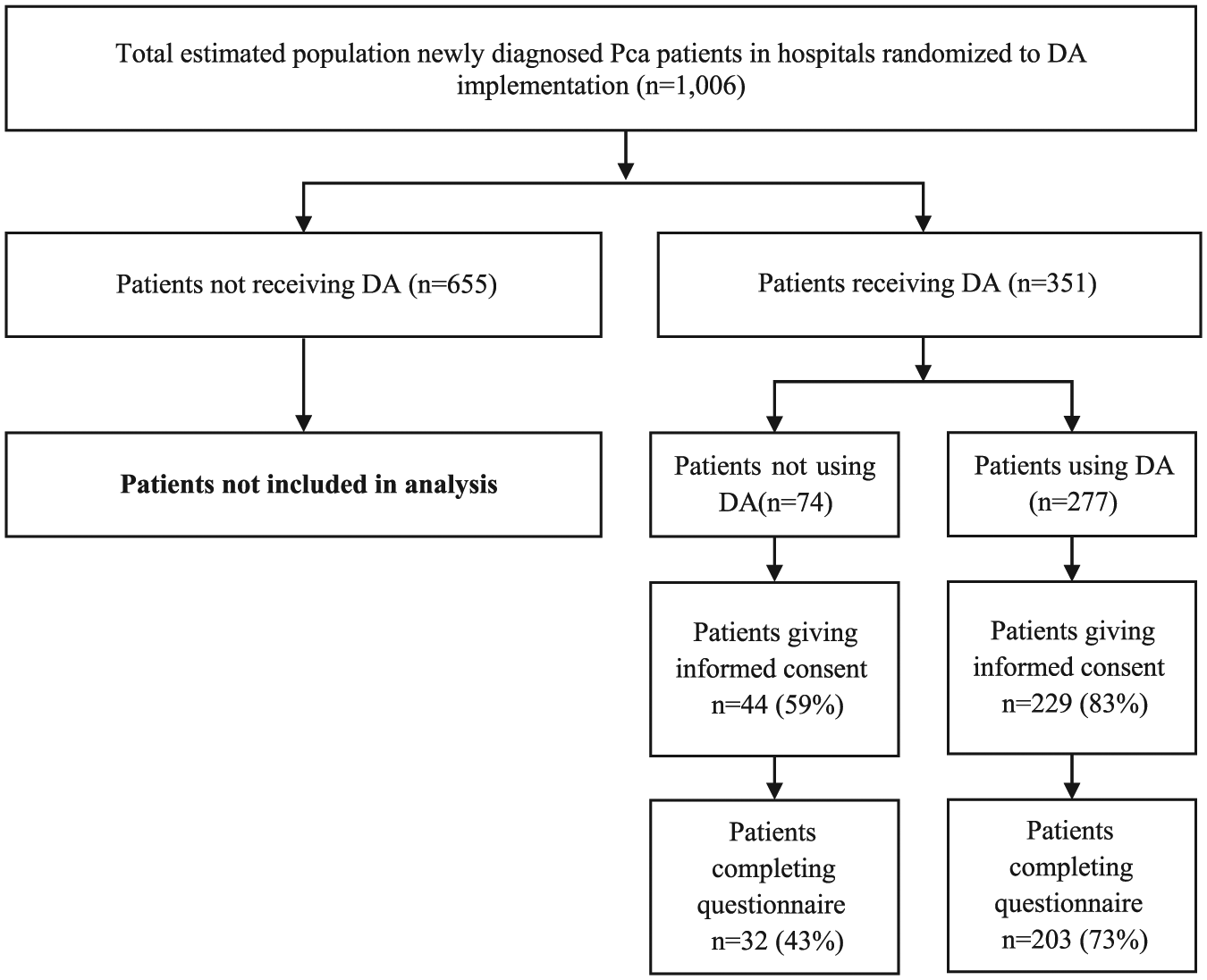
Flow diagram of patients included in the Prostate Cancer Patient Centered Care (PCPCC) trial.
From the questionnaire responders who used the DA (N = 193), 137 responders (71%) indicated that the summary obtained from the DA was discussed with their urologist in a subsequent consultation (Table 1). Most respondents indicated that the DA was received within a week from diagnosis (154/208, 74%), and 158 respondents (85%) felt the DA was sufficiently explained (28 respondents indicated no or insufficient explanation was received). One out of four patients received the DA from an (oncology) nurse, ranging between hospitals from zero up to two out of every three patients. In all other occasions, the DA was received from the urologist. A majority of the questionnaire responders (163/209, 78%) indicated that the online format of the DA also was the preferred format and 46 responders (22%) would have preferred a paper DA instead (Table 3). Figure 2 presents a flowchart of the most common workflow for DA dissemination across participating hospitals.
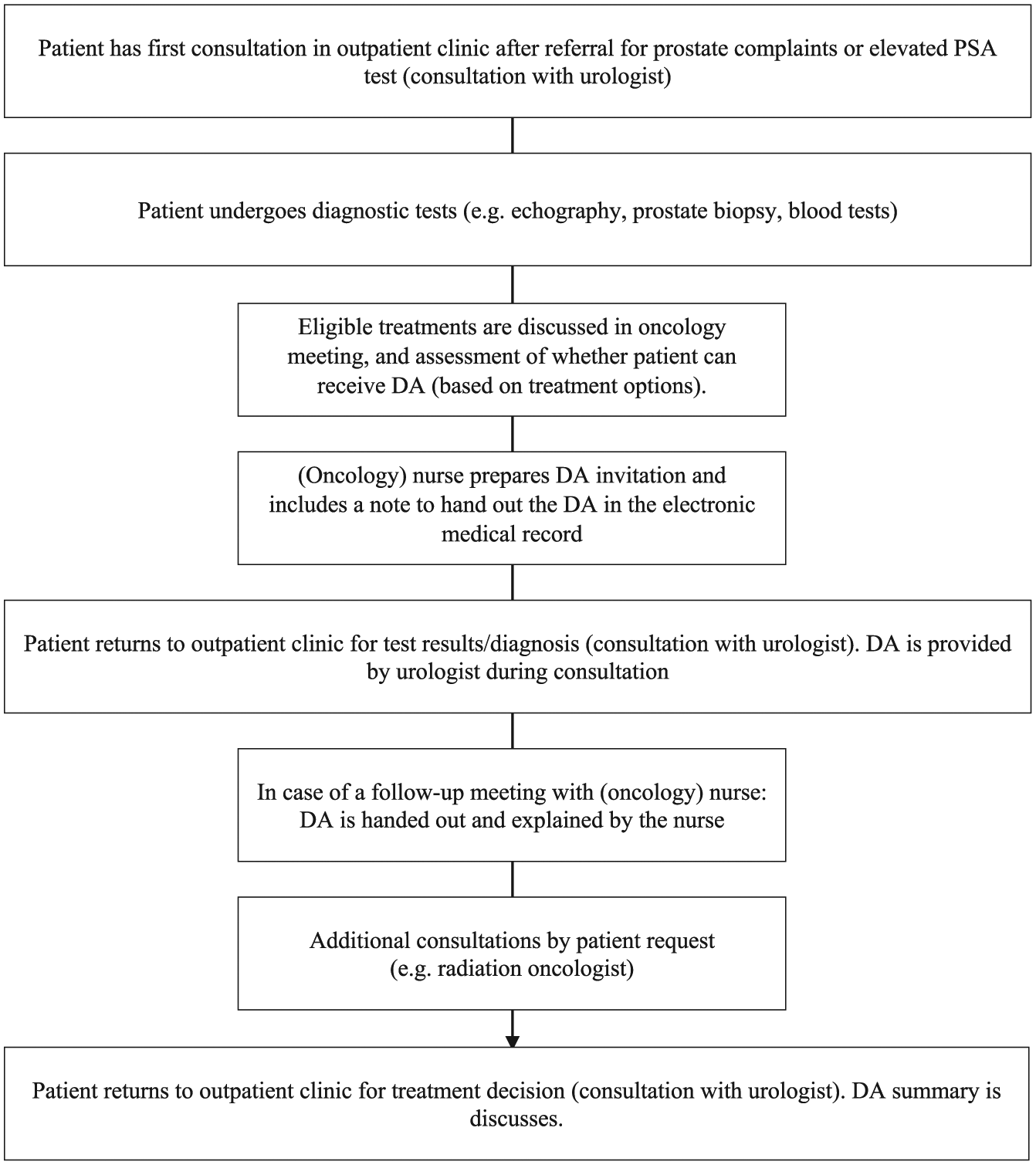
Flowchart of most common workflow for DA dissemination in participating hospitals.
Discussion
This study structurally evaluated implementation of a web-based Pca treatment DA in nine Dutch hospitals. All hospitals included the DA as part of their information routine following Pca diagnosis. On average, one in every three newly diagnosed Pca patients received a DA, but large differences were observed in implementation rates per hospital. Once a DA was distributed, most patients accessed the DA, indicated a treatment preference and discussed the summary obtained from the DA with their urologist.
Implementation was highest in hospitals of care providers who were also involved in the development of the DA and by random assignment enrolled in the intervention arm. Involvement during DA development may have increased intrinsic motivation to implement the DA. This motivational approach is identified as an important driver for implementing change in health care and applied in other DA development processes as well.19,20 To increase intrinsic motivation for care providers from institutions who were not involved in the DA development, it could be useful to offer opportunities to become more involved with the DA. For example, customization of the DA to match hospital layout as well as the possibility to integrate the DA with existing hospital materials could improve adoption of the DA by care providers in other hospitals.
Most patients received the DA from their urologist within the first week after diagnosis and felt that the DA was sufficiently explained to them. In most hospitals, the role of nurses in this process of distributing DAs was limited. In an earlier study among care providers, we found that nurses and urologists consider themselves equally suitable to provide patients with a DA and that distributing DAs should not be an exclusive clinician task. 21 Therefore, overall implementation could be further improved if nurses become more involved in the process of distributing DAs. As in hospitals that implemented best, nurses either prepared DA distribution prior to the diagnosis consultation or handed out the DA themselves in a subsequent follow-up consultation (Figure 2).
Respondents with a Gleason grade 6 tumor used the DA more often compared to respondents with a Gleason grade 7 tumor. Gleason scores provide a prognosis of oncological outcomes; a Gleason 6 score (or lower) represents the most favorable condition. 22 Consequently, patients with a Gleason grade 6 more often had all four available treatment options to choose from, while the choice set of eligible treatments for patients with a higher Gleason grade 7 tumor was often reduced. Choosing from a smaller choice set can be perceived as less difficult, resulting in a different information search behavior compared to patients who need information about more options.23–25 Therefore, the DA could have been perceived as less needed by patients with a Gleason grade 7 tumor.
DA users differed in the amount of information that they read and which VCEs they answered within the DA. This could indicate that DA users differed in their information needs or did not fully understand how to navigate within the DA. However, in the questionnaire, a large majority of responders (both DA users and non-DA users) felt the DA was sufficiently explained, and in previous usability tests, easy navigation was confirmed. 16 It is therefore more likely that the DA was used for specific information, beyond the information that was already known from other sources. The selective answering of VCEs could be because patients were undecided on the VCEs that were not answered or instead, already had a clear treatment preference and did not feel the need to provide an explicit answer to the VCEs. This is confirmed by the large majority of DA users who were able to indicate a treatment preference. Follow-up research is needed to determine whether the degree of DA completion has led to differences in patient-reported outcomes (e.g. information satisfaction and regret).
A majority of DA users indicated that the DA summary was discussed with their clinician in a subsequent consultation. The summary reported the patient’s VCE responses and treatment preference. With such a summary on paper during consultation, patients were encouraged to overcome the barrier of feeling unable to engage in SDM. 13 To further stimulate SDM in routine clinical care and engage patients and care providers in discussing preferences and values, distribution of the DA among eligible patients should be further optimized. One way to increase delivery could be to automate DA delivery, for example, by having the electronic medical record automatically signal if a patient should receive a DA. 26 Alternatively, eligibility for the DA could be included as part of the multidisciplinary team meeting, where all newly diagnosed patients are discussed, and in the preparation of the consultation (e.g. “did the patient already receive a DA?”).
A major strength of this study is that it is one of the first to analyze DA implementation in a structured manner, with data on every step from DA distribution to treatment choice (number of DAs distributed, log file data on DA usage and a post-decision evaluation). This study covers a largely neglected area in (Pca) DA implementation studies, that is, actual DA usage data. 27 Our results showed that implementation of DAs should not only be evaluated based on the number of distributed DAs, but require a more thorough investigation of the distribution procedures (e.g. role of nurses) and usage of specific DA elements (e.g. VCEs completed).
A limitation of this study was that distribution of the DA was linked to trial participation measuring the DA’s effectiveness. 17 Next to introducing the DA, care providers also had to explain enrollment in the trial and had to obtain informed consent to have patients participate in the questionnaire study. Although patients were informed that the DA could also be used without participation in the trial, it could have served as a barrier for DA use for some patients. Also, determination of the total number of eligible patients was estimated based on the number of Pca patients registered in the NCR in previous years (2009–2013). Regional trends (hospital mergers and changes in offered treatments) could potentially have caused some inaccuracy in the estimation of the number of patients during the trial per hospital. Nevertheless, as these number are fairly stable over time, it is unlikely that this would have a large influence on our findings. Moreover, the estimation was based on tumor stage only (cT1 and cT2), which could mean that the registry data included patients who would not have met the DA inclusion criteria due to comorbidities or other clinical characteristics. Therefore, the calculated degrees of implementation could underestimate actual implementation slightly. This, however, is likely to occur in all hospitals and could therefore contribute to a margin of error within the estimation that is equal for all hospitals and as such is unlikely to explain variations in implementation rates observed across hospitals.
Conclusion
While many studies have provided evidence that DAs can be effective tools to support SDM in clinical practice, this study is one of the first to provide a detailed analysis of the implementation results and usage rates of a DA in clinical routine. Eight out of nine hospitals involved in this study succeeded to implement a novel Dutch web-based Pca treatment DA in clinical routine within the trial period. Uptake between these hospitals varied from incidental to structural and patients varied in the extent to which they utilized all DA components. Most patients expressed a treatment preference and used the DA summary to talk about values and preferences with their urologist. Based on our results, strategies to further improve DA adoption by care providers and patients can be targeted more specifically.
Footnotes
Acknowledgements
The authors thank all patients participating in this study and all hospitals and involved staff from Elisabeth-TweeSteden Hospital, Tilburg and Waalwijk; Catharina hospital, Eindhoven; Maxima Medical Center, Veldhoven; St. Antonius hospital, Nieuwegein; Zuyderland Medical Center, Heerlen and Sittard; Haga Medical Center, The Hague and Jeroen Bosch hospital, ‘s-Hertogenbosch; all in the Netherlands. The authors would also like to thank Zorgkeuzelab for the technical development of the DA and providing the DA log data needed for this study. Finally, the authors thank Dr Linda van Mierlo of CZ for her role in facilitating this study. Trial registration: Dutch Trial Register, NTR-4554.
Author’s Note
Maarten Cuypers is now affiliated with Radboud University Medical Center, The Netherlands.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by a grant from CZ Fund (Grant 2013-00070). The funding agreement ensured the authors’ independence in designing the study, interpreting the data and writing and publishing the report. The DA development was funded by Delectus Foundation.
