Abstract
Early and accurate diagnoses of sepsis enable practitioners to take timely preventive actions. The existing diagnostic criteria suffer from deficiencies, such as triggering false alarms or leaving conditions undiagnosed. This study aims to develop a clinical decision support system to predict the risk of sepsis using tree augmented naive Bayesian network by identifying the optimal set of biomarkers. The key feature of our approach is that we captured the dynamics among biomarkers. With an area under receiver operating characteristic of 0.84, the proposed model outperformed the competing diagnostic criteria (systemic inflammatory response syndrome = 0.59, quick sepsis-related organ failure assessment = 0.65, modified early warning system = 0.75, sepsis-related organ failure assessment = 0.80). The richness of our proposed model is measured not only by achieving high accuracy, but also by utilizing fewer biomarkers. We also propose a
Keywords
Introduction
Sepsis is a
The prognosis of sepsis is a complicated process due to its complex physiological mechanism. The complication involves the effect of individual variables as well as the random variations. A proper understanding of dynamics among variables is important to explain the state of the disease. Recently, the new definition of sepsis has been introduced by Sepsis-3 in an effort to simplify the diagnosis of sepsis. 1 This definition proposed a paradigm shift by moving the focus of sepsis diagnoses from inflammatory response to organ failures. The Sepsis-3 study also tied the diagnosis of sepsis in suspected infected patients to the risk of mortality. 5
Most of the existing sepsis diagnostic criteria still rely on the old definitions and lack in performance: systemic inflammatory response syndrome (SIRS) are known to have poor specificity,6–8 modified early warning system (MEWS) have limited accuracy, 9 and sepsis-related organ failure assessment (SOFA) are highly complex and require knowledge of four laboratory results that can be difficult to measure in resource constrained environment. 10 Although a new diagnostic criteria, known as quick sepsis-related organ failure assessment (qSOFA), was proposed with the announcement of new sepsis definitions, it was not widely accepted in medical communities due to its poor sensitivity. 11 Hence, there exists a need for a diagnostic model that is easy to use and delivers high performance.
Bayesian network modeling techniques, with their characteristic to capture the conditional dependencies among variables, can be suitable to describe the relationship among biomarkers for the better prediction of mortality. Bayesian networks are very popular for capturing the uncertain knowledge in medicine and have been extensively used in the diagnosis of diseases.12–14 The tree augmented naive (TAN) Bayesian network is a specific class of Bayesian networks used for classification problems and applied widely in health care. 15 The TAN Bayesian networks perform better than alternate classifiers when biomarkers are correlated, and at the same time maintain the mathematical simplicity.16,17
In this article, we developed a clinical decision support system to predict the risk of sepsis using TAN Bayesian network. Following the Sepsis-3 study, the main hypothesis of our study is that mortality and sepsis have a close relationship among patients with suspicion of sepsis. Therefore, we used mortality to infer about the diagnosis of sepsis. The framework to develop the model includes the following steps: data preprocessing, model learning, and evaluation. The data preprocessing phase includes imputation, variable selection and discretization. In the model learning phase, the interactions among selected biomarkers were captured by utilizing the TAN Bayesian network. Unlike other studies where biomarkers were considered independently, 18 our study captured probabilistic interactions among biomarkers to better estimate the risk of mortality. Finally, along with discussing results with practitioners, the performance of the proposed model was compared to popular diagnostic criteria used in practices such as SIRS, 19 qSOFA, 1 MEWS 20 , and SOFA. 21 Our predictive model outperforms existing sepsis diagnostic criteria while using fewer biomarkers.
This study makes the following contributions:
Develop a sepsis predictive model with high sensitivity and high specificity by utilizing the new sepsis definitions.
Identify important biomarkers that enable easy and quick diagnosis.
Capture probabilistic interrelations among biomarkers rather than considering them independent to better evaluate the risk of mortality.
Propose an imputation method that is suitable for electronic medical records where percentage of missing data is dominant.
The remainder of the article is organized as follows. “Literature review” section includes a synopsis on relevant studies. “Method” section discusses data, preprocessing of data, and applied methodology along with an explanation of the proposed imputation approach. “Results and discussion” section includes the comprehensive set of experiments. “Conclusion” section elaborates on the results and incorporates concluding remarks.
Literature review
Many studies have been dedicated to the development of decision support systems for sepsis identification to aid practitioners in diagnosis. Mathe et al. 22 developed a sepsis surveillance system that monitors specific lab and vital signs in real time, and visualizes them on a screen. The system notifies health care providers in case of observed abnormalities. However, this tool does not analyze the information recorded and provide any suggestion about the future outcome. Brandt et al. 23 derived a flow chart for sepsis diagnosis and developed an electronic sepsis surveillance system. The major drawback of the system was that it had low specificity. Amland and Hahn-Cover 24 designed a computerized early warning tool for sepsis diagnosis. This tool generates two kinds of alerts: SIRS alert and sepsis alert. The results showed that the system was able to alert physicians with the median of 8.6 h earlier than the time of suspicion of infection noted by doctors. The limitation of this tool was that it triggered many false-positive alarms. Only 25% of patients that were alerted required the physician’s attention. Henry et al. 25 proposed a Target Real-time Early Warning (TREW) score to predict septic shock using the cox-proportional hazard model. This support system triggers a warning of septic shock based on the change in clinical variables over time. The proposed model achieved discrimination ability of 0.83. The limitation of this analysis is that it does not capture the effect of previous condition of disease and also does not comment about the mortality risk. Most expert systems for sepsis consider clinical variables independently. However, it is highly unlikely that physiological variables are independent.
A few attempts have been made to apply Bayesian network to understand the dynamics of sepsis. Mani et al. 26 developed a predictive model using noninvasive clinical observations for sepsis prediction among neonatals (infants during the first month). The authors compared the performance of nine machine-learning algorithms and found that naive Bayes, with area under receiver operating characteristic (AUROC) curve of 0.79, outperformed other learning techniques. Gultepe et al. 12 applied the Bayesian network structure learning algorithm (hill-climbing) to unearth the interaction among clinical variables used for sepsis diagnosis. Along with developing the Bayesian network structure to understand the dependencies among variables, the authors also proposed a model for mortality prediction using naive Bayes and support vector machine (SVM). With AUROC of 0.73, SVM-based model performed better than naive Bayes. The possible reason for poor performance of naive Bayes model was that the data set was small (number of patients was 741 where only 151 had sepsis and others were of control group). Nemzek et al. 27 applied Bayesian networks to the protein and cellular components of sepsis-related organ failure and elucidated the intricate relationships among the variables that cause lung dysfunction. Although this study provided detailed insights on development of sepsis-related lung dysfunction, it used the protein and cellular components that are not directly accessible and required laboratory analysis.
Some studies use the variant of Bayesian networks known as dynamic Bayesian networks (DBNs) to improve the performance of sepsis diagnostic system. Peelen et al. 13 proposed a clinical support system for medical practitioners to predict organ failures in intensive care units (ICUs). They developed three different models of the problem, and each model is an advancement of the previous. Model 1 takes into account the number of organ failures, Model 2 considers the transition of one organ failure to other, and Model 3 incorporates the dependency of one organ failure to other organs, that is, independent assumption was relaxed. The authors captured the dynamic nature of patients’ recovery in ICU using a Markov chain. The states in the Markov chain denote various conditions of patient’s stay in the ICUs. The transition probabilities are calculated using logistic regression. The limitation of the study is that the authors used data only from ICU admissions; therefore, it was not clear to what extent the findings could be generalized to non-ICU admissions. Another study by Sandri et al. 28 developed a model to predict the sequence of organ failure in a patient using DBNs. The main limitation of this study was that the organ dysfunctional time was not known, therefore, authors used orders of organ failure based on physicians’ recommendations.
In this study, we capture important interactions among biomarkers. The inclusion of dynamics among biomarkers differentiates our model from existing models. In addition, the existing sepsis diagnostic models lack in providing balance between sensitivity and specificity.6,11 Due to the nature of unbalance between sensitivity and specificity, both qSOFA and SIRS were criticized and considered unhelpful. Therefore, practitioners require a more sophisticated diagnostic system. MEWS provides a better alternative, but its sensitivity, specificity, and AUROC are not strong. Although SOFA provides high performance accuracy, its complexity is the biggest challenge in resource-constrained environments. 10 In this article, we aim to develop an easy-to-use expert system using Bayesian network classifier that captures the interaction among clinical variables and facilitates a balance between sensitivity and specificity by selecting the optimal set of biomarkers.
Method
In this study, we used the following steps to build a predictive model: (1) data extraction, (2) data preprocessing (imputation, feature selection, and discretization), and (3) building TAN Bayesian network (Figure 1).
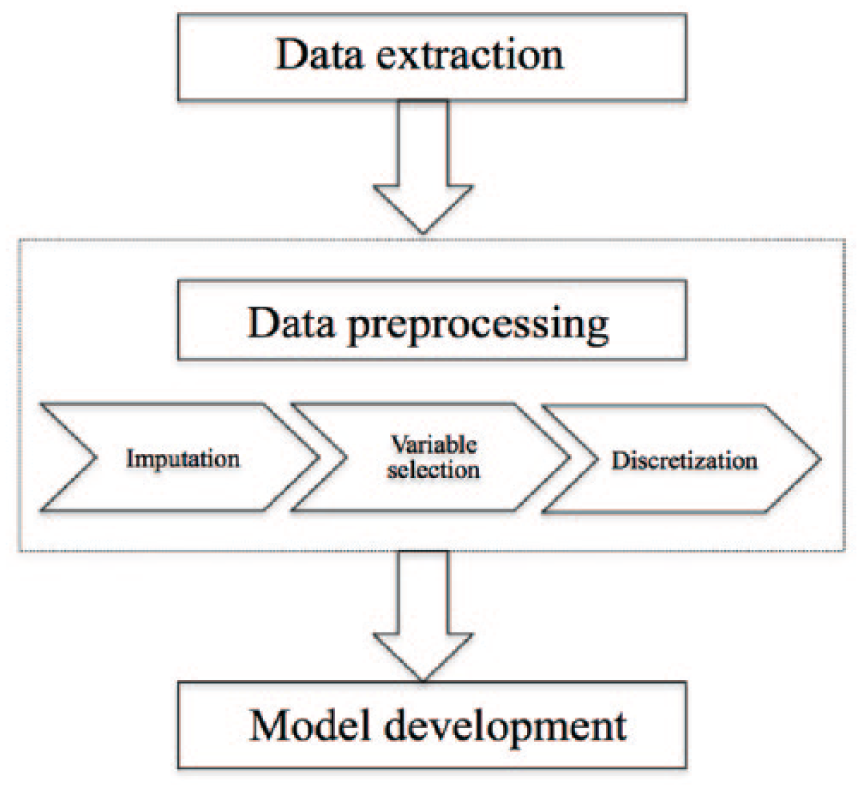
Systemic procedure to develop model.
Data extraction
This study used deidentified data from Cerner Corporations HIPAA-compliant Health Facts database. The database comprises electronic health records (EHRs) for 379 million patient visits, about 63 million patients, from 480 affiliated hospitals across the United States. These comprehensive clinical records for individual patient visits include demographic, vital signs, laboratory results, medication, diagnosis codes (both ICD-9 and ICD-10), pharmacy, procedures performed, and date- and time-stamped information on admission and discharge. This data set is one of the largest electronic medical records in the United States. 29
The population of suspected infection was selected using the gold standard explained in Sepsis-3. 5 The gold standard includes a combination of culture drawn (Event 1) and antibiotics administration (Event 2) within a defined time interval. There are two scenarios: (1) Event 1 occurs first, then Event 2 occurs within 72 h, (2) Event 2 occurs first, then Event 1 occurs within 24 h. The study cohort includes patient visits that satisfy either of the two aforementioned scenarios. The time of infection is the time of occurrence of the first event. The selection of culture and antibiotic was based on the consultation with practitioners. In the final data set, we had 16,909 patient visits. The outcome variable was discharge type (expired or nonexpired).
Data preprocessing
Data preprocessing includes three main steps. First, the missing values in the data were imputed using the proposed
Imputation
This section explains the proposed
In the proposed imputation method, we utilized observed biomarker measurements to impute unobserved biomarkers of individual patient’s visit. For this study, we divided the patient’s length of stay after the first suspicion of infection into 24 h (or 1 day) windows. We selected 24 h windows because the popular existing criteria such as SOFA uses the worst value recorded in 24 h.
21
The imputation procedure is easy to explain with the example shown in Figure 2. In Figure 2,
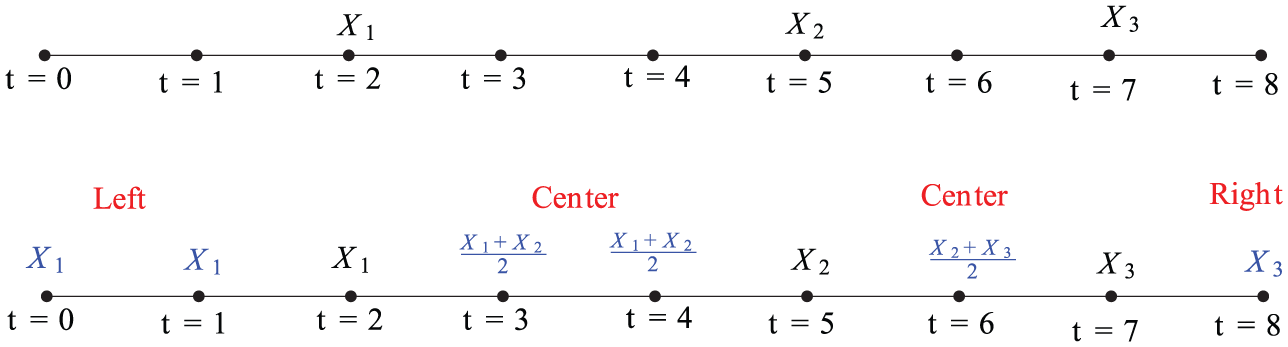
We compared the imputed data generated from the proposed imputation method to the
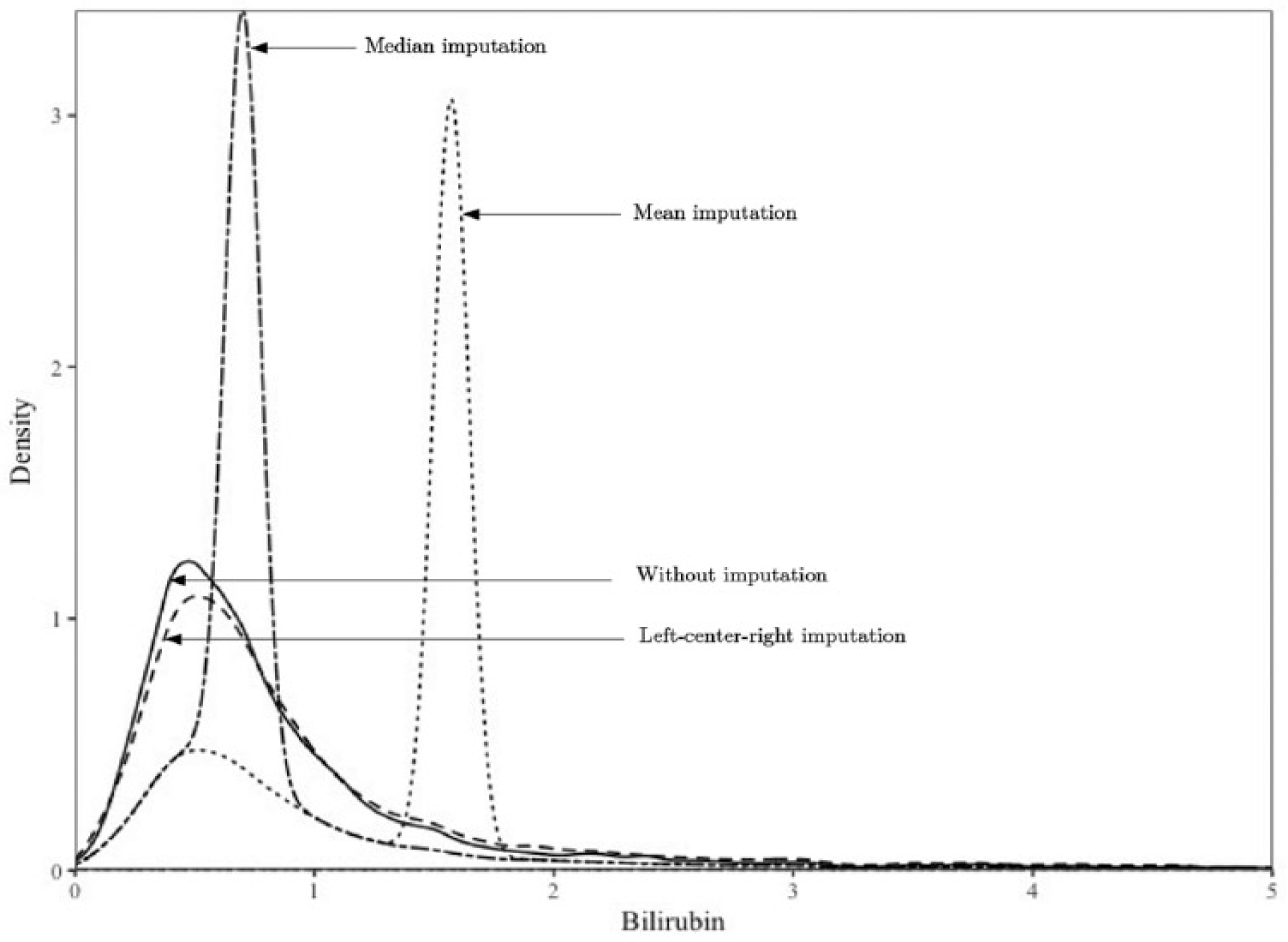
Density plots for different imputation techniques.
Variable selection
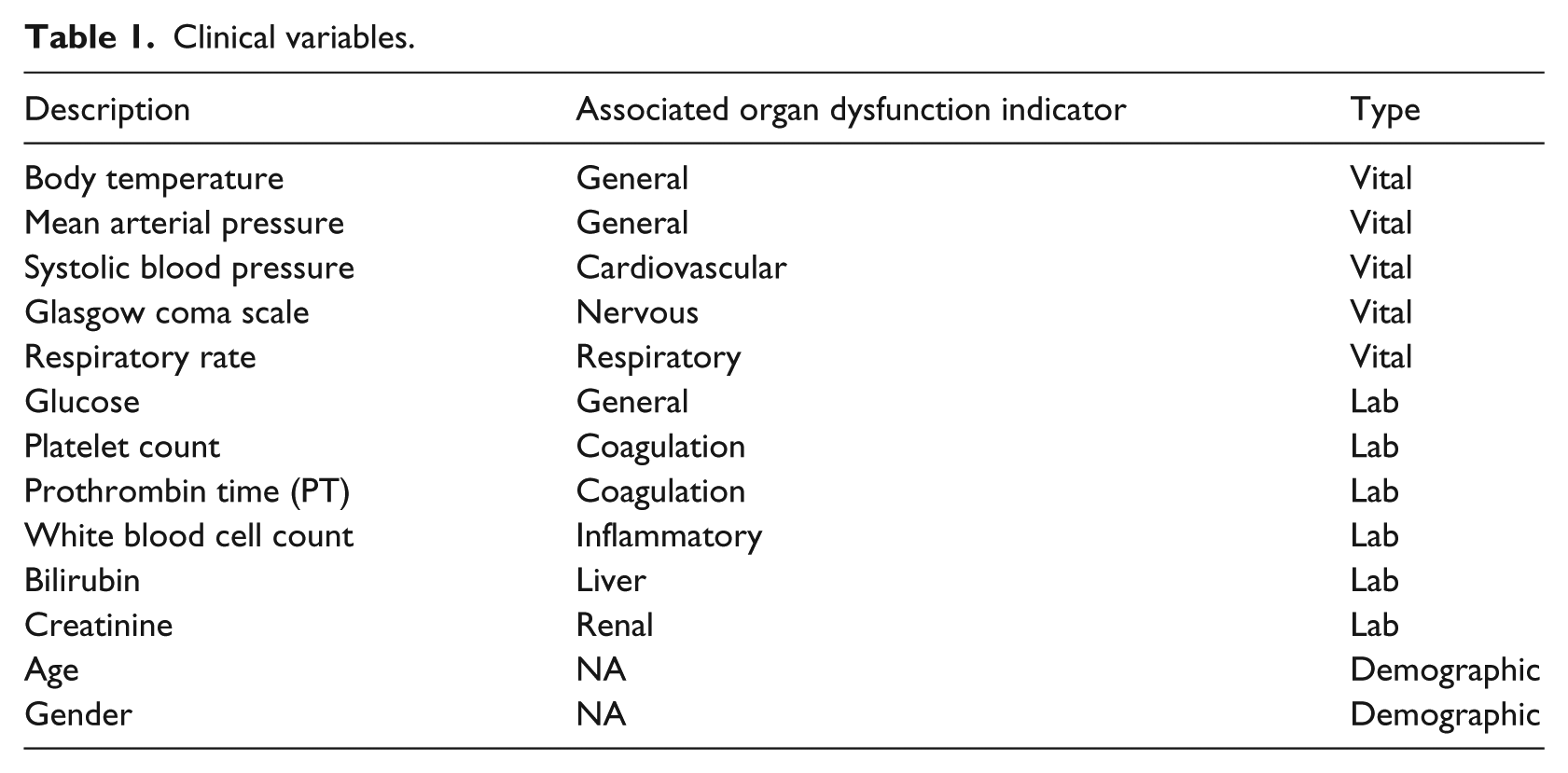
Variable selection is the most critical part of the analysis because most clinical results have lead time and cost associated with it. Also, Capan et al. 31 has shown that not all organ failures are equally associated with sepsis mortality. Therefore, to keep our model simple, and at the same time maintain high performance, we need to establish the best subset of variables. Table 1 includes the names and types of the variables initially selected in this study. The primary selection of biomarkers was based on the literature and discussion with practitioners. Later, the best subset of biomarkers among the primarily selected biomarkers was obtained using the machine learning techniques.
Clinical variables.
We used both elastic net 32 and recursive partitioning 33 for variable selection. The two methods stand on different principals. The elastic net pulls the coefficients of unimportant variables toward zero by minimizing the sum of square errors and regularization term. We used the elastic net rather than the stepwise selection because it facilitates shrinkage and avoids overfitting using the regularizing factor. The shrinkage implies reducing the complexity of the model by dragging the weights of unimportant biomarkers to zero. Shrinkage is an important characteristic for clinical applications because each biomarker (lab test or vital signs) is associated with a significant cost. The recursive partitioning focuses on minimizing the impurity present in the data by partitioning. For the final model, a conservative approach was applied and only variables that were suggested by both variable selection methods were used.
Zou and Hastie
32
proposed elastic net regression that utilized both
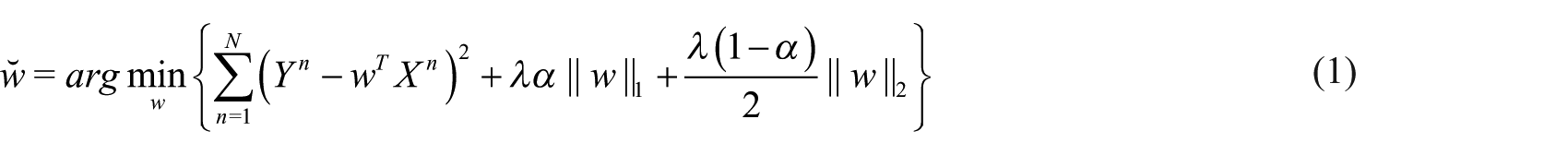
Here
To obtain an optimal set of variables, we searched for different
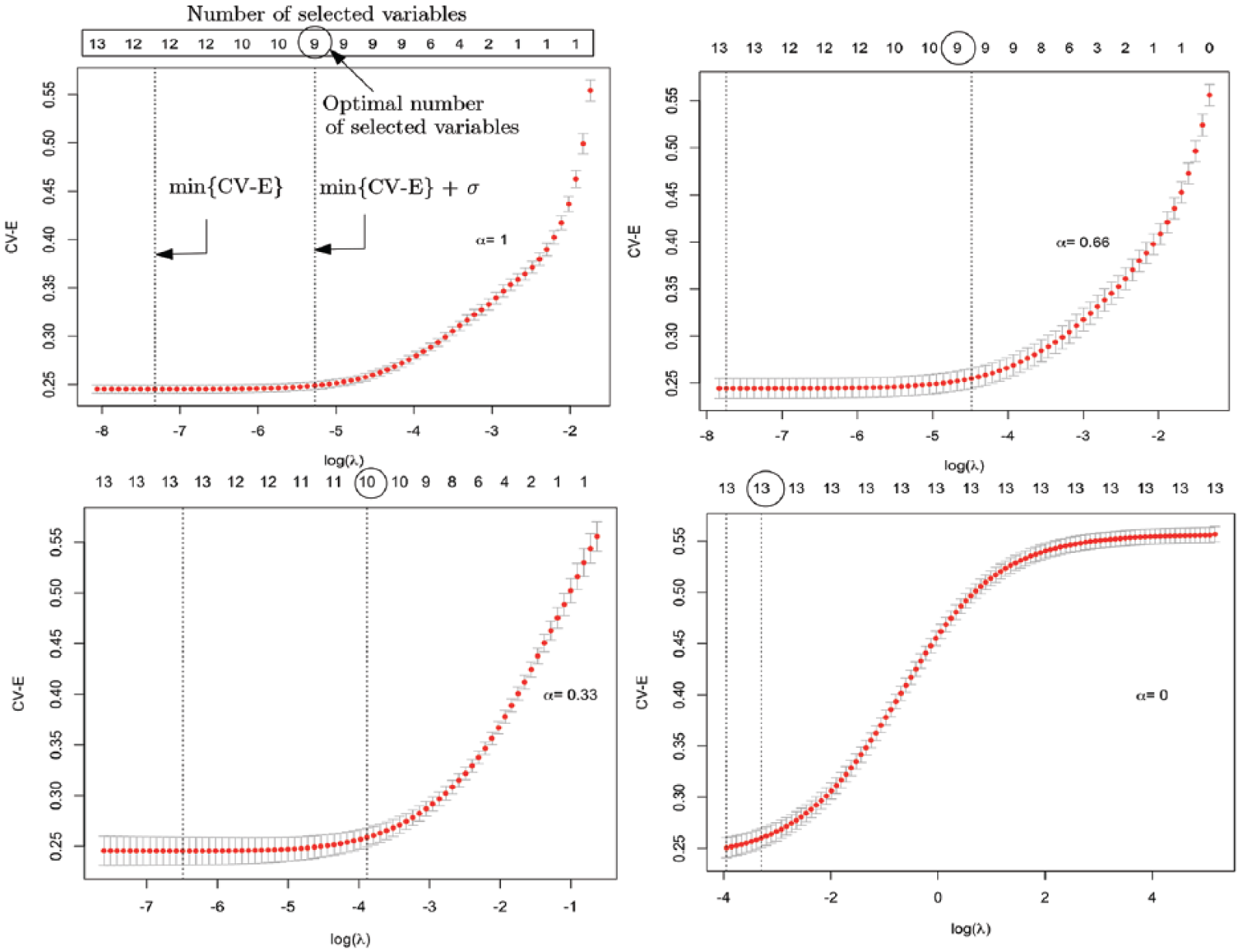
CV-E versus
Variable weights for

Italicized entries indicate variable having zero weight.
In recursive partitioning, the data are recursively partitioned to reduce the impurity present in the data. Each step of the recursive partitioning includes the splitting of data into two subgroups. The split is determined by the variable that reduces the impurity most. We used two methods to measure the impurity:


Where
The recursive partitioning can be summarized as follows:
1. Select variable that provides highest information gain (or reduction in impurity). The information gain is calculated using the following expression

2. Split data using the selected variable.
3. Repeat 1 and 2 until information gain is greater than a predefined threshold.
For our data, recursive partitioning using both splitting criteria resulted in the same classification accuracies (0.96). Since we had the same classification accuracies and the research has shown that both criteria disagree only in 2% cases, 34 we selected information index for further explanation of the results.
The variable importance of each biomarker obtained by recursive partitioning using information index is shown in Figure 5. Figure 5 illustrates a significant drop in variable importance from respiratory rate (RR) to mean arterial pressure. Therefore, we performed a sensitivity analysis to inspect the change in accuracy for two scenarios: considering only first five important variables (dotted variables in Figure 5), considering all variables with nonzero variable importance. The classification accuracy of the model (0.96) with five variables (dotted variables in Figure 5) was the same as the classification accuracy of the model (0.96) with nine variables (both dotted and shaded variables in Figure 5). Therefore, we considered only five biomarkers from recursive partitioning: Glasgow coma scale (GCS), creatinine, blood pressure, white blood cell (WBC), and RR.
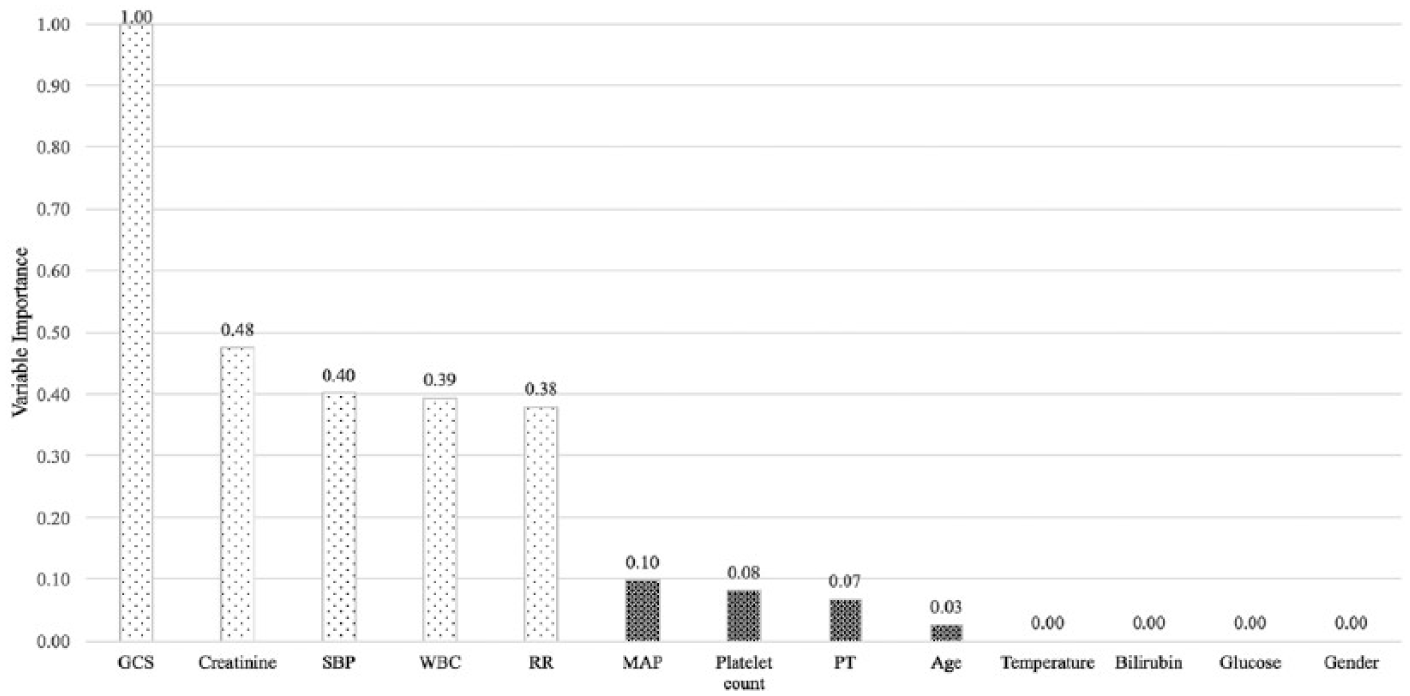
Variable importance from recursive partitioning.
We summarized the variables selection using elastic net (
Selected variable.
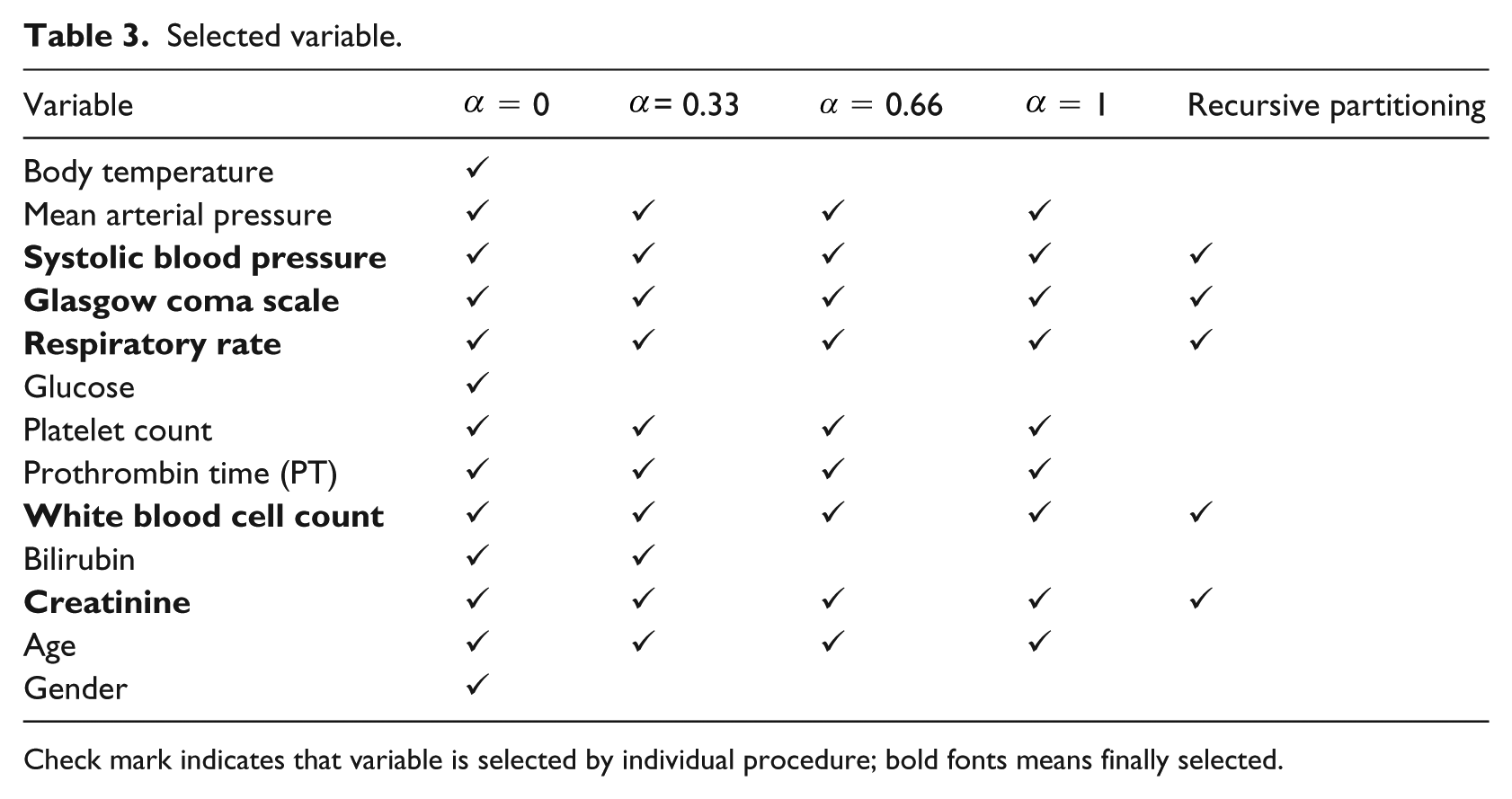
Check mark indicates that variable is selected by individual procedure; bold fonts means finally selected.
Discretization
Many methods have been developed to discretize continuous variables, and the performance of these methods depends on the shape and the nature of the data. Some approaches such as quantile and equal intervals utilize variables in isolation, while others such as Hartemink 30 consider relationship among variables to decide the cut-offs.
The correlation analysis uncovers significant dependencies among biomarkers. Figure 6 illustrates correlations among biomarkers. The size of the circle represents the strength of the correlation between biomarkers. The cross signs show that the correlation was nonsignificant. Therefore, a relationship among biomarkers should be utilized for discretization to avoid the loss of information. Hence, we selected Hartemink method to efficiently compute the discrete levels of biomarkers. Another reason why we chose it is that research has shown that Hartemink 30 discretization performs better with Bayesian classifiers.

Correlation analysis among biomarkers.
Hartemink discretization assumed that the continuous variables are normal by distribution. Therefore, we inspected the density plot of each variable. Using the Box-Cox method,
35
the transformed variables were obtained when original variables were deviating from normal, except for the GCS sore. For example, the original distribution of platelet count and its transformed distribution are shown in Figure 7. The GCS score was heavily left skewed, therefore, no better distribution was found using the Box-Cox method. Hence, the cut-offs
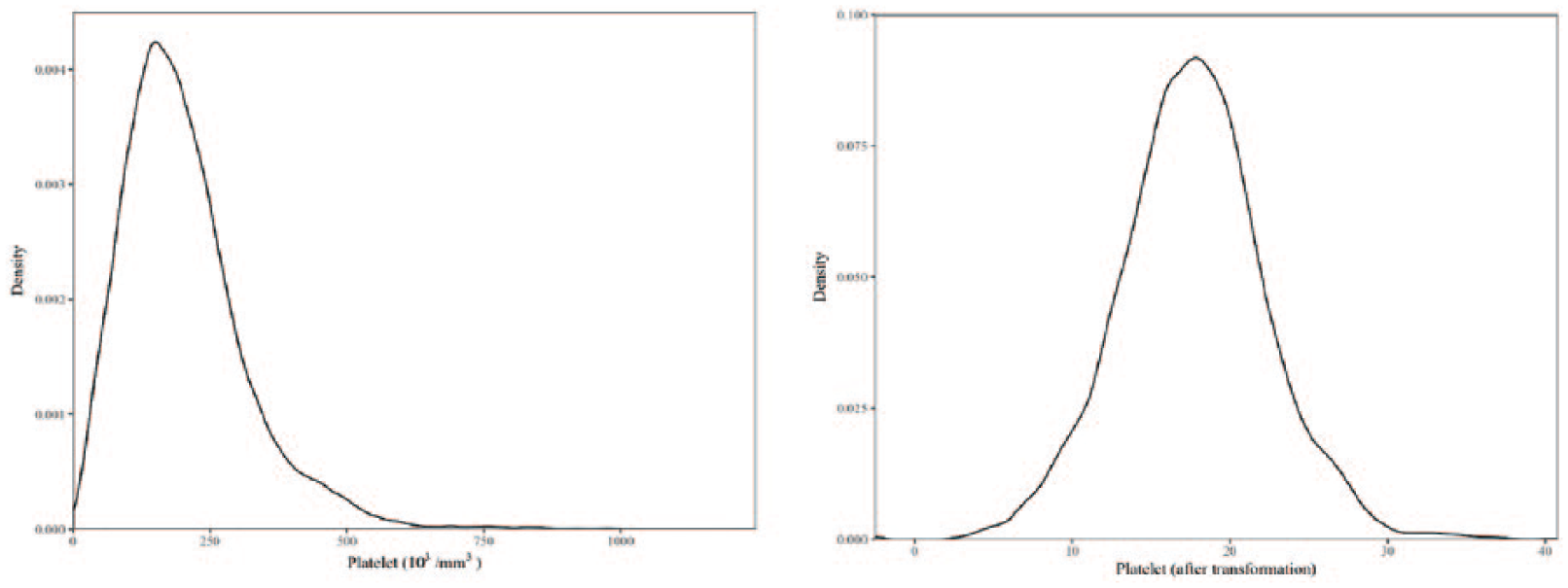
Platelet (before and after transformation).
In the Hartemink method, each continuous variable initially is segregated into a large number of levels using any of the traditional methods such as equal intervals. This large number of levels reduces to a specific number, defined by the users, by coalescing selected neighboring pair to a single level. The mutual information (MI) between the selected variable and other variables is calculated. The levels that result in a minimum loss of information with any of the other variables is coalesced with neighboring level. For our experiments, we first divided each variable into 10 levels and then coalescing was done with the Hartemink procedure until each variable reached three levels. Based on physicians’ opinion, three discrete levels facilitate simplicity and meaningful clinical interpretation. Some biomarkers are considered as
The computed discrete levels of the selected variables provide important insights. The patterns of mortality over different discrete levels of biomarkers (shown in Figure 8) are supported by practitioner’s knowledge. For example, practitioners suggested that both low and high RRs are associated with increased mortality. The discrete levels obtained from our analysis also showed the same pattern. However, there exists slight differences in the standard cut-offs and cut-offs obtained from discretization. The standard RR for a normal patient is 12 to 20 breaths per minute. However, we obtained least mortality for RR between 16 to 22 breaths per minute. It is possible that generated cut-offs work well for sepsis specifically because the upper cut-off is supported by the recent Sepsis-3 study, where RR greater than 22 was proposed as the cut-off for increased risk of mortality.
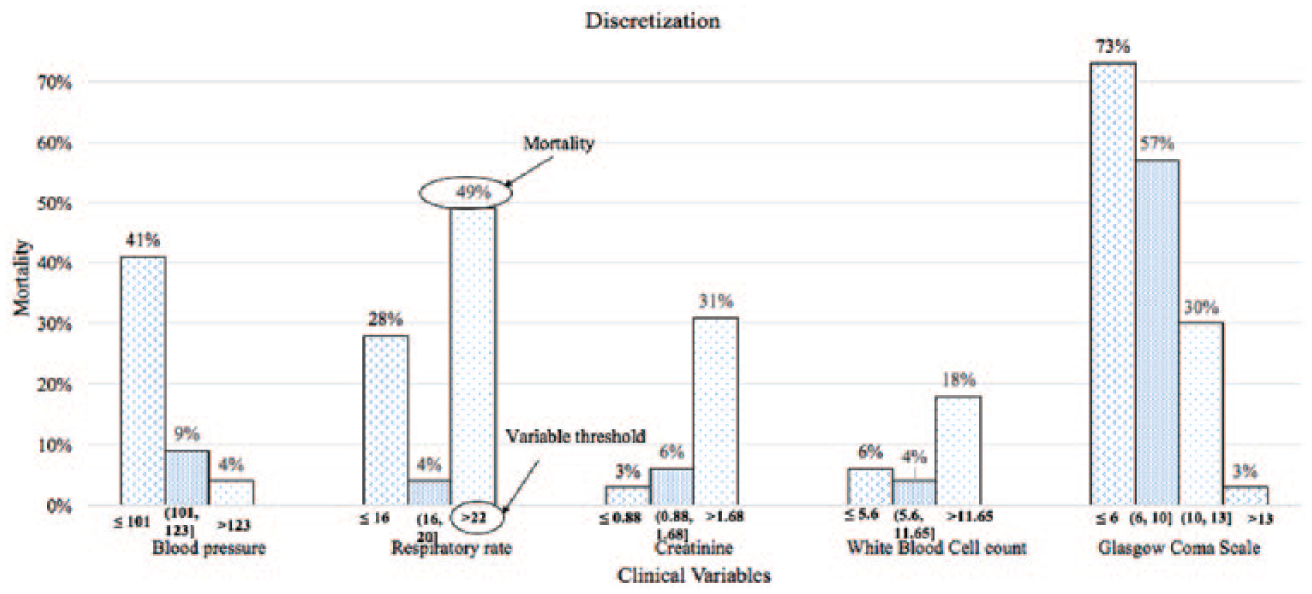
Discrete levels of selected biomarkers and corresponding mortality rate.
Model development
Bayesian network is a probabilistic model with directed acyclic graph. Combination of nodes and arcs is used for the graphical representation. Each node represents a random variable, and each arc implies conditional dependencies between variables. Bayesian networks are popularly used in medical research because they capture the uncertainties and nonlinearities presented in data using graphical representation that is easy to understand. 36
There are two popular Bayesian classifiers: Naive Bayesian and TAN Bayesian. The Naive Bayes classifiers are simple and assume conditional independence between variables given the outcome variable. TAN improves Naive Bayes classifier to capture important interactions among variables and at the same time maintains the mathematical simplicity. In TAN, the outcome variable (mortality risk) has no parent, and each predictor can have at most two parents: class variable and any other biomarker. The procedure to learn the structure of the proposed model is explained as follows. 37
Step 1: Calculate the MI between each pair of biomarkers using the following equation
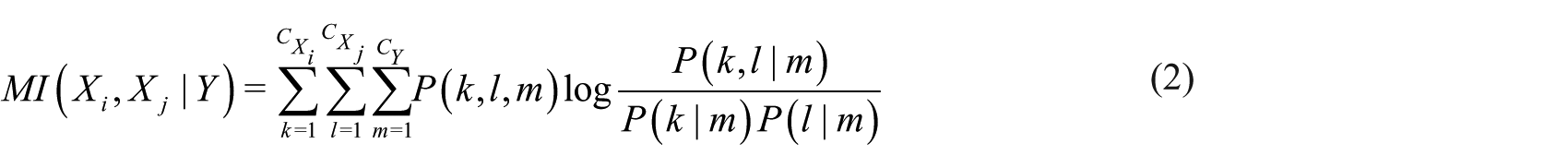
where
Step 2: Build a complete undirected graph with the MI as the weight of edges.
Step 3: Find the maximum weighted spanning tree.
Step 4: Transform the undirected tree obtained from Step 3 to a directed one by choosing a root variable either arbitrarily or by expert’s opinion, and setting the direction of all edges to be outward from it. In this study, the selection of root node was performed using an expert’s opinion such that the selection does not violate any domain knowledge. The establishment of direction does not change the data likelihood because MI(
Step 5: Include new node (mortality risk) and connect it with the rest of the graph obtained in Step 4.
The steps to construct TAN Bayesian network for the current sepsis model are shown in Figure 9.
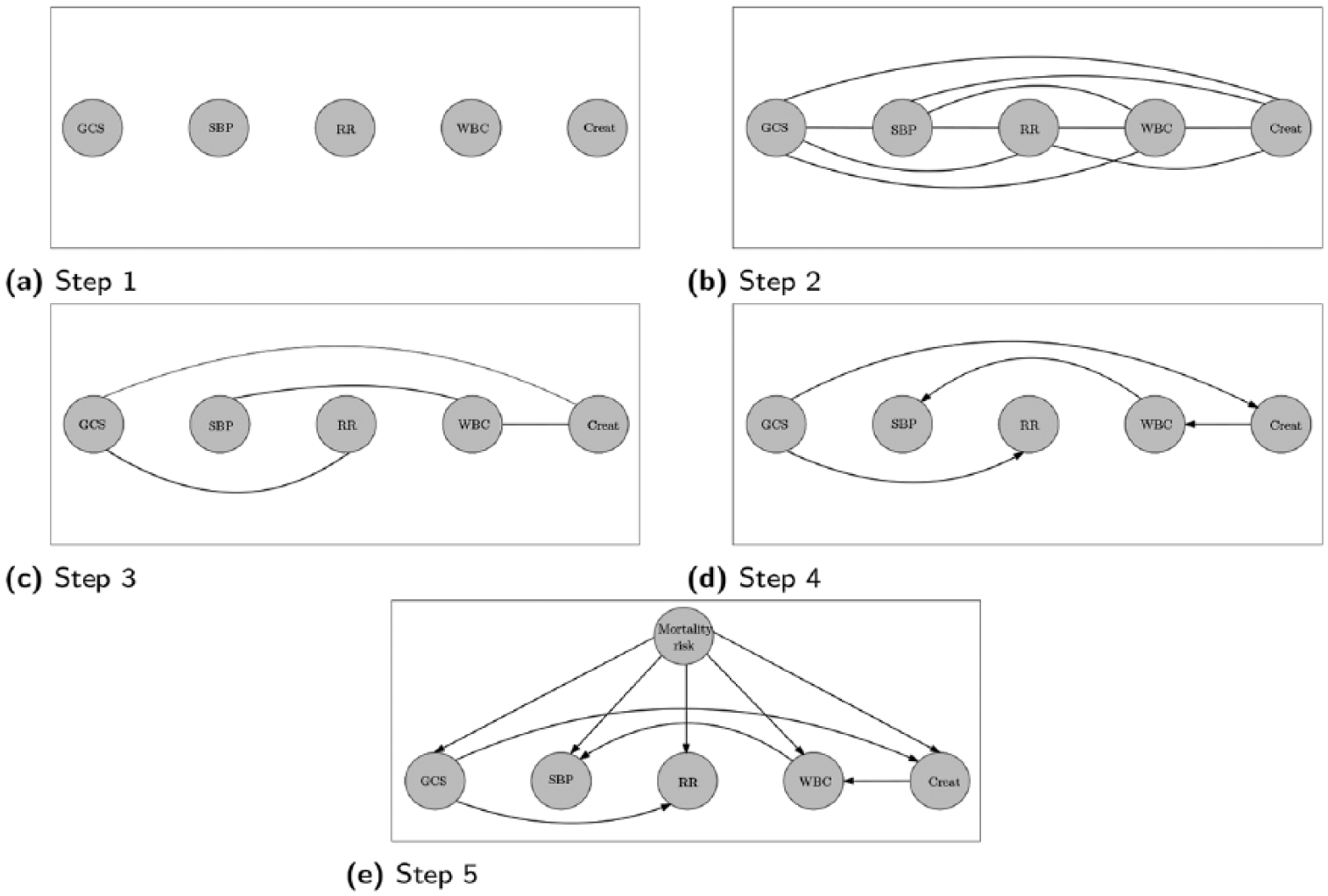
Structure learning of the TAN Bayesian network model.
After learning the TAN structure, the conditional probabilities are calculated using equation (3)
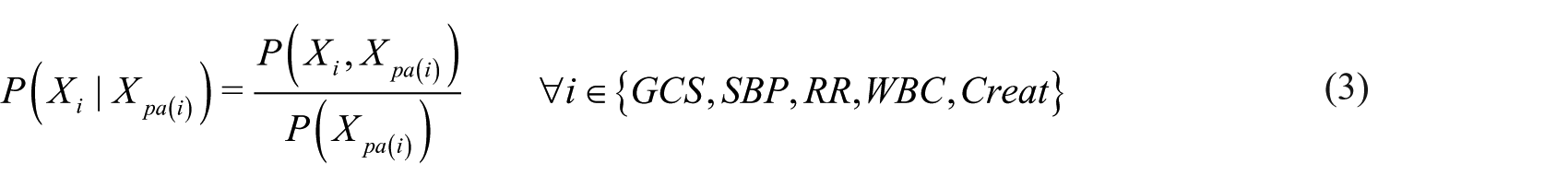

The estimated conditional probabilities were used for the inference. For our model, the inference can be derived using Bayes’ theorem as follows
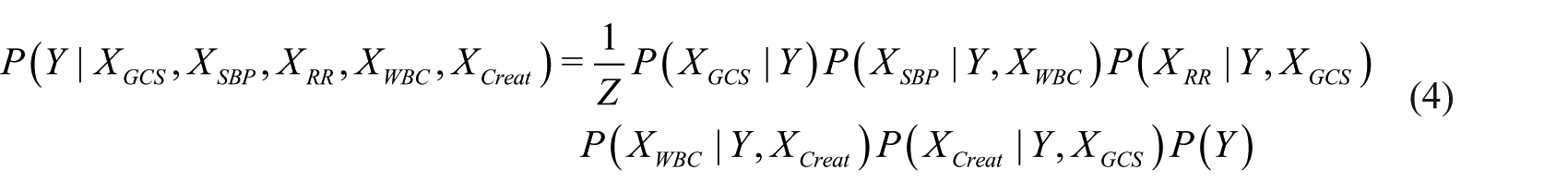
The inclusion of interactions among biomarkers differentiates our model from others. Most relationships discovered by TAN Bayesian network were supported by practitioner knowledge except the relationship between WBC and blood pressure. Based on expert’s opinions, the GCS is most likely to relate with RR and creatinine, and creatinine possibly affects WBC; but the relationship between WBC and blood pressure is not so apparent. Our results show eliminating the arc between WBC and blood pressure will marginally lower the overall performance of the model. Furthermore, we inspected the correlation coefficient between WBC and blood pressure and observed statistically significant negative correlation (
Results and discussion
In this section, we compare the performance of the proposed model with SIRS, qSOFA, MEWS, and SOFA. It is worth noting that MEWS and SOFA are not designed to predict mortality. MEWS is used to detect criticality of ill patients, and SOFA is used to understand the health of body organs. However, these criteria are commonly used in medical settings and provide a good benchmark to evaluate the performance of a model. 38
The metrics to evaluate the performance of a classifier are AUROC, sensitivity, specificity, and geometric mean (G-mean). The AUROC is most commonly used in medical literature to evaluate the diagnostic capability of a classifier as its discriminative threshold is varied. The AUROC of 1 corresponds to an ideal model, while 0.5 corresponds to the worst. Sensitivity is the true positive rate, and specificity is the true negative rate. G-mean is the geometric mean of sensitivity and specificity.

G-mean is often used to inspect the balance between sensitivity and specificity. 15
Our model estimates the risk of mortality. The estimated probabilities were translated into categorical outcome by using a cut-off point. This cut-off point governs the trade-off between sensitivity and specificity, therefore, the selection of cut-off point was given special attention. Instead of using default decision threshold (0.5), we explored previous studies to understand the best approaches to find the optimal cut-off point without considering the misclassification cost and prevalence. There exist two popular methods to find the optimal cut-off point: (1) point in receiver operating characteristic curve closer to (0, 1); first index represents false positive rate and second represents true positive rate, and (2) Youden index.39,40 Youden index determines the cut-off at which (sensitivity + specificity: –1) is maximum. Perkins and Schisterman 39 compared these two methods and identified advantages and disadvantages of selecting one over the other. The ideal model passes through the point (0, 1). Therefore, any point in receiver operating characteristic curve closer to (0, 1) is visually more meaningful. 39 Hence, cut-off point corresponding to the point closer to (0, 1) was considered as an optimal cut-off point and was used to estimate sensitivity and specificity.
The model validation was performed using fivefold cross-validation. Figure 10 illustrates the validation procedure. We divided the data into five equal folds. One fold was used for the validation and the remaining were used to train the model. This procedure was carried out iteratively to consider each fold for validation. The final performance measures (AUROC, sensitivity, specificity and G-mean) were estimated by taking the average of performance measures obtained from five mutually exclusive data folds.
Model validation approach.
We compared the AUROC of the proposed model against the alternate models (SIRS, qSOFA, MEWS, and SOFA) over different time instances prior to discharge. Figure 11 illustrates the AUROC of our model and competing models preceding the discharge time. The result indicates that our model performed better than competing models over all time instances before the discharge. The percentage differences in AUROC between our model and the alternate models (SIRS, qSOFA, MEWS, and SOFA) ranged from 30%–50%, 22%–32%, 8%–11%, 3%–5%, respectively. This longitudinal analysis presents results in favor of the robustness of our model.
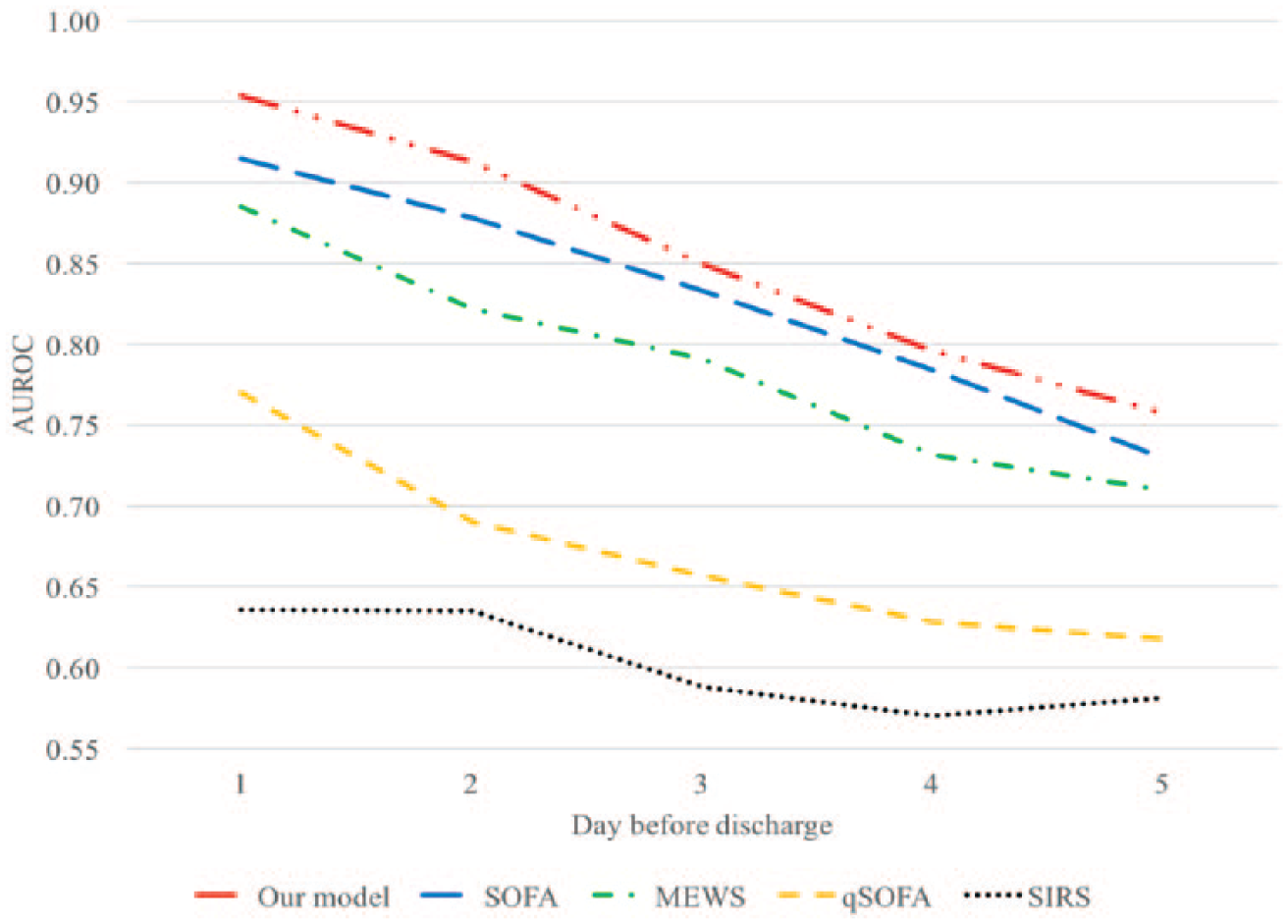
AUROC over the different time before the discharge.
We also compared the time-average performance of our model to alternate models (SIRS, qSOFA, MEWS, and SOFA). The sensitivity, specificity, G-mean, and AUROC of our model and competing models are shown in Figure 12. With AUROC of 0.84, our model outperformed other criteria (SIRS = 0.59, qSOFA = 0.65, MEWS = 0.75, SOFA = 0.80). In addition, our model provides the best balance between sensitivity and specificity, and this can be seen evidently using G-mean. The G-mean of our model (0.75) is greater than SIRS (0.55), qSOFA (0.58), MEWS (0.70), and SOFA (0.73).
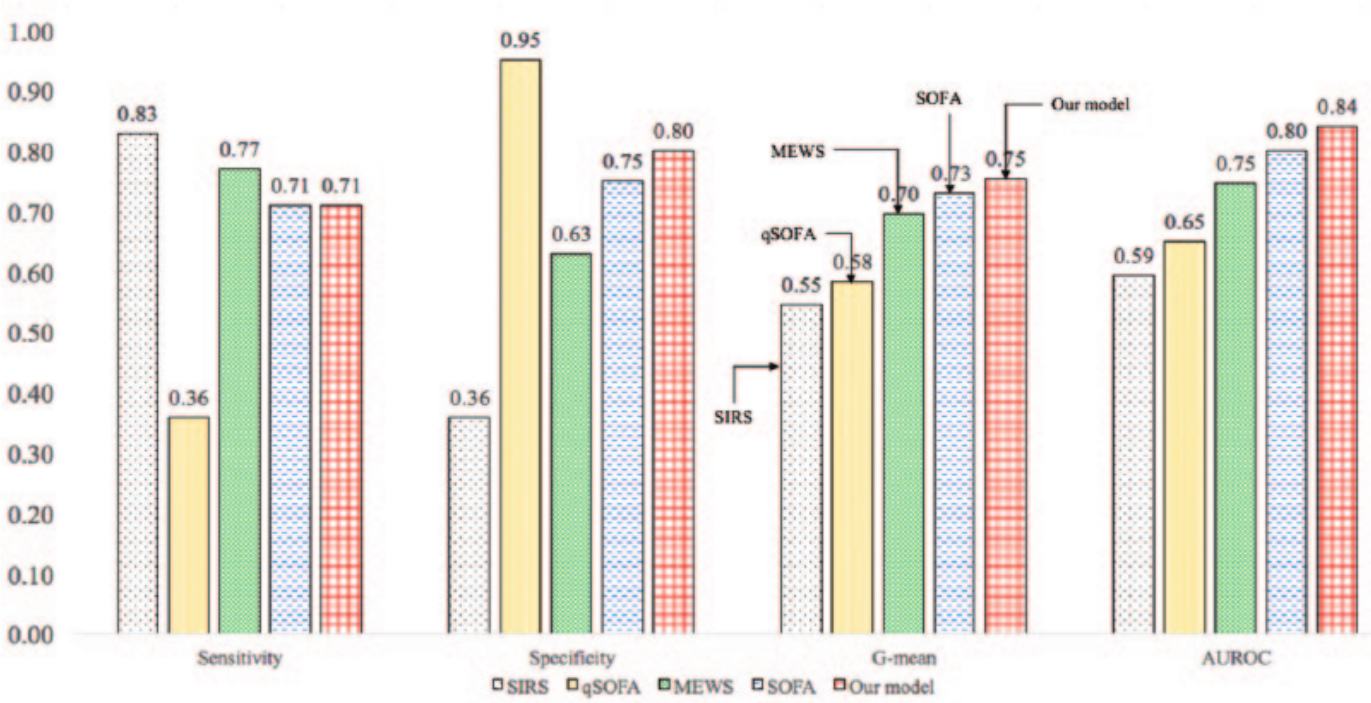
Comparison of performance of different diagnostic criteria.
Many clinical applications of a model necessitate high sensitivity. Therefore, for the same AUROC (0.84), Table 4 reports specificity on varying values of sensitivity. The results show that at very high sensitivity (0.99), the model loses the specificity significantly. Depending on the requirement on sensitivity, we can increase the specificity.
Specificity on varying values of sensitivity.

The primary reason for the better performance using TAN Bayesian network is the presence of correlations among biomarkers. TAN considers correlations, while other modeling techniques treat all variables as independent. TAN Bayesian network usually performs better when variables are correlated. 17 The physiological variables more often than not show correlation among biomarkers. For example, in our EHR data, the correlation between WBC and blood pressure is –0.12. Therefore, the performance gain of our model could be attributed to the existence of correlation among biomarkers.
Table 5 summarizes selected biomarkers and the performance characteristics of each model. Although our model uses more variables than SIRS and qSOFA, it performs significantly better than these models. SOFA score requires the knowledge of four laboratory results. Our model with only two laboratory results performs marginally better than SOFA. The use of fewer laboratory results enables practitioners to predict the risk of sepsis easily and quickly. The performance gain of our model using TAN Bayesian network is attributed to the presence of correlation between biomarkers in EHR data.
Biomarkers and the performance characteristics of different sepsis prognostic models.
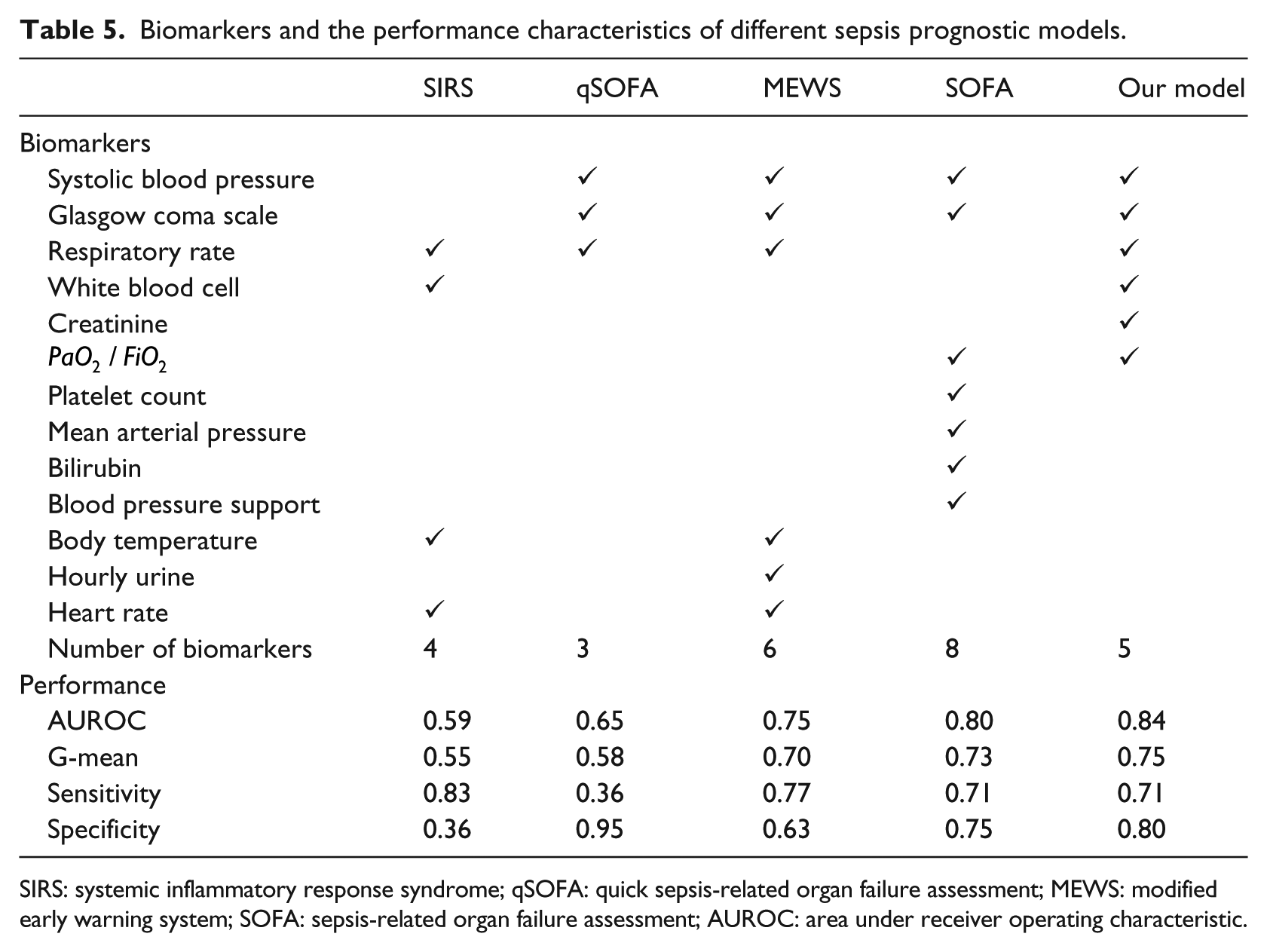
SIRS: systemic inflammatory response syndrome; qSOFA: quick sepsis-related organ failure assessment; MEWS: modified early warning system; SOFA: sepsis-related organ failure assessment; AUROC: area under receiver operating characteristic.
Conclusion
In this article, we developed a predictive model using TAN Bayesian network that inherently captures the important interactions among biomarkers and compared the performance of our model with SIRS, qSOFA, MEWS, and SOFA. The proposed model was trained with the population selected using the new sepsis definitions. The model development process includes the following main steps: data extraction, data preprocessing, learning of TAN Bayesian network, and model evaluation. The variable selection identifies biomarkers meaningful to predict sepsis. Out of a set of 13 variables, five variables were selected (SBP, GCS, RR, WBC count, and creatinine) to develop the TAN model.
The structure of the TAN network facilitates visual interpretation of the interactions among biomarkers. The model identified relations that were in align with practitioners’ knowledge. The comparative analysis showed that our model outperformed alternate models. The results underline the robustness of our model by comparing the AUROC of various models over different time instances prior to discharge. The proposed model eliminates the existing problem of unbalanced sensitivity (SIRS = 0.83, qSOFA = 0.36) and specificity (SIRS = 0.36, qSOFA = 0.95) and delivers balanced sensitivity (0.71) and specificity (0.80). The key characteristic of our model is that it outperformed MEWS and SOFA and uses fewer variables than them. The use of fewer lab results enables quick prognosis of sepsis. In comparison with SIRS, qSOFA, MEWS, and SOFA, our model provides the best alternative and can easily be integrated in EHR environment and autonomously identify the patient at high risk of sepsis.
The limitation of this study is that the data come only from hospitals using Cerners EHR system. So, there could be potential sources of bias in the data. Although we included data from hospitals located in all four U.S. census regions, we cannot generalize the results to all U.S. hospitals as there could be distinct differences between hospitals using the Cerner system and other hospitals. The possible reason of difference is that the Cerner’s EHR is mainly used in big hospitals because it is expensive to implement. We did not consider the effect of intervention by assuming that each patient was subjected to a similar intervention.
Footnotes
Authors’ Note
This work was conducted with data from the Cerner Corporation’s Health Facts database of electronic medical records provided by the Oklahoma State University Center for Health Systems Innovation (CHSI). Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the Cerner Corporation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
