Abstract
We use prescription of statin medications and prescription of warfarin to explore the capacity of electronic health record data to (1) describe cohorts of patients prescribed these medications and (2) identify cohorts of patients with evidence of adverse events related to prescription of these medications. This study was conducted in the WWAMI region Practice and Research Network (WPRN)., a network of primary care practices across Washington, Wyoming, Alaska, Montana and Idaho DataQUEST, an electronic data-sharing infrastructure. We used electronic health record data to describe cohorts of patients prescribed statin or warfarin medications and reported the proportions of patients with adverse events. Among the 35,445 active patients, 1745 received at least one statin prescription and 301 received at least one warfarin prescription. Only 3 percent of statin patients had evidence of myopathy; 51 patients (17% of those prescribed warfarin) had a bleeding complication. Primary-care electronic health record data can effectively be used to identify patients prescribed specific medications and patients potentially experiencing medication adverse events.
Introduction
Electronic health record (EHR) data are increasingly used for research, though much of the evidence is from research conducted using EHR data from large health systems. EHRs are designed for billing, patient care documentation and communications (e.g. e-prescribing).1,2 Potential challenges to conducting research with EHR data are well documented and include assessing and accounting for missing data, inconsistencies in data entry among clinical providers and inability to easily obtain data stored in free text. 1 Despite these limitations, use of EHR data for research offers unprecedented opportunity to address critical scientific questions that may be otherwise difficult to answer using claims data or traditional clinical research. 3 Medication adverse events is a field especially well suited for research with EHR data. 4 Study of medication adverse events often requires large numbers of potential subjects and the ability to identify relatively rare patients who might be experiencing adverse events. Primary care settings, the majority of which use EHR systems, are optimal for understanding epidemiology and clinical implications of medication adverse events, because of the prevalence of prescription medication use in primary care settings and continuity of care permitting longitudinal observations. EHR data from primary care systems can also allow inclusion of research priority populations, such as rural residents, who may not participate in medical research conducted exclusively in academic health centers. EHRs typically capture medication data because physicians routinely order and transmit prescriptions to pharmacies electronically (www.healthit.gov). EHRs additionally capture patient demographics, clinical laboratory and diagnosis information linked to prescription data. In contrast, claims or billing data have limitations which may create significant barriers for research, such as exclusion of patients without insurance and inability to link prescriptions to clinical data such as diagnoses or laboratory results.5,6
We use EHR data from primary care practices to explore two key classes of medications, both of which are commonly prescribed in primary care and have associated potential adverse events—statins and warfarin. Statins are commonly prescribed medications that are effective in treating hyperlipidemia and reducing cardiovascular disease risk.7–11 Statin-induced myopathy is an adverse drug-related condition that includes myalgia, myositis and rhabdomyolysis due to the use of statin medications. 12 The incidence of statin-induced myopathy reported in clinical trials ranges from 0.44 to 5.34 per 10,000 person years.13,14 Warfarin is an effective anticoagulant that reduces the risk of thromboembolism in patients with atrial fibrillation.15,16 Treatment with warfarin requires regular blood test monitoring and dosage adjustment to achieve adequate therapeutic response and minimize risk of complications. We focused on warfarin, rather than newer oral anticoagulants, which were not in widespread use during the time period covered by the EHR data extract. Overtreatment with warfarin may lead to adverse events due to excessive bleeding risk, such as hemorrhagic stroke or gastrointestinal hemorrhage.
The rationale for this study is to explore the capacity of EHR data within a network of community-based primary care practices to achieve three objectives: (1) identification and description of cohorts of patients prescribed statin or warfarin medications, (2) characterization of the types of prescription information available in the EHR data and (3) identification of cohorts of patients prescribed statins or warfarin with evidence of adverse events relevant to these medications. Given the widespread availability of EHR data for research purposes, further description of methods for conducting research with these data as well as elucidation of the limitations are needed. The findings from this study provide approaches for future clinical researchers studying medication adverse events, potentially supporting observational clinical research or recruitment of patients from primary care settings.
Methods
Setting and data source
This study was conducted in the WWAMI region Practice and Research Network (WPRN) region Practice and Research Network (WPRN), a network of more than 60 primary care practices across Washington, Wyoming, Alaska, Montana and Idaho (WWAMI). At the time of this study, a subset of nine clinics participated in Data Query Extraction Standardization Translation (Data QUEST), an infrastructure for sharing outpatient primary care EHR data for research. 17 Data QUEST practices represent diverse primary care practices, including federally qualified health centers and rural-serving primary care practices. Data QUEST includes patient-level data stored securely within local practices’ firewalls in aligned data repositories and uses a federated data-sharing infrastructure to support regulation-compliant governance of data between primary care partners and researchers.17,18 Data QUEST’s infrastructure and governance maintain compliance with Health Insurance Portability and Accountability Act (HIPAA) and Institutional Review Board (IRB) regulations. Data QUEST targets extraction from main data domains in the EHRs, including demographics, vital signs, diagnosis codes, diagnostic test results, social and family history, prescriptions and physical examination findings.
For this project, we worked with two clinical organizations from the WPRN that participate in Data QUEST. These organizations were selected based on data availability at the time of the research. The two organizations represent six clinics. Each clinical organization is an independent entity, with distinct instances of CentricityTM EHR systems. The Human Subjects Review Board at the University of Washington reviewed and determined this study to be exempt.
Time period
This study required continuous EHR data over a 2-year period so that we could both identify patients prescribed a medication and have sufficient observation time to capture potential adverse events. For organization A, the observation period we used was from 1 December 2010 to 30 November 2012, and for organization B, the observation period we used was from 1 September 2012 to 31 August 2014. The time periods were selected based on the most recent two-year period during which each organization had continuously available EHR data. Organization A had 6 years of EHR data (1 July 2006 to 30 November 2012) and organization B had 5 years (1 July 2009 to 31 August 2014) of EHR data.
Measures
Active patients
We defined active patients as those with either a blood pressure recorded by the clinic or a Current Procedural Terminology (CPT) code for an in-person primary care office visit at any time during the 2-year study period noted above. Only patients who were aged 18 years or older on the first day of the study period were included.
Demographics
Demographics included patients’ gender, age and race/ethnicity (American Indian/Alaska Native, Asian, Black or African American, Caucasian, Hispanic or Latino, Multiracial, Native Hawaiian or Pacific Islander or No information). Race/ethnicity data were combined into a single field in both EHR systems that contributed data for this study.
Prescribed medications
Using EHR-prescribed medications data, we identified patients with at least one prescription ordered for either a statin medication or warfarin during the study period. We created string-based search terms that included all brand and generic names for the medication groups (warfarin and statins) to identify relevant medication prescription data. We used a wild card search strategy to identify complete or partial medication names within the medication fields that would be indicative of either statin or warfarin use. Coded medication data, such as national drug code (NDC) was not available in either EHR system at the time of this study.
Statin or warfarin-related medication adverse events
For inclusion as a statin- or warfarin-related adverse event, the adverse event had to occur after the first prescription in the 2-year study period for the selected medication. Adverse events were selected based on clinical relevance and feasibility of measurement using outpatient EHR data. For detection of selected laboratory abnormalities, we used published reference ranges when available 19 or calculated the mean value in our dataset and designated three times the mean value as the cut off for abnormal. Among patients prescribed a statin medication, we included the following adverse events: (1) a diagnosis of myopathy or statin myopathy using appropriate International Classification of Diseases (Ninth Revision; ICD-9) codes (Appendix 1) and (2) laboratory evidence of elevated creatine kinase (creatine kinase ⩾486 units, based on 3× dataset mean), elevated aspartate amino transferase (AST; AST ⩾ 78, based on 3× dataset mean) or elevated alanine amino transferase (ALT; ALT ⩾ 87, based on 3× dataset mean). Among patients prescribed warfarin, we included the following adverse events: (1) a bleeding complication using an ICD-9 code (Appendix 1), (2) laboratory evidence of elevated international normalized ratio (INR; INR > 5), 19 and (3) receipt of vitamin K, based on evidence of a vitamin K medication electronic order.
Results
We counted the number of prescriptions per patient for both the statin and warfarin cohorts and reported the mean number of prescriptions per patient. To characterize the types of prescription information available in the dataset, we calculated the proportion of prescriptions that contained structured data reporting the dose, unit (e.g. milligrams), quantity (e.g. 30), signetur instructions (sig) or numbers of refills. For all patients in each of the medication cohorts, we calculated the proportions with evidence of medication-related adverse events.
Objective 1: identification and description of patients prescribed statin or warfarin medications
The dataset included 35,445 active, adult patients. Table 1 reports overall and individual organization counts of patients on statin (n = 1,745, 4.9% of patients seen during the time period) or warfarin (n = 301, 0.8%) medications.
Description of adults taking either statin medications or warfarin across two primary care organizations.
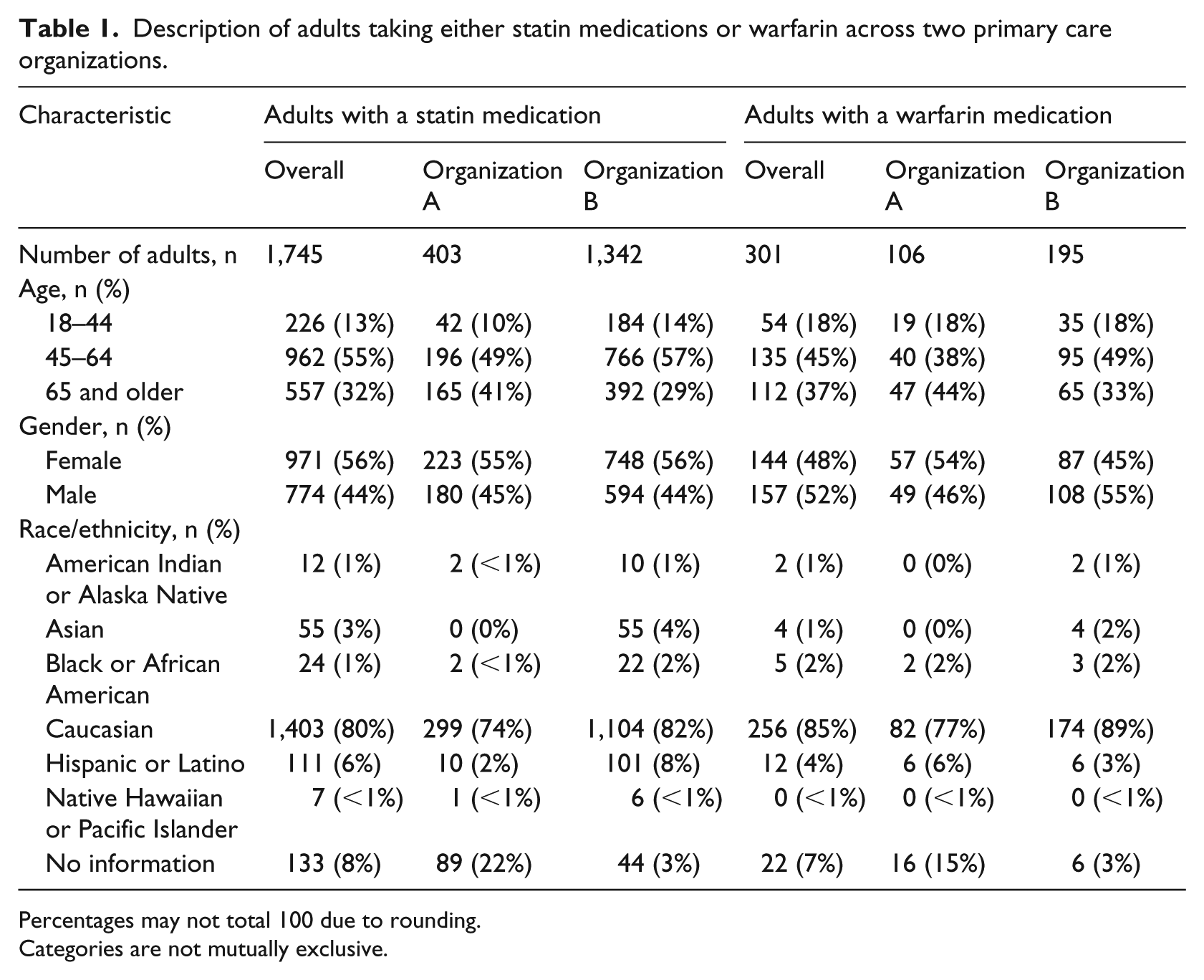
Percentages may not total 100 due to rounding.
Categories are not mutually exclusive.
Objective 2: characterize the types of prescription information coded in discrete data fields in the EHR
The EHR data were limited only to prescription data and did not include pharmacy fill data. We obtained all discrete field medication data. We were able to confirm the presence of information in the directions (sig) of all but 1 percent of statin prescriptions and 1 percent of warfarin prescriptions. One percent of statin prescriptions and 4 percent of warfarin prescriptions were missing information about either medication dose or medication unit; 36 percent of statin prescriptions and 51 percent of warfarin prescriptions were missing information about quantity of medication dispensed. And 36 percent of each statin or warfarin prescriptions were missing refill information.
Objective 3: adverse events relevant to medication prescriptions
We found that 6 percent of patients prescribed statins and 28 percent of patients prescribed warfarin medications had evidence of adverse events commonly associated with these medications (see Table 2).
Prevalence detected within the EHR of adverse events among patients prescribed statin medications or warfarin in two primary care organizations.
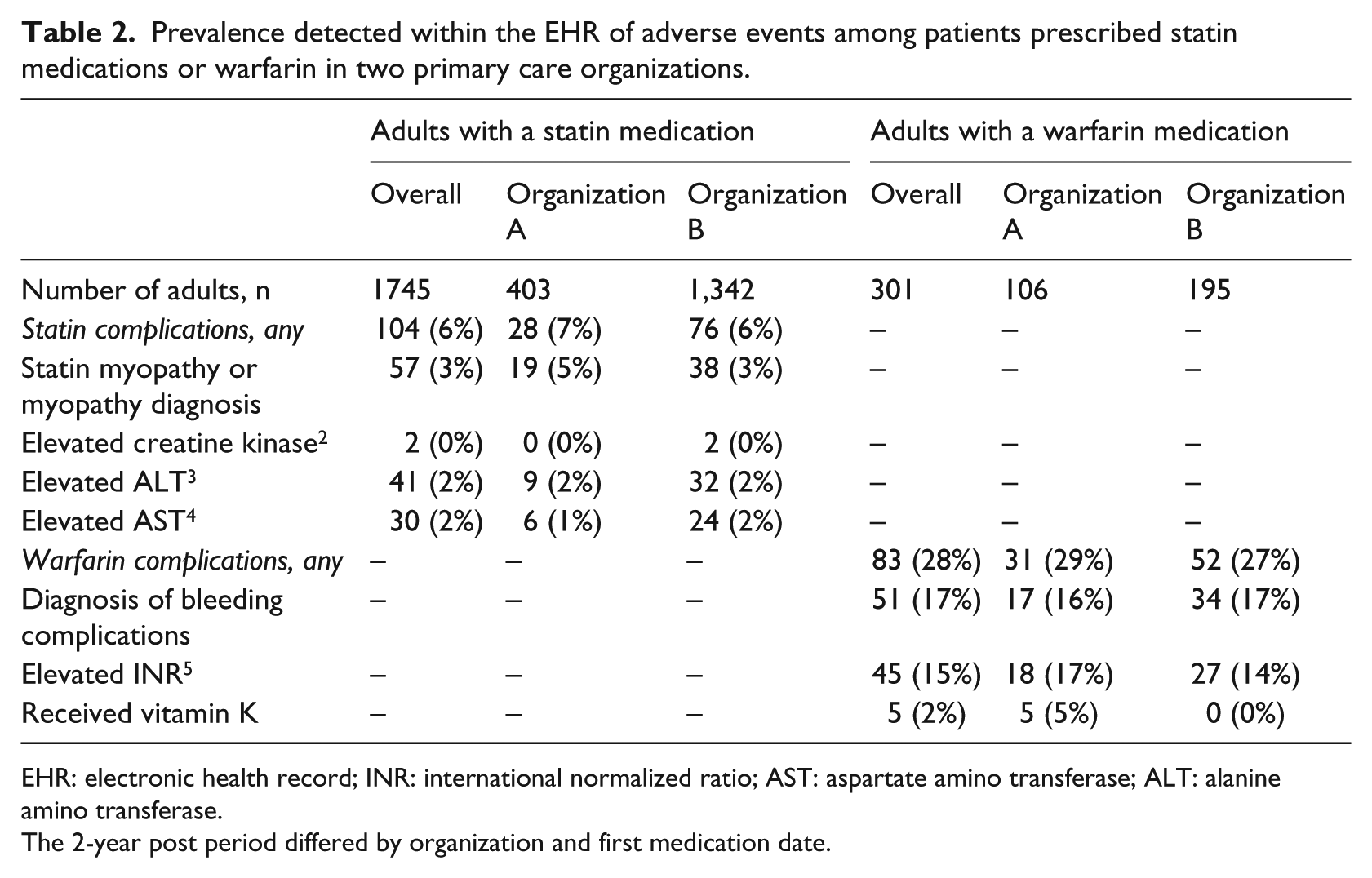
EHR: electronic health record; INR: international normalized ratio; AST: aspartate amino transferase; ALT: alanine amino transferase.
The 2-year post period differed by organization and first medication date.
Among active patients prescribed a statin medication during the first year of the study period, 104 (6%) had evidence of statin myopathy or a myopathy diagnosis coded within 1 year after the medication prescription date. Among active patients prescribed warfarin, 83 (28%) had evidence of a warfarin-related complication within 1 year after the medication prescription date.
Discussion
Across two distinct community-based primary care organizations and EHR systems, we were able to identify and describe cohorts of patients prescribed statin medications or warfarin. The population rates of statin use were similar to what has previously been reported using primary care EHR data.20,21 We also identified patients with potential adverse events related to these medications. Our approach to identifying cohorts of patients, using string-based search terms of medication names, may be particularly useful for both clinical or quality improvement purposes, particularly in smaller primary care practice settings, where EHR systems may be less sophisticated and NDC information may not be easily accessible. The ability to initially identify patients experiencing medication-related adverse events is a critical step for future interventions to reduce these adverse events. These strategies may also be useful for broader clinical research, including cohort identification for clinical trials and epidemiologic research.
We discovered that the EHR-based medication data were frequently missing elements needed to characterize medication treatment course. While EHR-based medication data frequently contained information about the dose of medications, details about the quantity and refills were more often missing. Details for medication instructions were not easily captured and reported and EHRs did not track fill data or patient reports of medication adherence. Without dosing instructions or quantity, the exact amount of medication prescribed cannot be ascertained and the clinical course of treatment is therefore difficult to assess. Improvements in EHR data capture of medication information, such as use of structured data fields for quantity and sig information, could address this limitation of EHR data. These limitations may be specific to the EHR systems included in this study.
We were able to identify patients who may have experienced adverse events relevant to either prescription of statin medications or prescription of warfarin. The rate of adverse event identification for both statins and warfarin were comparable to what has previously been reported: in published data, approximately 8–9 percent of statin users report symptoms consistent with myalgia or myositis, but the rate of severe myositis or rhabdomyolysis is much lower. 22 Our rate of adverse statin-relevant event detection (6%) may have been somewhat lower than published reports due to our relatively short observation period. Using ICD-9 and laboratory data, we were able to identify patients with evidence of myositis or rhabdomyolysis, but patients experiencing the symptoms of myalgia may be more readily identified through analysis of free text documentation where nonspecific symptoms are more likely to be reported. We were able to identify 51 patients (17% of those prescribed warfarin) with diagnoses suggestive of any bleeding complication. This is similar to the rate of reported hemorrhage among patients prescribed warfarin. 23 We were not able to assess the severity of bleeding complication or whether transfusion treatment was required, which is a limitation of the type of data available. The rate of elevated INR among patients prescribed warfarin (15%) is lower than what has been previously reported in the literature, where 20 percent of patients prescribed warfarin in a primary care setting have had laboratory evidence of elevated INR. 24 This difference may be due to the limited observation period (2 years) or differences in our patient population or standards of clinical practice in the participating practices. We did not do manual chart review to validate the accuracy of our adverse event detection. This would be an important step to further validate our approach for future research or clinical care applications.
While EHR data may be successfully used to identify cohorts of patients prescribed specific medications or a subset of patients potentially experiencing adverse events related to medication use, additional analysis and exploration is needed to confirm associations between medications and adverse events. Expanded algorithms, which combine natural language processing of text notes with laboratory or diagnostic code fields, may be more successful in identifying cohorts of patients or cases of adverse events with more precision. Exploration of alternate strategies to identify potential outliers, or those at risk for potential adverse medication events, might include searching for patients prescribed a particularly high or low dose of medications or those with early discontinuation of what is typically a chronic medication.
Because EHR data are collected primarily for clinical care and billing, EHR data present certain limitations when applied to research.1,2 To address some of these limitations, our team found that an understanding of the clinical context of the data was critical in interpreting our results. For example, the EHR data in Data QUEST are limited to that entered in primary care settings. Patients prescribed a statin or warfarin medication by a specialist may not have been identified using our strategies, unless the medications had been entered in the EHR by the primary care provider. In addition, complications diagnosed by a specialist, emergency room or hospitalization would also not be discovered using primary care data alone.
Our experience using Data QUEST’s EHR data demonstrates the power of EHR data to support clinical research, in particular creating a better understanding of medication adverse events. Based on our experience, EHR data can be used to preliminarily identify a subset of patients potentially experiencing adverse events related to medication use. However, we also highlight important limitations of use of EHR data, such as difficulty assessing medication dose and instructions, which would be needed to accurately describe patients’ treatment courses and identify treatment “outliers” or other important patient subgroups. Our findings should be considered in the future planning and interpretation of clinical research using EHR data related to medications and associated adverse events.
Footnotes
Appendix
9th International Classification of Diseases (ICD-9) codes used for identifying patients with diagnosis of statin myopathy or bleeding complication.

| ICD-9 Codes | |
|---|---|
| Statin myopathy | 359.24, 359.4, 359.89, 359.9 728.88, 728.89, 728.9 729.1 |
| Bleeding complications | 280, 280.0, 280.00, 280.9 285.1, 285.9 360.43 372.72 379.23 380.31 423 430 431 432, 432.0, 432.1, 432.9 455.2, 455.5, 455.8 456, 456.2 459 530.7, 530.82 531, 531.01, 531.2, 531.21, 531.4, 531.41, 531.6, 531.61 532, 532.01, 532.2, 532.21, 532.4, 532.41, 532.6, 532.61 533, 533.01, 533.2, 533.21, 533.4, 533.41, 533.6, 533.61 534, 534.01, 534.2, 543.21, 534.4, 534.41, 534.6, 534.61, 535.41, 535.51, 535.71 537.83, 537.84, 562.12, 562.13, 568.81, 569.3, 569.85 578, 578.1, 578.9 596.7 599.7, 599.71, 599.72 626.2, 626.6, 626.87, 626.8 719.1, 719.11, 719.12, 719.13, 719.14, 719.15, 719.16, 719.17, 719.18, 719.19 770.3 779.92 782.7 784.7, 784.8 786.3, 786.31, 786.39 964.2 998.11, 998.12 |
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Institutes of Health under Awards Number UL1 TR000423 and U01 GM092676-05. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. This publication [or project] was supported by the DARTNet Institute, a not-for-profit 501(c)3 organization that conducts research, supports collaboration among health care providers and organizations and hosts data sets of health information for quality improvement and research. The content is solely the responsibility of the authors and does not necessarily represent the official views of the DARTNet Institute.
