Abstract
The Penn State Heart Assistant, a web-based, tablet computer-accessed, secure application was developed to conduct a proof of concept test, targeting patient self-care activities of heart failure patients including daily medication adherence, weight monitoring, and aerobic activity. Patients (n = 12) used the tablet computer-accessed program for 30 days—recording their information and viewing a short educational video. Linear random coefficient models assessed the relationship between weight and time and exercise and time. Good medication adherence (66% reporting taking 75% of prescribed medications) was reported. Group compliance over 30 days for weight and exercise was 84 percent. No persistent weight gain over 30 days, and some indication of weight loss (slope of weight vs time was negative (−0.17; p value = 0.002)), as well as increased exercise (slope of exercise vs time was positive (0.08; p value = 0.04)) was observed. This study suggests that mobile technology is feasible, acceptable, and has potential for cost-effective opportunities to manage heart failure patients safely at home.
Keywords
Introduction
Heart failure (HF) is a major public health concern, responsible for high rates of hospitalization and re-hospitalization. 1 It is the most common reason for hospital admission in the United States and Europe for patients over the age of 65 years, and is also associated with the highest rate of readmission compared to all other medical and surgical causes of hospitalization.2,3 Nearly, 25 percent of patients discharged with a primary diagnosis of HF are readmitted within 30 days. 4 In addition to the morbidity of repeat hospitalizations, the cost of hospitalizations is high. 5 In 2012, the total economic burden in the United States from HF was estimated at US$31 billion, with US$21 billion coming directly from medical expenses and 80 percent of that from the cost of hospitalization.2,6,7 Accordingly, there is a strong motivation both clinically and economically to improve HF patient outcomes.
Generally, hospital readmission is driven by disease progression and inadequate self-care. 8 HF self-care involves patient adherence to the treatment plan (such as medication and exercise recommendations) and careful monitoring of changes in weight. 9 HF programs which include frequent provider contact have demonstrated reduction in re-hospitalization rates and even improve mortality.10,11 Such programs, however, are expensive, not widely available and typically do not provide long-term care. 12 A major reason for the limited access to these programs is the need for time-consuming one-on-one interventions from highly trained personnel to educate HF patients, motivate them to engage in self-care, and monitor them regularly. Moreover, the benefits of current self-care programs are not generally sustained over time.10,11
Mobile technology provides an opportunity to improve both the efficiency and quality of HF management in cost-effective ways. 13 Both the World Health Organization 14 and the American Heart Association 15 are focused on understanding the impact of mobile technology on health, but also on using these new technologies to transform healthcare. Specifically, technology promises the opportunity to “remotely” provide patients with consistent education, motivation to become engaged in their own self-care, and assistance in monitoring on a regular basis.
There have been a number of large, rigorous, technology clinical trials in HF management.16–19 However, a series of recent systematic reviews13,20–22 reveal great variability in type of technology (i.e. telephone, email, text, web portal); timing (i.e. frequency of contact, whether at set times or as needed); interface (i.e. automated, live or mixed); uni-directional or bi-directional transfer of health information; and content. Patient outcomes in these studies generally involve mortality, 13 hospital readmission, 23 or both.19,24 To date, the potential mechanisms, such as self-care, driving these patient outcomes in HF have rarely been examined.
Two studies, similar to our own, bear closer examination. Lyngå 25 qualitatively assessed patients’ responses to a telephone-based, weight monitoring randomized control trial (RCT). In a sample of older adults (n = 20; age range: 61–86) with New York Heart Association (NYHA) class II-IV HF, the patients reported that knowing they were being monitored activated them to engage in their own care. In a recently completed clinical trial, Better Effectiveness After Transition-Heart Failure (BEAT-HF), Ong et al. 18 tested a telephone coaching and telemonitoring intervention which focused on self-care education and daily monitoring via a wireless transmission pod. This large (n = 1437), 2-arm, multicenter trial resulted in no significant differences between the intervention and control group in 30-day readmission rates or 180-day mortality rates. Two significant study limitations noted by the investigators were lack of integration of the trial into the clinical practice sites caring for the patients and uneven protocol adherence rates. However, they did not report patient responses to the intervention itself, so the impact of monitoring on patient activation is not known. In general, processes and behaviors around patient motivation and activation are currently understudied in HF self-care technology intervention studies despite being identified as a key concept in chronic illness self-care.25,26 Taken together, these two studies suggest a need for better understanding the role that the patient and the potential to enhance motivation to engage in self-care when using technology plays in common HF outcomes.
Cognizant of the technological/utilization challenges identified in past HF technology studies, in the present study we used an easy to use wireless computer tablet. This type of technology is widely available, affordable, and easy for patients to use. Accordingly, we anticipated that this would enhance both the ease of use and uptake of the program.The purpose of this study then was to conduct a proof of concept test of a custom, web-based, secure application delivered via tablet computer to improve HF patient self-care. The specific goals of the intervention were to educate the patient through an educational video session, motivate the patient to become involved in their self-care by providing feedback on their progress, and allow them to self-monitor their condition, in the form of graphs of their weight and activity. In addition, the program allowed providers to monitor the patient remotely via a secure server.
Methods
Project goals and research design process
The project was developed by an interdisciplinary team comprised of three cardiologists, two HF nurse practitioners, three HF clinical specialist nurses, a social worker, the chaplain for our Heart and Vascular Institute, an epidemiologist, a health economist, a psychologist, a public health sciences data manager, and a web developer/programmer. Social cognitive theory (SCT) 27 provided the broad theoretical framework for the intervention. Self-efficacy, an important component of SCT, has been found to moderate the effect of determinants of self-care on self-care in previous studies conducted by members of our group.28–30 Our intervention thus was designed to include opportunities for observational learning for the patients, development of mastery in carrying out self-care tasks, and feedback in the form of personal data graphs. Each of these features have been linked to improved self-efficacy. 31
The team identified patient inclusion and exclusion criteria and a tablet computer assisted-intervention which included (1) daily recording of medication use, weight, and time using an exercise program and (2) an educational video of one of the cardiologists teaching about HF causes and symptoms. We used a prospective, nonrandomized, proof of concept study for 30 days with 12 HF patients recently discharged from the hospital. Patients would be asked to enter into the tablet computer what medications were taken that day, their daily weight measured on a home scale, and how many minutes they spent that day in a simple step exercise using an aerobic stepper. The information would then be transmitted via home wireless router to a secure server. A professional programmer with experience in developing health technology applications took the design plan and developed the software program. After alpha prototypes were developed, two research assistants (RAs) tested two tablet computers over a 3-day period, discovering minor technical issues which were corrected by modest software adjustments.
Special considerations of the study population
As HF occurs primarily in older adults who are less likely to be familiar with navigation on mobile devices, we used the following five concepts to make the overall program as user-friendly as possible. First, we created a series of simple screen prompts to lead the patient through their daily program using large font/icon size and colors easily seen by older adults. Second, we used these screen prompts to create a hard copy step-by-step instruction manual, with instructions in large font size. (Readers may contact the corresponding author directly for access to this instruction manual.) Third, we created a 5-min introductory video which featured an older adult, acting the part of a HF patient, using the tablet computer at home and going through each of the daily tablet computer-directed activities. The video was used during recruitment activities when the potential participants were still in-patients in acute care in our hospital. Fourth, an RA visited the home of each consented patient, brought the tablet computer to be loaned to that patient and went over it and the instruction manual, and left the tablet computer and the manual with the patient. The RA also left a dedicated phone number that the patient could call if she/he needed help with any part of the program.
Security of personal health information
To achieve the desired level of security of personal health information (PHI), required by federal regulations, the team selected Research Electronic Data Capture (REDCap), a secure, web-based application designed to support data capture for research studies. REDCap is maintained by the REDCap Consortium (http://project-redcap.org/) which includes over 600 institutional partners including Penn State University. REDCap is Health Insurance Portability and Accountability Act (HIPAA) compliant; all data are encrypted and stored on a secure server; access to databases requires user authentication with password and data access is based on the individual’s role on project. Logging and audit trails are kept on all data interactions. REDCap can remove identifiers from a data set prior to exporting for analysis to create either a limited data set or a safe harbor data set.
Participants
Inclusion criteria are as follows: adults, alert and oriented, documented NYHA class II-III HF in the electronic health record, currently hospitalized with a HF-related admission, reliable home wireless Internet access, English speaking and willing and able to participate. Exclusion criteria are as follows: chronic cognitive impairment documented in the electronic health record, dialysis patients, discharge disposition to a skilled facility, refusal to participate, and NYHA class I or IV or weight greater than 300 lbs (due to the exercise component).
After obtaining Institutional Board Approval, potential study patients were approached in the critical care unit by one of the team physicians or an HF nurse practitioner. Potential participants were shown in the 5-min introductory video. Patients who agreed to take part in the study and provided informed consent before leaving the hospital were contacted by the RA who arranged to meet them at their home between 24 and 48 h after their hospital discharge.
Procedures
The RA hand-delivered the tablet computer and an aerobic stepper to the patient’s home and at that time the RA created the patient’s specific medication table in REDCap in consultation with the patient and with the patient’s current cardiovascular-related medications in front of them (prescriptions and/or pill bottles). In this pilot study of 12 patients, medications prescribed for HF remained constant over 30 days and it was not necessary to adjust any of the individualized patient medication table in REDCap. Each patient supplied demographic data that the RA then entered into REDCap. The demographic data consisted of age, gender, marital status, living arrangement, education, employment status, smoking history, and body mass index (BMI); left ventricle ejection fraction (a measure of heart health) was also collected. The RA then went over the use of the tablet computer, demonstrated the use of the aerobic stepper, instructed the subject to use the stepper daily, left the subject with the illustrated instruction manual, the aerobic stepper, and a telephone number which the patient was to use to contact the RA directly if they had questions. For each of the next 30 days, the patient was to enter their daily medication use, daily weight, and time spent using the aerobic stepper into REDCap. Patients were enrolled between 26 June and 3 August 2015 and all completed their 30-day use of the Penn State Heart Assistant by 4 September 2015.
Quantitative analysis
Proof of concept was assessed by analyzing group compliance in completing three daily tasks: (1) recording medication intake, (2) recording daily weight, and (3) exercise. Compliance with reported medication intake was measured as the patient selecting the “yes” radio button each day on the tablet computer for a particular medication entered by the RA into REDCap. Compliance with daily weight monitoring was measured as the patient entering a weight each day from their home scale. Compliance with reported exercise was measured as the patient entering their time using the aerobic stepper each day. With respect to medication adherence, for each patient we calculated the number of medications taken per day according to the REDCap medication table and then summed over all days for a total number of doses taken during the study period. Overall, medication compliance per patient was calculated as the total number of doses taken divided by the total number of doses the patient should have taken based on his/her list of prescribed medications. The overall medication compliance was calculated as the sum of the total doses taken over all patients divided by the sum of total doses prescribed over all patients. Linear random coefficients models were fit to assess the relationship between weight and time, as well as exercise and time over the 30-day period. 32 The magnitude of the relationship was quantified using the slope (i.e. rate of change) of the regression line. The linear random coefficients model accounts for the repeated measurements per patient due to daily acquisition of data. Residual diagnostics were assessed to determine the appropriateness of the model fit.
Results
Proof of concept was supported as follows. We enrolled 12 qualified patients prior to hospital discharge who had a primary diagnosis of HF after approaching 56 patients. In all, 44 of the 56 patient approached were excluded for one of the following reasons: no Internet access, 17 educational barrier, 7 language barrier, 4 NYHA Stage 4, 7 or refused. 9 Demographic characteristics for the study patients are presented in Table 1.
Participant descriptive statistics.
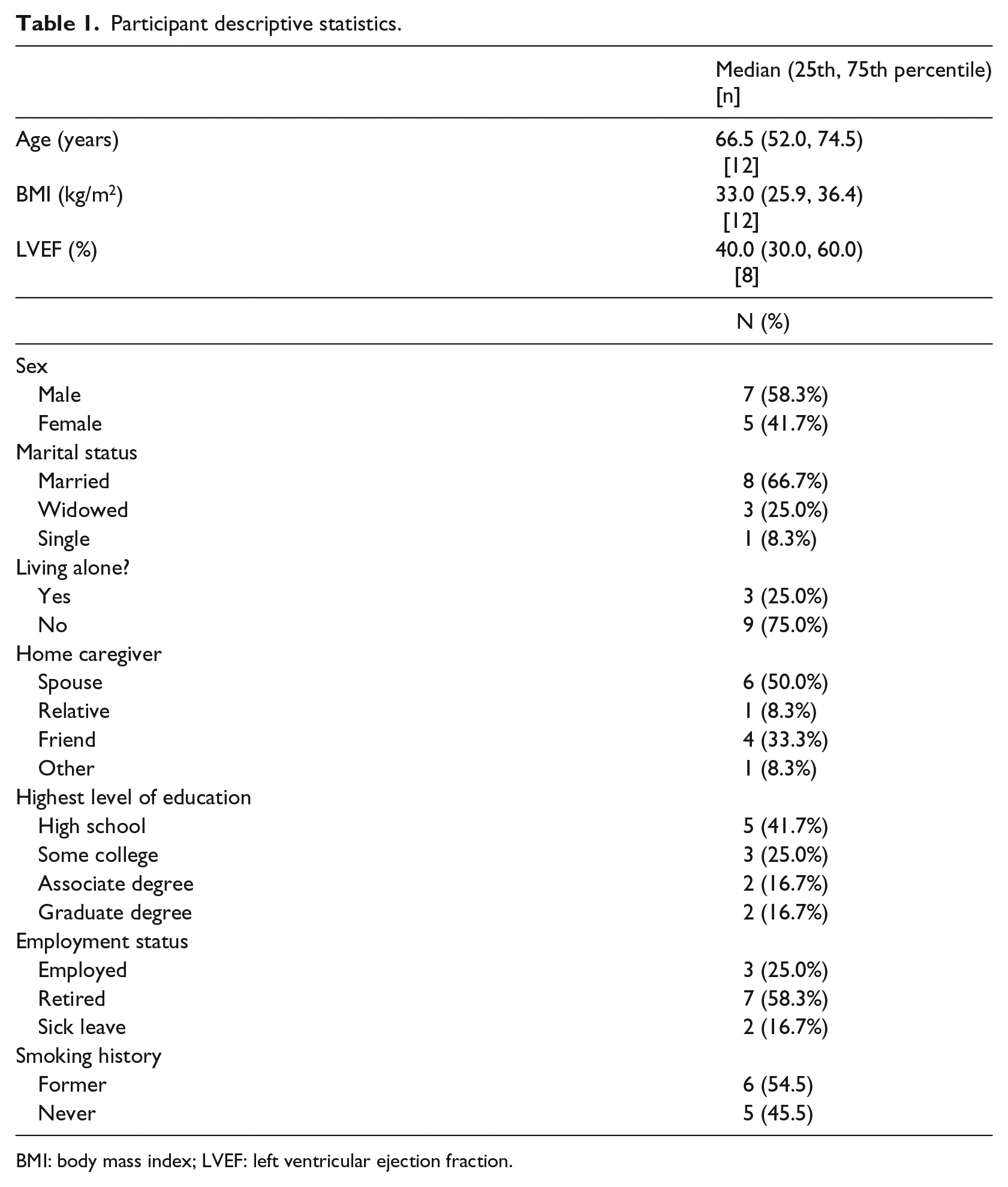
BMI: body mass index; LVEF: left ventricular ejection fraction.
Detailed results are presented in a three graphics (Figures 1 to 3) with each patient identified by a separate shading code. Overall, the group compliance in completing each day’s individualized tasks was 84 percent. Linear random coefficients models were fit to assess the relationship between weight and time as well as exercise and time. Figure 1 shows the individuals’ daily recorded weights, and it is notable that none of the patients showed a persistent weight gain (often a harbinger of re-hospitalization). In fact, overall, patients reduced their weight at a rate (i.e. the slope) of 0.17 lbs per day (95% confidence interval, CI: (−0.26, −0.08); p = 0.002; Figure 1). This translates to a mean weight loss of about 5.1 pounds over the 30 days of the intervention (although this appears to be driven primarily by larger weight loss among a subset of users). Figure 2 shows the number of minutes that patients completed stepper exercise each day. Overall, patients slightly increased their activity time at a rate of 0.08 min per day (95% CI: (0.004, 0.15); p = 0.04; Figure 2). This mean change in use of the aerobic stepper translates to a mean increase in 2.4 min over the 30 days. Figure 3 shows that two-thirds of the group reported taking 75 percent or more of their medications as prescribed.
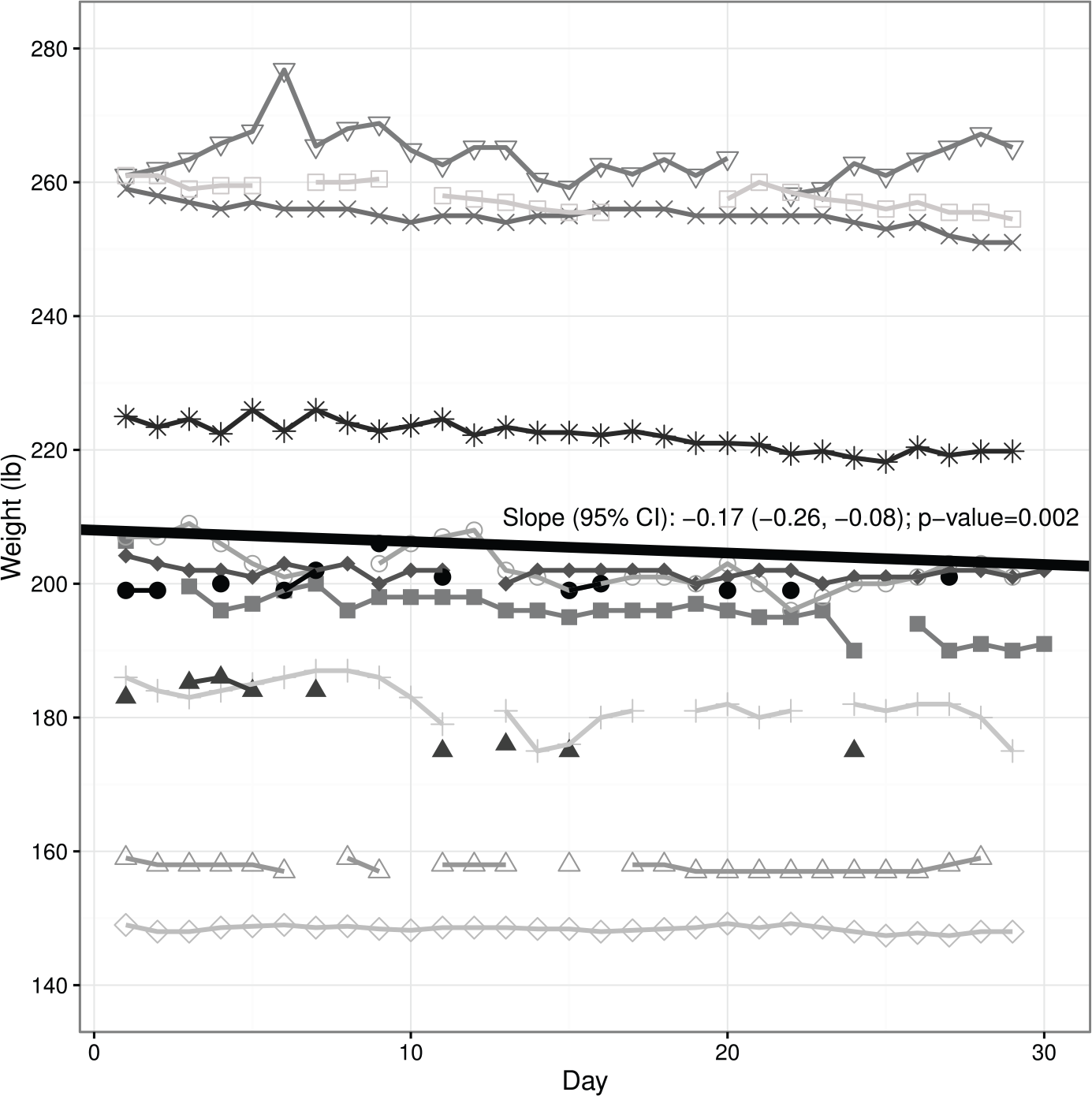
Participant daily weights over the 30 day study period.
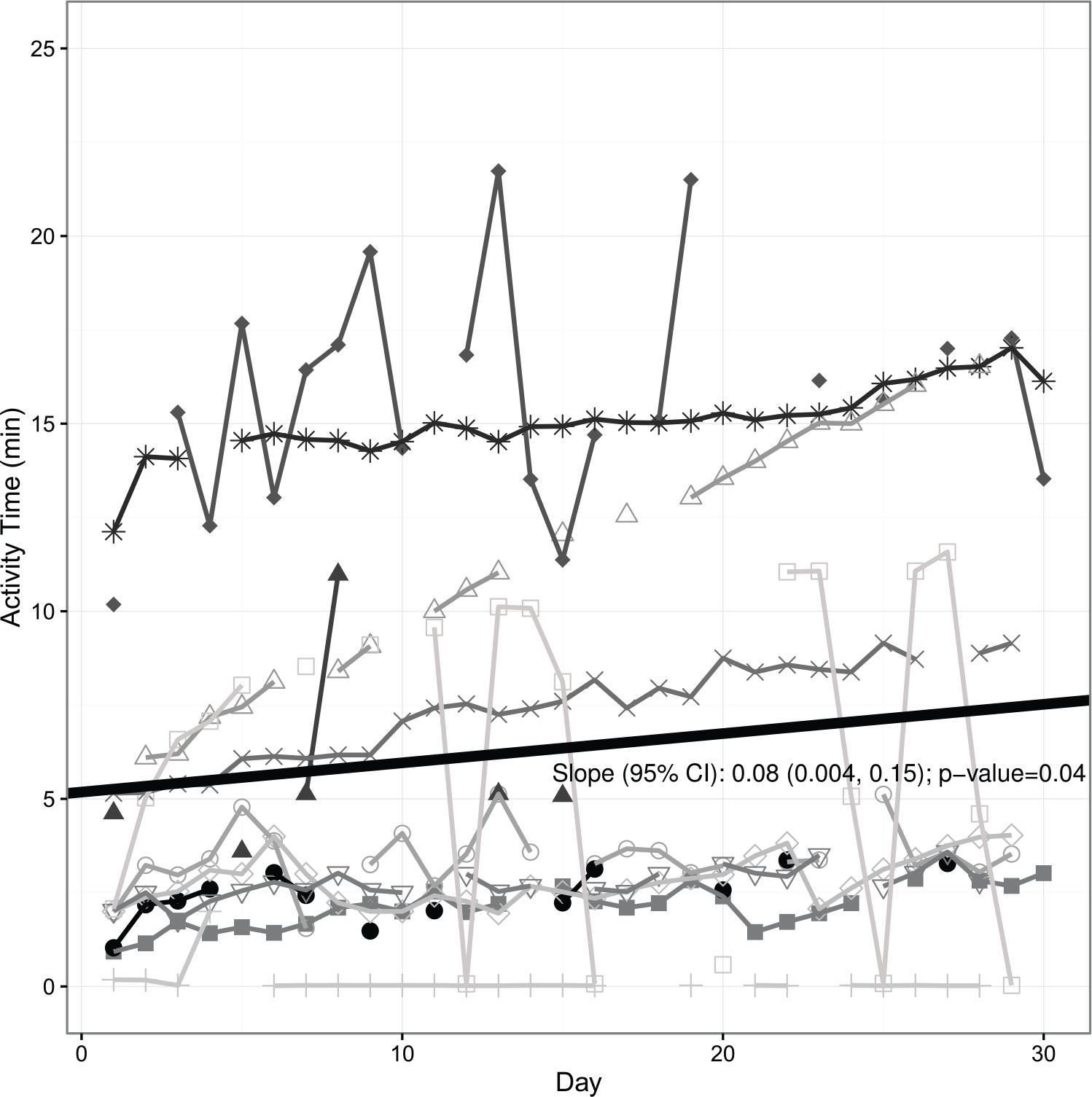
Participant aerobic stepper use in minutes over 30-day study.
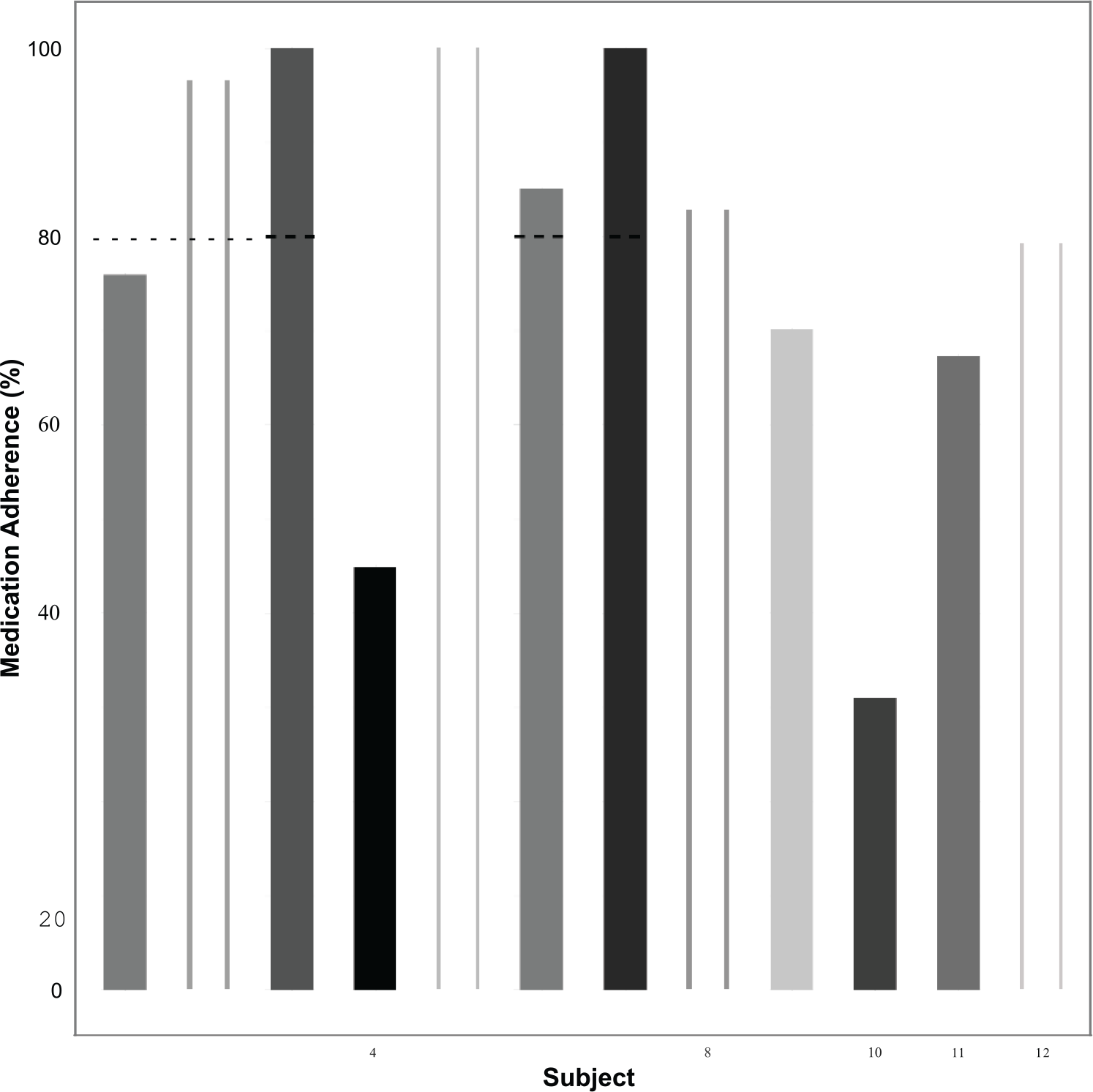
Participant medication adherence, as percentage of prescribed.
Discussion
The purpose of this study was to conduct a proof of concept test of a custom, web-based, secure application delivered via tablet computer to improve HF patient self-care. The software program consists of user-friendly icons to step the patient through the daily tasks of recording his/her weight, medication use, and daily activity. Several of the entry screens are electronic surveys designed to transmit patient data to a centralized REDCap database.
In this article, we have shown that our intervention and study procedures are feasible and appropriate for future testing in a fully powered, larger study. Although we were not powered to assess significance, and our sample size was modest, we were still able to provide preliminary evidence that our tablet-delivered intervention may have enhanced patients’ motivation and capacity to improve their adherence to self-care practices (although noting considerable variability both between patients and within patients over time). Several things bear further discussion in light of the existing literature.
First, two-thirds of our patients reported taking 75 percent or more of their medications as prescribed using our technology. This is comparable with the findings of a recent meta-analysis of technology-based medication adherence studies (n = 16 randomized clinical trials) which found that medication adherence rates improved from 50 percent to almost 68 percent with use of technology. 33 Even when taking into account social desirability in reporting medication adherence, this suggests that our technology perhaps helped patients to remember to take their medication, and motivated them to act upon the information. Equally interesting was that almost one-third of our sample felt comfortable reporting non-adherence; although speculative, this suggests that technology-based adherence assessment may actually improve the accuracy of self-report assessment in medication adherence as has been found in other populations. 34 It could be that patients feel more comfortable reporting their actual practice online rather than to a human who they fear would judge or reprimand them. We suggest that this be examined further in future studies, as this has clear implications for the design of self-reported adherence in clinical trials.
Second, the patients were generally very consistent in use of the aerobic stepper and, overall, slightly increased the number of minutes they performed over time. This is a very interesting finding. First, it is known that functional impairment and hospital readmission rates are highly correlated. 35 Therefore, anything that improves functional status is projected to decrease readmission rates. Exercise was added to the American Heart Association Class I recommendations 36 because it has been shown that exercise does improve functional status and as importantly, patient quality of life in HF.37,38 So although we set out to determine whether a simple exercise intervention was feasible in recently discharged hospital patients, we also were able to show patient improvement over time suggesting that our patients did not find this simple exercise onerous. Moreover, it is possible that they may have experienced improved functional status and quality of life as a result of this intervention although we did not measure these outcomes in this pilot study.
A final issue bears further reflection. The results of the BEAT-HF 18 trial were published during the conduct of our study. BEAT-HF was designed as a 2-arm randomized trial using both telephone health coaching and technology transmission of patient information (weight, blood pressure, heart rate, and responses to three symptom questions) with threshold triggers versus usual care. However, no significant differences between the two arms were found on the primary or secondary outcomes (readmission and mortality). Particularly concerning is the finding that a little over half of the participants were only 50 percent adherent to the protocol. This supports a similar finding from an earlier large trial where 14 percent of the intervention patients never used their equipment and only 55 percent were still using the system at the end of the study (180 days out from baseline). 19 These results add to the body of literature that suggests that technology solutions to HF management can be ineffective, yet the reasons may have little to do with technology or intervention per se but rather with the delivery, engagement, and utilization of the intervention elements. Yet, more and more chronic illness management is delivered using some form of telehealth. We suggest that it is important and informative to engage patient stakeholders early in design phases of technology programs, keep them engaged as co-researchers in clinical trials, and listen to the technology end-user voice when evaluating technology programs in order to better understand how to create engaging and effective mHealth interventions and programs. We hope that our study, with its assessment of the end-user experience, is a small first step in this direction.
Limitations and future directions
A number of limitations to this preliminary work should be kept in mind when reviewing our findings. First, this was primarily a feasibility study; as we did not have a comparison group, we can only speculate that the positive findings we observed were due to the intervention. Given the uncontrolled nature of this trial, an essential next step is testing of this very promising technology in an appropriately powered RCT of HF patients, and extending the study to 3 or 6 months in length, and ideally collecting objective indicators of health and healthcare utilization (including costs, readmissions, and mortality). We also believe that it will be important to evaluate the intervention for use with patients and caregivers in a more diverse sample at a different site. As noted above, stronger causal inferencing will be possible following such an RCT. Second, our sample size was very small and homogenous, raising concerns about selection bias (e.g. due to its convenience sampling frame and to the possibility that patients motivated to improve their health may have been more likely to volunteer) and generalizability. We also recruited only about one in four of the HF patients approached in the hospital. The largest reason for exclusion, however, was lack of Internet access. Were this not a barrier, and assuming most of those lacking access would have otherwise been interested, it is plausible that our enrollment would have been close to 50 percent of those approached. We hope to address this barrier in the next phase of the project by providing portable WiFi Hotspot devices (along with the intervention tablets) to patients without home Internet access. Third, our only measure of exercise was by the aerobic stepper; future studies should use additional measures, such as by wearable actigraphy.
Conclusion
In summary, we suggest that mobile technology offers cost-effective opportunities to manage HF patients safely at home. In our study, we showed that the Penn State Heart Assistant tablet computer, a custom, web-based, secure application, is a feasible and acceptable way to educate HF patients and motivate them to become engaged in their own self-care while monitoring them on a regular basis. Moreover, the use of the program provided exciting preliminary evidence of benefit, resulting in improved medication adherence, weight monitoring, and daily aerobic activity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Penn State Social Sciences Research Institute, the NIH Grant UL1 TR000127, the I.O. Silver Endowment Fund of the Penn State Heart and Vascular Institute and the Department of Public Health Sciences.
