Abstract
Objective
Prior research has not assessed the value of remote patient monitoring (RPM) systems for patients undergoing anticoagulation therapy after cardiac surgery. This study aims to assess whether the clinical follow-up through RPM yields comparable outcomes with the standard protocol.
Methods
A crossover trial assigned participants to SOC-RPM or RPM-SOC, starting with the standard of care (SOC) for the first 6 months after surgery and using RPM for the following 6 months, or vice-versa, respectively. During RPM, patients used the Coaguchek© to accurately measure International Normalized Ratio values and a mobile text-based chatbot to report PROs and adjust the therapeutic dosage. The study assessed patients’ and clinicians’ experience with RPM and compared direct costs.
Results
Twenty-seven patients participated. The median time in therapeutic range (TTR) levels during RPM were 72.2% and 50.6% for the SOC-RPM and RPM-SOC arms, respectively, and during SOC, they were 49.4% and 58.4% for SOC-RPM and RPM-SOC arms, respectively. Patients and the clinical team reported high trust and satisfaction with the proposed digital service. Statistically significant differences were only found in the cost of RPM in the RPM-SOC, which was higher than SOC in the SOC-RPM arm.
Conclusions
Portable coagulometers and chatbots can enhance the remote management of patients undergoing anticoagulation therapy, improving patient experience. This presents a promising alternative to the current standard procedure. The results of this study seem to suggest that RPM may have a higher value when initiated after a SOC period rather than starting RPM immediately after surgery.
Trial registration: ClinicalTrials.gov NCT06423521.
Keywords
Introduction
Patients diagnosed with conditions such as atrial fibrillation, congenital heart disease, or recurrent venous thromboembolism require appropriate values of the International Normalised Ratio (INR), a standardised measure of blood clotting time, primarily used to monitor and adjust the dosage of anticoagulant medications. 1 Vitamin K antagonists (VKAs) such as warfarin are low-cost and widely applied in anticoagulant therapy.2,3 However, while VKA oral medication effectively reduces thromboembolic events, it requires frequent INR monitoring, requiring patients to visit clinical analysis centres frequently to be tested. 4 Recently, new oral anticoagulants (NOACs) like rivaroxaban have been proposed as an alternative to VKAs, as they do not require dose adjustment and have predictable anticoagulant effects. Still, NOACs are unsuitable for some patients with mechanical valves, liver or renal dysfunctions.5–8 VKAs remain the standard treatment for this group of patients. A risk of this treatment is that ensuring safe drug administration can be difficult because of its narrow therapeutic range, unpredictable biological effects, and susceptibility to interactions with other medications and food. 3 Therefore, the measurement of the INR is essential for effective VKA management and to optimise benefits while minimising risks. 2 The assessment of the proportion of INR values that fall within the therapeutic range, commonly referred to as the time in therapeutic range (TTR), can provide valuable insights into anticoagulation control, as high TTR is linked with decreased occurrences of adverse bleeding and thromboembolic events.3,9
As the need to keep a high TTR for a safe anticoagulation therapy, regular monitoring of the INR values is critical to support self-management and medical dosage prescription. Personalised and home-based care interventions that empower patients and caregivers for self-management are necessary, especially considering the ageing population and increasing demand for healthcare professionals. 10 Advances in self-testing and self-management technologies within digital healthcare have significantly benefited patients. 11 Portable coagulometers allow patients to monitor their coagulation parameters independently with fingertip blood samples, reducing their dependency on testing facilities. Previous studies demonstrated that the results obtained from portable coagulometers are comparable to those from hospital laboratories. 10 Portable coagulation monitors for testing prothrombin time-international normalised ratios have recently gained popularity. 4 INR self-testing based on portable coagulation monitors and doctor-patient communication can include many clinical and economic advantages over standard of care.4,12 The patient can report the INR reading to healthcare professionals, who will prescribe and communicate medication adjustments, avoiding unnecessary hospital or healthcare centre visits.
Remote patient monitoring (RPM) services are rapidly growing, enhancing communication between health professionals and patients, facilitating drug monitoring, and offering greater flexibility in anticoagulant self-management practices. 4 Patients are now considered active rather than passive regarding healthcare consultation models since technological advances have given patients access to more information and tools, allowing them to participate and actively interact with their health professionals. 13 However, it is crucial to acknowledge that successful self-management interventions involve several factors, such as education, training, and effective communication with healthcare providers.1,14 These conditions may not be achieved for all individuals, especially those with complicated medical conditions or circumstances that make self-management unsuitable or unsafe. Recently, a meta-analysis found that using portable coagulometers and telemedicine for management significantly improved test frequency and TTR compared to the traditional model. 4 Moreover, it has significantly improved adherence due to increased patient awareness to monitorization. 15 A recent review has explored the use of eHealth tools in anticoagulation management after cardiac valve replacement. The study found that computer-based support systems, telemedicine platforms, and mobile apps can improve the time spent in therapeutic range and life satisfaction. However, further research is needed to evaluate their economic benefits and potential to reduce adverse events 16 by evaluating health outcomes, patient and clinical team experiences and costs.
Despite these advances, there remains a gap in effective communication and management between patients and healthcare professionals. Our study aims to address this gap by proposing an innovative system that enhances communication by collecting patient-reported outcomes and sending medication adjustments via an SMS chatbot. The main objective of this pilot study was to evaluate the outcomes and costs of a digital clinical service designed to support patients with anticoagulant therapy after cardiac surgery. This study employed an RPM system with a portable coagulometer for clinical follow-up and self-management of INR control, and it compared this approach with the standard of care (SOC). Additionally, the study sought to assess the satisfaction and experiences of both patients and clinical teams, as well as the economic cost evaluation of the proposed RPM-based system.
Methods
Study design
A clinical trial was conducted utilising the crossover design, as depicted in Figure 1, to investigate the relationship between intervention and target outcomes. The sample size was estimated using G*Power software (version 3.1.9.7), utilising T-Test and the effect size (Cohen's D) was calculated from similar studies (0.76).4,17 Then, a total sample size of 27 subjects was obtained, accounting for a 5% dropout rate, which will provide adequate power (>0.80) to achieve statistical significance at an alpha level of 0.05. This sample size is considered appropriate for the context of our intervention. A total of 27 patients from the Department of Cardiothoracic Surgery at Hospital de Santa Marta receiving anticoagulant therapy were enrolled in the study. Inclusion criteria were patients ≥18 years old and with a smartphone. The exclusion criteria were analphabetism, poor health, low digital literacy level, and inability to use the RPM equipment alone or with caregiver support. During the first visit, the healthcare professional assessed patients’ eligibility for anticoagulation self-management therapy based on their clinical information. To characterise the sample and analyse costs, health indicators such as age, sex, and place of residence were collected. Additionally, patient-reported outcomes, experience, and satisfaction were evaluated. The trial was conducted under the Declaration of Helsinki. The ethics committee of the Centro Hospitalar Universitário de Lisboa Central approved this study (n° 1057/2021). The trial was registered on ClinicalTrials.gov with the identifier NCT06423521. All participants gave written informed consent to participate in the study. Patient records were anonymised in the database.

Flow diagram of the crossover pilot trial of SOC-RPM and RPM-SOC for guiding RPM versus SOC.
Participants were assigned into two arms: one would follow the SOC for the first six months and then receive the RPM intervention for the following six months (SOC-RPM); the other group would receive the intervention (RPM) for the first six months and then follow the SOC for the remaining six months (RPM-SOC). Patients were recruited by the clinical team during the surgery discharge appointment. They were assigned to the RPM-SOC group if the coagulometer devices allocated for this study were available; otherwise, they were assigned to the SOC-RPM arm, which would commence using RPM after the follow-up appointment 6 months post-surgery. Blinding was not feasible due to the nature of the trial, and both patients and the clinical team were aware of the follow-up being conducted. In this study, it was decided that a washout period was not required. This was because withdrawing effective follow-up care for a washout period is not possible, as patients need to be constantly monitored to ensure effective treatment, thus preventing thromboembolic events. The study was conducted for twelve months, as follows:
Patients received a kit with a Coagulometer-CoaguChek® (Roche Diagnostics, Switzerland) and the necessary test strips for use during the period established, as well as written instructions on how to take the measurements, and were registered on the monitoring platform. Patients received periodic text messages on their smartphones to report the INR value, and they responded to messages regarding their symptoms related to anticoagulant therapy and the INR value. The clinical team received notifications if patients’ reports had been assessed outside therapeutic standards and then sent a text message back with the medication adjustment.
Endpoints
The primary endpoint was the patient's TTR for INR values among the programs, i.e., a comparison between SOC and RPM over time. The TTR for INR value was self-measured by the portable monitor and calculated by the Rosendaal method of lineal interpolation.18,19 To obtain reliable results, collecting more than two measurements per participant was necessary. However, seven participants did not fulfil this requirement during the SOC intervention, resulting in being considered as missing data. Our design protocol expected an estimated frequency of one test every 15 days in RPM. However, the clinical team could adjust this frequency upwards if the values fall outside the therapeutic range limits according to their criteria, allowing for more effective therapeutic adjustment and reducing the time needed to normalise INR values.
Secondary endpoints were the number of events of hypocoagulation symptoms. During the RPM intervention, these were assessed periodically by a chatbot-based questionnaire through patients’ self-reported messages on the phone for six months. A questionnaire with 13 self-reported questions was used to evaluate patient experience and satisfaction. The first four questions measured experience, while the following nine focused on satisfaction. The responses were rated on a scale of disagree to agree, with 1–4 points assigned to each response. To prevent ceiling and floor effects in our tool, the questions numbered ‘2’, ‘11’, and ‘12’ were calculated as “inverted,” and an optional open question was included. The questions were presented to the patients in Portuguese and translated to English for reporting purposes of the study (Appendix I and Appendix II). The average cost of patient care was analysed as described in the section Cost Analysis.
Web platform for data collection and patient follow-up using a participatory action research approach (PAR)
The RPM solution for managing oral anticoagulation therapy was developed using a PAR approach. This method has shown legitimacy in the collaborative development of RPM solutions and ensures stakeholder involvement at every stage. 20 The development team was multidisciplinary, consisting of three nurses, two physicians, two software developers, and five researchers, enabling a comprehensive integration of clinical and technical expertise. The PAR approach was structured into five main phases, following a non-linear and recursive process of action and reflection. 1) Problem Definition: Two focused meetings were held with nurses and physicians to understand the existing standard of care and identify challenges for the new web platform; 2) Planning: A subsequent meeting outlined the technical specifications and functional requirements of the platform; 3) Action: The team developed multiple iterations of the web platform, incorporating features identified in previous phases; 4) Observation: Testing was carried out by nurses and physicians to assess usability and effectiveness in a clinical environment; 5) Reflection: Meetings were conducted to reflect on testing outcomes and prioritize necessary modifications using the MoSCoW method (Must Have, Should Have, Could Have, and Will Not Have this time). 21 The result of this collaboration led to the proposition of integrating a coagulometer with a mobile text message-based RPM system. This combination would allow patients to remotely self-report therapy-relevant outcomes and to receive drug dosage adjustments from medical doctors. The RPM system is comprehensively explained in. 22
The design of the chatbot mentioned above considers the following features: (1) text understanding, (2) dialogue management, (3) data management and (4) text generation. The interaction begins with a greeting message, followed by a prompt for the patient to start the questionnaire. This prompt displays the answers for the first measure, as illustrated in the flow diagram presented in Figure 2. Patients are encouraged to interact with a chatbot that delivers multi-step questions and securely stores validated patient-reported data used for clinical monitoring. Patients are provided with a coagulometer device to measure INR and the chatbot's phone number. The assessment plan consists of seven patient-reported items: the INR value measured using the coagulometer and responses to 6 questions regarding medication, bruises, bleeding, faeces, nausea, and hospital/health centre visits, all taken periodically. Detailed information regarding the conversational agent used for data collection is also available in. 22
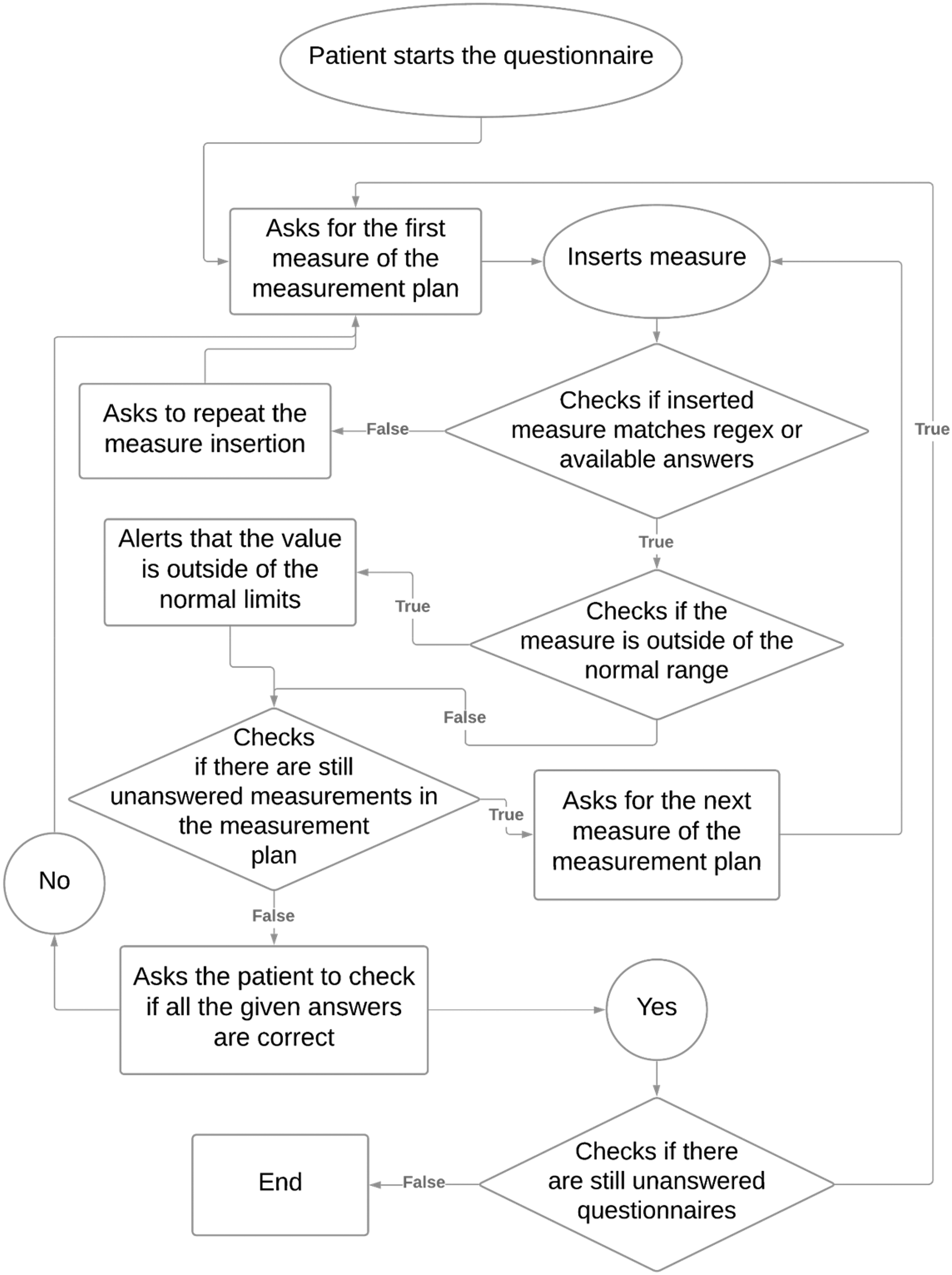
“Data collection flow:” the patient is represented by a circle, the finite state machine by a diamond, and the chatbot by a rectangle.
Cost analysis
The average cost per patient in the SOC versus RPM was compared using the Time-Driven Activity-Based Costing (TDABC) method and the perspective of the Public National Healthcare Service (NHS). 23 The method involved mapping patient pathway activities and identifying the direct costs, such as healthcare professionals and facilities, equipment and technology (ET), and consumables. 24 A literature review was first conducted, followed by interviews with healthcare professionals to validate the findings.23,25,26 Healthcare professional costs were obtained from Portugal's Public Salary Scale, while ET costs were determined based on their market acquisition value and depreciation rate. The manufacturer provided the cost of the coagulometer and the testing strips. Facility costs were based on the cost per square meter multiplied by the minimum required size defined by Portuguese legislation for medical facilities. It was assumed that all human resources had an 80% effective work time. Each resource cost rate was operationalised as the cost per minute. 23 The cost of INR testing in private labs was considered the amount charged by private providers to the NHS.
Travel costs incurred by patients to test INR during SOC were considered. Patients identified the healthcare institution they went to during the six-month SOC period, and it was assumed they would go by personal car. Travel costs were estimated considering the Portuguese legislation for travel expenses in 2023, which defines a cost of 0,36 €/km. 27 No absenteeism costs were taken into account. Patients with less than three measures in the SOC period were not considered for cost analysis due to the assumption of missing data. All monetary values are in Euros as of 2023.
Statistics
The statistical analyses were performed using Python (version 3.9.7). Count and proportion were reported for categorical variables. Continuous data were presented as the mean ± standard deviation or median with the interquartile range (IQR), depending on the normality distribution of the data, verified by the Shapiro–Wilk test. The Analysis of Variance (ANOVA) and the Kruskal Wallis tests were used to explore differences in TTR between the two groups and the crossover combinations. The analysis used the median of the percentage of TTR. In addition, the t-test was used to compare costs. The Friedman test, a non-parametric test for repeated measures, was employed to assess the differences in TTR percentage and the occurrence of hypocoagulation events across the different months of the study. Statistical significance was demonstrated at p < 0.05.
Results
Patient characteristics
In this study, 27 patients were initially enrolled. However, one patient from the SOC-RPM arm dropped out because the clinical team agreed with the patient to terminate the study early, due to non-compliance with the measurement guidelines and medication dosage given by the clinical team, leaving 26 participants who completed the study. Of these, 13 were females, and 13 were males, with an average age of 55.65 ± 13.30 years. Most participants had mechanical aortic or mitral prostheses, with fewer experiencing conditions such as atrial fibrillation, pulmonary thromboembolism, Bentall procedure, or mitral valvuloplasty. Most patients had an INR goal range of 2.0 to 3.0 (detailed in Table 1). However, INR is highly dependent on each patient's specific condition. During the RPM program, patients answered 94.6% of the questions asked by the chatbot, with a median (IQR) time between order submission and patient response of 23.0 (2.0 to 153.5) minutes.
Characteristics of the study population.

Time in the therapeutic range
Table 2 displays a comparative analysis of the median and interquartile range (IQR) of the percentage spent within the TTR for INR values across each study arm and both programs, targeting specifically the TTR. The differences were tested using the Kruskal-Wallis test, revealing no statistically significant differences (p = 0.07).
Comparative median (IQR) of the TTR for INR range by program type between groups.

Table 3 provides a comparison of the percentage of TTR for INR values across six months of RPM within each study group. Each row displays the monthly TTR values for a specific arm, organised in columns. Statistical analyses indicated no significant temporal variations in TTR for the group receiving SOC followed by RPM (SOC-RPM) (p = 0.068). Similarly, the Friedman test revealed no significant differences across the months for the group with RPM followed by SOC (RPM-SOC) (p = 0.385).
Monthly comparison of median TTR percentages for SOC-RPM and RPM-SOC groups over six months.

Hypocoagulation events
Data analysis identified 59 hypocoagulation events in the RPM programs, with 18 occurring in the SOC-RPM arm and 41 in the RPM-SOC arm. The analysis revealed that the SOC-RPM arm had a median value of events by the patient of 1.0 with an IQR of 0.0 to 2.0, while group RPM-SOC had a median value of 2.0 with an IQR of 1.0 to 5.0. The Mann-Whitney test indicated non-significant differences (p = 0.054) between the two study arms, with group SOC-RPM showing a more positive impact regarding fewer events observed. Moreover, the Friedmann test results revealed a non-significant difference between the number of events over the six months during the RPM programs for both study arms (p = 0.887 for SOC-RPM and p = 0.089 for RPM-SOC).
Patients’ feedback, experience, and satisfaction
Patient feedback was collected using a questionnaire to assess satisfaction and experience, as described in the Methods section, and also from open comments. In terms of experience, the patients’ answers had a mean and SD of 3.54 ± 0.36; for satisfaction, it was 3.62 ± 0.25. The full results for each question are available in the Appendix I. The open comments showed that most responses favoured the proposed RPM system (Appendix II).
Costs
Of the 26 patients, nine were not considered eligible for inclusion in the cost analysis as, during SOC, seven reported less than three INR measures. Additionally, one patient did not provide where he was tested during SOC, and one was tested at home. Table 4 presents a comparison of per-patient costs between RPM and SOC follow-up for the two study arms. The costs associated with INR testing were categorized either as fixed or variable costs. Only when the patients were followed through RPM were fixed costs incurred. Generally, estimated costs were higher in the first semester. Accordingly, RPM was more expensive than SOC in the RPM-SOC study arm while less costly in the SOC-RPM arm. However, only the RPM-SOC study arm observed statistically significant differences between RPM and SOC. Additionally, when comparing the costs between groups, the cost of RPM in the RPM-SOC was statistically significantly higher than SOC in the SOC-RPM arm, as p < 0.001, with a mean difference of 32,97€.
Average (standard deviation) patient INR monitoring cost (€) for each arm during six months of intervention.

Discussion
The present research analysed using a chatbot and a portable coagulometer as an RPM system versus SOC in patients’ follow-up receiving anticoagulation therapy after cardiothoracic surgery. The study aimed to assess the impact of RPM on TTR for INR values, hypocoagulation events, patient experience, patient satisfaction, and costs following a value-based healthcare approach. Our study suggests that the RPM system is more successful than SOC intervention, regarding the median percentage of TTR for INR values, when RPM is initiated six months after surgery. This finding may be because when VKA therapy is initiated or resumed after surgery, the values of INR fluctuate until the ideal personalised dose is determined for each patient. This personalised dose can vary significantly between patients, and the required INR ranges can also differ. Additionally, RPM results could have been better after the SOC period because the initial phase of higher INR variability occurred during SOC. RPM was initiated during a period of higher stability, when the ideal dose for each patient is already clinically assessed.
During RPM interventions, the TTR levels were 72.2% for the SOC-RPM arm and 50.6% for the RPM-SOC arm, which may have influenced the number of hypocoagulation events in the SOC-RPM arm, which was significantly lower than in the RPM-SOC arm. Previous studies have suggested that spending more time outside the therapeutic range can have serious health consequences. According to a study by Lee et al. (2021), a 10% increase in time spent outside the TTR can result in a 29% increase in mortality risk and a 10%–12% increase in the risk of ischemic stroke and other thromboembolic events. 28 Our results align with a meta-analysis comparing telemedicine-based oral anticoagulation management with usual care, in which it was found that telemedicine-based systems, compared to usual care, had similar rates of major bleeding and mortality and improved TTR, indicating the potential of RPM as a promising alternative to standard procedures for the management of anticoagulation therapy. 29
The present study utilised a portable coagulometer and a digital platform to create a communication channel between patients and the clinical team. This allowed prompt reporting of outcomes, symptom management and structured data collection. A web platform through a chatbot was used to report PROs and INR measures from the coagulometer and to receive medication dosage recommendations from the clinical team. Such a combination of portable coagulometers with patient-doctor communication has been previously proven to support better TTR maintenance, especially when complemented with patient education. 6 The patients and clinical team involved in our study reported a positive experience with our digital healthcare service, expressing trust and satisfaction. The clinical team has highlighted the ease of access to medical prescriptions and the comfort of performing the INR test remotely as the principal benefits of the proposed solution. The successful implementation of our RPM system may be attributed to the relevance of co-creation methodology for service design30,31 and the PAR approach used for developing this platform. 20 The dynamic, iterative nature of the research process enabled stakeholders to address various aspects—from design through execution and implementation—simultaneously rather than sequentially. This collaborative and iterative method not only garnered stakeholder buy-in but also improved the functionality and user acceptance of the final product. Such methodology facilitated the adoption and confidence of the patients through the design of a simple interface technology and the education support from the nurses. Considering the target group of patients in cardiac surgery, in a public hospital in Portugal, this digital service addressed key barriers for technology adoption in elderly population, as studied in Frishammar et al. (2023). 32 The use of eHealth strategies has many benefits, including improved healthcare access for remote populations and more effective anticoagulation management.4,29 Remarkably, the patients answered (on average) 94.6% of the questions made by the chatbot. This result demonstrates that the methods used in our study mitigated the risk of low adoption and usage of digital services in healthcare. In fact, previous research has shown that PAR approach improves patients’ engagement with medical services, enhances the interactions between patients and providers, fosters shared decision-making and strengthens relationships between them. Hence, the alliance between stakeholders, patients, and developers can result in more effective interventions, satisfied patients, and reasonable costs.7,20
The RPM period was associated with increased costs compared to SOC in the RPM-SOC arm, possibly due to a higher number of INR measures that led to increased spending. This might be, as previously described, due to higher INR variability during the initial postsurgery period, when patients need closer clinical monitoring and reassurance. Contrarily, in SOC, the cost of transportation for outpatient visits must be considered. Despite the fact that we did not accurately assess the indirect costs due to lack of data, previous studies suggest that telemedicine may be more cost-effective over a long period when considering the cost of transportation and absenteeism for outpatient visits. 4 This study primarily examined the direct monetary costs of INR testing in SOC and RPM periods of care. However, the cost impact extends beyond testing costs, including costs with clinical complications and indirect costs. 33 A systematic review 12 concluded that when accounting for the costs associated with complications, self-testing may be less costly than SOC due to its potential to decrease thromboembolic and bleeding events via improved INR control. They also conclude that RPM could enhance cost savings by reducing outpatient clinic visits, allowing healthcare professionals to dedicate more time to other patients. 12 Additionally, including indirect costs, such as productivity losses when patients attend in-person tests in clinical settings, would increase the costs of SOC. Moreover, in countries like Portugal, where a high proportion of informal caregivers also participate in the workforce, the indirect costs rise with their work absenteeism, emotional strain, and the opportunity costs of caregivers’ time. 34 Thus, future research should consider the potential cost savings of home-based testing regarding the costs of severe complications, the additional time healthcare professionals can devote to other activities, and indirect costs.
This study has several limitations. The small sample size may reduce the statistical power of the analysis. Nevertheless, the crossover design allowed us to conclude that future studies with higher sample sizes should focus on stages when patients have already passed the acute post-surgical instability. Besides, the lack of a washout period between treatments could have led to biased results and increased variability. In addition, we identified limitations related to the exclusion of patients with low digital literacy which may affect the generalisability of our findings. The questionnaire used to assess experience and satisfaction is not validated, precluding external validity. Still, it was used due to the lack of instruments in the Portuguese language to assess the experience and satisfaction of digital health RPM-based systems. The open comments from patients and the clinical team gave valuable insights and corroborated the questionnaire results. To enhance the reliability and precision of the experience and satisfaction outcomes, future research could integrate the application of validated questionnaires along with mixed methods. This approach would enable the collection of comprehensive and reliable data, ensuring the results are highly accurate and trustworthy. Moreover, forthcoming studies can consider controlling confounding variables identified as potentially affecting the outcomes. As previously documented, these variables may include drug interactions 35 and nutritional status. 36
Further research is required to gather real-world evidence on the outcomes and costs of digital health services, specifically for follow-up procedures after cardiac surgery. 37 Electronic health records and billing data can provide real-world evidence that complements traditional clinical trials. 38 Therefore, they can help develop digital health services that provide higher value to patients. The current study can serve as a starting point for the development of an RPM system for anticoagulation therapy, empowering patients to report outcomes and INR measures, from home, with clinical supervision for symptoms management and dosage adjustment.
Conclusions
Anticoagulation therapy management can pose a challenge due to the need for frequent monitoring of INR values and TTR. However, a RPM system that tracks these clinical outcomes can provide adequate therapy control. For patients undergoing oral anticoagulation therapy after cardiac surgery, self-testing and RPM techniques have been shown to be viable options for INR management. This study validates the use of a chatbot and a portable coagulometer as a viable method to improve the management of patients undergoing anticoagulation therapy after cardiac surgery. The proposed RPM-based system has the potential to complement the current standard procedure and may be especially useful when RPM is initiated after a SOC period.
Footnotes
Acknowledgements
This study was supported by Roche Diagnostics which is the manufacturer of the coagulometer kit.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was co-funded by Roche and by the public national funding PRR 01/C05-i02/2022.P173: Missão Interface–renovação da rede de suporte científico e tecnológico e orientação para o tecido produtivo.
Ethical approval
The Ethics Committee of the Centro Hospitalar Universitário de Lisboa Central approved this study (REC number: 1057/2021). The trial was registered on the ClinicalTrials.gov with the identifier NCT06423521.
Guarantor
AL.
Contributorship
AL, PC and SA researched literature and conceived the study. AL, PD and PC was involved in protocol development, gaining ethical approval. TS, PC, HS and CV were involved in patient recruitment. FG, EO, TS, PC and AL were involved in data analysis. FG and EO drafted the manuscript. All authors reviewed, edited the manuscript, and approved the final version of the manuscript.
Patient experience ratings (questions 1–4) and the satisfaction ratings with the chatbot (questions 5–13).

| Question | Do not know | Strongly disagree | Disagree | Agree | Strongly agree |
|---|---|---|---|---|---|
| 1 - I felt well looked after with this service. | 0.0 | 0.0 | 0.0 | 26.1 | 73.9 |
| 2 - The service interferes with my daily routine. | 0.0 | 21.7 | 60.9 | 17.4 | 0.0 |
| 3 - The service should be recommended to people with a health condition similar to mine. | 0.0 | 0.0 | 0.0 | 34.8 | 65.2 |
| 4 - I am satisfied with this service. | 0.0 | 0.0 | 0.0 | 26.1 | 73.9 |
| 5 - The explanation given at the time of delivery of the equipment was sufficiently enlightening. | 0.0 | 0.0 | 0.0 | 34.8 | 65.2 |
| 6 - It was easy to learn how to use the equipment. | 0.0 | 0.0 | 4.3 | 34.8 | 60.9 |
| 7 - The time it takes me to take the daily measurements is acceptable. | 0.0 | 0.0 | 4.3 | 39.1 | 56.5 |
| 8 -Whenever I make a mistake using the equipment, I get through it with ease. | 13.0 | 0.0 | 0.0 | 52.2 | 34.8 |
| 9 -The equipment is reliable and has few technical problems. | 0.0 | 0.0 | 0.0 | 26.1 | 73.9 |
| 10 -The team is quick to respond when technical problems occur. | 34.8 | 0.0 | 0.0 | 17.4 | 47.8 |
| 11 -The equipment I received invades my privacy. | 0.0 | 65.2 | 30.4 | 4.3 | 0.0 |
| 12 - The equipment makes me feel uncomfortable physically or emotionally. | 0.0 | 69.6 | 30.4 | 0.0 | 0.0 |
| 13 - The equipment allows professionals to better monitor my condition from a distance. | 0.0 | 0.0 | 0.0 | 26.1 | 73.9 |
The values are presented in percentage (%).
Patient Feedback on Experience and Satisfaction (Open-ended).

| "Very useful equipment, if handled and used well” |
| ” I recommend it, and I think that everyone with this problem should always benefit from the equipment. It saves us time and convenience, as we don't have to go to hospitals and waste hours. It also allows us to take just one drop of blood instead of being monitored in hospital.” |
| “I would like this system to be made available to everyone who needs it, as it really helps with measurements. It saves me the hassle of going to a hospital, wasting time, and bothering nurses, as I can do it myself at home without bothering anyone.” |
| "The equipment was very good, it was a shame to have to give it away, as it made measurements easier and saved me and the hospital time" |
| "I would like to get the machine if possible." |
| "Everything was good. I only have good things to say about the device” |
| "I am very interested in the device, as it makes day-to-day measurements easier. The only reason I didn't buy the device was the price, not only of the device but also of the tapes, which are quite expensive." |
| "I have already bought a similar machine to have at home" |
No participant reported a negative experience.
