Abstract
Early detection of patients at risk seems to be effective for reducing the prevalence of lifestyle-related chronic diseases. We aim to test the feasibility of a novel intervention for early detection of lifestyle-related chronic diseases based on a population-based stratification using a combination of questionnaire and electronic patient record data. The intervention comprises four elements: (1) collection of information on lifestyle risk factors using a short 15-item questionnaire, (2) electronic transfer of questionnaire data to the general practitioners’ electronic patient records, (3) identification of patients already diagnosed with a lifestyle-related chronic disease, and (4) risk estimation and stratification of apparently healthy patients using questionnaire and electronic patient record data on validated risk estimation models. We show that it is feasible to implement a novel intervention that identifies and stratifies patients for further examinations in general practice or behaviour change interventions at the municipal level without any additional workload for the general practitioner.
Keywords
Background
Interventions that contribute to a reduction in the increasing prevalence of lifestyle-related chronic diseases such as type 2 diabetes (T2DM), chronic obstructive pulmonary disease (COPD), and cardiovascular disease (CVD) are highly warranted. 1 Reducing the prevalence of chronic diseases calls for primary disease preventive efforts such as health-promotion activities and early detection of people at risk for lifestyle-related chronic diseases.2,3 Recent systematic reviews of general practice–based health checks suggest that people at high risk for a chronic disease may benefit from a targeted approach to health checks.4,5
To identify patients at high risk, the general practitioner (GP) needs systematically registered information on lifestyle risk factors in the electronic patient record (EPR).3,6–11 However, GPs in Denmark and elsewhere lack systematically collected information on lifestyle.12,13 This study aims to test the feasibility of a novel population-based risk stratification, comprising individual lifestyle intervention in primary care in Denmark for early detection of lifestyle-related chronic diseases using a combination of questionnaire and EPR data.
Methods
The intervention comprises four elements: (1) collection of information on lifestyle risk factors using a questionnaire with 15 validated items sent to patients listed with a GP, (2) electronic transfer of questionnaire data to the GPs’ EPR, (3) identification of patients already diagnosed with a lifestyle-related chronic disease, and (4) risk estimation and stratification of apparently healthy persons using questionnaire and EPR data on validated risk estimation models (see Figure 1).
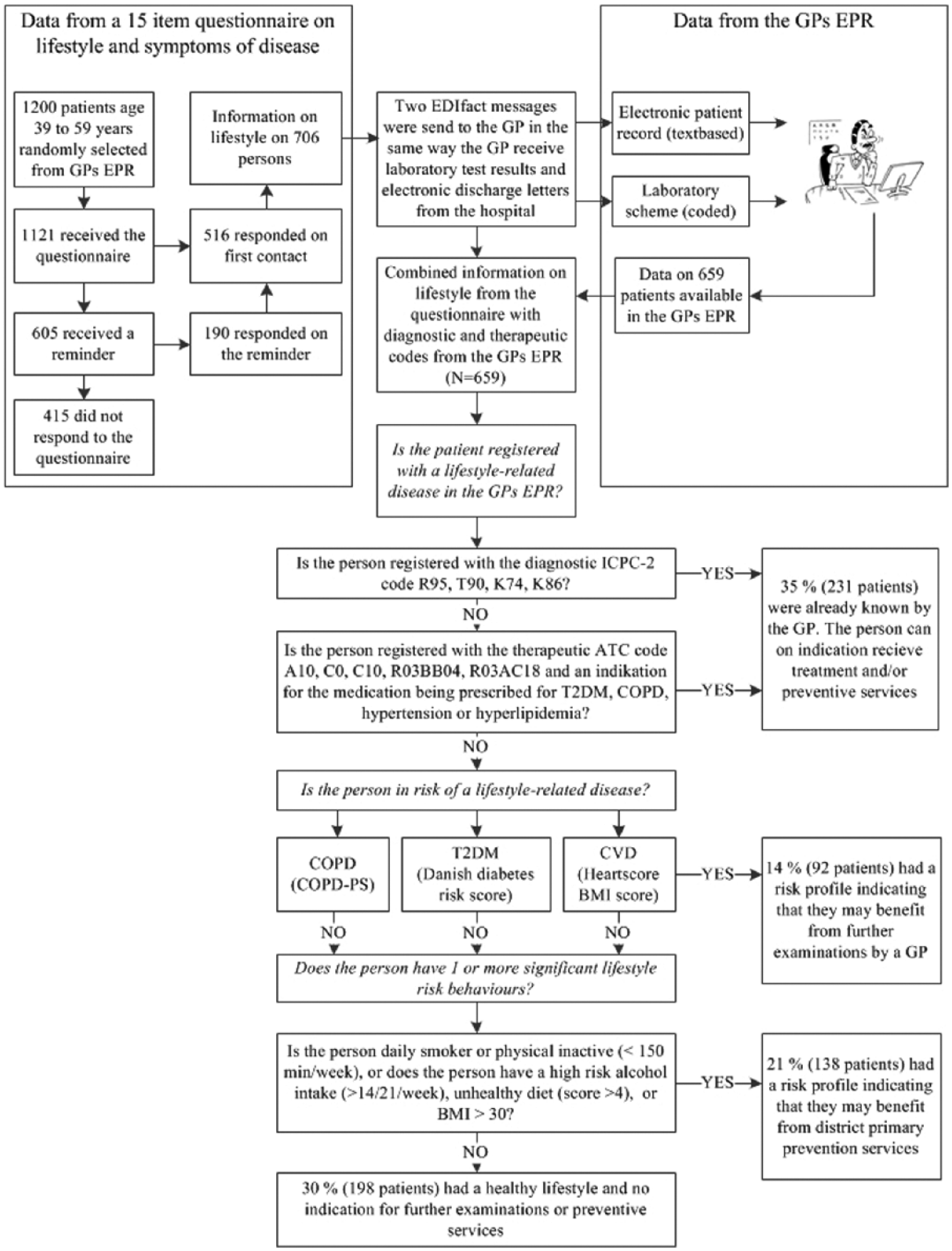
Flow diagram of the intervention.
A total of 1200 individuals aged 39–59 years were randomly selected from the patient lists of four clinics in three regions in Denmark. No disease-related criteria for excluding a patient were defined prior to the study. Danish general practices are organized as publicly financed private clinics with a list system and an average of 1600 patients registered per GP and two GPs per clinic. Approximately half of the practices have one GP. 14 A total of 11 EPR systems for primary care are certified to comply with national standards for cross-sectional communication. All Danish GP clinics have fully implemented EPR systems in daily practice. The four participating GP practices consist of two or three GPs. One of the practices is situated in a provincial capital city with 250,000 inhabitants. The other three practices are in cities with populations of 3000 to 44,000 inhabitants. One practice is in an area mainly populated by persons with low socioeconomic status, another is in an area mainly populated by persons with high socioeconomic status, and the other two practices are located in areas with diverse populations. To increase the probability of reaching the aim of the feasibility study, the four clinics were chosen based on their former experience and engagement in research and quality development.
Collection of information on lifestyle risk factors using a questionnaire
The questionnaire was developed based on a literature search for validated questions used in the selected risk estimation models. Items on smoking habits, alcohol consumption, physical activity, weight, height, and family disposition for lifestyle-related disease (first-degree relatives >70 years of age) were drawn from the Danish National Health Survey. 15 Items on diet were drawn from the Swedish National Guidelines for Disease Prevention and translated directly from Swedish into Danish as the two languages are very similar. 16 Items on symptoms of COPD were drawn from a validated COPD population screener (PS),7,16 translated from English to Danish using forward and back translation, and pilot tested among fellow research colleagues (see Appendix 1 for an English version of the questionnaire).
Questionnaire logistics
A letter of invitation and questionnaire were sent by the Danish Quality Unit of General Practice to participating patients by regular mail on behalf of each patient’s GP. In the invitation letter, the patient was invited to fill in the questionnaire on paper and return it by mail or to fill it in electronically using the survey software solution SurveyXact (www.datafabrikken.dk). The invitation letter stated the purpose of the study, that it was voluntary to fill in and return the questionnaire, and that the respondent was consenting to participate when returning the questionnaire. Non-response triggered a reminder after 3 weeks. The reminder was sent by regular mail. No further action was taken to increase the response rate. The participating GPs were consulted on the content and set-up of the invitation letter and questionnaire through mail correspondence and subsequent approval.
Transfer and synthesis of questionnaire and EPR data
The raw questionnaire data were processed to render them usable in a clinical context. Body mass index (BMI) was calculated based on self-reported weight and height (weight (kg)/height2 (m)), and a dietary score and a score on physical activity were calculated following the recommendations in the Swedish National Guidelines for Disease Prevention. 16 The results from the questionnaire were subsequently transferred to the GPs’ EPR systems as electronic data interchange (EDI) messages. For each patient, an EDI text message similar to a hospital discharge letter was transferred to the EPR, and an EDI coded message similar to biochemical laboratory results was transferred to the laboratory scheme. The coded message consisted of codes from the International Union of Pure and Applied Chemistry (IUPAC)/Nomenclature for Properties and Units (NPU) nomenclature and specific national codes for cross-sectional electronic communication within the Danish health-care system. The NPU terminology is a coding system and terminology for identification and communication of examination results from clinical laboratories in the health area and is supported by the International Federation of Clinical Chemistry (IFCC)-IUPAC (Sub)committee on NPU.
The coded EDI message contained information on smoking behaviour, BMI, units of alcohol consumption per week, minutes of physical activity per week, dietary score, and risk scores together with the result of the stratification stipulating whether the patient had been recommended to consult the GP or not.
Identification of patients already known to the GP
In 2005, the computer program Sentinel Data Capture was developed for Danish GPs with the purpose of collecting data for quality development and research.17,18 Sentinel Data Capture is installed on a GP’s PC or server and is designed to collect patient-specific data from the EPR. The collected data consist solely of structured data such as Anatomical Therapeutic Chemical (ATC) therapeutic codes on prescribed drugs, National Health Service disbursement codes, IUPAC/NPU laboratory codes, and International Classification of Primary Care (ICPC-2) encounters. Sentinel Data Capture was first used to collect key patient-specific data on diabetes care and seems to have contributed to improved care of patients with diabetes in general practice in Denmark. 18 The GPs have the possibility of using the whole range of ICPC-2 codes and are encouraged to assign ICPC-2 codes to all encounters.
In this study, patients already diagnosed with a lifestyle-related chronic disease or conditions such as hyperlipidaemia and hypertension were identified using a combination of ICPC-2 codes and therapeutic ATC codes for prescribed medicine, together with the indication for prescribing the medicine. Adding both the ATC therapeutic code and the indication for prescribing the medicine increased the likelihood that the medication was prescribed for, for example, COPD and not asthma. Patients with COPD were identified using the ICPC-2 code R95 or the ATC code category R03BB04 (tiotropium bromide) or R03AC18 (indacaterol) together with the indication text ‘obstructive’ or ‘COPD’ (see Table 2).
Validation algorithms.
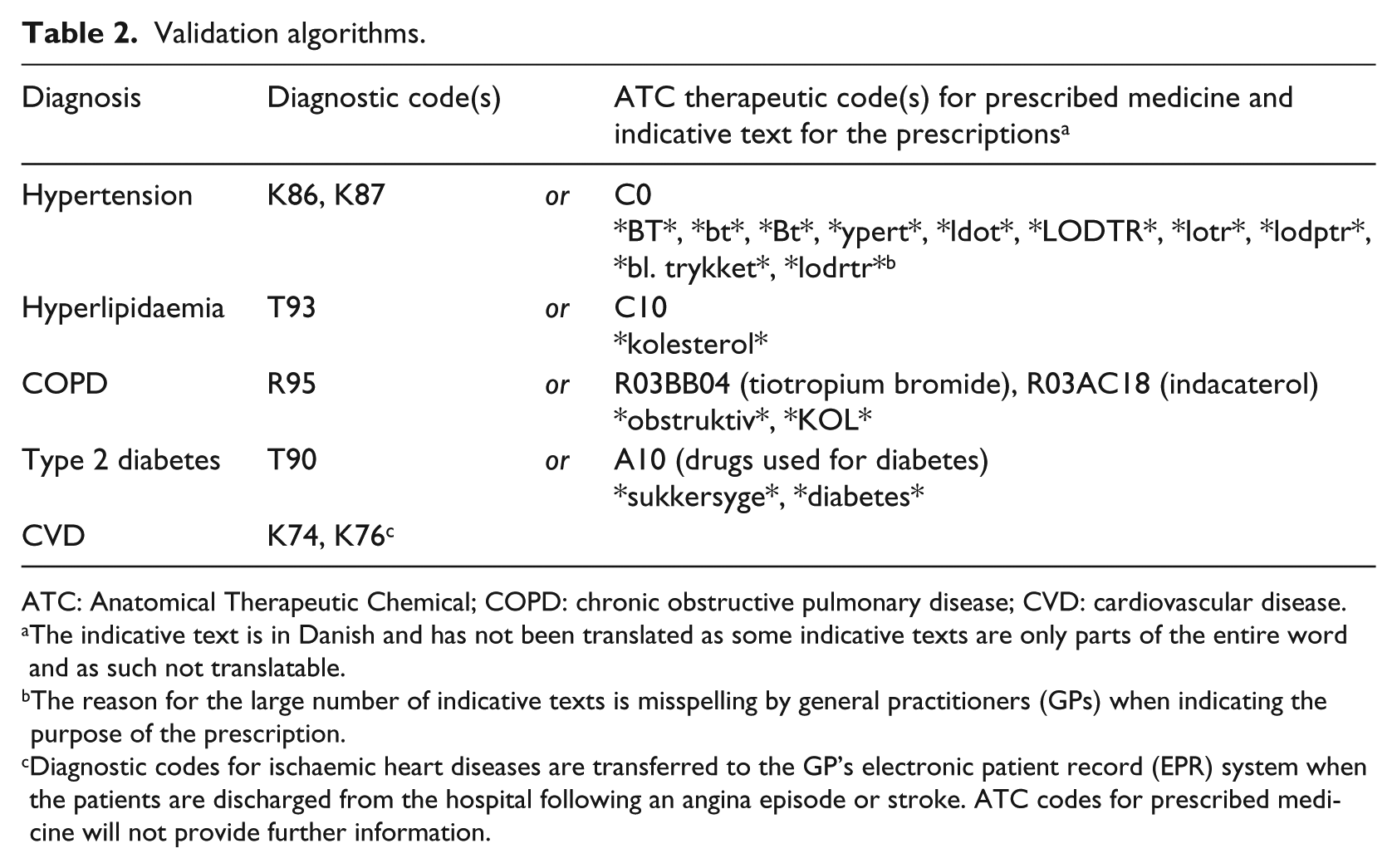
ATC: Anatomical Therapeutic Chemical; COPD: chronic obstructive pulmonary disease; CVD: cardiovascular disease.
The indicative text is in Danish and has not been translated as some indicative texts are only parts of the entire word and as such not translatable.
The reason for the large number of indicative texts is misspelling by general practitioners (GPs) when indicating the purpose of the prescription.
Diagnostic codes for ischaemic heart diseases are transferred to the GP’s electronic patient record (EPR) system when the patients are discharged from the hospital following an angina episode or stroke. ATC codes for prescribed medicine will not provide further information.
Patients identified by the validation algorithms were stratified to a group already known to be at risk for or have a lifestyle-related chronic disease and were as such assumed to be in some kind of treatment, with either medication or behaviour change. These patients were not included in the subsequent estimation and stratification of people at risk for a lifestyle-related chronic disease.
Risk estimation and stratification of persons at risk
The subsequent stratification of persons at risk consisted of two steps. In the first step, patients at risk for a lifestyle-related chronic disease were identified using three validated risk scores: the COPD-PS screener, the Danish Diabetes Risk model, and the Heartscore BMI score.7,9,19 All three models identify people at risk based on lifestyle risk factors and current EPR information only. It is important to keep in mind that risk estimation models such as the above are not intended to diagnose diseases. Diagnosis of disease calls for further examinations.
A COPD risk score was calculated based on the COPD-PS screener algorithm. 7 The COPD-PS screener uses an algorithm consisting of age, lifetime use of cigarettes, and symptoms from smoking to identify patients who may be offered spirometry to examine for COPD. The diabetes risk score was calculated based on the Danish Diabetes Risk score, 6 which uses an algorithm involving age, sex, BMI, known hypertension, physical activity, and parents having diabetes. The cut-off for being at risk for T2DM and COPD follows the recommendations in the Danish Diabetes Risk model and the COPD-PS screener, respectively. Patients with a COPD-PS score of 5 or above were considered eligible for spirometry. Patients with a diabetes risk score of 31 or above were considered eligible for HbA1c measurement.
Cardiovascular risk was calculated from the Heartscore BMI score, 9 which uses age, sex, smoking status, and BMI. The European Society of Cardiology recommends that this score be used to give a preliminary estimate of cardiovascular risk based on lifestyle risk factors alone, and as such, it is not a substitute for the full Heartscore. The Heartscore BMI score provides a figure for a 10-year CVD risk in the same way as the full Heartscore. In this study, the cut-off for being at risk was established as a 5 per cent risk of dying from CVD within the next 10 years, which is the cut-off for determining whether the patient may benefit from further examinations and possibly also pharmacological treatment.
When one or more of the risk estimation models indicated a risk for a lifestyle-related chronic disease, the patient was stratified to further examinations by the GP to confirm or invalidate a possible diagnosis or provide further information for planning a treatment, whether behaviour change or pharmacological treatment. Patients stratified for further examinations by the GP were not included in the second step.
The second step identified patients with an unhealthy lifestyle with one or more risk factors. An unhealthy lifestyle was defined as daily smoking, alcohol intake of more than 14/21 (male/female) units of alcohol per week, an unhealthy diet (diet score <4 on a 12-point score drawn from the Swedish National Guidelines on Disease Prevention), 16 BMI >30, and/or physical activity <150 min/week. A patient with an unhealthy lifestyle with one or more risk factors was stratified to behaviour change interventions at the municipal level.
Finally, patients with no lifestyle-related chronic disease, no risk for a lifestyle-related chronic disease, and no unhealthy lifestyle risk factors were stratified to a group with a healthy lifestyle with no indication for further examination or intervention.
Ethics approval
The study was approved by the Danish Data Protection Agency. According to Danish regulations, approval from a health research ethics committee is not required for questionnaire surveys.
Results
Questionnaire response rates
A total of 1200 persons were selected for the study – 300 from each of the four clinics. Because the GPs receive a fee from the subsequent consultations, the Danish Consumer Ombudsman’s view is that this kind of proactive contact with patients listed with the GP falls under the Danish Regulations on Marketing. We were therefore obliged to respect people who have actively chosen not to receive marketing by mail. Some 62 persons had actively chosen not to receive marketing by mail, and 17 persons had an unknown address. Hence, a total of 1121 persons received a questionnaire. Of these, 706 (63%) responded, 46 per cent at first contact and 17 per cent after one reminder. Of the 706 persons who returned the questionnaire, 62 per cent returned it in an enclosed envelope. The remaining 38 per cent filled in the questionnaire electronically.
The response rates of women and men were 67 and 59 per cent, respectively, and the response rate increased with age. The response rate in the four clinics ranged from 58 to 66 per cent. Some 685 persons filled in the entire questionnaire (see Table 3). The 21 persons who did not fill in the entire questionnaire were not included in the analysis.
Results from the questionnaire.
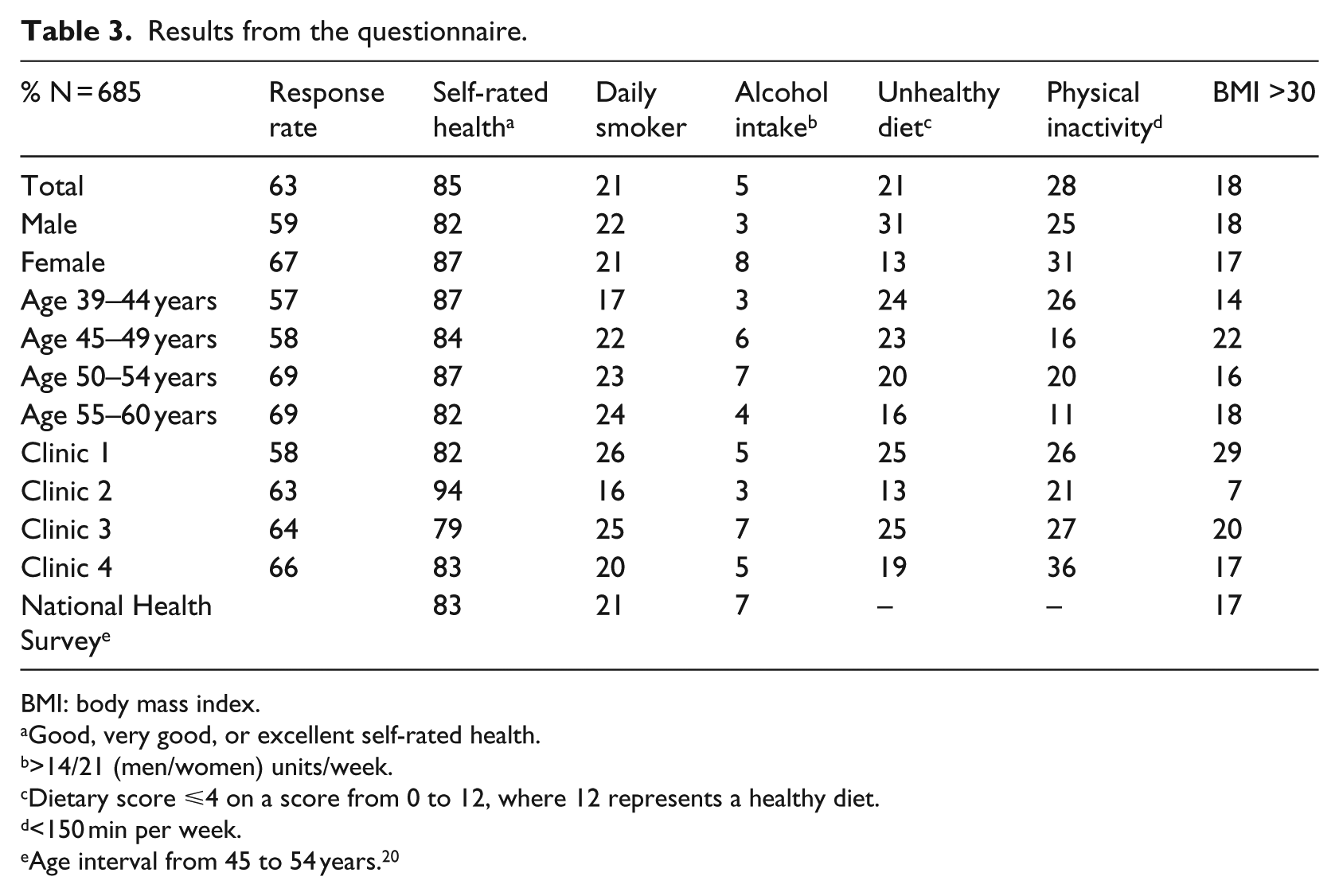
BMI: body mass index.
Good, very good, or excellent self-rated health.
>14/21 (men/women) units/week.
Dietary score ⩽4 on a score from 0 to 12, where 12 represents a healthy diet.
<150 min per week.
Age interval from 45 to 54 years. 20
Registration of lifestyle risk factors in GPs’ EPRs
Among the respondents, the systematic registration of smoking and BMI in the GPs’ EPR system was 17 and 23 per cent, respectively, prior to the intervention. Almost all respondents filled in the questionnaire, making it possible to increase the systematic registration of lifestyle risk factors in the EPRs up to 63 per cent (see Table 4).
Number of responses on the individual risk factors.
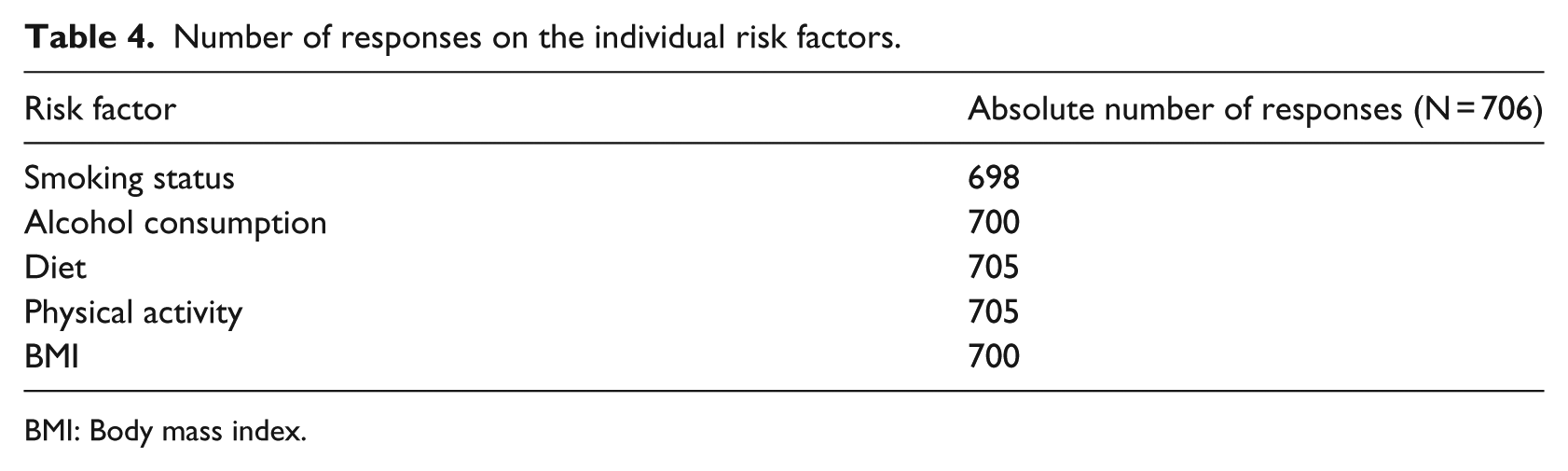
BMI: Body mass index.
Stratification of respondents according to diagnosis and risk
It was possible to stratify 659 of the 706 respondents based on both the questionnaire and EPR data. The 24 persons whom we could not stratify because of missing EPR data are not included in the following analysis. Some 35 per cent of the respondents were already known by the GP as a patient with a diagnosis or receiving treatment for T2DM, CVD, or COPD and/or receiving treatment for hyperlipidaemia and/or hypertension. A total of 66 per cent received treatment for one condition, 21 per cent for two conditions, and 13 per cent for three or more conditions. Of the 35 per cent who were already known by the GP, 83 and 37 per cent received treatment for hypertension and hyperlipidaemia, respectively. A total of 14 per cent were identified as being treated for T2DM and 11 per cent for COPD, while 4 per cent were identified with ischaemic heart disease.
Some 14 per cent of the respondents (92 patients) had a risk profile indicating that they might benefit from further examinations by a GP, 16 per cent of whom (15 patients) were estimated to be at risk for COPD. A total of 89 per cent (81 patients) were estimated to be at risk for T2DM and 24 per cent (22 patients) at risk for CVD. Some 74 per cent were estimated to be at risk by only one risk estimation model, 23 per cent by two models, and 3 per cent by all three models. A total of 21 per cent had a risk profile indicating that they might benefit from municipality primary prevention services but had no need for further examinations by the GP. Among these, 43 per cent were daily smokers, 8 per cent had a high-risk alcohol intake, 17 per cent had an unhealthy diet, 54 per cent were physically inactive, and 20 per cent had a BMI above 30. A total of 30 per cent (198 patients) had a healthy lifestyle and no indication for further examinations or preventive services.
Discussion
This study showed that it is feasible to stratify patients according to their risk of a lifestyle-related chronic disease without an additional workload for the GP and furthermore to target the services provided by the GP to one out of five ‘healthy’ patients.
Strengths and limitations
We managed to collect information on lifestyle from almost all respondents. We also managed to transfer information from the questionnaires to the GPs’ EPR systems using the established system for electronic communication as both a text message and a coded message.
The four participating clinics were selected based on their former engagement in quality development projects. The clinics thus are most likely not representative of Danish general practice in terms of the use of ICPC-2 coding, prompting the question of whether the intervention is feasible in Danish general practices with less comprehensive ICPC-2 coding practices and less experience with information technology (IT)-based quality development tools. In this study, we used EDI messages and national communications standards such as the IUPAC/NPU nomenclature, which all Danish GPs can receive and send. The only active involvement from the GP was to approve the incoming EDI messages in the EPR. A meta-analysis has shown that the effectiveness of feedback to the GP depends on the baseline performance. 21 The lower the baseline recording, the greater the effect, suggesting that the impact of the intervention could be even larger in other less optimally performing Danish GP clinics than those participating in this study.
Patients already diagnosed with a chronic disease were identified based on ICPC-2 codes, ATC therapeutic codes, and the indication given for prescribing the drug. The strength of this way of validating diagnoses is high sensitivity because it captures patients receiving therapeutic treatment for a condition or a disease. The weakness is low specificity; patients are not identified by this model if they are either not registered with an ICPC-2 code but should have been, or do not receive therapeutic treatment for the condition or diagnosis. On the one hand, the registration of ICPC-2 codes among Danish GPs is currently not comprehensive, and ICPC-2 codes cannot in themselves identify all patients with a diagnosed disease. Close to 100 per cent of Danish GPs are enrolled in Sentinel Data Capture, but only 50 per cent of them register more than 70 per cent of their face-to-face consultations with an ICPC-2 code (May 2014). On the other hand, ATC therapeutic codes, together with an indication for prescribing the drug, will identify only patients receiving therapeutic treatment. Laboratory test results could add important information because they are currently used as diagnostic criteria for the diseases and conditions included in the study.2,3,22 Algorithms using laboratory test results, ICPC codes, ATC therapeutic codes, and the indication for the prescription are thus warranted.
Even though we achieved a response rate of 63 per cent with a mail-distributed questionnaire, we lack information about lifestyle from 37 per cent of the patient population. Participation in lifestyle surveys has declined during the last decade, and a response rate of 63 per cent with a low effort to increase the response rate shows that the intervention is perceived as valuable to the target population. 23 Two similar interventions in the Netherlands reached a response rate of 33 and 75 per cent, respectively.24,25 According to the authors, the low response rate was primarily due to low confidence about online assessments among the respondents. 24
Almost all Danes consult their GP within a time span of 3 years and around 80 per cent within one calendar year. Similar studies from New Zealand and Sweden have shown response rates of 91 and 70 per cent, respectively, using questionnaires distributed in the waiting room.26,27 Questionnaires distributed in this way could, in combination with mail-distributed questionnaires, increase response rates and should be examined further.
Generalizability
The model is generalizable to other health-care systems, but it requires a primary care system with a highly developed IT infrastructure. This infrastructure should include ways to receive structured electronic information such as EDI messages from sources other than hospital-based laboratories and the possibility of combining this information with current EPR information to develop patient-specific risk profiles, either directly in the EPR or by a database. In the stratification of patients at risk for lifestyle-related chronic diseases, we focused our attention on lifestyle and lifestyle-based risk estimation models. Other questionnaires, risk estimation models, target groups, and focus areas such as cancer or mental illnesses can be used according to the specific purpose of the intervention.8,26,27 However, laws and regulations on the privacy of health-related data may limit the possibilities of implementing a model like the one presented here.
Conclusion
It is feasible to implement an innovative intervention that identifies and stratifies patients for further examinations in general practice or behaviour change interventions at the municipal level without additional workload for the GPs. Using this model at the national level holds the potential to support GPs in preventing and treating lifestyle-related diseases and targeting diagnostic and lifestyle interventions towards high-risk patients. A large randomized intervention study is being planned that will combine the described model with targeted interventions in GP practice and the municipality.
Footnotes
Appendix 1
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
