Abstract
This article describes the development and clinical impact of the Italian Regional ADHD Registry, aimed at collecting and monitoring diagnostic and therapeutic pathways of care for attention-deficit hyperactivity disorder children and adolescents, launched by the Italian Lombardy Region in June 2011. In particular, the model-based software used to run the registry and manage clinical care data acquisition and monitoring, is described. This software was developed using the PROSAFE programme, which is already used for data collection in many Italian intensive care units, as a stand-alone interface case report form. The use of the attention-deficit hyperactivity disorder regional registry led to an increase in the appropriateness of the clinical management of all patients included in the registry, proving to be an important instrument in ensuring an appropriate healthcare strategy for children and adolescents with attention-deficit/hyperactivity disorder.
Keywords
Introduction
Attention-deficit hyperactivity disorder (ADHD) is a neurobehavioral disorder characterized by maladaptive, and inappropriate, levels of inattention and/or hyperactivity and impulsivity in early childhood that can persist through adolescence, pervade across settings and lead to notable impairments in adulthood. 1 Current evidence defines ADHD as a disorder resulting from complex interactions between genetic and environmental factors. ADHD is diagnosed by the severity and persistence of symptoms, which are associated with high levels of impairment in family and social relationships and with a higher risk of developing co-occurring psychiatric disorders during the lifetime, such as mood, conduct and substance abuse disorders. 2
In recent years, there has been an increase in the clinical recognition of ADHD, with a corresponding increase in the number of children diagnosed and treated. 3 Worldwide, the prevalence of ADHD is estimated at 5.3 per cent, although there is wide variability between geographic locations. 4
In the Italian Lombardy region (the most populated and economically important region in Italy), the estimated prevalence was 9.5 per 1000 children and adolescents aged between 6 and 17 years, and less than a quarter of them received drug therapy. 5
Although there is growing evidence that stimulants for the treatment of ADHD both improve ‘core’ ADHD symptomatology and reduce the risk of developing psychiatric co-morbidities, psychosocial treatments such as psychoeducation, cognitive-behavioural therapy (CBT) and supportive training to help with organizing daily activities are also the first choice, alone or in combination with pharmacological therapy, according to clinical and environmental evaluation. Psychosocial treatments have been shown to be more effective than pharmacological therapy alone, in particular when both parents and teachers are involved. 6 According to the Italian guidelines on ADHD treatment (defined in 2003 through a consensus conference), 7 drug treatment should only be started after a child psychiatrist, who is an expert in ADHD, has thoroughly assessed the child or adolescent and confirmed the diagnosis. Moreover, once treatment has been initiated, the effectiveness and adverse effects need to be assessed carefully and regularly, and reported, by each reference centre’s team of clinicians.
Because of the large concern about the safety and rational use of psychotropic drugs in children, a national initiative was launched in Italy in 2007 to monitor the prevalence and appropriateness of drug therapy in the ADHD paediatric population: the National ADHD Registry. The registry was set up under the auspices of the Italian Medicines Agency (AIFA) and coordinated by the Italian National Institute of Health, following the reintroduction of methylphenidate on the market (as immediate-release tablets) and the registration of atomoxetine. These are the only two drugs with a specific indication for ADHD available in Italy, and are registered for use only in children aged 6–17 years. The Italian registry is a unique tool internationally; it is able to ensure the monitoring and evaluation of the safety and tolerability of methylphenidate and atomoxetine in children and adolescents with ADHD. 8
To create shared and feasible diagnostic and therapeutic pathways, however, an analysis of currently existing clinical and socio-anamnestic variables, such as type of ADHD or specific environmental life contexts, is a key step. This analysis then needs to be followed by a final evaluation of the process outcomes, including user satisfaction in relation to the health services provided and the quality of life of families. The data collected through the National ADHD Registry made it possible to clarify certain critical issues, but not to perform a finalized epidemiological analysis, nor to best plan effective strategies for improvement. In particular, few data concerning the families’ anamnesis are collected and no information about ADHD patients receiving psychological treatment or other psychotropic agents are collected.
In this context, the need to create a new database to monitor the diagnostic and treatment pathways for all children and adolescents with ADHD, not only those already receiving drug treatment, was apparent. This article describes the development and clinical impact of the Italian Regional ADHD Registry, an initiative aimed at ensuring an appropriate management plan for children and adolescents with ADHD and set up as part of the project ‘Sharing of diagnostic and therapeutic pathways for ADHD’. 9
Methods
The new ADHD database, the ‘Italian Regional ADHD Registry powered by PROSAFE’, allowed, through a flexible interface, the collection of information relating to
Anamnestic data;
Clinical assessment;
Diagnosis;
Therapeutic interventions, both pharmacological and non-pharmacological;
Follow-up visits.
The information collected was analysed monthly, and the results were presented and discussed at regular meetings between the participating centres and the coordinating centre (IRCCS – Istituto di Ricerche Farmacologiche Mario Negri), in order to assess whether any improvements were achievable. 9 The coordinating centre published all results in a monthly summary report sent to each participating centre and posted it on the ADHD project website (adhd.marionegri.it).
Before the project started one of the 18 centres was selected as a pilot site to test the system and to resolve emerging technical problems. After a 3-month running period, a technical support service was established through a dedicated phone number and monthly meetings with all clinician participants were organized for feedback.
PROSAFE
The ‘Italian Regional ADHD Registry powered by PROSAFE’ originated from the development of an already-existing model-based software called ‘PROSAFE’, a programme used for data collection in many Italian intensive care units (ICUs). 10 The modular structure and features of the software have made it possible to create a stand-alone Case Report Form (CRF) interface to collect data on the Italian Regional ADHD Registry, while continuing to use the skills and technical characteristics that distinguish PROSAFE.
The software architecture used is PROSAFE CLIENT-SERVER, which allows more software to be installed on the same network, according to the needs of each individual centre. In addition, each centre may use and store data even offline without an internet connection – an option that would have been impossible with a web-based software. The software is free of charge for the ADHD centres.
Eligibility of centres and users
Italian healthcare is provided free or at a nominal charge through a network of 148 local health units (LHUs). Child and adolescent neuropsychiatric services (CANPS) are part of the LHU and provide care at the hospital and community level for children and adolescents with neurologic and/or psychiatric and/or neuropsychological disorders (including developmental disabilities and intellectual disabilities), and for their families. CANPS are multi-professional, comprehensive community services providing diagnosis, treatment, and rehabilitation. In order to prescribe methylphenidate or atomoxetine to ADHD patients, Italian regulatory rules require a strict clinical assessment for the diagnosis of the disorder and a systematic patient’s monitoring during treatment. Since September 2007, local reference centres have been required to send patient information to the Italian National Registry dedicated to collecting data only on pharmacological treatment of ADHD patients aged less than 18 years. Regional health authorities are responsible for the accreditation of the reference centres in regional hospitals, which are linked to the CANPS located in the local communities. The reference centres are therefore the specialized hubs of the CANPS network on ADHD. In the Lombardy Region there are 18 ADHD reference centres and all participated in this project.
Children and adolescents aged 5–17 years who accessed any of the 18 local centres for a diagnosis of suspected ADHD were enrolled in the Regional ADHD Registry if (a) their first access to the service (first visit) was after 1 June 2011, regardless of the diagnosis or (b) they began their drug treatment after 1 June 2011 (if they were not already included in the national register).
Results
Access-history
The Italian Regional ADHD Register permitted an evaluation of the access to the ADHD reference centres by patients in the Lombardy Region, that is, the patients’ requests and waiting times (time elapsed between the first request and the first survey).
Questions were also asked on the environment of the families and the patients (with whom they were living, if they had siblings, or if there were twins) and on the social situation of the parents, and their history, in order to better understand if there was a possible familiarity for ADHD and if there were any genetic hypotheses.
Assessment
The evaluation page contained a whole series of tests, interviews, and examinations that the patients underwent for the diagnosis of ADHD. These tests were agreed on by a specific sub-group of the regional project, formed by the operators from each participating centre, which had the task of sharing their own diagnostic methodologies (guidelines and tests) in order to allow a greater uniformity among all centres in the diagnostic phase.
Specifically, during the diagnostic evaluation the following tests/information required were as follows:
Anamnestic data;
Clinical interview;
Neurological examination;
Intelligence Quotient (IQ);
Diagnostic Interviews: Kiddie-Schedule for Affective Disorders and Schizophrenia (K-SADS) or Development and Well-Being Assessment (DAWBA);
Evaluation of parents: Conners’ Parent Rating Scales (CPRS) and Child Behaviour Checklist (CBCL);
Evaluation of teachers: Teachers’ Parent Rating Scales (CTRS);
Clinical Global Impressions-Severity (CIGS) or Children’s Global Assessment Scale (CGAS).
Diagnosis
The diagnostic page contained questions on the patient’s symptoms and behaviour. Based on the answers entered the programme then processed the data entered by the clinician and classified the diagnosis according to the diagnostic criteria of the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM-IV-TR), assigning one of four possible results:
Patient not diagnosed with ADHD;
Patient diagnosed with ADHD type I (inattention);
Patient diagnosed with ADHD type H (hyperactivity-impulsivity);
Patient diagnosed with ADHD type C (combined).
Figure 1 shows an example of the data collection form.
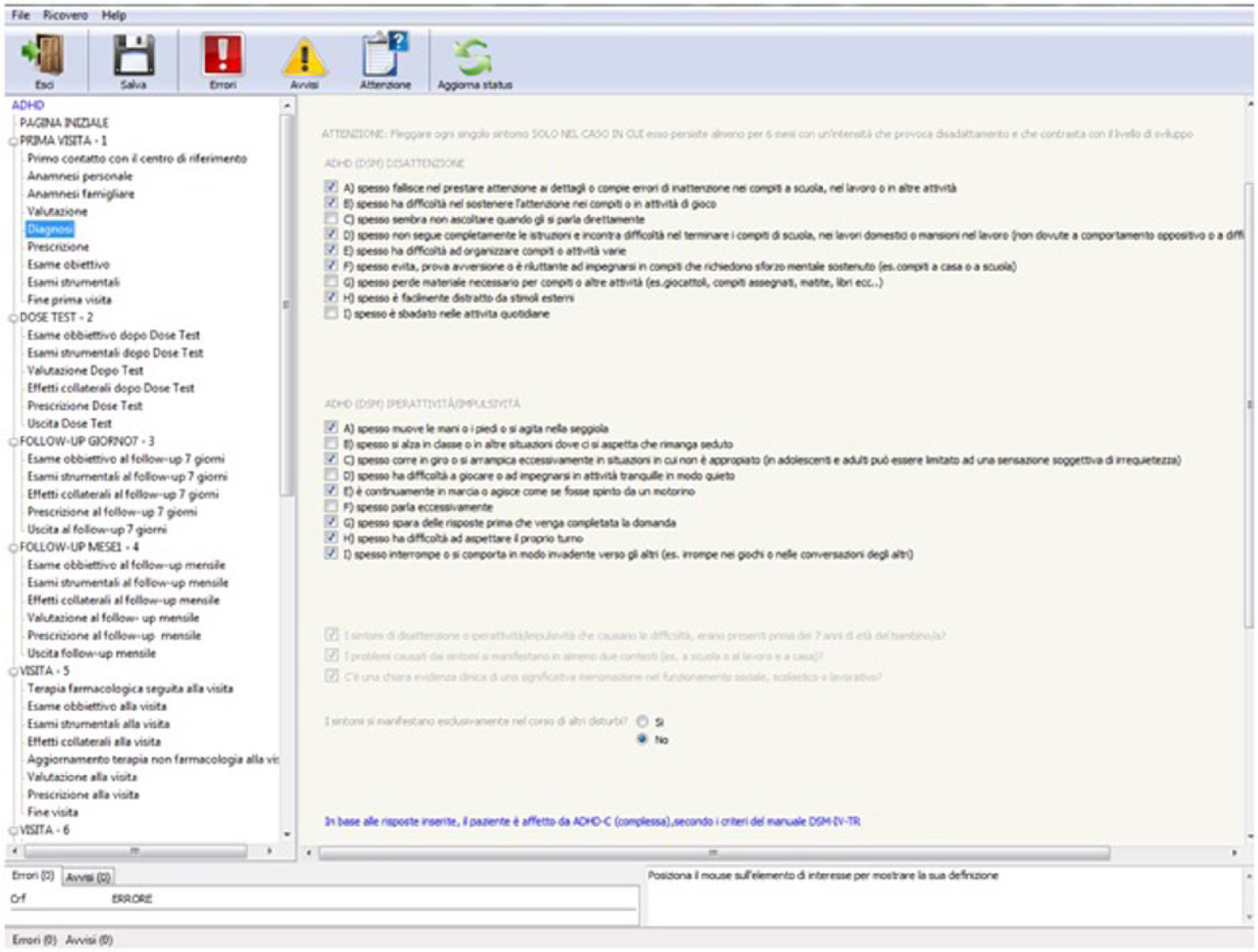
Data collection form in the diagnostic page.
In the case of an ADHD diagnosis, the centre’s operator was requested to proceed with filling in the fields of the pages following the diagnosis section, according to the prescribed therapy.
In cases in which the patient was not diagnosed with ADHD the operator was sent to a page stating ‘out of the registry’, in which the following potential causes for the interruption of the visits were indicated: ‘no ADHD’, ‘the patient does not show up at the visits’, ‘treatment interruption’ or ‘transfer to another ADHD centre’.
Therapy
According to the Italian Regional Registry the prescribed therapy was pharmacological, non-pharmacological, or both. The pharmacological treatment involved all the psychotropic drugs indicated for ADHD therapy: mainly methylphenidate or atomoxetine, and in a few cases other psychotropic agents.
Drug therapy was given in combination with non-pharmacological interventions, such as cognitive-behavioural therapy (CBT), child, parent, and teacher training, or counselling.
Once the therapy was prescribed the patient was seen for follow-up visits at given time periods, according to the type of prescribed therapy: in cases of methylphenidate or atomoxetine treatment, the patient was re-assessed after 7 days, while in cases of other psychotropic agents, after one month. This was different for non-pharmacological therapies, in which the type of prescribed therapy did not influence the timing of the subsequent visit if associated with a pharmacological treatment, if not, the follow-up visit was provided after 3 months of the start of therapy.
Follow-up visits
Following the diagnosis of ADHD, the register was designed to provide several, differently structured types of follow-up visits, based on the prescribed therapy, in order to optimize the compilation of the CRF and to therefore have clean and correct data. In particular, the planned follow-up visits were as follows:
First visit. The set of requests for anamnestic data of patients and families, assessment and formulation of diagnosis and prescription of therapy.
Dose test. A visit performed only in cases in which the patient was treated with methylphenidate for the first time, and it was therefore necessary to ensure that there were no adverse drug reactions.
7 days. A visit following the dose test, whose aim was to provide further checks, or a visit for those patients prescribed atomoxetine for the first time.
1 month-visit. A visit following the ‘7 days’ (for atomoxetine or methylphenidate), or a visit performed when an another psychotropic drug was prescribed.
3 month- and/or 6 month-visit. Control visits following the 1 month-visit to monitor all patients, whether or not on drug therapy.
Extra visit. An extraordinary (unplanned) visit performed due to the occurrence of side effects and/or adverse drug reactions, changes in therapeutic plan, or additional patient monitoring.
The order of the visits in the register was decided by the software, which followed an internal logic based on the drug therapy prescribed to the patient. The drug prescription on the first visit determined the first follow-up visit according to the written criteria indicated in the following Table 1.
Drug prescription on the first visit.

The calculation of the follow-up visits took into consideration the following data:
Type of previous follow-up visit;
Drug therapy prescribed on the previous visit;
Presence of extra visits not provided in the therapeutic plan;
Type of current follow-up visit;
Prescribed therapy in the current follow-up visit;
Patient’s compliance to the prescribed therapeutic plan.
Figure 2 shows the calculation of the follow-up visits, when the patient was in a normal situation without extra visits, and when the patient changed drug (or did not use it) and there was an extra visit.
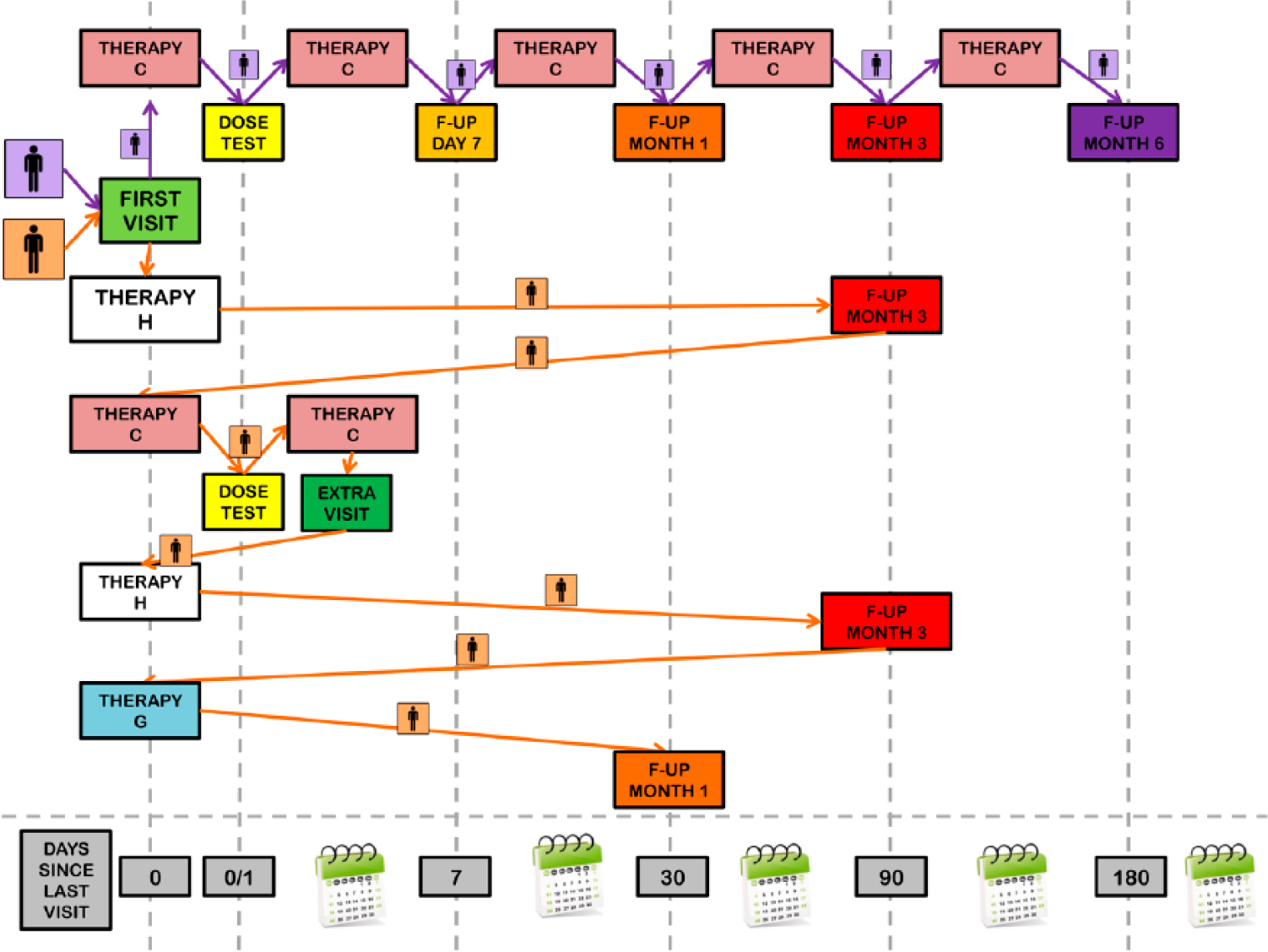
Follow-up visit logic.
The possible combination therapies in Figure 2 are outlined in Table 2 below.
Possible combination therapy.

Data collection
At the time of analysis, data from the Italian Regional ADHD Registry were collected and referred to the diagnostic and therapeutic pathways of 1338 children and adolescents. All of the 18 centres adopted the initiative and completed the patient records in the registry, with a range of 34–187 patients per centre (average 74). The number of patients per centre was related to the resident paediatric population.
Of the 1338 children and adolescents (86.7%), 1160 with suspected ADHD completed the diagnostic assessment, and 751 (64.7%) met criteria for ADHD. In all, 115 patients (15.3% of those diagnosed with ADHD) had been treated with at least one psychoactive drug: 107 with methylphenidate (5–60 mg daily), 19 with atomoxetine (10–80 mg daily) and 10 with other agents. A total of 63 adverse events were reported in 28 patients treated with drugs (rate: 24.3%), and headache, decreased appetite, asthenia and drowsiness were the leading events.
Of the children with ADHD who were treated, only 9 discontinued the drug prior to 1 year of treatment, none of whom because of adverse events. Although the medications for ADHD are generally well tolerated, the Lombardy Region ADHD registry could be a useful tool to improve the rational use of drugs in children and adolescents by disseminating and monitoring evidence-based practices and by monitoring the safety and efficacy of treatments in both the short and long terms. 5
Data processing and monitoring
The IRCSS – Mario Negri Institute for Pharmacological Research, as coordinating centre – monitored and processed the data entered in the Italian Regional ADHD Registry from all the centres and sent a monthly summary report to each participating centre (Figure 3).
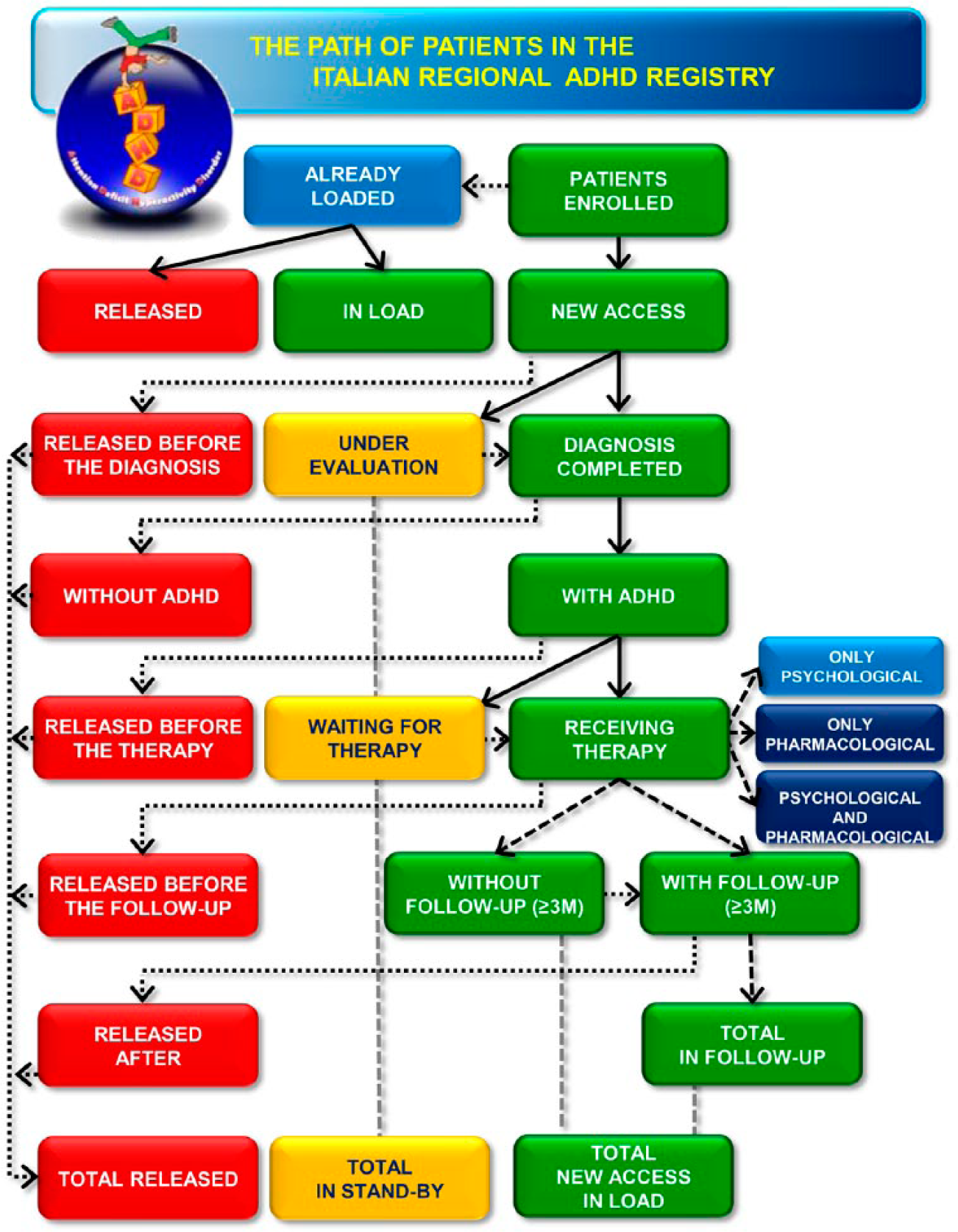
Report structure.
The report contained a flow-chart illustrating the paths of the all patients enrolled in the registry, with the corresponding values achieved, and consisted of 3 sections:
General trend of all participating centres;
Trend of the each single centre, referring only to the patients of the centre receiving the report;
Inconsistent and/or missing data detected for the patients enrolled by each single centre. The centre receiving this indication was requested to correct its data.
Discussion
The differences between the centres (different management, structure, staff, etc.) led to many problems in drafting the CRF shared by all the centres. Consequently, it was necessary to create a series of software releases subsequent to the first one in order to standardize the data collection form. This was possible thanks to the programme’s flexibility, because every single change could be made without affecting the normal compilation of the registry by the participating centres.
The use of diagnostic–therapeutic pathways and the follow-up visit logic controlled by the programme based on the data entered, as well as the parallel management of other resources supporting the software such as the monthly summary data report for each centre, the periodical newsletter sent to the centres and the website (adhd.marionegri.it) content update, led to an increase in the appropriateness of the clinical management of all patients included in the registry.
Initially, the use of the software by the ADHD centres was sporadic, mainly due to the lack of staff organization and capacity in filling out a computerized CRF. Continuous support activities performed by the coordinating centre, such as providing quick replies to the centres’ queries by telephone or email and holding monthly meetings with the ADHD centres, led to a large improvement, over a 1-year period, in the technical management of the programme and to a consequent improvement in the quality of data entered as well as to an increase in the frequency with which the centres connected to the programme.
The use of the software increased the centres’ ability to use computer technology for clinical data collection, especially in centres that were not previously registering data systematically. Currently, most of the centres are connecting daily or weekly, with only a few centres connecting monthly. Finally, thanks to the ongoing development of the PROSAFE software, the registry can be continuously integrated and upgraded so as to bring constant improvements to the CRF and to the data collection in general, without causing any disturbance to users.
The identification of centres and the software installation were carried out through a web portal, which monitored access and authentication by the different centres involved in the project. This project has demonstrated how the software has improved the appropriateness of diagnostic and therapeutic pathways for patients enrolled in the Regional ADHD Registry. Specifically, the software described allowed a greater ability to edit, check, and manage the diagnostic and therapeutic pathways, thanks to a flexible internal frame. In particular, the calculation of the follow-up visits was automatic, based on certain data values. The programme gave the centres access to the system for later follow-ups in a manner consistent with the diagnostic–therapeutic data entered previously.
Conclusion
The Italian Regional ADHD Registry is an efficient tool to estimate the prevalence and incidence of ADHD, to evaluate the patients’ psychopathological profile and comorbid psychiatric conditions, and to monitor the clinical outcome of the prescribed therapy (psychological, pharmacological treatments or both) and potential adverse events and/or side effects of pharmacotherapy in the Lombardy Region’s paediatric population. The software is free of charge and is potentially available for other regions and/or LHUs.
The Italian Regional ADHD Registry therefore represents a distinctive tool to promote a collaborative experience between several ADHD centres and the coordinating centre, that is unique in the international context and that assures the appropriate care and safety of drug use in ADHD children, according to recent evidence-based practices and guidelines.
Footnotes
Acknowledgements
The authors would like to thank Luca Antiga (OROBIX S.R.L) Daniele Crespi and Guido Bertolini (IRCCS – Istituto di Ricerche Farmacologiche Mario Negri) for their contributions to the development of the key aspect of this research project, Chiara Pandolfini for the English revision, and Daniela Miglio for manuscript editing. The authors also wish to thank the Italian Regional ADHD Registry Group participants who took part in the project: Stefano Conte, Valeria Renzetti and Laura Salvoni (Bergamo); Massimo Molteni and Sara Trabattoni (Bosisio Parini, LC); Paola Effedri, Elena Filippini, Elisabetta Pedercini and Edda Zanetti (Brescia); Nadia Fteita (Como); Daniele Arisi and Roberta Mapelli (Cremona); Simona Frassica, Simonetta Oriani and Christian Trevisan (Garbagnate Milanese, MI); Susanna Acquistapace, Ottaviano Martinelli and Davide Villani (Lecco); Emanuela Binaghi, Andrea Deriu and Ernesta Ricotta (Legnano, MI); Arianna Borchia and Paola Morosini (Lodi); Maddalena Breviglieri, Giuseppe Capovilla and Roberto Segala (Mantova); Claudio Bissoli, Maria Paola Canevini, Antonella Costantino, Isabella Cropanese, Emiddio Fornaro, Silvia Merati, Alberto Ottolini, Monica Saccani, Roberto Vaccari, Vera Valenti and Alessandra Valentino (Milano); Umberto Balottin, Matteo Chiappedi and Elena Vlacos (Pavia); Corrado Meraviglia, Maria Grazia Palmieri and Gianpaolo Ruffoni (Sondrio); Francesco Rinaldi and Federica Soardi (Vallecamonica–Sebino, BS); Chiara Luoni, Francesca Pavone, Giorgio Rossi and Cristiano Termine (Varese). All authors contributed to this work in terms of conception and design of the study, analysis and interpretation of data and paper writing. M.B. was involved in drafting the manuscript and revising it critically for important intellectual content. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is part of the ‘Sharing diagnostic-therapeutic approaches for ADHD in Lombardy’ project partially funded by the Lombardy Region (D.G. sanità n.3250, 11/04/2011).
