Abstract
We demonstrate how to develop a simulation tool to help healthcare managers and administrators predict and plan for staffing needs in a hospital neonatal intensive care unit using administrative data. We developed a discrete event simulation model of nursing staff needed in a neonatal intensive care unit and then validated the model against historical data. The process flow was translated into a discrete event simulation model. Results demonstrated that the model can be used to give a respectable estimate of annual admissions, transfers, and deaths based upon two different staffing levels. The discrete event simulation tool model can provide healthcare managers and administrators with (1) a valid method of modeling patient mix, patient acuity, staffing needs, and costs in the present state and (2) a forecast of how changes in a unit’s staffing, referral patterns, or patient mix would affect a unit in a future state.
Introduction
Adequate staffing levels in hospitals are critical to daily operations and for ensuring optimal care delivery, patient safety, and patient and staff satisfaction. Inadequate staffing increases adverse patient outcomes, including pneumonia, shock, cardiac arrest, and urinary tract infections; 1 it also increases the likelihood of staff turnover. Hospitals spend US$22,000–US$64,000 per registered nurse who leaves the workforce and is replaced, leading to US$12 billion in excess costs each year.2,3
To predict staffing needs, managers use (1) the number of patients admitted and discharged per day and per year to a unit or service, (2) the acuity of the patient population and an inventory of needed resources, and (3) the estimated growth or decline in these factors over time. But managers must often base planning on insufficient data and are further affected by uncertainty regarding the effects of local and national policy changes, such as the US Affordable Care Act 4 which has increased access to care. Moreover, some localities mandate particular staffing levels, and it would be helpful for managers to have tools that would allow better prediction of staff needs based upon patient population. Discrete event simulation incorporates probability distributions estimated from existing data as well as previous experiences to model complex, dynamic systems. Discrete event simulation can help nurse managers better predict staffing needs, thereby potentially improving patient safety, health outcomes, and predictability of nursing assignments.5–7 It may also reduce stress associated with understaffing and ultimately reduce costs and improve the efficiency of care. Although it is impossible to account for all variables affecting daily fluctuations in census and acuity at hospitals or clinics, staffing needs in 1 unit can be simulated based upon historical data of patient admissions, discharges, transfers, and acuity over days, months, and years. Furthermore, a simulation model can be adjusted to predict needs related to growth by addressing strategic questions such as the following:
If two hospitals partner, can current staffing levels tolerate 100 additional transfers each year?
If population growth continues for 10 years, how many new beds will be needed?
If new nursing assistants free each registered nurse to take one additional patient and the maximum nurse:patient ratio drops from 1:3 to 1:4, how will the cost structure change?
This study demonstrates how we developed and piloted a discrete event simulation model of nursing staff needed using data from a neonatal intensive care unit (NICU) to help managers better meet patients’ needs. We developed the simulation by combining historical data elements from our hospital and US national-level outcomes data. 8 We then assessed model validity and accuracy using two different staffing levels (26 and 28 nurses) and comparing the model’s output with historical data.
Methods
Model development: data elements
To develop our model, we used various data elements, including the hospital bed size, number of staff, patient flow structure, nursing practices, and de-identified, retrospective patient outcomes from the Duke University Hospital NICU from January 2008 to June 2013. An existing database maintained by the unit’s administrative staff included patients’ demographic information, diagnoses (date of specific disease states or therapies), billing and coding details by each day of patients’ stay, and discharge status (sent home, transferred to another unit or facility, or deceased). Following approval by the Duke University Institutional Review Board, data were de-identified, analyzed, and combined with published US national-level outcomes data 8 to estimate admission and morbidity inputs. The final probability distributions were anonymized. No simulated patients were based directly on any actual patient.
Discrete event simulation: process flow
A discrete event simulation model requires a defined “process flow,” which we refer to here as a simulated NICU (Figure 1). The objects or entities that flow through the simulated NICU represent babies (“simulated patients”).
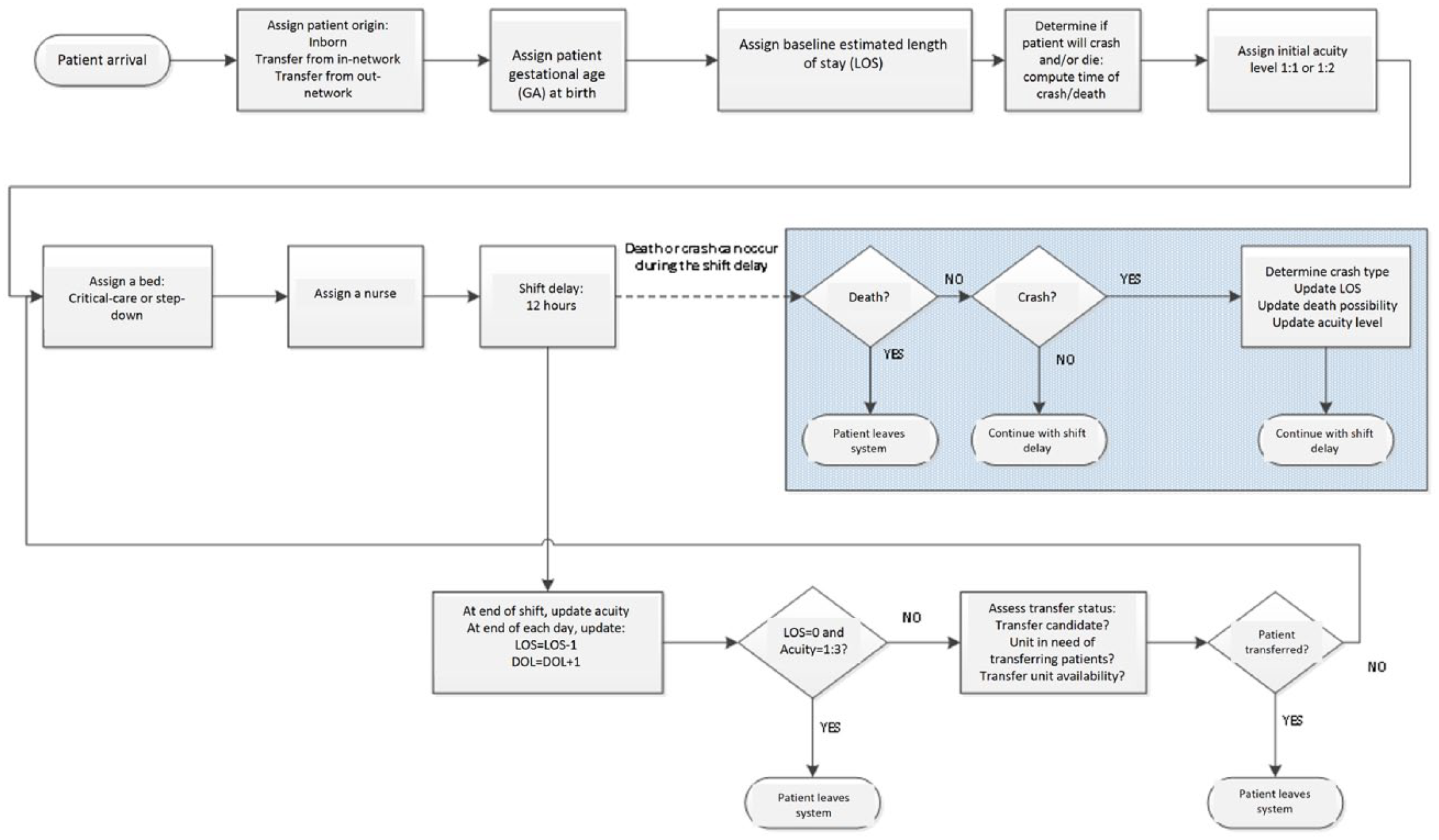
Final process map: simulated patients in the NICU.
The number of simulated patients admitted to the simulated NICU each day was sampled from a probability distribution based on the hospital’s historical data. We distributed the arrival times of the simulated patients uniformly across a 24-h interval. Each arriving simulated patient was randomly assigned one of the three admission types: inborn (same NICU; 66%), outborn-in-network (different in-network birthplace, transferred into the NICU; 9%), or outborn-out-network (different out-of-network birthplace, transferred into the NICU; 25%). We created different admission types for inborn and outborn babies because their admission frequencies differ in this setting and could influence strategic staffing decisions.
Based on admission type, simulated patients were assigned a gestational age (number of weeks (range: 22–42 weeks) of gestation completed before birth), which was randomly sampled from an empirical distribution. Gestational age is a critical determinant of length of stay (LOS) in the NICU. Babies born close to term (40 weeks) with mild illnesses may be discharged home within days; however, babies born at 24 weeks have a mandatory stay of many weeks in any NICU, even in the absence of significant long-term medical problems of prematurity. Figure 2 shows the historical distribution of gestational age of inborn admissions.
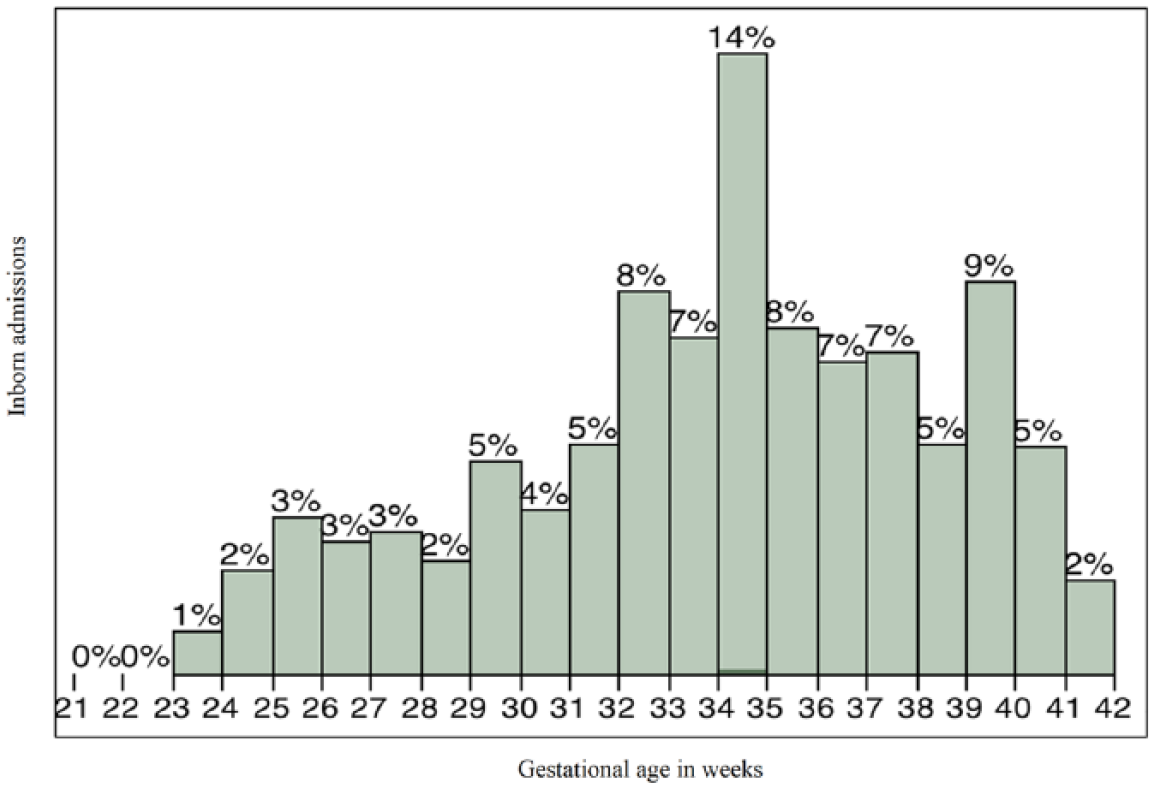
Gestational age (weeks) for inborn admissions (N = 3307).
Using gestational age, an estimated baseline length of stay (bLOS) in days for each simulated patient was calculated as follows: if gestational age ⩽29 weeks, then bLOS = (37 − gestational age) × 7 days; if 29 ⩽ gestational age ⩽ 33 weeks, then bLOS = (35 − gestational age) × 7 days; if gestational age >33 weeks, then bLOS = 14 days.
Gestational age also determined the simulated patient’s initial acuity level, defined as nurse:patient ratios of 1:1, 1:2, and 1:3. A 1:1 simulated patient was the only patient a nurse cared for during that shift; a 1:3 simulated patient could be assigned to a nurse who had two other 1:3 patients or one additional 1:2 patient, and so on. If gestational age was <28 weeks, then the simulated patient’s initial acuity level = 1:1; if gestational age was 28–38 weeks, acuity = 1:2; if gestational age was ⩾ 39 weeks, there was a 50% chance that acuity = 1:1; otherwise, acuity = 1:2.
Based on acuity level, each simulated patient was assigned either a “critical-care” or “step-down” bed. As in our NICU, the simulated NICU included 47 critical-care and 21 intermediate-level beds. Critical-care beds allowed patients assigned 1:1, 1:2, or 1:3 ratios; step-down beds allowed only patients assigned a 1:3 ratio.
Within the simulated NICU, shift changes occurred every 12 h to simulate our actual NICU shift changes at 07:00 and 19:00. Because admission, crash (acute decompensation in clinical status), and death could occur at any point during a nursing shift, each simulated patient’s acuity was recalculated at the end of each shift. At that time, patients could remain in their current status, transfer out of the unit, or move between critical-care and step-down beds in either direction, as appropriate. Based on recalculated acuity, nurses were likewise reassigned every 12 h to only as many patients as appropriate for the acuity level of his or her most acute simulated patient.
bLOS was updated according to five major morbidities that influence NICU LOS: (1) sepsis (infection in the bloodstream), (2) necrotizing enterocolitis (a deadly neonatal intestinal condition), (3) patent ductus arteriosus (a neonatal cardiovascular anomaly requiring medical or surgical intervention), (4) retinopathy of prematurity (a condition affecting blood vessels in the eye that leads to blindness if not promptly treated), and (5) intraventricular hemorrhage (bleeding into the internal structures of the brain). Because the probability of each condition varies by gestational age, 8 separate empirical distributions were estimated and sampled to determine which simulated patients would experience one or more of these conditions, and how LOS, acuity, and potential for mortality would be affected. For example, necrotizing enterocolitis is several times more likely in infants born at 24 versus 28 weeks and effectively absent in infants born close to term. Table 1 describes the effects of each morbidity on outcomes. The LOS effect data in Table 1 are based on a multivariate regression model using internal data incorporating each morbidity into the model in addition to gestational age, while temporal effect on acuity was based on subject-matter experts from neonatologists at the authors’ institution.
Defining morbidities for individual patients in the discrete event simulation model.
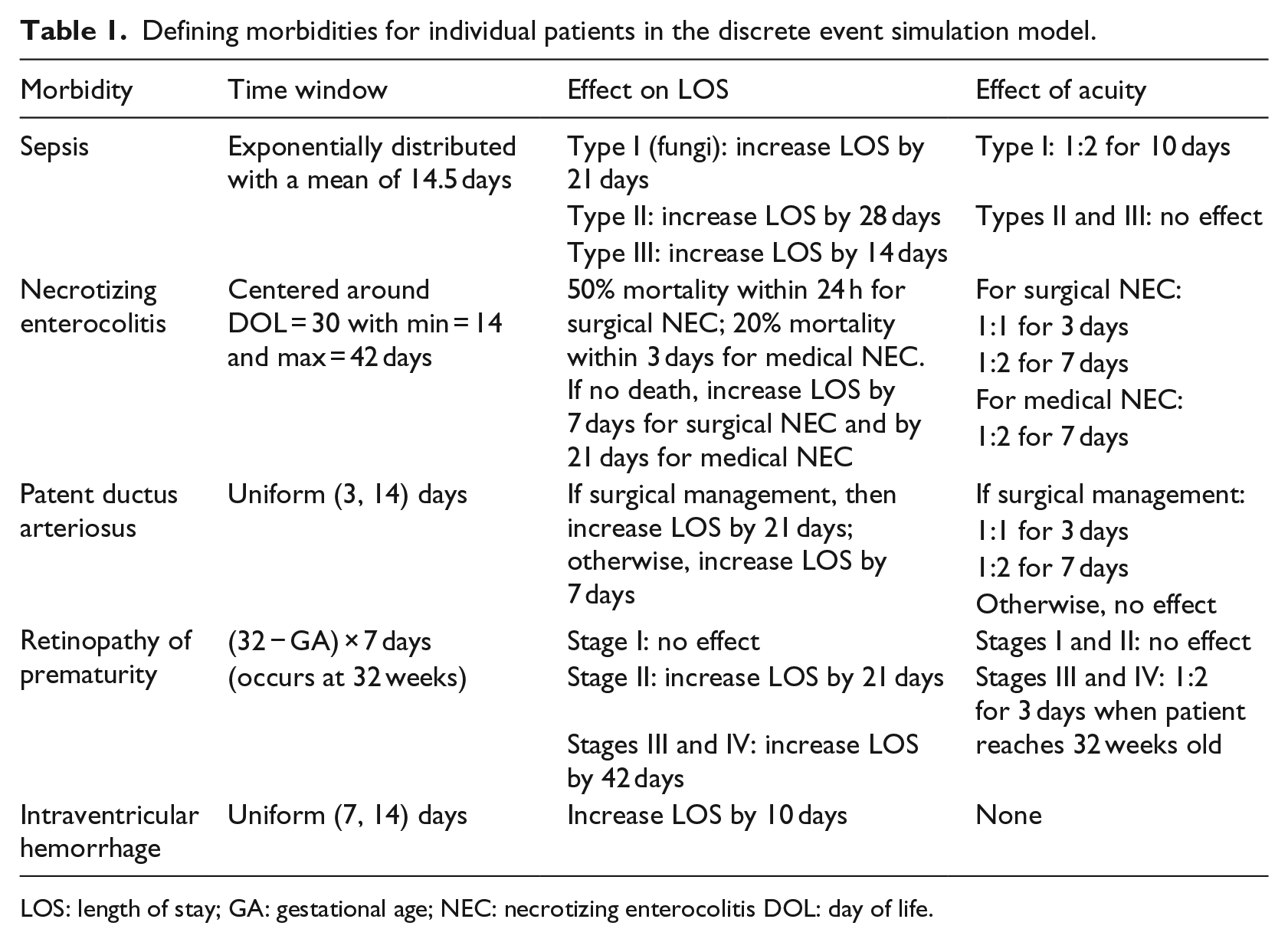
LOS: length of stay; GA: gestational age; NEC: necrotizing enterocolitis DOL: day of life.
The anonymized admission and morbidity distributions predefined each simulated patient’s entire course of admission to departure from the simulated NICU. This included whether and at what hour a crash would occur, shift-to-shift variations in acuity, and ultimate LOS, which was reduced by 1 day at the end of each simulated day. A simulated patient exited the simulated NICU by (1) succumbing to a morbidity identified on admission, (2) dying from another aspect of prematurity not crash-related, or (3) surviving to discharge or transfer.
Criteria for discharge were acuity = 1:3 and LOS = 0; indicating that the infant needs less care and is getting healthier. Discharge could occur at any shift change when a simulated patient met these criteria. Criteria for transfers included benchmarks for gestational age, days of life (days since birth), and postmenstrual age (gestational age × 7 + days of life). Postmenstrual age is the time elapsed between the first day of the last normal menstrual period and the day of delivery. A transfer was calculated as follows: if gestational age <9 weeks, then postmenstrual age ⩾238 days, acuity = 1:3, and no retinopathy of prematurity; if 29 ⩽ gestational age ⩽ 31 weeks, then postmenstrual age ⩾224 days, acuity = 1:3, and no retinopathy of prematurity; if gestational age ⩾32 weeks, then days of life ⩾7, acuity = 1:3, and no retinopathy of prematurity. Transfers required simulated patients to meet individual criteria, the simulated NICU to meet census-based requirements for transfers, and a transfer bed to be available based on a probability distribution at the start of each day. At the end of a shift, if a simulated patient had not crashed and his or her postmenstrual age ⩾210 days, the acuity level was set to 1:3.
Data analysis
The process flow was translated into a discrete event simulation model using SAS Simulation Studio (SAS Institute Inc., Cary, NC, 2013). Figure 3 shows a high-level view of the simulation model. The model collected the following information from de-identified, retrospective data from <<blinded>> Hospital, using a simulation run-length of 1 year: (1) number of admissions, (2) number of admissions with gestational age <28 weeks, (3) number of transferred patients, and (4) number of patients who died. Additional information included average daily census, average LOS of all patients, and average LOS for those whose gestational age was <28 weeks. This information was estimated using steady-state analysis methods along with statistically valid confidence intervals.
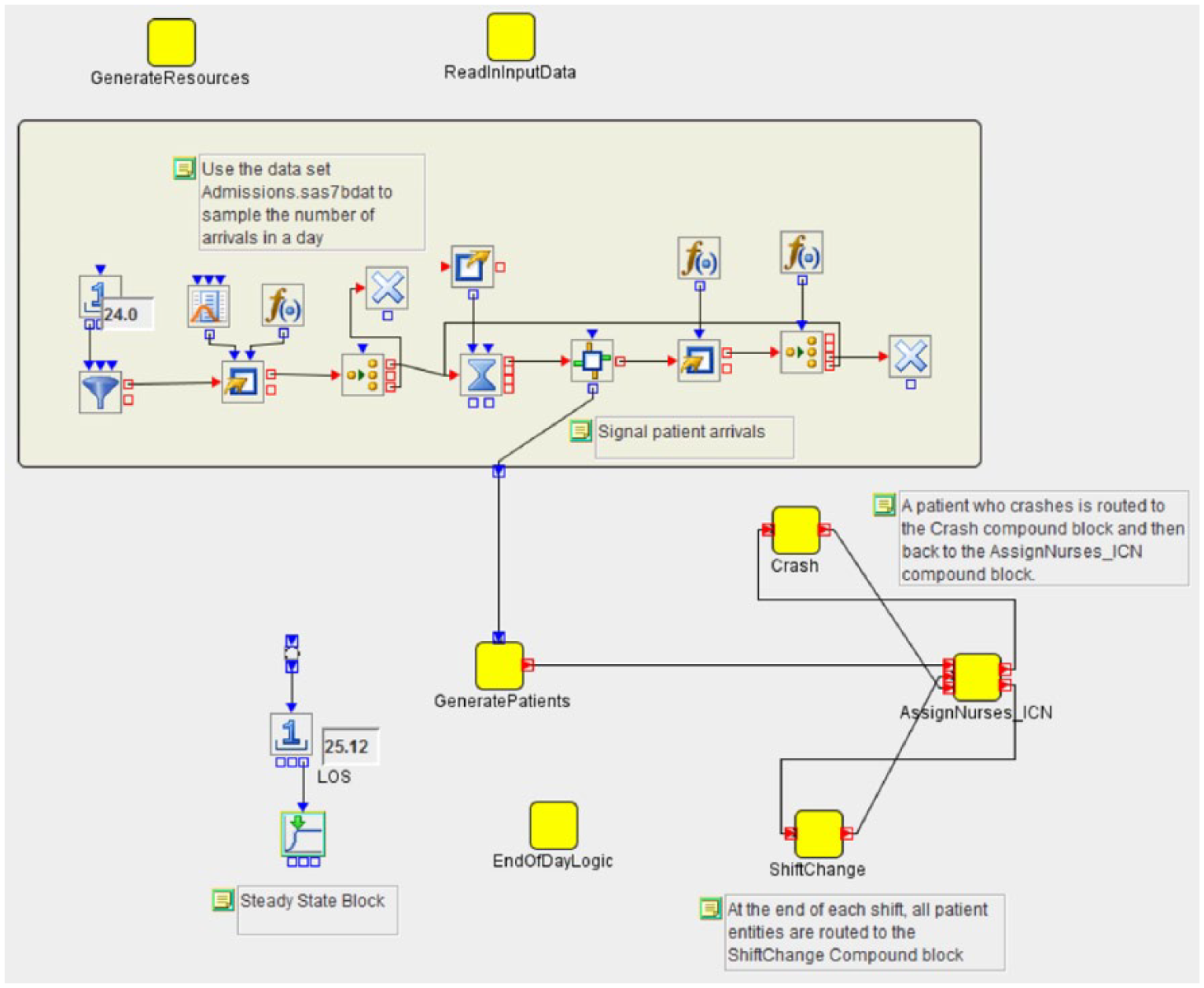
SAS Simulation Studio model. 9
System “warm-up”
It is typically difficult to start a simulation in steady-state operation in which there is no change in the data. This would require knowing exactly what steady-state behavior looks like ahead of time so that the model could be prepopulated with an appropriate number of patients in various states. Thus, the model was started empty and idle with no patients in the system. Patients were then added to the system over time until, as determined by a statistical test for randomness in the output data, we could verify whether it had reached steady-state operation in the simulation. When the end of this warm-up period was reached, only observations collected after the end of the warm-up period were included in the analysis to avoid biasing response point estimates. For nurses (n = 30), 64 simulation days were required to reach steady-state; however, we used a conservative warm-up length of 120 days and ran the simulation for 485 days.
Results
Model results
Results averaged from 50 independent replications of the simulated NICU model were compared with averaged actual hospital NICU data from 2008 to 2013. During this period, our actual NICU was generally staffed with 26–28 nurses per shift. Consequently, we staffed our simulated NICU with 26, 27, and 28 nurses per shift; the results for 26 and 28 nurses are shown in Table 2. The 28-nurse model demonstrates how the addition of two nurses per shift would allow for an increased daily census of approximately 4 patients (56.5–60.3) and an increased yearly census of approximately 45 patients (824–870). Adding two nurses per shift would allow for an additional five to six patients with a gestational age <28 weeks per year (126.7–132.6). Although one might expect that more infants with a gestational age <28 weeks might increase LOS due to their acuity, the model showed that this would have no effect on average LOS (25.4–26.0 weeks, overlapping confidence intervals).
Discrete event simulation model predictions versus actual data from <<blinded>> NICU, 2008–2013.
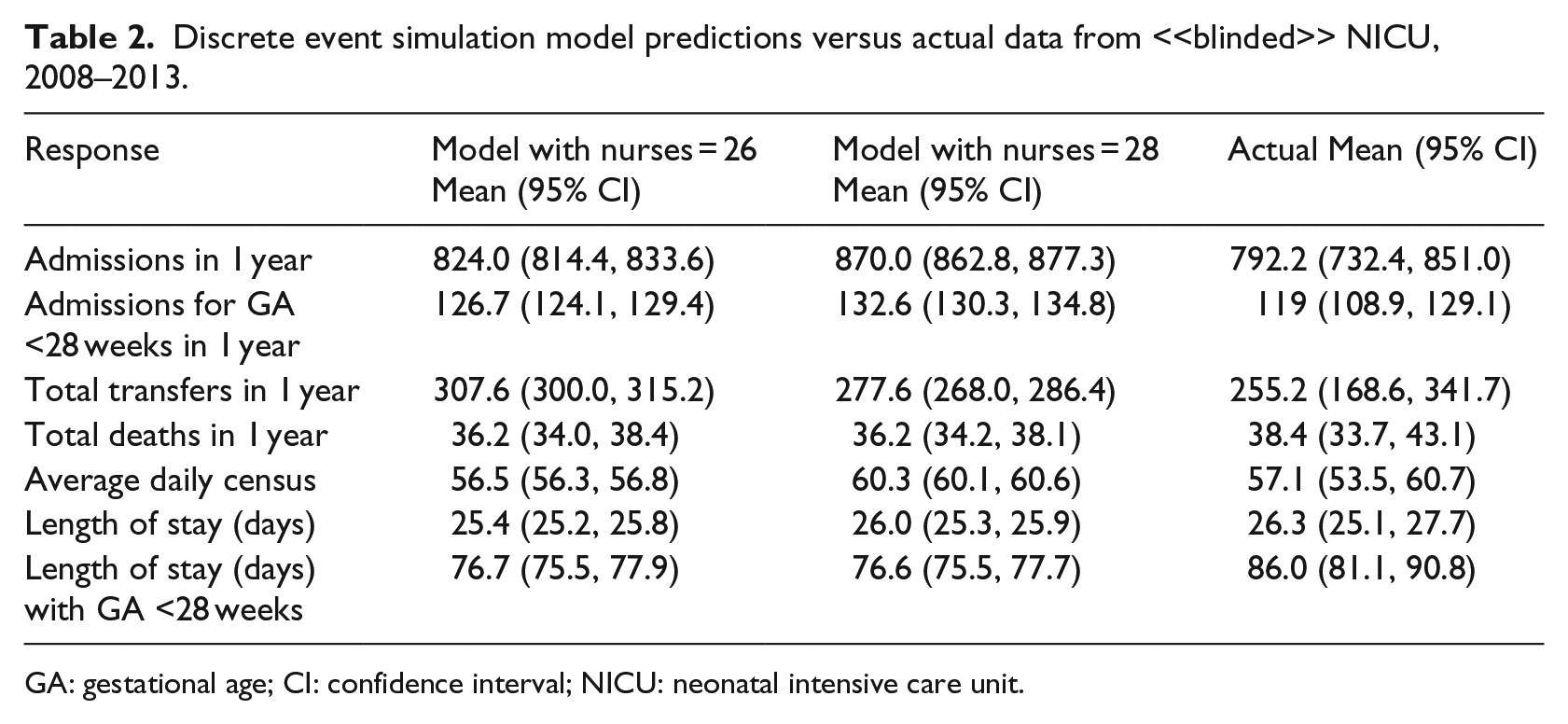
GA: gestational age; CI: confidence interval; NICU: neonatal intensive care unit.
Discussion
These results demonstrate that it is possible to develop a simulation that managers and administrators might use when staffing a hospital unit. The model demonstrates an accurate method of simulating patient mix, patient acuity, staffing needs, and costs in the present state, and it also forecasts how changes in a unit’s staffing, referral patterns, or patient mix would affect a unit in a future state. Our results demonstrated the validity of the present-state and future-state modeling as reflected in the actual data from the unit’s real patients over a 66-month period.
Although present-state models alone have significant value for hospital administrators to estimate staffing needs, accurately modeling future states is even more valuable. This value, in addition to a unit’s proprietary cost information, becomes clear as management contemplates the return on investment for additional hiring per added patient and staffing the unit for growth. For example, managers could calculate how many nurses to hire for each bed added during a growth phase, or how variations in bed allocation (step-down versus critical-care) would affect the flow of patients over a week, month, year, or decade. Furthermore, this tool could be valuable in situations of uncertain staffing needs (e.g. disaster responsiveness, mass casualty, flu outbreak).
Others have demonstrated the validity of analytical models for predicting hospital needs, including physician residents in surgical units, 5 LOS in geriatric adult patients with schizophrenia 6 and acute psychiatric hospitals, 7 hospital-associated mortality for Medicare patients, 10 bed unit utilization in obstetric hospitals, 11 and understanding patient characteristics in NICUs. 12 Our model expands these by including additional elements for predicting staffing needs such as patient acuity, referral patterns, and LOS. Furthermore, using probability distributions based on real data to simulate individual patients creates a more accurate and useful tool.
Like others, 13 we have demonstrated that predictive modeling can be refined and made more accurate by using factors tailored to a unit’s patient population, such as later-occurring morbidities. With modifications to input variables specific to the unit (e.g. patient age or morbidities), our model is easily transferable to other NICUs. More broadly, the discrete event simulation structure underlying this model could be used in any clinical setting that can access appropriate data: patient acuity; LOS; demographics; and historical admission, transfer, and discharge frequencies. Although the major drivers of admission, discharge, staffing, and LOS will differ by unit, data on these factors are likely immediately available to clinical managers.
Our next steps are to test this model in real time by comparing its forecasting with prospective hospital data. This will allow us to further validate our model and then translate it into practice for managers and administrators as a decision-making tool.
Limitations
This model is predicated on 66 unique past months of data. Policy changes may produce effects not captured in these findings. Furthermore, we have not yet implemented the model to obtain an understanding of its utility in a real-world planning environment. Nevertheless, this model may provide managers with better data with which to make staffing judgments.
Implications and conclusions
As medical technologies have advanced over time, patients of lower acuity who previously would have received inpatient medical care have been moved to outpatient settings. From 1980 to 2000, the average length of an inpatient hospital stay fell from 7.5 to 4.9 days. 14 Thus, patients who would have spent the early stages of their recovery in the hospital are now discharged to skilled nursing facilities or home. By implication, hospitals have a higher overall concentration of sick people who need more care and thus more overall staff and resources. To maintain standards of quality care and to prevent adverse events, adequate numbers of staff are critical. 15 This model could be used to investigate the effects of different treatment protocols for morbidities on patient outcomes in the NICU. While we presented an example from nurse staffing, this model can be easily altered to plan for other staff, including physicians, pharmacists, and social workers and to estimate physical needs and facilities support. Predicting and preparing for staffing levels and physical space needs are challenging and may impact patient outcomes, hiring, and capital expenses. Discrete event simulation modeling is a tool that can be applied to any hospital unit, empowering leadership to make more informed, data-driven staffing judgments and can give managers better operational awareness as the healthcare landscape changes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
