Abstract
There is a lack of research on the use of electronic tools that guide patients toward reducing their cardiovascular disease risk. We conducted a 9-month clinical trial in which participants who were at low (n = 100) and moderate (n = 23) cardiovascular disease risk—based on the National Cholesterol Education Program III’s 10-year risk estimator—were randomized to usual care or to usual care plus use of an Interactive Cholesterol Advisory Tool during the first 8 weeks of the study. In the moderate-risk category, an interaction between treatment condition and Framingham risk estimate on low-density lipoprotein and non-high-density lipoprotein cholesterol was observed, such that participants in the virtual clinician treatment condition had a larger reduction in low-density lipoprotein and non-high-density lipoprotein cholesterol as their Framingham risk estimate increased. Perceptions of the Interactive Cholesterol Advisory Tool were positive. Evidence-based information about cardiovascular disease risk and its management was accessible to participants without major technical challenges.
Keywords
Introduction
Cardiovascular disease (CVD) is the leading cause of death in the United States, and the total cost of CVD in 2009 was estimated to be more than US$475 billion. 1 Annually, more than 800,000 deaths from coronary heart disease occur in the United States, and 65.5 percent of sudden deaths from coronary artery disease occur without a prior diagnosis of coronary heart disease. 2 Coronary heart disease and atherosclerosis have long asymptomatic phases that offer opportunity for intervention to lower risk, and health behaviors and pharmacologic agents have important risk-reduction effects. 3 However, a large proportion of patients and medical providers do not take full advantage of these treatments, as there is a gap between treatment recommendations and actual clinical practice 4 although statin therapy has been shown to be safe and guidelines exist for its use. Indeed, there are many barriers that explain non-adherence to statin therapy5,6 and patients not initiating and maintaining healthy behaviors to lower CVD risk. 7
Recently, several important advances have been made that may support the ability of both patients and clinicians to lower CVD risk, including (1) categorization of patients according to whether or not a recommendation is made to take a statin medication; (2) improvement and validation of 10-year global risk estimators for CVD events, including myocardial infarction and stroke; (3) validation of the Self-Determination Theory Model of Health Behavior 8 and its application to outcomes that are related to CVD; and (4) improvements in virtual technology that enhance clinical reach in the population. However, little priority has been given to incorporating evidence-based approaches that are part of national guidelines in a tool that (1) patients find engaging and (2) can reduce practitioner burden, and few (if any) such tools exist for use by adults with no known history of CVD, its equivalent, or high CVD risk. We propose that patients can be engaged in interactive learning about CVD risk using virtual clinician (VC) avatars and that the online delivery of a standardized clinical experience can facilitate patients making therapeutic lifestyle changes and using medications to lower CVD risk as recommended by the American College of Cardiology and the American Heart Association. 9 Herein, we present results from a clinical trial that was designed to examine whether a VC tool that offers 10-year risk estimators for CVD events 10 can lead to reductions in low-density lipoprotein (LDL) and non-high-density lipoprotein (HDL) cholesterol.
Methods
Participants, study design, and conditions
We recruited 123 participants with no known history of CVD or its equivalent (including diabetes mellitus) for a study that examined whether health information and education about cholesterol can be delivered effectively online via computer, which we refer to as the Interactive Cholesterol Advisory Tool (ICAT). The focus of the ICAT is on achieving healthy cholesterol goals and their maintenance10,11 through therapeutic lifestyle change and statin medication use. The ICAT is a consumer-facing, web-based application that provides medical information about CVD and its subsequent events, risk assessment, and therapeutic plan-building within an interactive environment designed to facilitate optimal motivation by supporting participants’ basic psychological needs for autonomy, competence, and relatedness. An avatar provides all medical information (with accompanying text) in the ICAT, and this information is evidence-based and interactive with content provided by the user, including personal preferences, cholesterol values, and other CVD risk factors. The ICAT consists of seven modules (viz. introduction, therapeutic lifestyle, physical activity, medications, dietary supplements, omega-3 fatty acids, and weight loss), as well as a personalized plan for therapeutic lifestyle change and medication initiation and maintenance for participants to review and confirm with their primary care practitioner. In sum, the ICAT was designed for use by medical providers to assist in offering to patients consistent, evidence-based information in lieu of the time required to provide such information in a face-to-face setting.
Participants were recruited using signs in physicians’ offices and using advertisements in the community, as well as at biometric screening programs for University of Rochester employees. Eligible participants were at least 18 years of age and fluent in English, willing to complete informed consent and attend five visits over 9 months, and at low or at moderate CVD risk based on the National Cholesterol Education Program III’s 10-year risk estimator. Participants were excluded for any of the following reasons: on a statin medication at the time of study participation; terminal illness; pregnant or lactating; contraindications for statin medications; triglyceride levels above 500 mg/dL; secondary cause of hypercholesterolemia that is uncontrolled (e.g. diabetes mellitus, hypothyroidism); and diagnosis of CVD, CVD equivalent (including diabetes mellitus, peripheral vascular disease, abdominal aortic aneurysm, and cerebral vascular disease), liver disease, or psychosis. This study was approved by the University of Rochester Human Subjects Review Board, and it conforms to the principles embodied in the Declaration of Helsinki as well as the International Ethical Guidelines for Biomedical Research Involving Human Subjects and the International Guidelines for Ethical Review for Epidemiological Studies. Its timeline is depicted in Figure 1.
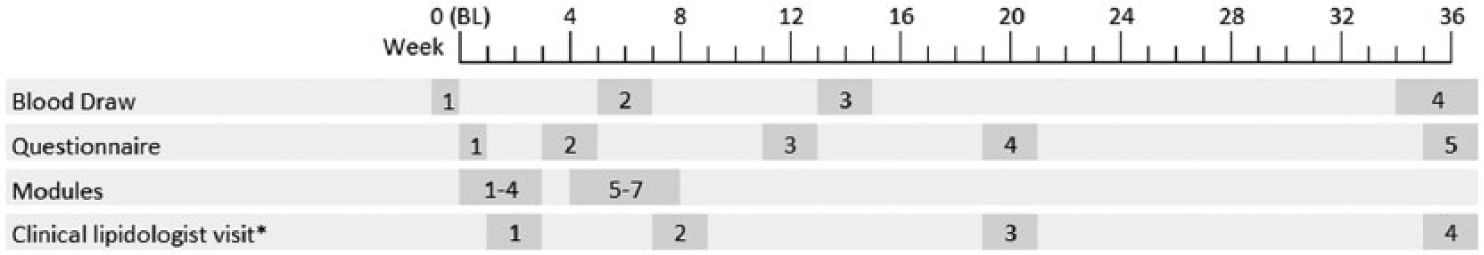
Study timeline.
Participants who were at low CVD risk (n = 100; defined as having a 10-year Framingham risk estimate for CVD events less than 10%) 10 were randomized either to usual care (n = 50) in which they received their fasting lipid profile and a recommendation to see their primary care practitioner to discuss the results, or to usual care plus completion of seven VC modules (n = 50) during the first 8 weeks of the study. Participants who were at moderate CVD risk (n = 23; defined as having a 10-year Framingham risk estimate for CVD events from 10% to 20%) saw a board-certified lipidologist for initial consultation (50 min); had four 30-min follow-up visits at weeks 6, 12, 20, and 36 for assessment and management of lipids; and were randomized to completion of seven VC modules (n = 11) during the first 8 weeks of the study, or to no completion of VC modules (n = 12). In line with the Self-Determination Theory Model of Health Behavior, 8 the lipidologists were trained to provide support for autonomy, competence, and relatedness around lifestyle change and medication use in order to lower LDL cholesterol to less than 100 mg/dL during the 9-month study period. All randomization was done using a random number generator. Each randomization block consisted of 10 participants, 5 of whom were randomized to the VC condition and 5 of whom were randomized to the non-VC condition. Adherence to completion of VC modules was tracked electronically and non-adherent participants were encouraged to complete the VC modules.
Regardless of CVD risk category and treatment condition, all participants were informed of their 10-year Framingham risk estimate and given the recommendation that their optimal target for LDL cholesterol was less than 100 mg/dL. As well, all participants were encouraged to meet with their primary care practitioner, if desired, and were encouraged to have the clinical notes from the lipidologist sent to the primary care practitioner. Participants who were at moderate risk were told that therapeutic lifestyle changes, dietary changes, and statin medication use can lower LDL cholesterol and risk of heart attack, stroke, and premature death, and they were given the recommendation to lower their LDL cholesterol in order to reduce their CVD risk. All participants completed a set of questionnaires that assessed demographic information and constructs that are relevant to the Self-Determination Theory Model of Health Behavior. 8
Analytic overview
Baseline characteristics of the study participants were described according to CVD risk category (low risk vs moderate risk) and further stratified according to treatment condition (usual care vs usual care plus completion of VC modules) using relative frequencies for categorical variables and using means and standard deviations (SDs) for continuous variables. Homogeneity was tested between treatment conditions within the low-risk and moderate-risk categories using χ 2 tests for differences in proportions, and using generalized linear models for differences in means. Two-sided p values for comparisons with expected counts less than 5 were calculated using Fisher’s Exact Test.
Measures of LDL and non-HDL cholesterol were plotted as mean and SD at each time point for the total sample and stratified according to CVD risk category and treatment condition. Mean levels of cholesterol at each time point (relative to baseline) were compared within treatment conditions using paired-samples t-tests. Mean levels of cholesterol at each time point were compared between treatment conditions using independent samples t-tests. A sensitivity analysis using generalized estimating equations was conducted to model the longitudinal effect of the VC (relative to non-VC) on LDL and non-HDL cholesterol during the 9-month study period and to assess whether the 10-year Framingham risk estimate 10 moderates this association. Partial correlations (controlling for LDL and non-HDL cholesterol at baseline) were computed to assess the longitudinal associations among Framingham risk estimate and LDL and non-HDL cholesterol at weeks 6, 14, and 35.
Participants who were at low risk reported whether or not they discussed their cholesterol with their primary care practitioner, and the frequency of and mean time (in minutes) spent having these discussions were compared between treatment conditions using χ2 tests and independent samples t-tests, respectively. Self-reported statin medication use was compared between treatment conditions within the low-risk and moderate-risk categories using Fisher’s Exact Test. Mean levels of dietary activity and physical activity at each time point were tested for linear trends within treatment conditions during (1) the time at which the ICAT was administered (i.e. from baseline to 3 months) and (2) the time period of the study (i.e. from baseline to 9 months). Finally, several questions were asked that assessed participants’ perceptions of the ICAT.
All analyses were conducted using SAS version 9.3 (SAS Institute, Cary, NC, USA), and p values less than 0.05 were considered to be statistically significant.
Results
Baseline characteristics of the study participants are shown in Table 1. Participants who were at low risk had a mean age of 47.6 years (SD = 12.6 years). A majority of these participants were females (71.0%), Caucasian (84.0%), married (65.0%), employed (83.0%), and at least college educated (82.0%). A small percentage of these participants had hypertension (12.0%), while a majority had high cholesterol (LDL cholesterol ≥100 mg/dL) (60.0%). One difference (in amount of education; p = 0.01) was noted between treatment conditions in the low-risk category. Participants who were at moderate risk had a mean age of 55.4 years (SD = 9.1 years). A majority of these participants were males (65.2%), Caucasian (78.3%), not married (56.5%), employed (60.9%), and at least college educated (60.9%). A majority of these participants had hypertension (60.9%) and high cholesterol (69.6%). One difference (in amount of education; p = 0.08) was noted between treatment conditions in the moderate-risk category.
Baseline characteristics of the study participants.
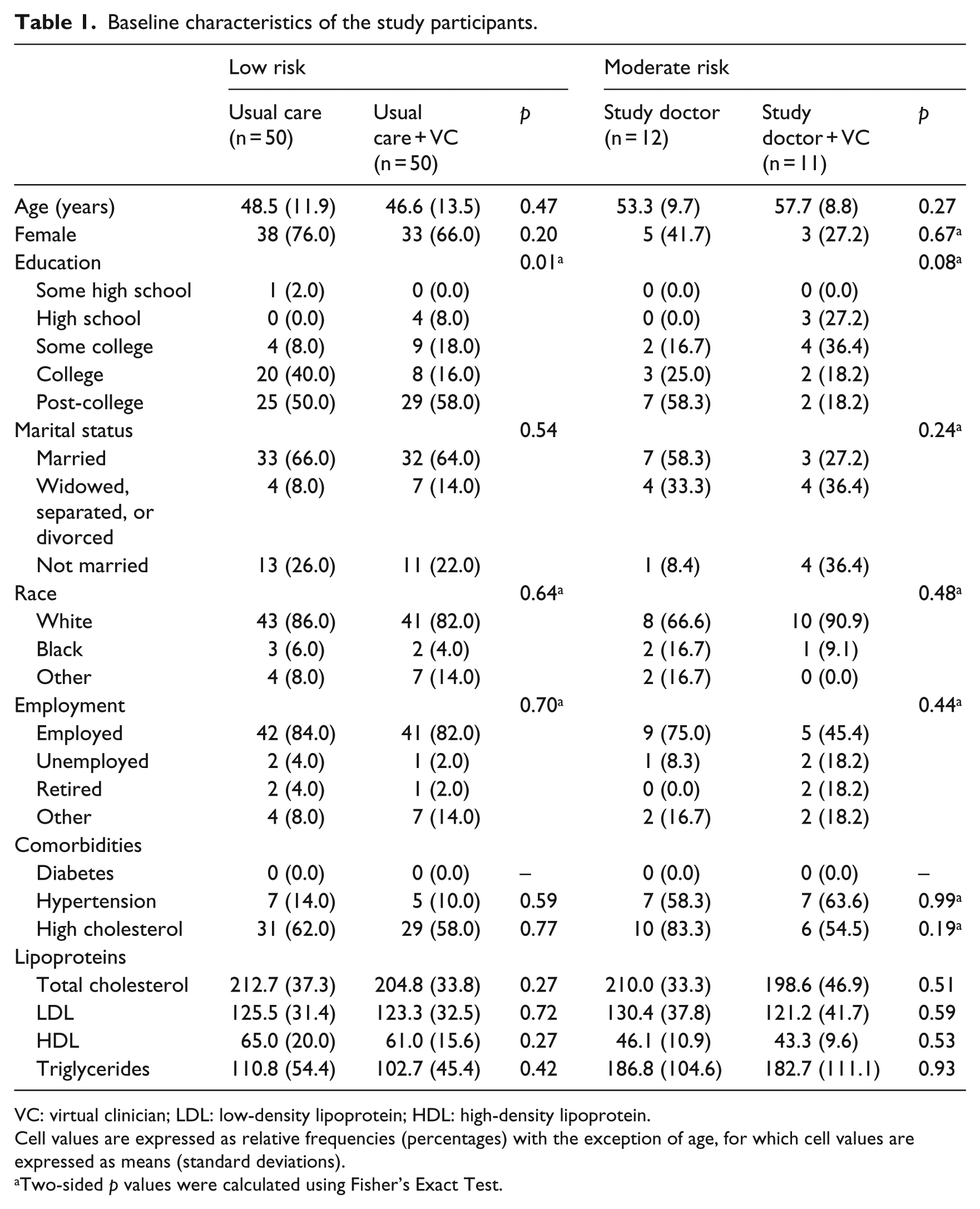
VC: virtual clinician; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Cell values are expressed as relative frequencies (percentages) with the exception of age, for which cell values are expressed as means (standard deviations).
Two-sided p values were calculated using Fisher’s Exact Test.
Changes in LDL and non-HDL cholesterol are shown in Table 2. Collapsed across risk categories (i.e. total sample), participants in the non-VC treatment condition had reductions in LDL cholesterol at weeks 6 (p = 0.05) and 14 (p = 0.03) after baseline, along with reductions in non-HDL cholesterol at weeks 6 (p = 0.03), 14 (p = 0.01), and 35 (p = 0.06) after baseline. No changes were noted among participants in the VC treatment condition, and no mean differences were noted between the non-VC and VC treatment conditions in the total sample. In the low-risk category, participants in the usual care treatment condition had a reduction in LDL cholesterol at week 6 (p = 0.09) after baseline, along with a reduction in non-HDL cholesterol at week 6 (p = 0.07) after baseline. Participants in the usual care plus VC treatment condition had an increase in non-HDL cholesterol at week 14 (p = 0.10) after baseline. No mean differences were noted between the usual care and usual care plus VC treatment conditions in the low-risk category. In the moderate-risk category, participants in the study doctor treatment condition had reductions in LDL cholesterol at weeks 14 (p = 0.07) and 35 (p = 0.03) after baseline, along with reductions in non-HDL cholesterol at weeks 14 (p = 0.03) and 35 (p = 0.03) after baseline. No changes were noted among participants in the study doctor plus VC treatment condition, and no mean differences were noted between the study doctor and study doctor plus VC treatment conditions in the moderate-risk category.
Measures of low-density lipoprotein and non-high-density lipoprotein cholesterol at each time point.
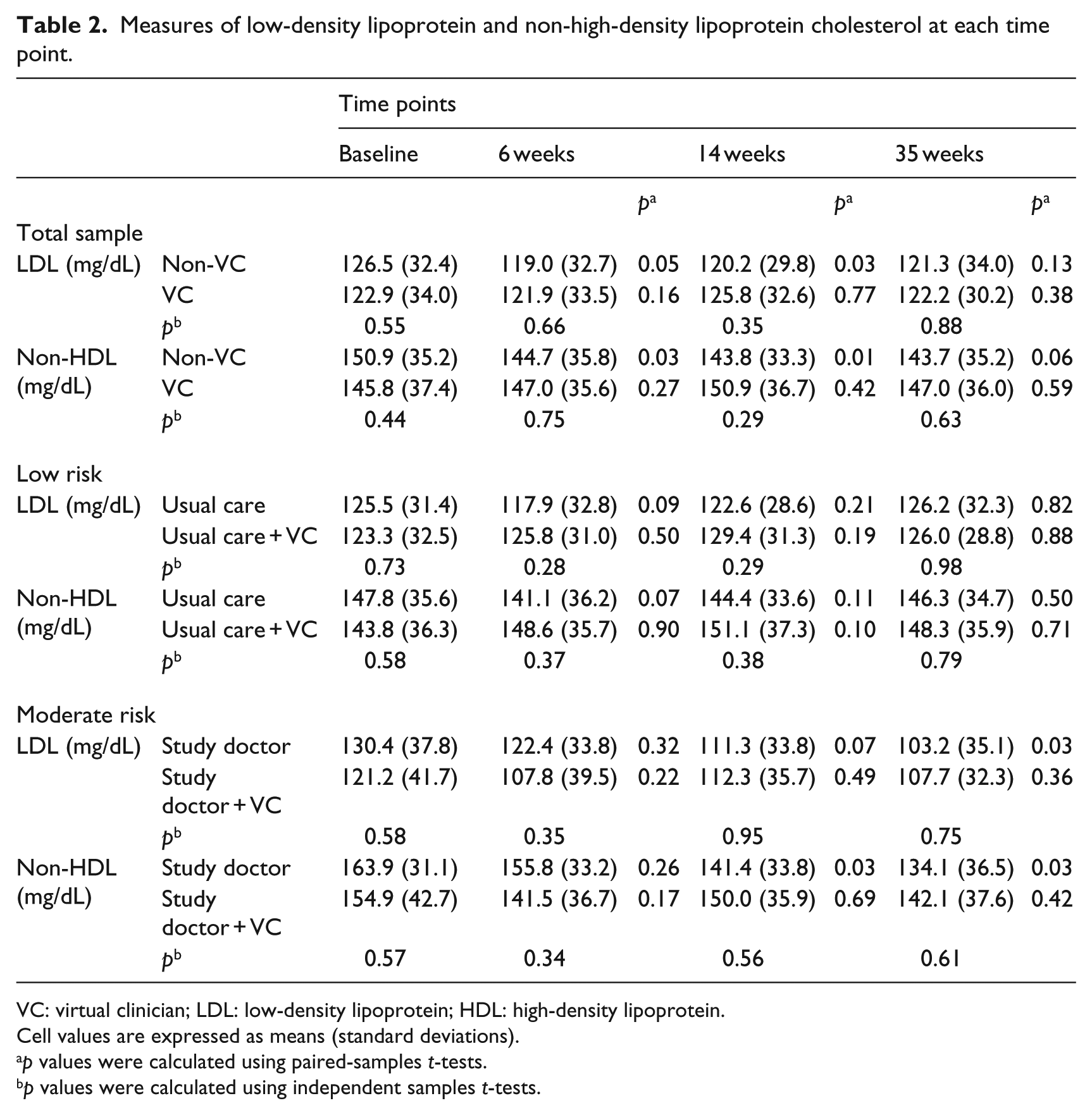
VC: virtual clinician; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Cell values are expressed as means (standard deviations).
p values were calculated using paired-samples t-tests.
p values were calculated using independent samples t-tests.
We conducted a sensitivity analysis using generalized estimating equations to assess whether the 10-year Framingham risk estimate moderates the longitudinal effect of the VC (relative to non-VC) on LDL and non-HDL cholesterol during the 9-month study period. In the total sample, there were no differences in LDL and non-HDL cholesterol during the 9-month study period among participants in the non-VC and VC treatment conditions after controlling for the interaction between treatment condition and Framingham risk estimate. Stratification by risk category revealed that these (null) findings were due to a lack of effect in the low-risk category. In the moderate-risk category, participants in the study doctor plus VC treatment condition (relative to study doctor) had reductions in LDL (p = 0.05) and non-HDL (p = 0.02) cholesterol during the 9-month study period after controlling for the interaction between treatment condition and Framingham risk estimate. For every 1 percent increase in the Framingham risk estimate, 10 on average there was a 2.9- and a 2.6-point reduction in LDL (p = 0.08) and non-HDL (p = 0.07) cholesterol, respectively. Of importance, there was an interaction between treatment condition and Framingham risk estimate on LDL (p = 0.04) and non-HDL (p = 0.02) cholesterol in the moderate-risk category, suggesting that participants in the VC treatment condition (relative to non-VC) had a larger reduction in LDL and non-HDL cholesterol as their Framingham risk estimate increased.
Partial correlations were computed between Framingham risk estimate and LDL and non-HDL cholesterol at weeks 6, 14, and 35 (controlling for LDL and non-HDL cholesterol at baseline) in order to understand better the associations among these variables. In the low-risk category, participants in the usual care plus VC treatment condition had positive partial correlations between Framingham risk estimate and LDL cholesterol at weeks 6 (p < 0.01) and 35 (p = 0.01), along with a positive partial correlation between Framingham risk estimate and non-HDL cholesterol at week 6 (p < 0.01). No partial correlations were noted among participants in the usual care treatment condition. In the moderate-risk category, participants in the study doctor plus VC treatment condition had a positive partial correlation between Framingham risk estimate and LDL cholesterol at week 6 (p = 0.07), along with a positive partial correlation between Framingham risk estimate and non-HDL cholesterol at week 6 (p = 0.08). No partial correlations were noted among participants in the study doctor treatment condition.
Among participants who were at low risk, the frequency of and mean time (in minutes) spent having discussions about cholesterol with a primary care practitioner are shown in Table 3. There were increases in the frequency of these discussions among participants in the usual care (p < 0.01) and the usual care plus VC (p < 0.01) treatment conditions from 1 to 9 months. No mean differences (in frequency) were noted between the usual care and usual care plus VC treatment conditions. In contrast, there were no increases in the time spent having these discussions from 1 to 9 months, and one difference (in time) was noted between the usual care and usual care plus VC treatment conditions at 1 month (p = 0.08).
Discussion of cholesterol with primary care practitioner.
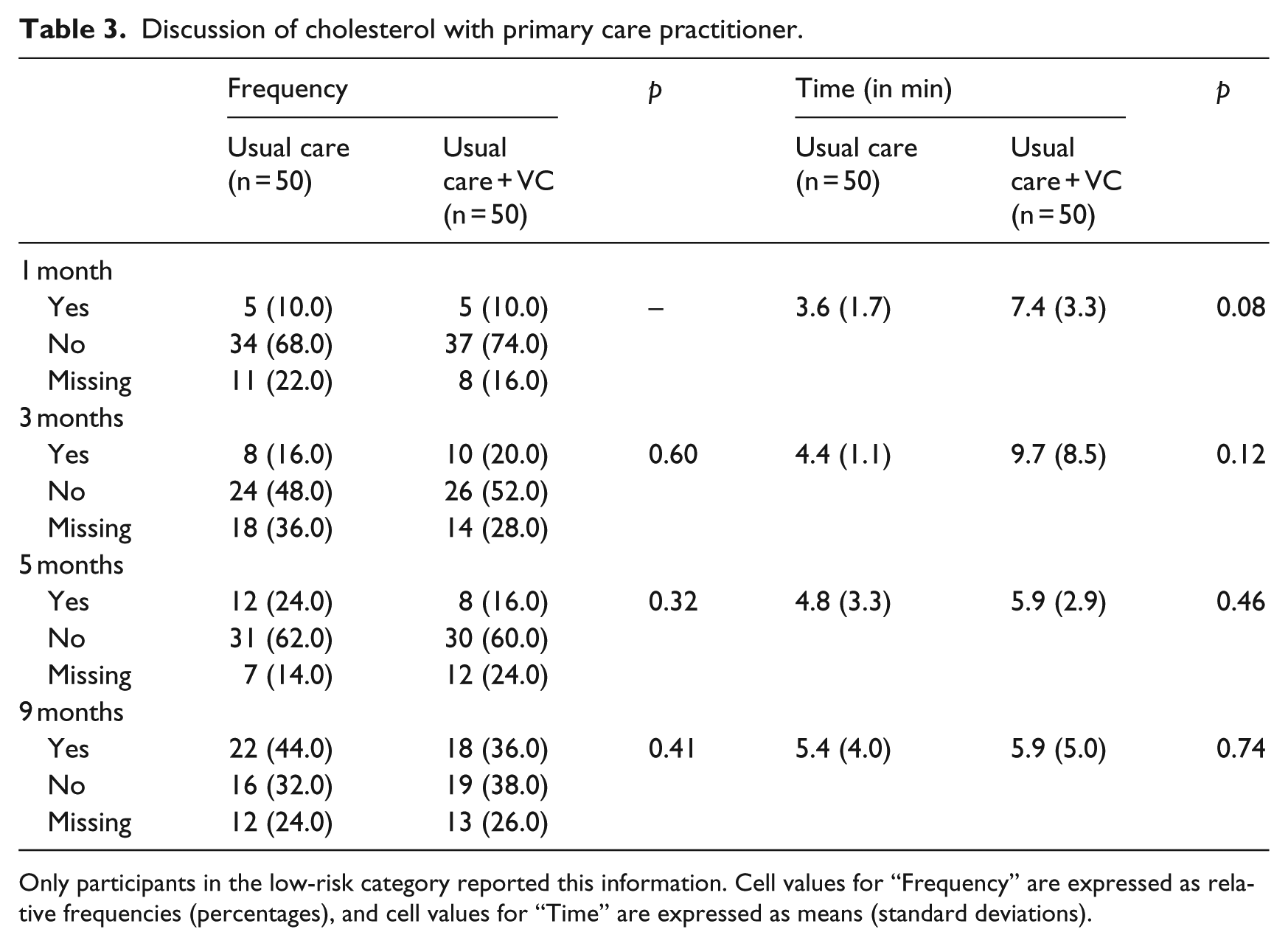
Only participants in the low-risk category reported this information. Cell values for “Frequency” are expressed as relative frequencies (percentages), and cell values for “Time” are expressed as means (standard deviations).
Self-reported statin medication use is shown in Table 4. In the low-risk category, there were no increases in statin medication use from baseline to 9 months among participants in the usual care treatment condition or the usual care plus VC treatment condition. In the moderate-risk category, there was an increase in statin medication use from baseline to 9 months among participants in the study doctor treatment condition (p = 0.06), but not in the study doctor plus VC treatment condition. No mean differences were noted between the usual care and usual care plus VC treatment conditions (in the low-risk category) or between the study doctor and study doctor plus VC treatment conditions (in the moderate-risk category).
Self-reported statin medication use.
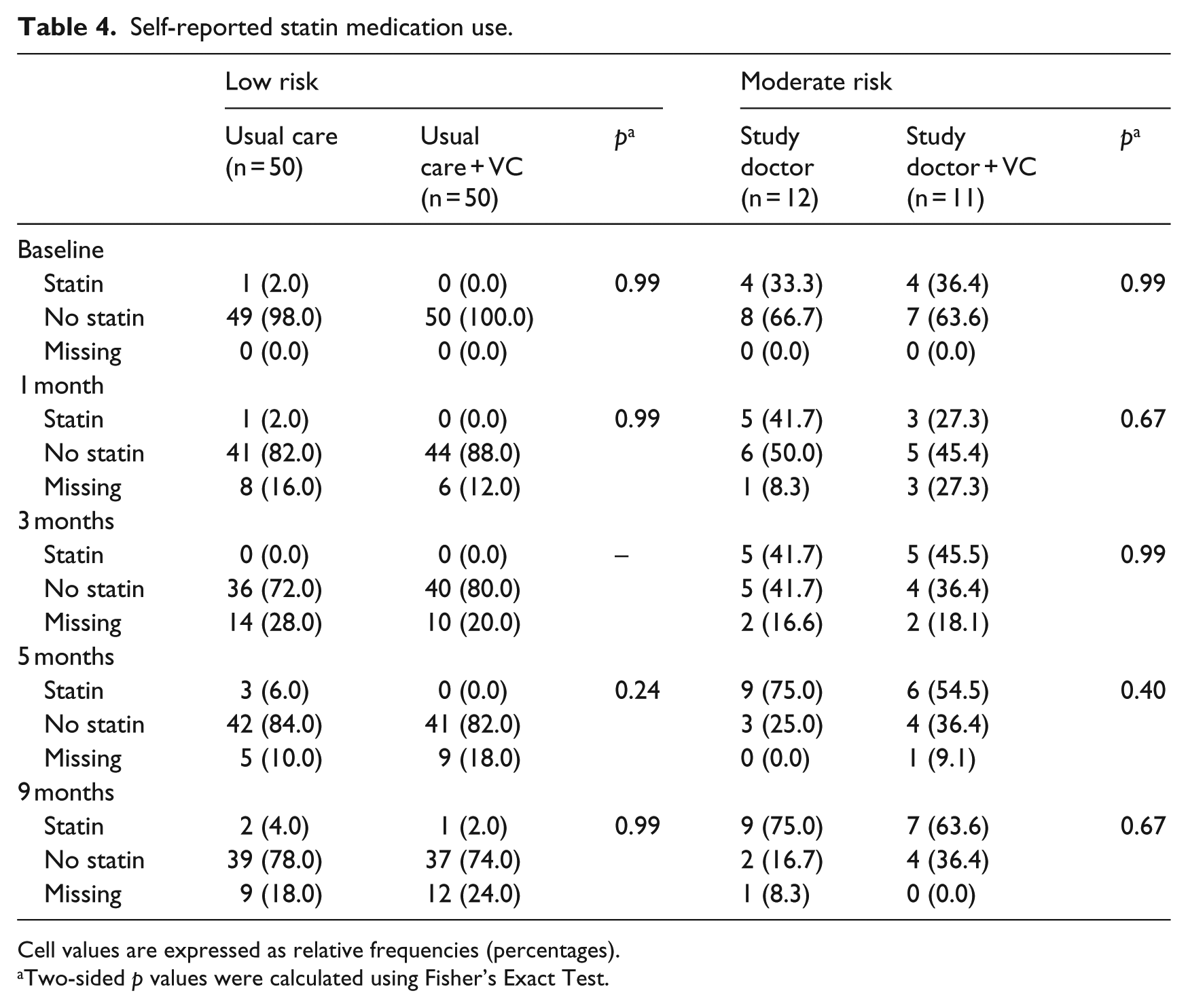
Cell values are expressed as relative frequencies (percentages).
Two-sided p values were calculated using Fisher’s Exact Test.
Changes in dietary activity and physical activity are shown in Tables 5 and 6, respectively. There were no increases in healthy dietary activity or healthy physical activity from baseline to 9 months. However, during the time at which the ICAT was administered (i.e. from baseline to 3 months), there were reductions in “saturated fat content” (p = 0.06) and “use (of) regular salad dressing and mayonnaise instead of low-fat or fat-free” (p = 0.08) among participants in the VC treatment condition. As well, there were reductions in “add butter, margarine or oil to bread, potatoes, rice or vegetables at the table” (p = 0.09) and “do less than 30 total minutes of physical activity 3 days a week or more” (p = 0.10) among participants in the non-VC treatment condition.
Measures of dietary activity at each time point.
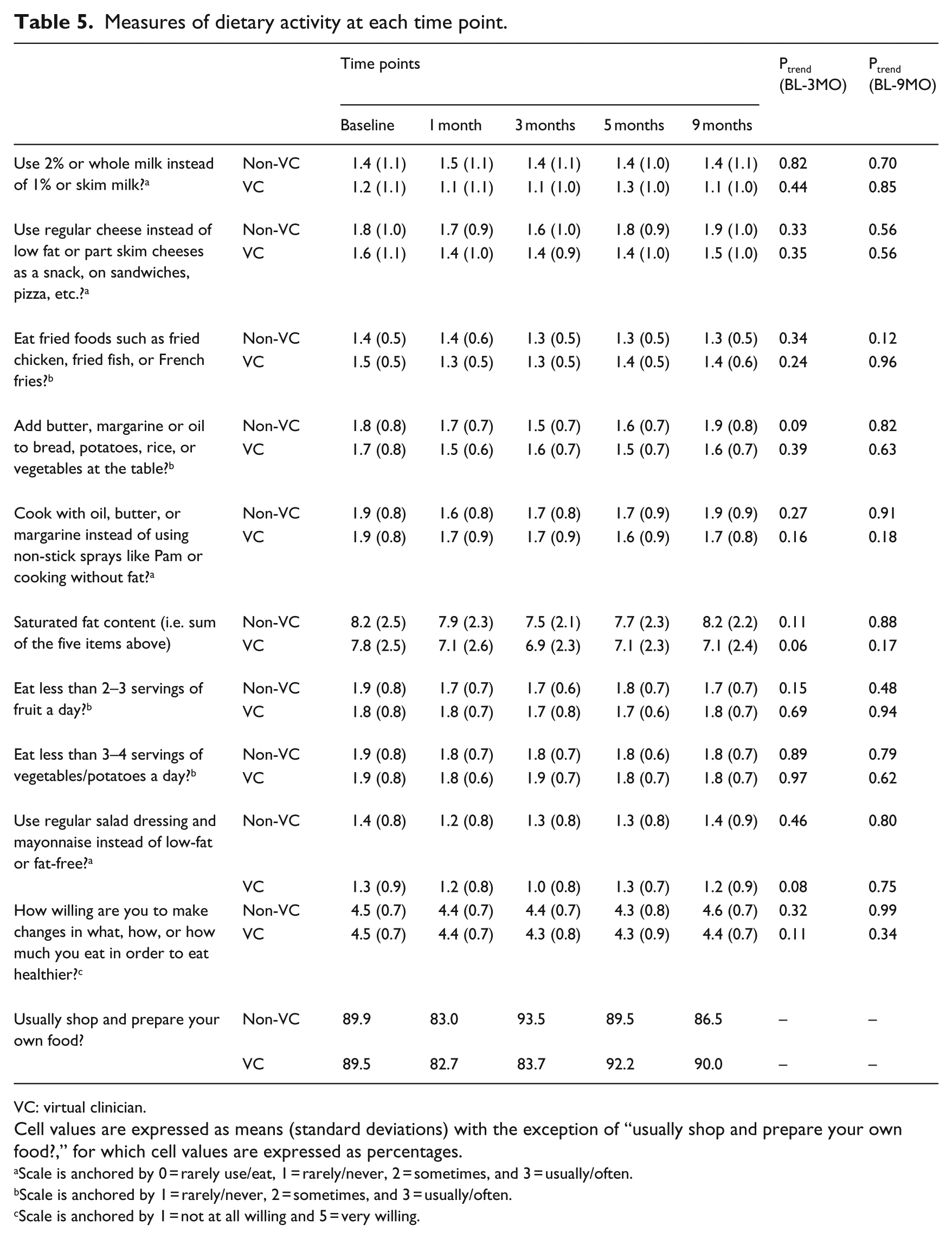
VC: virtual clinician.
Cell values are expressed as means (standard deviations) with the exception of “usually shop and prepare your own food?,” for which cell values are expressed as percentages.
Scale is anchored by 0 = rarely use/eat, 1 = rarely/never, 2 = sometimes, and 3 = usually/often.
Scale is anchored by 1 = rarely/never, 2 = sometimes, and 3 = usually/often.
Scale is anchored by 1 = not at all willing and 5 = very willing.
Measures of physical activity at each time point.
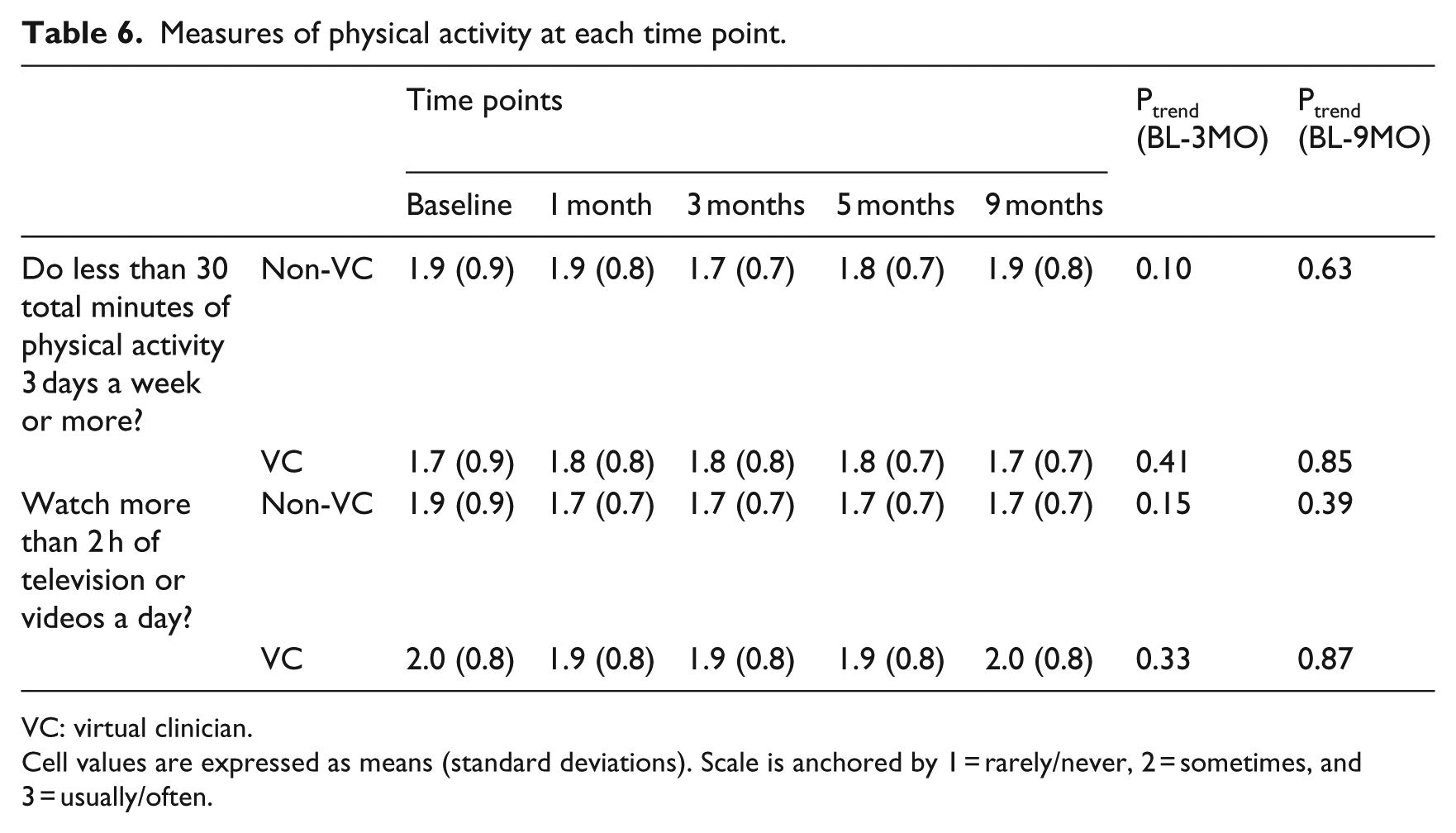
VC: virtual clinician.
Cell values are expressed as means (standard deviations). Scale is anchored by 1 = rarely/never, 2 = sometimes, and 3 = usually/often.
Study participants’ perceptions of the ICAT are shown in Table 7 and tended to be positive.
Study participants’ perceptions of the Interactive Cholesterol Advisory Tool (at 14 weeks).
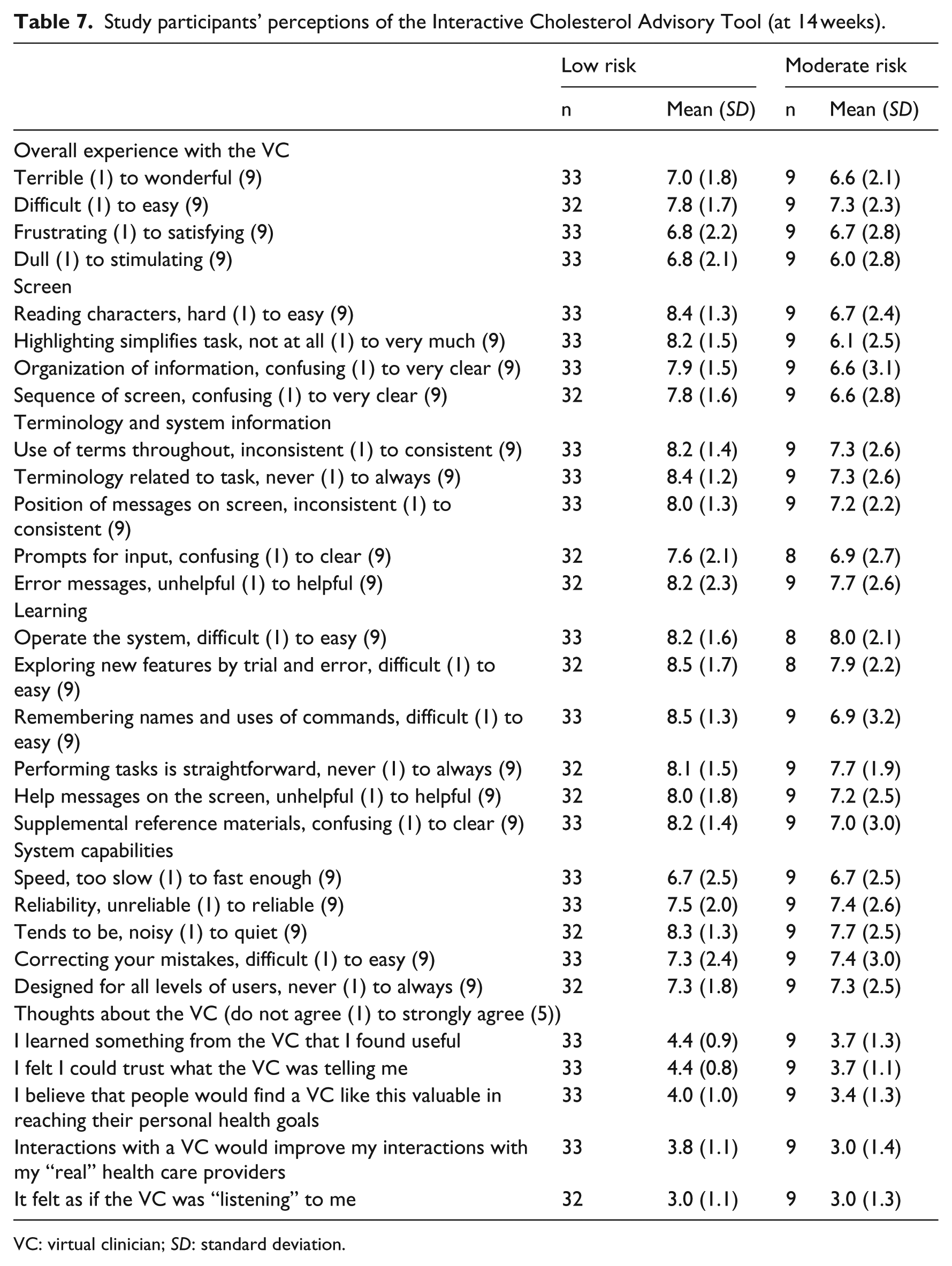
VC: virtual clinician; SD: standard deviation.
Discussion
In this clinical trial, we examined whether a computerized VC tool that offers 10-year risk estimators for CVD events, which we refer to as the ICAT, can lead to reductions in LDL and non-HDL cholesterol in adults with no known history of CVD, its equivalent, or high CVD risk. Also, we tested the effect of the ICAT on a variety of health-related behaviors and we assessed participants’ perceptions of the ICAT. To our knowledge, this study is the first to investigate the health effects of interactive virtual modules that provide education about cholesterol and CVD as well as personalized risk estimators for CVD events in adults with no known history of CVD, its equivalent, or high CVD risk.
In the total sample and in the moderate-risk category, there were reductions in LDL and non-HDL cholesterol among participants in the non-VC treatment condition but not among participants in the VC treatment condition. There were no reductions in LDL or non-HDL cholesterol among participants in the low-risk category. It is important to consider possible explanations for the lack of beneficial effect of the ICAT on LDL and non-HDL cholesterol. We believe that participants who were at low CVD risk—a majority of our sample—may have been unmotivated to make lifestyle changes and/or take statin medication after learning of their low CVD risk status. Although other explanations are likely to exist, this possibility warrants careful examination in light of the finding that the 10-year Framingham risk estimate moderated the longitudinal effect of the VC on LDL and non-HDL cholesterol in the moderate-risk category. That is to say, participants in the VC treatment condition (relative to non-VC) had a larger reduction in LDL and non-HDL cholesterol as their Framingham risk estimate increased. More research is needed to replicate and explore this finding further, yet for now this result speaks to the potential of the VC to lower LDL and non-HDL cholesterol among individuals who are at moderate risk for CVD (and who may be more motivated to make lifestyle changes and/or take statin medication). Indeed, we believe that this is the first demonstration that knowledge of CVD risk can lead to a reduction in LDL and non-HDL cholesterol among individuals who interact with a VC avatar.
Among participants in the low-risk category, there were increases in the frequency of discussions about cholesterol with a primary care practitioner in the usual care and the usual care plus VC treatment conditions although no mean differences were noted between treatment conditions. Self-reported statin medication use did not increase over time or differ between treatment conditions. Finally, during the time at which the ICAT was administered, there were reductions in “saturated fat content” and “use (of) regular salad dressing and mayonnaise instead of low-fat or fat-free” among participants in the VC treatment condition, thereby suggesting that the ICAT may be able to promote changes toward more healthy dietary activity. More research is needed to examine this possibility further.
This clinical trial is not the first to examine CVD prevention through the use of health information technologies (HIT). That being said, in 2013, a National Lipid Association workshop concluded that randomized clinical trials designed to determine the effect of HIT on lipid control are limited, and only a minority of such trials have demonstrated benefits for clinical outcomes such as lipids and statin medication use. 12 As well, a recent systematic review found more evidence for a beneficial effect of patient-level computerized decision-making tools that connect patients to the health care system, relative to provider-level tools. 13 We decided to develop a patient-level tool that could reduce clinical burden while providing consistent, evidence-based information in an interactive virtual environment. We are aware of other trials that examined patient-level tools, two of which provided risk estimators and/or heart age. One trial 14 enrolled patients who were at high CVD risk, provided information on vascular age and CVD risk at four times over 12 months (via paper form), and found effects on reduction in LDL cholesterol and proportion of patients who met their LDL goal. A second trial 15 enrolled patients who were at high CVD risk, provided a self-directed web-based decision aid coupled with education that was delivered in a clinic setting and in follow-up mailings, and found effects on self-reported medication use and coronary heart disease risk. In contrast to these trials and others,16–21 we decided to enroll adults with no known history of CVD or its equivalent because this population is the focus of primary prevention that represents a very large proportion of the population in the United States and often is not prioritized for risk reduction by medical care providers. As noted above, however, our focus on participants who were at low CVD risk (and possibly unmotivated to make lifestyle changes and/or take statin medication) may have obscured a beneficial effect of the ICAT on LDL and non-HDL cholesterol.
As noted previously, this study was informed by the recent validation of the Self-Determination Theory Model of Health Behavior 8 and its application to outcomes that are related to CVD. Indeed, the ICAT was designed to provide consistent, evidence-based information about CVD risk and its management in a way that is supportive of patients’ basic psychological needs for autonomy, competence, and relatedness. Such information was found to be accessible to participants without major technical challenges, and more research is needed to determine whether the use of an ICAT can lead to reductions in LDL and non-HDL cholesterol. The importance of developing a VC tool that is perceived as need supportive is underscored by recent data showing that such perceptions predict motivation for managing weight loss among patients with type 2 diabetes, which in turn predicts weight-loss outcomes. 22 Currently, we are examining whether perceptions of the ICAT as need supportive predict changes in cholesterol, motivation, dietary and physical activity, and psychological health.
Several limitations and strengths deserve mention. One limitation is that this study represents pilot research and, as a result, our sample size in both the low- and moderate-risk categories was small. A second limitation is that the ICAT modules were designed for use in this study, and thus their relevance in other paradigms is not known. One important strength is that the ICAT modules were designed using information about CVD risk and its management with strong empirical support, and this information was found to be accessible to participants without major technical challenges. Now that the 2013 AHA/ACC Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Disease in Adults 9 is available, it is important to examine the effect of a VC tool (similar to the ICAT) on relevant clinical outcomes in a larger sample.
Footnotes
Acknowledgements
Avi Dressler served as the study coordinator and data manager, and Nobiru Suzuki contributed to data entry. Trial Registration: Clinicaltrials.gov: NCT01890031.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Geoffrey C. Williams and C. Scott Rigby are shareholders in Self-Determined Health, Inc., the company to which the SBIR grant was awarded. Drs Williams and Rigby were involved in the development of the VC and contributed only to the introduction and methods sections of this report. Dr Williams managed subjects in both of the moderate-risk groups, and those subjects were informed (via the study consent form) of Dr Williams’ interest in the company. Drs Williams and Rigby did not have access to the data or contribute to the interpretation of the results.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was made possible by Grant Number 4R44HL097506-02 from the National Heart, Lung, and Blood Institute, which was awarded to Dr C. Scott Rigby, and by Grant Number T32HL007937 from the National Heart, Lung, and Blood Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
