Abstract
This study was aimed to investigate the effects of computerized decision support system in improving the prescription of drugs for cardiovascular prevention. A total of 197 Italian general practitioners were randomly allocated to receive either the alerting computerized decision support system integrated into standard software (intervention arm) or the standard software alone (control arm). Data on 21230 patients with diabetes, 3956 with acute myocardial infarction, and 2158 with stroke were analysed. The proportion of patients prescribed with cardiovascular drugs and days of drug–drug interaction exposure were evaluated. Computerized decision support system significantly increased the proportion of patients with diabetes prescribed with antiplatelet drugs (intervention: +2.7% vs. control: +0.15%; p < 0.001) or lipidlowering drugs (+4.2% vs. +2.8%; p = 0.001). A statistically significant decrease in days of potential interactions has been observed only among patients with stroke (−1.2 vs. −0.5 days/person-year; p = 0.001). In conclusion, computerized decision support system significantly increased the use of recommended cardiovascular drugs in diabetic patients, but it did not influence the exposure to potential interactions
Keywords
Introduction
Practice guidelines recommend the concurrent use of at least three pharmacologic agents to reduce the occurrence of adverse cardiac events among high-risk individuals, specifically antithrombotic, antihypertensive, and lipid-lowering drugs (LLDs).1–3 However, research in clinical practice has shown that preventive therapies remain largely underutilized, 4 although they have been associated with decreased morbidity rates and lower medical care costs.5,6
In primary care, short visits, competing visit objectives, lack of proactive systems for disease surveillance and alerting support, and challenges associated with managing treatments with multiple medications are all potential barriers to appropriate use of drugs.7–9 Polypharmacy is one of the main risk factors for drug–drug interactions (DDIs), and it has been estimated as a preventable cause of at least 0.5–4.7 per cent of all the hospital admissions.10,11
Computerized decision support system (CDSS) has been suggested as a potential means for facilitating the translation of research into practice by enhancing physicians’ adherence to recommended care standards.12–14 They have been shown to improve the decisions of health-care professionals in screening for cancer, 15 vaccination, 16 management of diabetes, 17 and laboratory test ordering. 18 Moreover, a recent study reported that Dutch general practitioners (GPs) have judged CDSS useful to improve their prescription quality. 19
However, few studies conducted in primary care explored the use of CDSS for the optimization of multiple drug treatments, 13 while the effects of such tools on medication adherence have been generally limited to simple advice for dosing and prescribing of single drugs.20,21
Several factors appear to be critical to the successful implementation of CDSS in daily practice: providing automatic support as part of the clinician’s workflow, providing decisions at the exact point of care, and providing recommendations rather than just warnings. 14
We, therefore, performed a cluster-randomized controlled trial to explore whether a CDSS, automatically prompted during daily practice, may improve the pharmacological management in high-risk cardiovascular patients. Specifically, we tested whether the CDSS may increase the prevalence of preventive therapies, as recommended in the treatment guidelines, and reduce the number of days of exposure to potentially interacting drugs among high-risk cardiovascular patients.
Methods
We chose, as chronic illnesses, type 2 diabetes mellitus (T2DM), acute myocardial infarction (AMI), and stroke because such diseases affect over 8 per cent of the adult population, and the management of these chronic illnesses is mostly undertaken by GPs.
General practice setting
At the time the study was conducted, the Health Search Network was a research group of nearly 800 GPs representative of each Italian geographic area in terms of patient population, which sent (from 1998) all clinical information from its patient list to Health Search Cegedim Strategic Data Longitudinal Patient Database (HS-CSD-LPD). GPs voluntarily agreed to collect patient information and attend specified training courses for data entry into a specific designed software used for managing patient information during their routine practice.
The HS-CSD-LPD contains patient demographic details that are linked through the use of an encrypted patient code with medical records (diagnoses, tests, and tests results), drug prescription information (medication name, date of filled prescription, and number of days’ supply), prevention records, hospital admissions, and date of death.
To be considered for participation in this study, GPs had to meet ‘up-to-standard’ pre-defined quality criteria pertaining to the completeness of coding, prevalence of well-known diseases, mortality rates, and years of recording. This approach has been extensively used in other databases, 22 as well as in several other epidemiological studies using information from the HS-CSD-LPD.5,23–25
Study design and power
Our study was a before and after, cluster-randomized open-label controlled trial allocating participating GPs to one of the two groups by a computerized randomization process stratified by age and geographic location (i.e. north-east, north-west, central, southern, major islands); such features are in fact the most relevant predictors of physician prescribing behaviour in Italy. 26 The random allocation of GPs was performed using STATA software, version 10.1 (STATA Corp., College Station, TX, USA).
Before the index date, both groups received the evidence-based recommendations for the appropriate use of cardiovascular drugs in patients with T2DM, AMI, and stroke.1–3 They were also provided with a list of 446 potentially serious DDIs, which involved antiplatelet drugs (APs), oral anticoagulants (OAs), LLDs, angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and angiotensin II receptor blockers (ARBs). DDIs were identified using leading drug information sources (i.e. DRUGDEX from Micromedex, Drug Interaction Facts, and Thesaurus des interactions medicamenteuses). Agence Française de Sécurité Sanitaire des Produits de Santé (AFSSAPS)).27,28 We have also considered the updated summary of product characteristics (SPCs) collected in Farmadati database (http://www.farmadati.it/), the source of information recommended by the Italian Medicine Agency.
According to these sources of information, only well-documented and potentially serious DDIs have been identified and distinguished into (a) contra-indicated and (b) not recommended. The list of potential DDIs also included the description of the possible adverse clinical outcome, the level of evidence, and related recommendations.
Both groups were provided with standard software aimed to transmit electronically all relevant clinical information on selected patients. The intervention group also received an automatically prompted CDSS integrated within their standard software system. The characteristics of the CDSS are described in Table 1. During the follow-up, the CDSS could be deactivated according to physician preferences.
Characteristics and specific recommendations of the CDSS at the time of its activation.
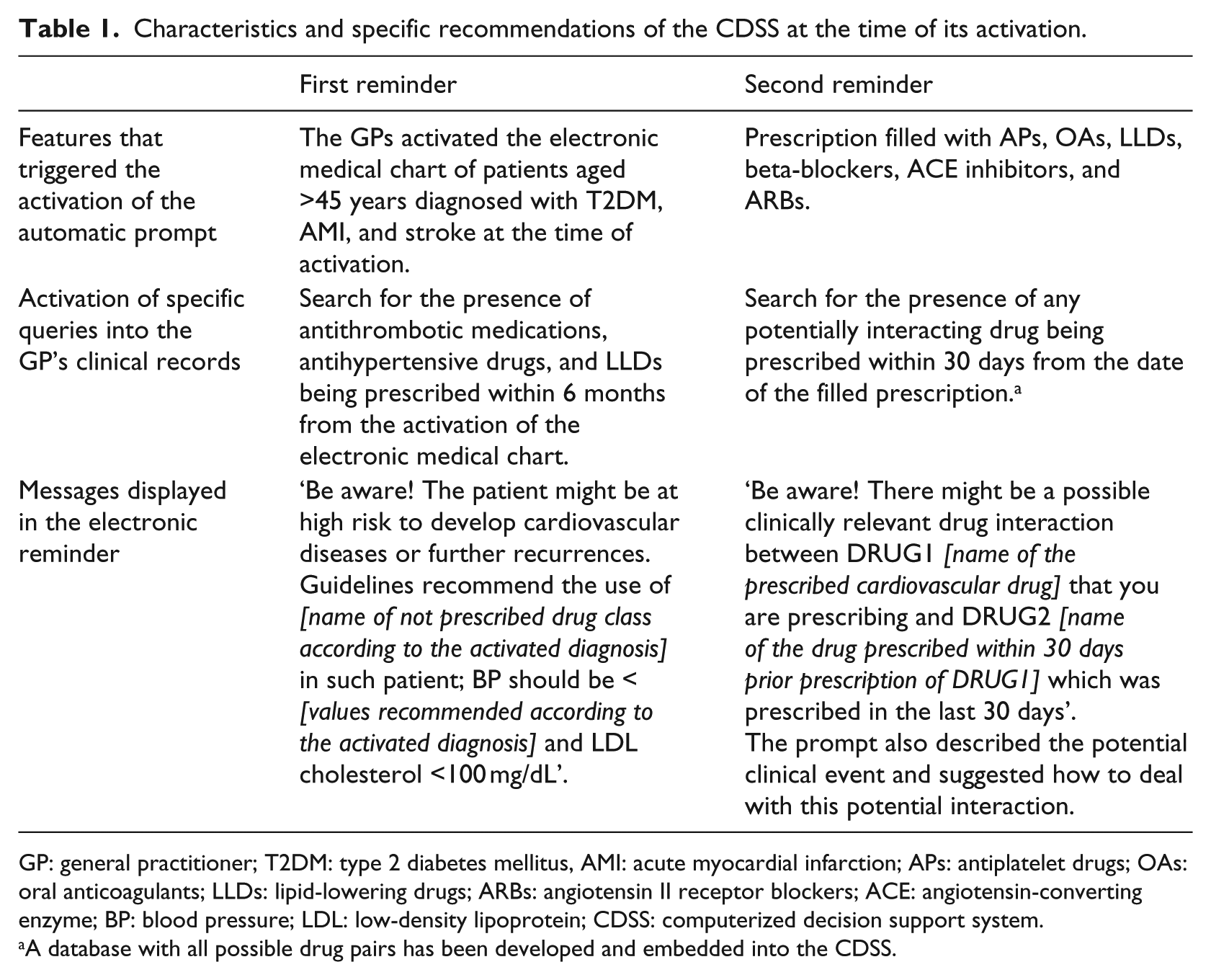
GP: general practitioner; T2DM: type 2 diabetes mellitus, AMI: acute myocardial infarction; APs: antiplatelet drugs; OAs: oral anticoagulants; LLDs: lipid-lowering drugs; ARBs: angiotensin II receptor blockers; ACE: angiotensin-converting enzyme; BP: blood pressure; LDL: low-density lipoprotein; CDSS: computerized decision support system.
A database with all possible drug pairs has been developed and embedded into the CDSS.
To detect an absolute difference of 5 per cent between intervention and control groups for the primary outcomes, with a power of 80 per cent and taking into account an intra-class correlation coefficient for ‘taking medications in general practice’ of 0.0099, 29 our trial required at least 144 GPs. In order to detect this difference, with enough power in any disease under evaluation, the following numbers of patients were required: at least 2120 patients with T2DM, 1095 patients with AMI, and 1076 patients with stroke in each group. All these figures were derived from an algorithm created in STATA based on the study by Smeeth and Ng. 29
Cohort definition
The date of the standard software activation for data transmission was defined as the start date of the study. Study patients were those registered with the participating GPs for at least 1 year prior to index date, aged 45 years or older, and with a diagnosis of T2DM (International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) codes 250.x, excluding 250.x1 and 250.x3), AMI (codes 410.x through 412.x), and ischaemic stroke (codes 434.x, 436.x, 438.x, 342.x) prior to the index date. Those patients’ categories were not mutually exclusive.
Subjects were followed up from the index date until the end of the study period (i.e. 2 years). Individuals who transferred out from GPs’ practices or died during the follow-up were censored.
Study outcomes
The primary endpoint was the evaluation of the effectiveness of the CDSS in increasing the prevalence of recommended cardiovascular drug use among high-risk patients after the first year of follow-up. More specifically, we assessed whether the intervention determined significant changes in (a) the prevalence of patients with T2DM who were treated with APs or LLDs; (b) the prevalence of patients with history of AMI who were treated with APs, OAs, LLDs, or antihypertensive medications; and (c) the prevalence of patients with history of stroke who were treated with APs, OAs, or LLDs.
The prevalence of treated patients has been defined as the number of patients requiring cardiovascular treatment and those actually treated, divided by the total number of patients requiring cardiovascular treatment. Patients were considered as treated when at least one of the cardiovascular medications of interest was prescribed within 1 year prior to the start date for the baseline evaluation or anywhere during the first year after the start date for the follow-up evaluation.
As a secondary endpoint, we evaluated the effectiveness of the CDSS in decreasing the mean number of days of exposure to potentially interacting drugs among cardiovascular patients with the outcomes of interest. Duration of exposure was based on the actual prescription duration, which was estimated by dividing the total amount of active drug in each prescription by the recommended defined daily dose. We estimated the total duration of potentially interacting drug exposure during the treatment with APs, OAs, LLDs, beta-blockers, ACE inhibitors, and ARBs, either at baseline or during the first year of follow-up.
Statistical analysis
We used the clinical information routinely gathered into the HS-CSD-LPD for the data analysis of all defined outcomes. The intention-to-treat approach was used to analyse data: only GPs who activated the CDSS during the follow-up were considered in the intervention group. As the intervention is delivered at the physician level but outcomes are measured at the patient level, we accounted for clustering effects in assessing the potential differences between the groups at the end of follow-up.
Baseline comparisons of patient characteristics between the control and intervention groups have been done using Student’s t-tests for continuous variables and chi-square tests for categorical variables. We expressed the effect size as the difference within the groups at baseline and at the end of follow-up. Between-group differences in the primary and secondary endpoints have been compared by generalized linear regression models that accounted for clustering at physician level, number of patients within each cluster, and baseline estimates of either prevalence of drug use or DDIs. We have also included in the models most of the documented risk factors of coronary heart disease and stroke,1–3 that is, familiar history of cardiovascular disease, blood pressure (BP) values, and cholesterol levels (calculated as the average between the last two separate measurements over the 12 months before the index date); obesity (body mass index ≥30 kg/m2 (last available value for the 12 months before the index diagnosis) or code 278.0); arrhythmias (code 427.x); peripheral vascular diseases (code 443.9); cancer (codes 140 through 199.x, 200 through 208.x); and chronic respiratory diseases (codes 490 through 496.x).
Multivariable selection of risk factors was done by a stepwise procedure after adjustment for age and gender. Statistical significance was defined as a two-tailed value of p < 0.05. Estimates of treatment effects, 95 per cent confidence intervals (CIs), and probability values were generated with STATA software, version 10.1 (STATA Corp.).
Results
Figure 1 illustrates the flow of participants into the trial. Among 285 GPs who expressed their interest in participating in the study, 230 have been randomly allocated to receive either the CDSS (intervention group: 115 GPs) or paper-based information (control group: 115 GPs). The remaining 55 GPs were excluded from the analysis due to issues in matching the intervention and control groups.
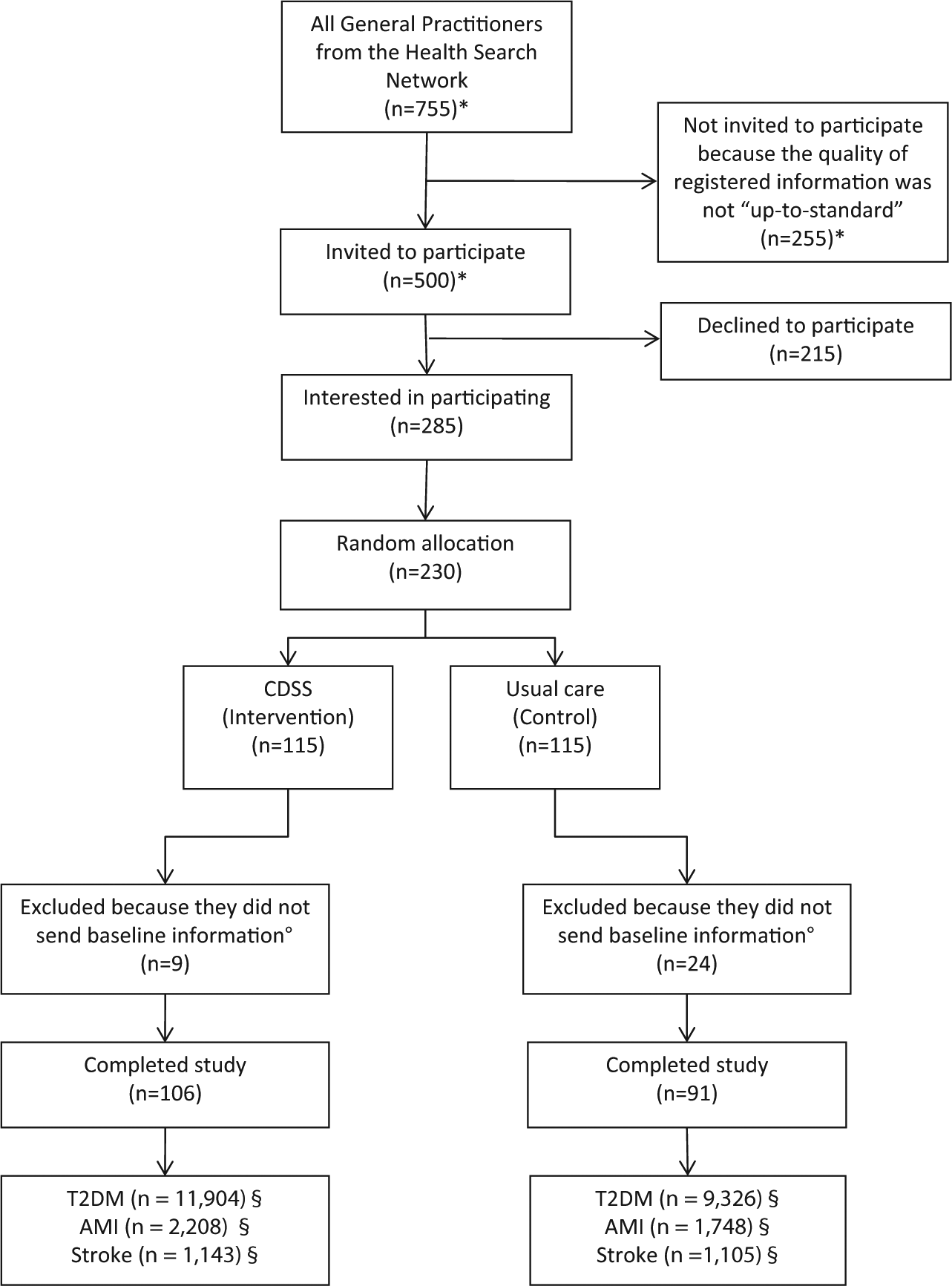
Flow diagram of the general practitioners through the trial and corresponding selected patients.
Finally, only 197 GPs (106 in the intervention group vs 91 in the control group) sent all baseline information about eligible patients. Characteristics of GPs (i.e. age, gender, and geographic location) did not significantly differ between intervention and control groups. Among 106 GPs in the intervention group, 65 (61.4%) truly maintained the CDSS during follow-up.
Overall, we selected 14,238 patients in the intervention group and 11,253 in the control group. These groups showed similar demographic characteristics, whereas statistically significant differences have been observed for congestive heart failure (p = 0.007), hypertension (p = 0.034), chronic obstructive pulmonary disease (COPD) (p = 0.001), Charlson index (p = 0.001), and the mean number of concurrent drug use (p < 0.001) (Table 2).
Baseline characteristics of patients with at least one of the selected diseases randomized to CDSS (intervention) or usual care (control).
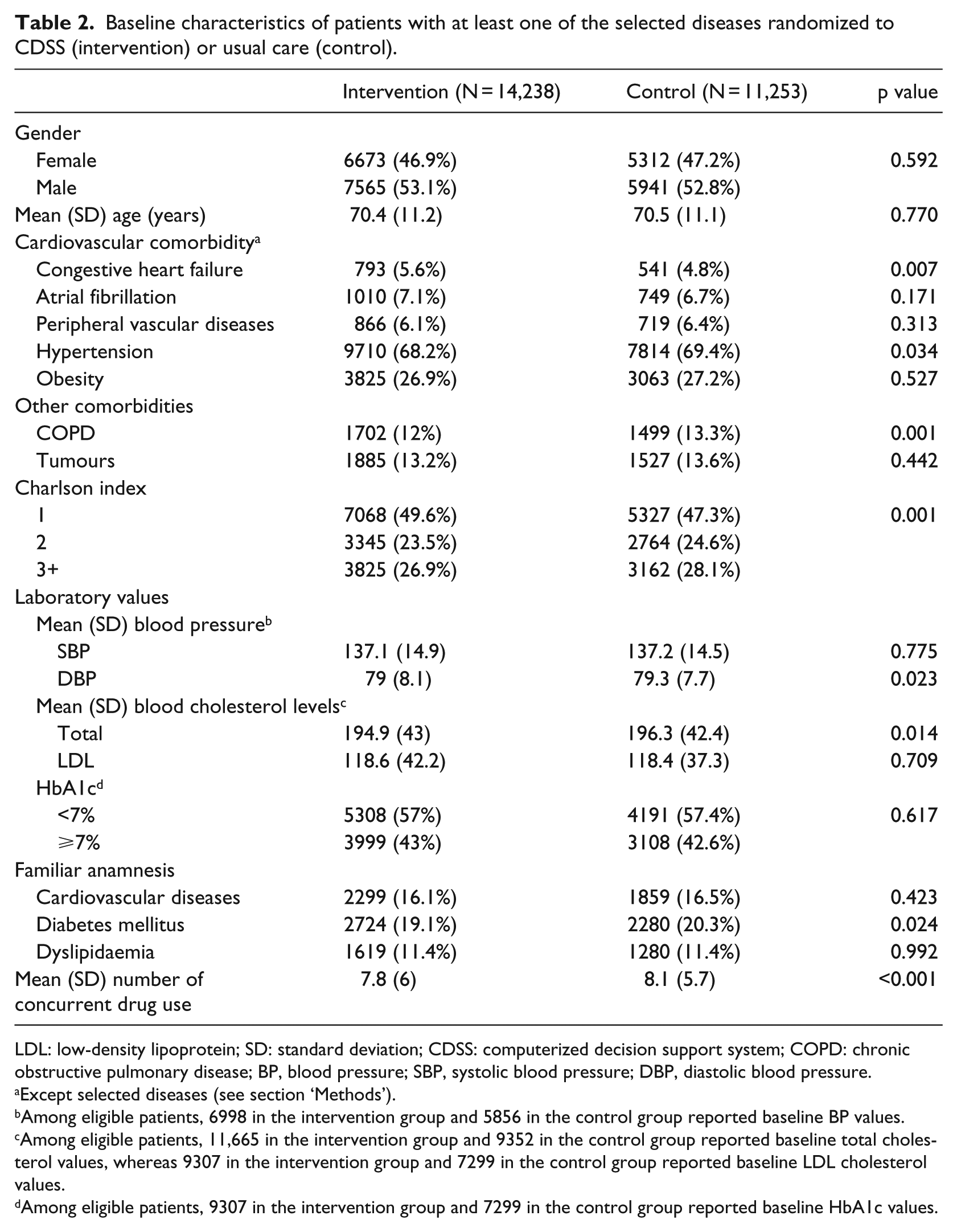
LDL: low-density lipoprotein; SD: standard deviation; CDSS: computerized decision support system; COPD: chronic obstructive pulmonary disease; BP, blood pressure; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Except selected diseases (see section ‘Methods’).
Among eligible patients, 6998 in the intervention group and 5856 in the control group reported baseline BP values.
Among eligible patients, 11,665 in the intervention group and 9352 in the control group reported baseline total cholesterol values, whereas 9307 in the intervention group and 7299 in the control group reported baseline LDL cholesterol values.
Among eligible patients, 9307 in the intervention group and 7299 in the control group reported baseline HbA1c values.
Prevalence of cardiovascular drug use
At baseline, around 40 per cent of patients with T2DM were treated with APs, whereas such prevalence was much higher among patients with AMI (intervention: 73.4% vs control: 78.7%) and stroke (intervention: 62.2% vs control: 61.5%) (Table 3). Similar prevalence was observed for LLDs among patients with T2DM and stroke, whereas higher estimates have been observed for AMI (intervention: 66.1% vs control: 68.8%)
Changes in the prevalence of cardiovascular drug use at baseline and during the follow-up among eligible patients.
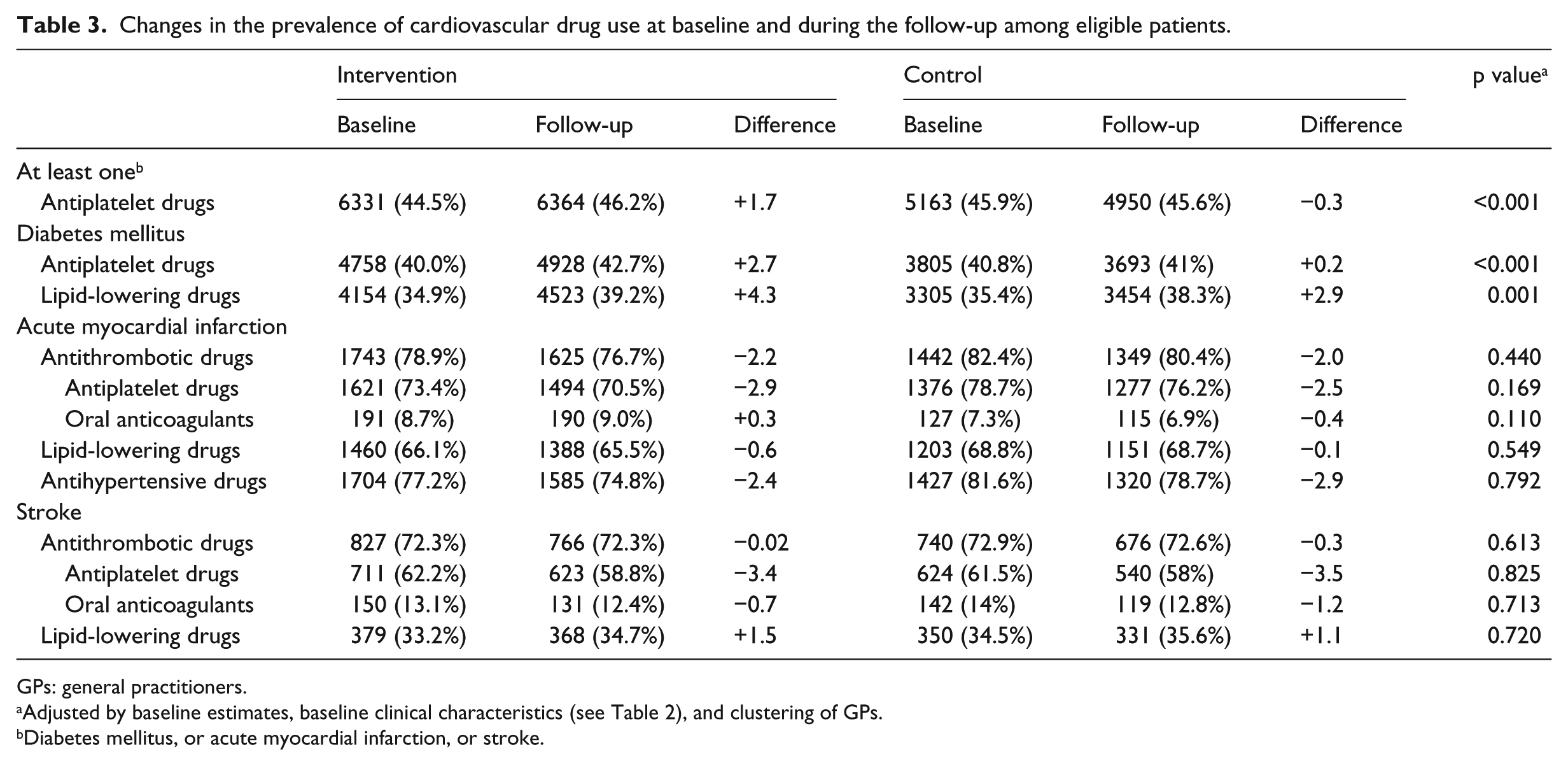
GPs: general practitioners.
Adjusted by baseline estimates, baseline clinical characteristics (see Table 2), and clustering of GPs.
Diabetes mellitus, or acute myocardial infarction, or stroke.
During the follow-up, the use of CDSS significantly increased prescription with APs (intervention: +2.7% vs control: +0.15%; p < 0.001) and LLDs (intervention: +4.3% vs control: +2.8%; p = 0.001) among patients with T2DM. No statistically significant effects have been observed among patients with AMI and stroke.
GPs were then stratified by tertiles according to the baseline prevalence of AP and LLD use in their patients’ population, regardless of their morbidity status. In the first tertile, the CDSS significantly increased either AP (p = 0.021) or LLD (p = 0.006) exposure, whereas in the second (p = 0.047) and third tertiles (p = 0.04), a statistically significant increased exposure has been observed only for APs (Figure 2).
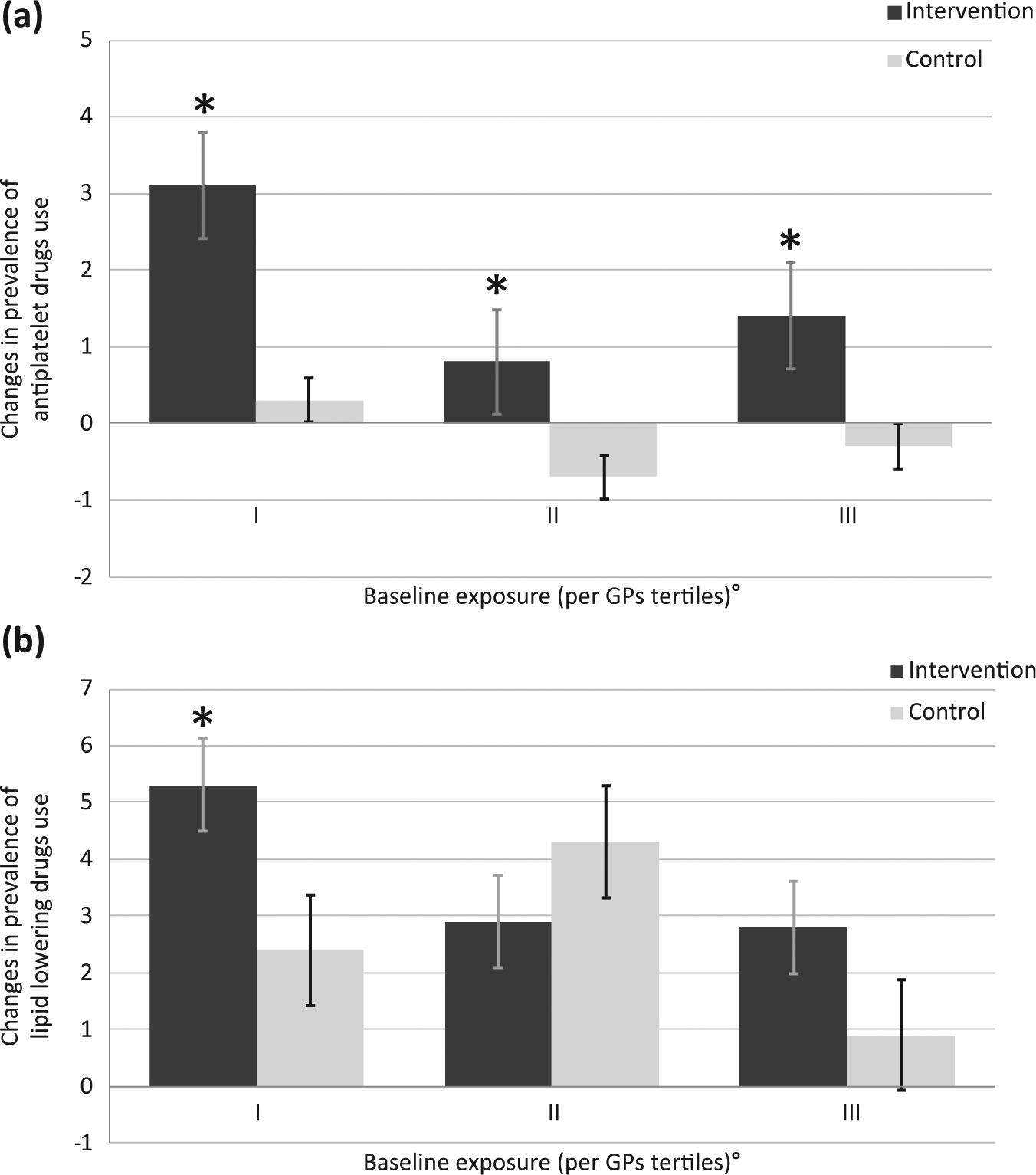
Changes in the prevalence (%) of (a) antiplatelet (b) and lipid-lowering drug use by baseline exposure and trial arm.
DDIs
Table 4 describes the mean number of days with concurrent interacting drugs among patients exposed to the observed drugs. Such estimates ranged from 4.7 to 8.1 mean days/person-year of exposure for non-recommended drugs and from 0.79 to 2.14 mean days/person-year of exposure for contra-indicated drugs. Overall, the use of CDSS significantly decreases the mean number of days of concurrent administration of cardiovascular drugs and non-recommended drugs due to DDI risk (p = 0.006), while no effect for contra-indicated drugs was observed. Among patients with stroke, both contra-indicated (p = 0.066) and non-recommended (p < 0.001) drugs at interaction risk were slightly, but significantly, reduced. When GPs were stratified by tertiles according to the baseline mean days of exposure with contra-indicated or non-recommended drugs in their patients’ population, a statistically significant (p < 0.001) reduction in the intervention group, compared with controls, has been observed only at the highest level of baseline exposure with non-recommended drugs (Figure 3).
Changes in the mean days of concurrent use of cardiovascular drugs and non-recommended or contra-indicated drugs due to drug–drug interaction risk (per person-year of cardiovascular drug exposure) among patients with diabetes mellitus, acute myocardial infarction, and stroke at baseline and during the follow-up.
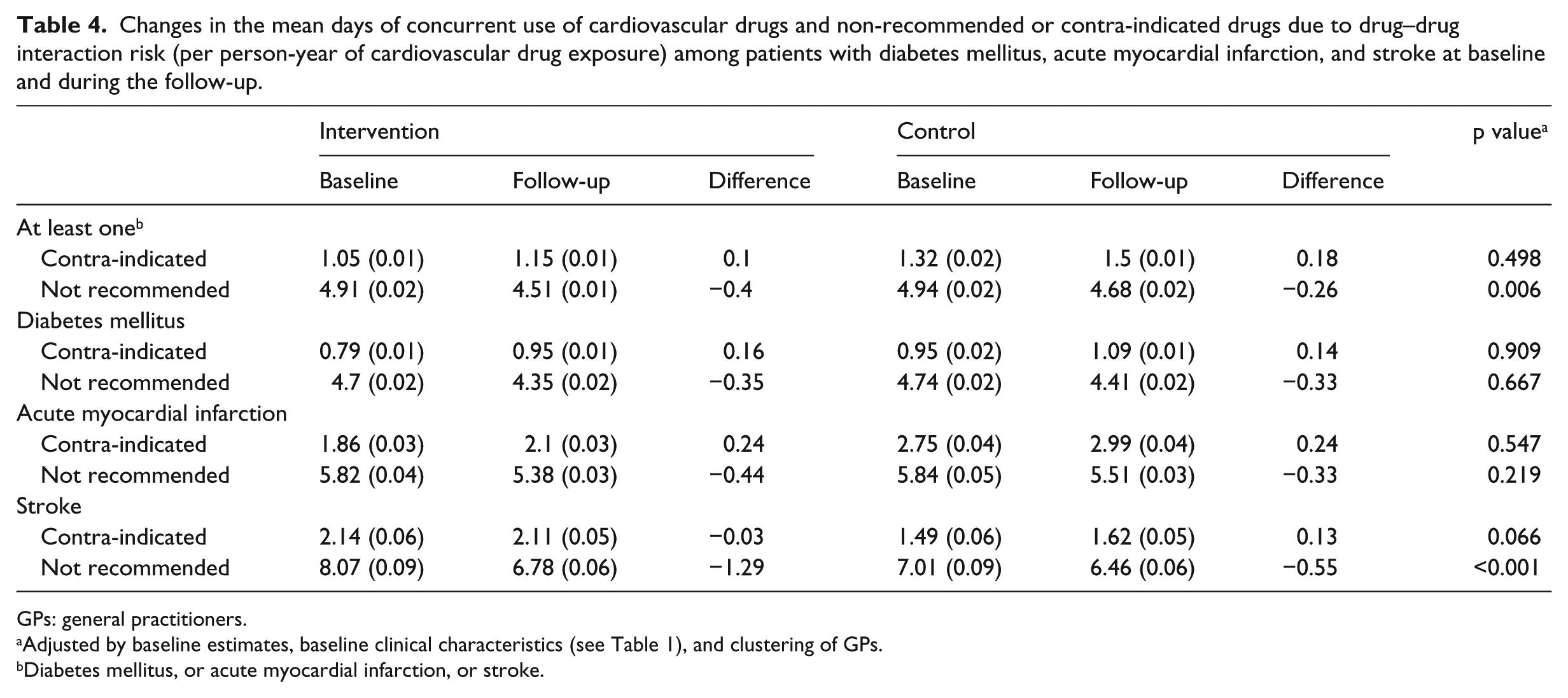
GPs: general practitioners.
Adjusted by baseline estimates, baseline clinical characteristics (see Table 1), and clustering of GPs.
Diabetes mellitus, or acute myocardial infarction, or stroke.
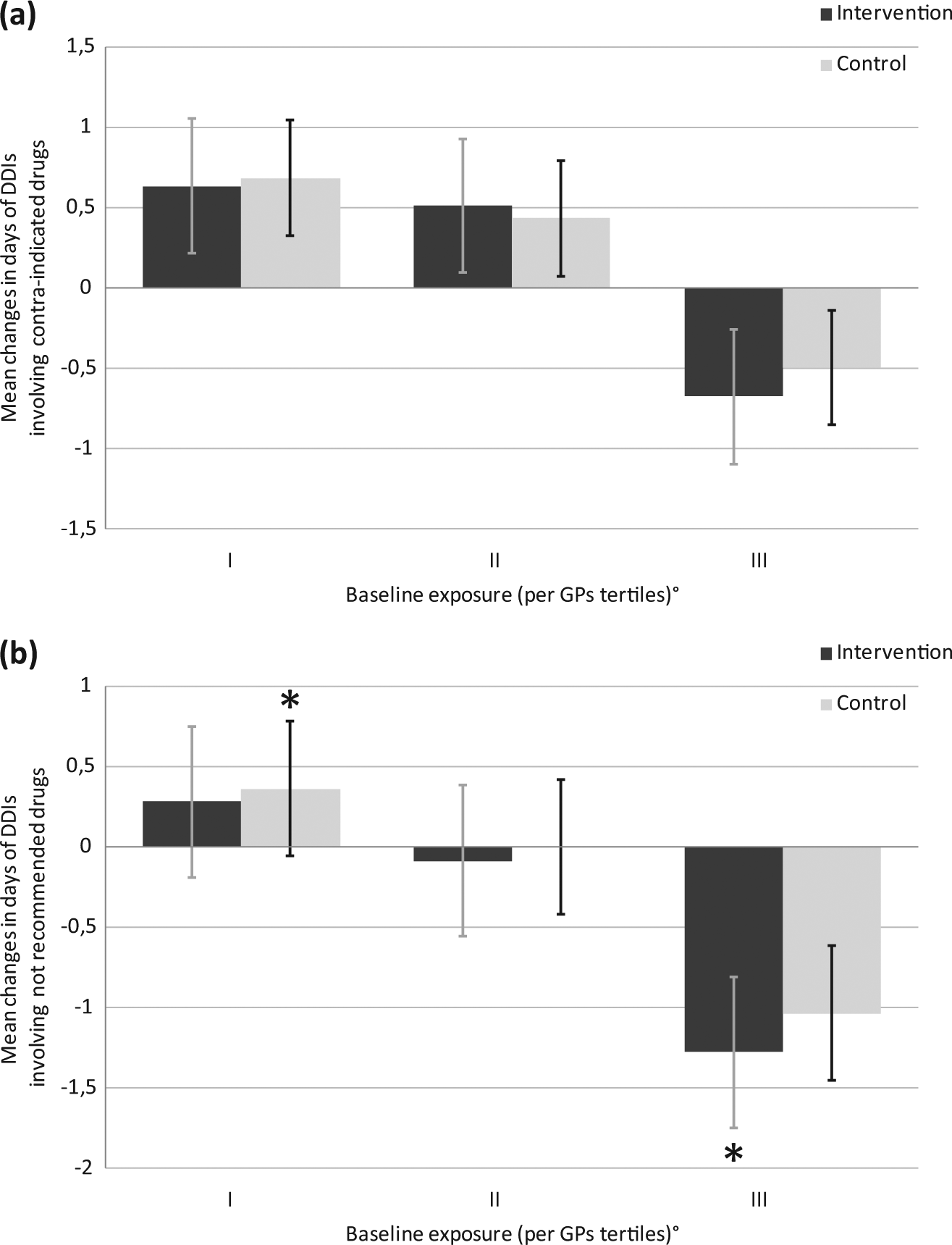
Changes in the mean days (per person-year of exposure) of (a) contra-indicated and (b) non-recommended drugs due to drug–drug interaction risk by baseline exposure and trial arm.
Discussion
In this cluster-randomized open-label controlled trial, we have shown that an automatically prompted CDSS may improve the pharmacological management of specific categories of high-risk cardiovascular patients in primary care. In particular, the CDSS significantly increased the use of ADs (+2.7% vs +0.2%; p < 0.001) and LLDs (+4.3% vs +2.9%; p = 0.001) among patients with T2DM, when compared with GPs who were only provided with paper-based advice. On the other hand, the effect of CDSS was not observed in patients with AMI and stroke, who had a baseline exposure to recommended cardiovascular medications higher than patients with T2DM.
Regarding the effect on the co-prescription of drugs at interaction risk in cardiovascular patients, this study showed a modest effect of CDSS. A statistically significant decrease in the mean number of days of concomitant exposure to cardiovascular medications and non-recommended drugs due to interaction risk (−1.29 vs −0.55 days/year; p < 0.001) was observed only among patients with stroke, who had at baseline the greatest exposure to interaction risk. Altogether, these findings suggest that CDSS is likely to improve physicians’ behaviour only in the presence of higher levels of inappropriate use of drugs. They also support previous hypotheses, which suggested that one of the key elements for the successful development of indicators in primary care is the potential for improvement.30,31 Further confirmation is provided in the analyses stratified by baseline exposure either to recommended drugs or to potentially interacting drugs, where statistically significant beneficial effect of CDSS has been observed only among most physicians not fully complying with guidelines.
Although our analysis considered available confounding factors (e.g. age, gender, cardiovascular comorbidity, other comorbidity, and Charlson index), we cannot exclude the possibility that the results may be partly affected by other unrecognized confounding factors (related to patient, GP, or system).
The ability of CDSS in improving the pharmacological management of chronic diseases has been previously explored in several randomized and non-randomized controlled trials, which overall documented the difficulties in achieving improvement in physicians’ performance. 32 These improvements, although small in absolute numbers, should be framed in the context of costs: compared to the low running costs of the CDSS, the improvements in clinical practice, with related savings for health services, make these systems useful tools for monitoring and improving prescribing practice.
A recent systematic review identified 100 randomized/non-randomized controlled trials that compared CDSS to routine care and evaluated clinical performance (i.e. measure of process of care) or patient outcome. 13 In detail, five of these studies assessed the effect of CDSS in improving adherence with multidrug prescribing and three in reducing potentially relevant drug interactions. Furthermore, three studies33–35 were specifically targeted on GPs, with one 35 reporting 18 per cent significant reduction in the number of new potentially inappropriate prescriptions, but no effect was observed in the prevention of interacting co-prescriptions.
CDSS improved physician performance in 65 per cent of the observed studies and in four of five studies on multidrug prescribing. Only one of them 36 assessed the effect of a computer-based physician workstation (PWS) designed to reduce adverse drug interactions in hospital setting, but no significant reductions were reported. Authors hypothesized that the small fractions of prescriptions (2.8%) prescribed with the support of the PWS were the most plausible explanation for the lack of results.
Our study has several limitations. First, in performing the study, we did not question about the validity of the guidelines nor the degree of acceptance from participating GPs. Thus, we believe that if the CDSS is based on well-accepted evidence-based guidelines, improvement in physician performance might be better than that observed, and it will result in improved patient outcomes. Indeed, in testing the CDSS, we assessed the potential challenges faced by the participating GPs in properly using the software system, but we did not test the impact of such system in routine practice. CDSS, for instance, was designed in our study to be prompted in the presence of DDIs with a high degree of scientific evidence, even those easily managed with a precautionary approach. There is probably the need to further limit software screening only to drug interactions judged by a consensus panel as likely to produce clinically relevant adverse effects.37,38 In our study, the main reason for discontinuation of the CDSS use was in fact the large number of DDI alerts. This reason raises a number of questions about the development and usability of these systems. However, this phenomenon is common, and it is well recognized in the Heath Informatics literature: the practice of ignoring (or stopping) the computer system alerts sometimes can lead to an increase in the risk of inappropriate drug prescribing. 39 Finally, all participating GPs, belonging to a research group already involved in epidemiological research, agreed to use the CDSS and were also aware of the study’s objective; both the intervention and the control groups had electronic and paper access to the treatment guidelines of cardiovascular diseases at the beginning of the study. Therefore, the results may have underestimated the effect of CDSS in practice generally and may not be generalizable to all primary care physicians (PCPs) who could have a limited knowledge and adherence to these guidelines, as shown in other studies. Although the open-label design was unique to our study, this could have generated an overestimation of the CDSS effects.
Conclusion
We developed a CDSS for improving pharmacological management in high-risk cardiovascular patients in primary care. Unlike some previous studies of electronic reminders that have been unsuccessful in improving care for diabetes, the effect of CDSS in our study was positive in diabetic patients, especially in improving the chance that they would receive recommended care. In our opinion, because of the high prevalence of diabetes and its significant morbidity, both patients and GPs could benefit from an advanced diabetes-specific CDSS.
Although we have demonstrated that evidence-based computerized decision support holds some promise resulting in a better prescribing behaviour among Italian GPs, we did not find compelling evidence of an effect on other study outcomes. Only a modest effect of CDSS on the reduction in exposure to potentially interacting drugs was observed.
The use of well-accepted guidelines, as well as targeting of specific issues, particularly where high levels of inappropriate use of drugs are likely to occur, should help to improve the effectiveness of CDSS.
Certainly, the implementation of this system cannot be considered the ‘magic bullet’ 34 that provides an answer to all questions, but may provide the basis for further interventions aimed at improving preventive measures in primary care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by funds from the Italian Medicines Agency (AIFA).
