Abstract
Patient safety is a matter of major concern that involves every health professional. Nowadays, emerging technologies such as smart pumps can diminish medication errors as well as standardise and improve clinical practice with the subsequent benefits for patients. The aim of this paper was to describe the smart pump implementation process in a paediatric intensive care unit (PICU) and to present the most relevant infusion-related programming errors that were prevented. This was a comparative study between CareFusion Alaris Guardrails® and Hospira MedNet® systems, as well as a prospective and intervention study with analytical components carried out in the PICU of Gregorio Marañón General and Teaching Hospital. All intravenous infusions programmed with a pump in the eleven beds of the unit were analyzed. A drug library was developed and subsequently loaded into CareFusion and Hospira pumps that were used during a three month period each. The most suitable system for implementation was selected according to their differences in features and users’ acceptance. Data stored in the pumps were analyzed to assess user compliance with the technology, health care setting and type of errors intercepted. The implementation process was carried out with CareFusion systems. Compliance with the technology was 92% and user acceptance was high. Vacation substitution and drug administration periods were significantly associated with a greater number of infusion-related programming errors. High risk drugs were involved in 48% of intercepted errors. Based on these results we can conclude that implementation of smart pumps proved effective in intercepting infusion-related programming errors from reaching patients. User awareness of the importance of programming infusions with the drug library is the key to succeed in the implementation process.
Introduction
The growing complexity of the ‘drug use circuit’ carries an ever increasing risk of medication errors throughout different stages. 1 Errors occurring in the administration phase are the hardest to intercept, 1 and guaranteeing safety in the administration of high-risk intravenous or narrow therapeutic range drugs to critical paediatric patients must be a priority in our health service.2–4
The use of infusion pumps has increased safety and precision in administering drugs requiring exhaustive flow control. Nevertheless, because these systems may give rise to programming errors, which in turn may become medication errors, 5 second-generation infusion systems known as ‘smart pumps’ play a fundamental role.
A smart pump is a conventional infusion pump with an incorporated ‘drug library’, that is a list of drugs with defined concentrations, dosage units and maximum and minimum infusion times for each. Based on these parameters, hard and soft limits are established, upper hard limit (UHL) and upper soft limit (USL) and lower hard limit (LHL) and lower soft limit (LSL), the objective of which is to avoid over- and under-dosage, respectively.
Therefore, if a user exceeds the defined rate for a soft limit due to a programming error, an alarm is triggered warning the user that the dose or infusion rate may not be adequate for a determined patient, but allows the infusion to continue. On the other hand, accidentally exceeding a hard limit will sound an alarm which cannot be ignored leading the user to cancel the infusion or reprogramme the administration. 6
Currently, different suppliers offer smart pump technology, and the differential features among systems may condition the success of the implementation of this technology. There is little information published regarding the results of smart pump implementation process, especially in paediatric populations, in terms of number and types of errors intercepted. This project may help other health-care professionals involved in paediatrics safety to develop their own smart systems adapted to their patients and clinical settings.
Objective
The aim of this work is to describe the implementation process of smart pump technology in a paediatric intensive care unit (PICU), from the manufacturer selection and the development of a drug library to the description of most relevant errors intercepted after 18 months of total implementation in the unit.
Methods
The PICU in which this study was performed belongs to a 223-bed maternity hospital, of which 140 are paediatric and 83 belong to obstetrics and gynaecology distributed over 5 floors of the hospital. The hospital operates computerised physician order entry (CPOE) associated with 18 automatic dispensing systems. The PICU comprises 11 beds and an average of 500 hospitalisations per year: 35 per cent cardiopathies, included in cardiac surgery postoperative, 30 per cent derived from other postoperatives and the remaining 35 per cent for other medical reasons. The nurse/patient ratio is 1.
A multidisciplinary team comprised two PICU paediatricians, two clinical pharmacists, the PICU chief nurse and a technician from each manufacturer involved in the system selection phase. Every member of the team actively participated in each phase of the implementation process. Pharmacists played a very important role in designing and coordinating the different phases of the study and also acted as a link among all health professionals involved in the project.
Drug library and back-up document development phase
The first version of the drug library was built up from June to December 2009 and was developed by the multidisciplinary team through weekly meetings during the first 4 months and fortnightly during the last trimester of this phase.
Most commonly used PICU intravenous drugs, high-risk drugs as well as those which could cause problems in administration due to lack of experience were included. For each drug, concentration and recommended infusion rates were determined according to the sources of information most used by PICU and Pharmacy Service.7–12 Drugs were distributed into profiles according to their therapeutic group.
A training profile (loaded with a sample of drugs from the other profiles) for exclusive use during learning periods was included to allow debugging of alarms for their further analysis and avoid bias being introduced into the results afterwards.
Alongside the development of the drug library, an intravenous drug administering guide was also elaborated, with information on intravenous mixture preparation, stability and storage instructions. Furthermore, a compatibility chart for drug candidates for infusion through the same line (Y-site administration), when patients lack available vascular access, was developed. Both documents were designed with the aim of supplying back-up and reference material for nursing staff.
System selection phase
A comparative, prospective, descriptive study was designed to be performed from January to June 2010. The infusion pumps were volumetric and syringe, Alaris Guardrails®, CareFusion with safety software, available at the hospital at the time of the study, and Plum A+®, Hospira volumetric pumps with MedNet® safety software, of interest due to some technological advantages offered regarding connectivity, compared to other systems on the market. 13
The hospital informatics service was involved for the necessary installations and connections in setting up the Hospira systems. Both types of pumps had been approved for paediatric use.
The drug library was uploaded in both systems for use during each corresponding trial period. Both periods had to be preceded by a training week for staff on every shift so as to train them on conventional use of Hospira pumps, as they were new to the unit, as well as programming using the safety software for both systems. Training was provided by the technicians from the manufacturing companies for all nursing shifts and with a variable duration depending on user demand. System user guides (electronic and paper) were delivered. The initial study schedule is detailed in Figure 1.
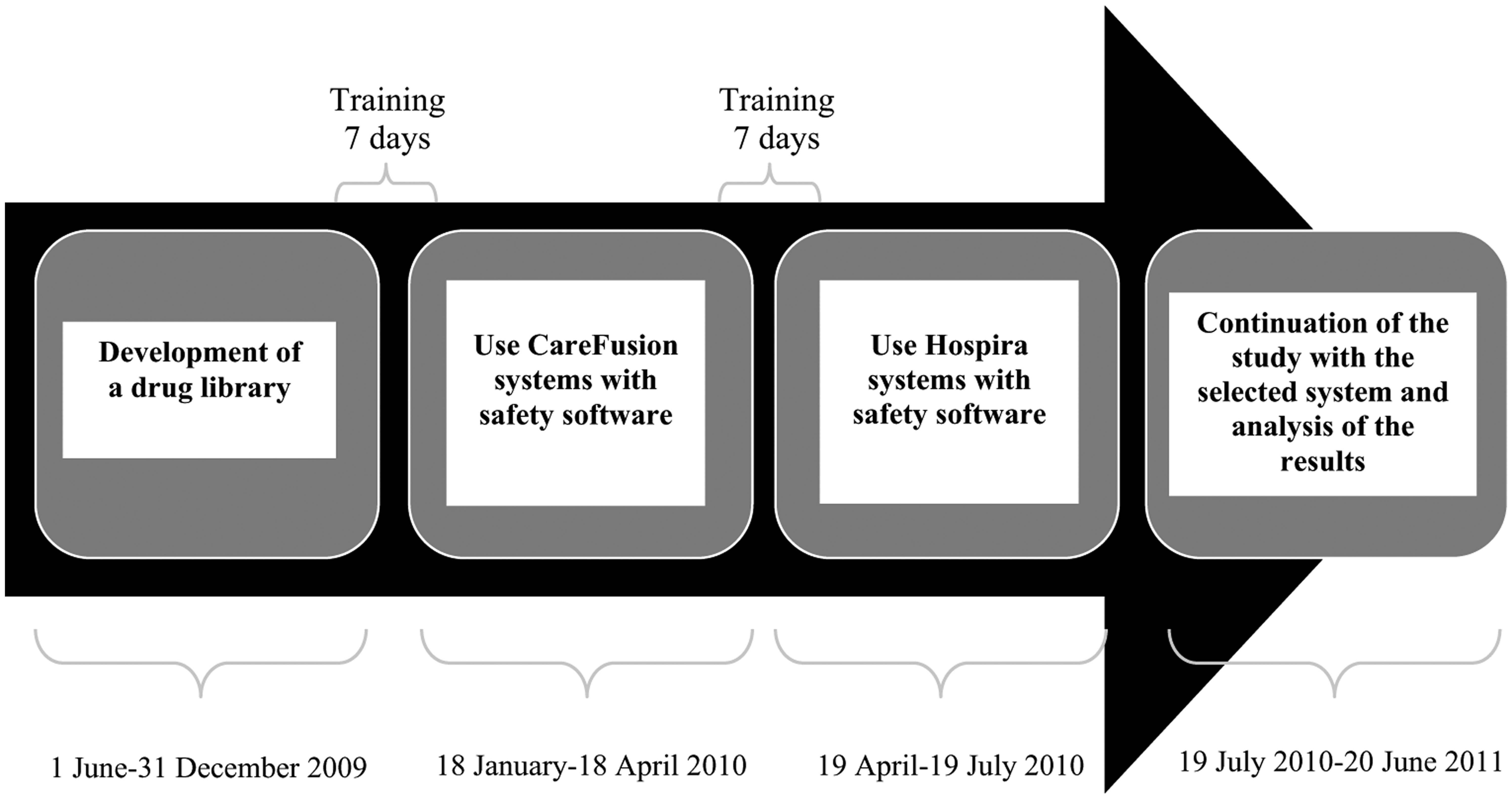
Initial study schedule.
On finalising the comparative phase between systems, the selected system would be the best one adapted to the PICU requirements with regard to differential characteristics and physical aspects, edition and data programmes, connectivity, technical support, price and degree of user satisfaction. The degree of user satisfaction was assessed through a questionnaire aimed at all nursing shifts available in the PICU 6 and 17 months after the implementation process began.
Analysis of stored information phase
The systematic analysis of all information stored in the pumps was performed using Guardrails® CQI v4.1 Event Reporter software, for CareFusion infusion pumps, or MedNet® v5.5 system, for Plum A+ Hospira volumetric pumps, depending on the system chosen.
Access to this information allowed for the identification of areas for improvement such as drugs included in the drug library, limits established and staff training, which usually lead to the increase of users’ compliance with this technology.
Every 6 months, the downloading of stored information and the updating of the database were performed by a clinical pharmacist and a paediatrician from the multidisciplinary team who were granted access to information independent of the supplier and taking suggestions from the PICU team into account.
The following variables were studied:
Quality of technology use. The percentage of programmed infusions using the drug library and the percentage of ignored warnings were analysed. Both variables detail user compliance with this technology.
Health-care setting. Alarm distribution by time of day, day of the week and month of the year was analysed.
Technology efficacy. The total number of intercepted infusion-related programming errors was determined based on the alarms triggered by the safety software. Only real programming errors that could have reached the patient were described. Alarms caused by incorrect use of the systems and not associated with a real risk of medication error were not included in the results.
In case an error reached a patient, it would be documented in the hospital self-reporting system. The study had approval from the Ethics and Clinical Investigation Committee of the Gregorio Marañón Hospital. It was not necessary to request a signed informed consent form from patients since the object of the study was the infusion systems and information related to patients was not provided; infusion pumps are routinely used on hospitalised patients, and the study did not involve any change to their therapeutic treatment.
Statistical analysis
For technology user satisfaction, a questionnaire was elaborated for PICU nursing staff from all shifts after 6 months from the implementation (June 2010) and at the end of the 17-month study period (June 2011) (see Table 1). The questionnaire was administered in Spanish, and Table 1 shows the translation of the original one into English.
Satisfaction survey aimed at PICU nursing staff.
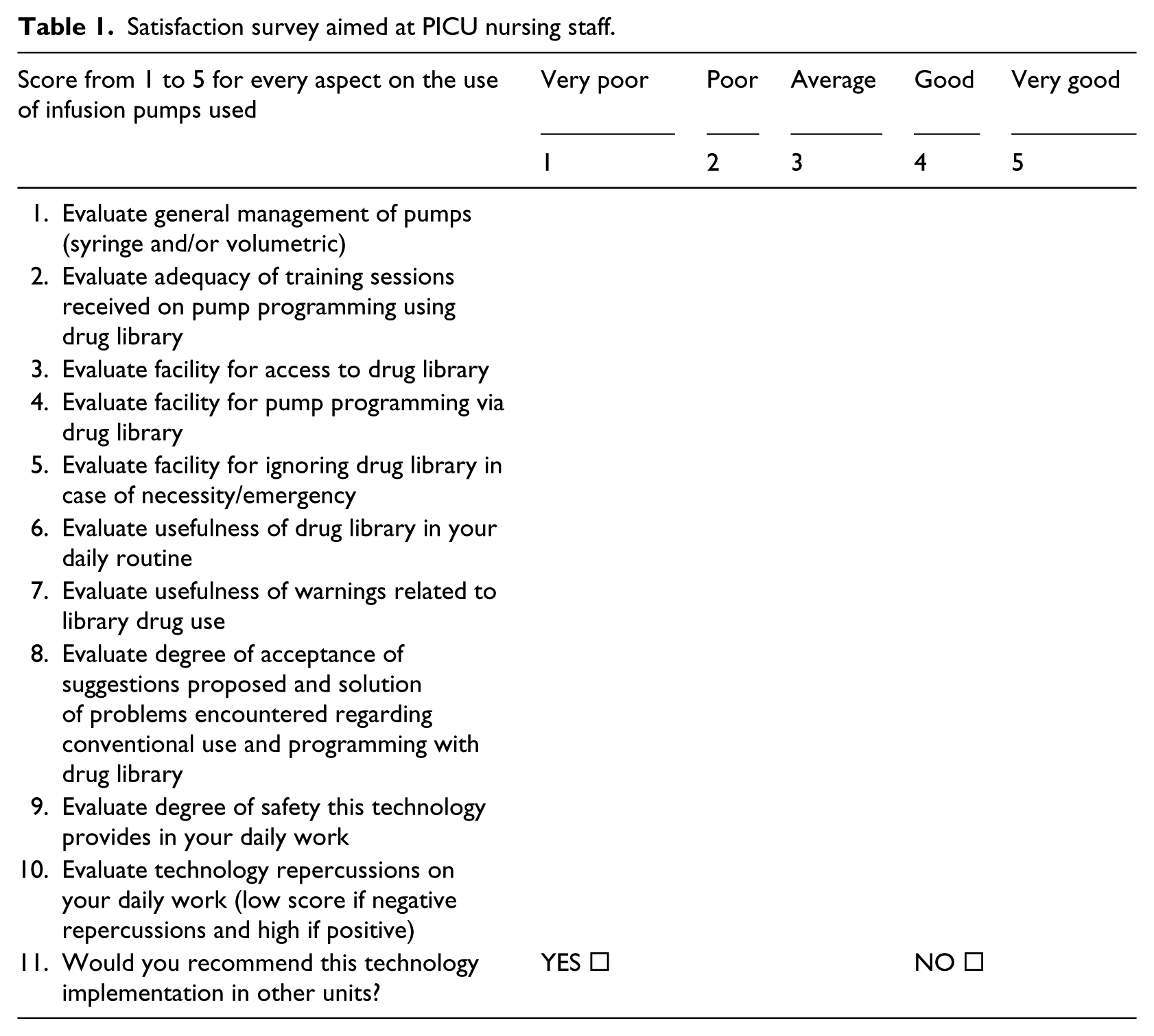
Comparison of the degree of nursing staff satisfaction with the technology during the two periods was performed using Mann–Whitney non-parametric test applied to answers to questions 1–10 and Fisher’s exact test applied to question 11.
For comparison of percentages of each variable studied, chi-square test was used. Differences were considered statistically significant if p < 0.05.
Results
Drug library and back-up document development
Between June and December 2009, the first version of the drug library was drawn up for the CareFusion system. From this date, changes to the initial library were periodically made derived from daily clinical practice and analysis of the information stored in the system, which gave rise to seven versions of the drug library. 14 The current drug library includes 105 drugs. 15
As the intravenous drug administering guide was elaborated alongside the drug library, all changes incorporated in the different versions were also included in the guide. The guide comprises 129 drugs, of which 81.4 per cent are included in the latest drug library version. The compatibility chart includes 47 drug candidates for Y-site infusion in PICU. 16
System selection phase
The results of the comparison between systems were influenced by an event on commencing use of the Hospira infusion system in April 2010. During the first infusion programmed with these systems, air bubbles were detected in the distal extreme of the infusion line, which posed a risk of gaseous embolism to the patient.
From this moment, Hospira 3-month trial period was cancelled, the systems were isolated and infusion tests were performed in the PICU as well as at Hospira technical service. Bubbles appeared in every case where flow rate under 5 mL/h was programmed and disappeared on incorporating 0.2-µm filter in the infusion line, a measure proposed by Hospira as a solution for continuing the study.
However, systematic use of filters for administering drug concentrations lower than 5 µg/mL is not recommended; it is not recommended when the amount to administer over 24 h is under 5 mg, due to risk of under-dosage if part of the drug were to remain in the filter membrane; also, it is not recommended to infuse oily mixtures due to a risk of emulsion breaking. 17 As many of the drugs administered in the PICU comprise some of these characteristics and infusion flows in paediatrics tend to be under 5 mL/h, Hospira systems were not considered safe for use in paediatric patients, and CareFusion systems were chosen to continue the implementation project. Figure 2 shows the final study schedule.
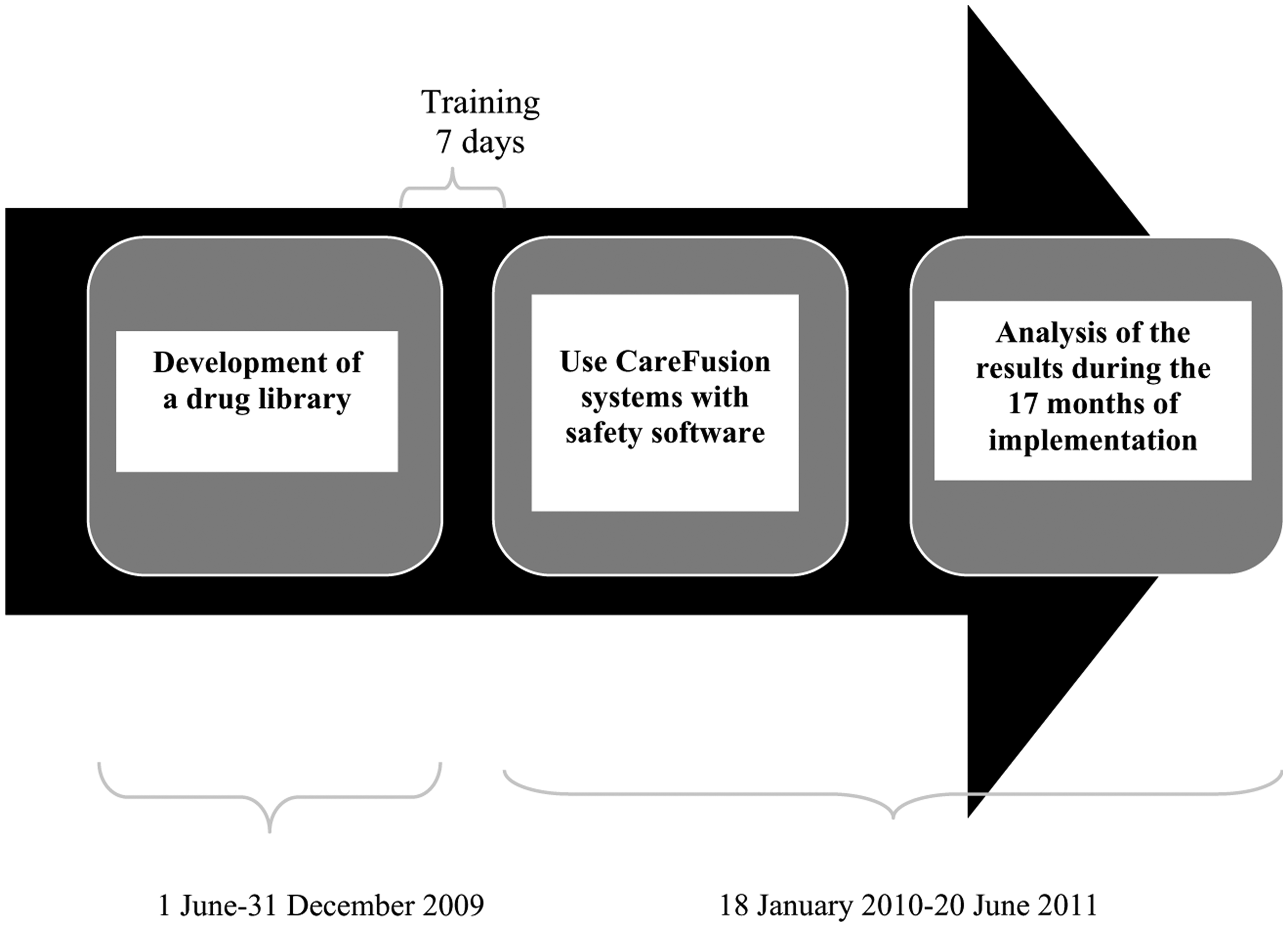
Final study schedule.
Despite this incident, some of the differential characteristics between both systems could be compared, even though the Hospira 3-month trial period could not be finished. Connectivity was the factor that favoured Hospira systems, as it allows real-time data transfer due to a server installation and the availability of Wi-Fi facilitating the unit work and the future integration of the technology,18,19 unlike CareFusion systems at the time of the study.
Regarding the nursing staff satisfaction survey, only the results corresponding to CareFusion systems are available as a consequence of suspending the Hospira study phase. After 6 months of use, 94 per cent of participants (96% participation) would recommend the technology implementation in other units, against 6 per cent who would not.
After 17 months of implementation, 97.6 per cent (78% participation) would recommend implementation in other units, against 2.4 per cent who were indifferent. Differences between both periods had no statistical significance (p = 0.252).
The percentage distribution of the remaining questions may be seen in Figure 3. Regarding the first 6 months of implementation, statistically significant differences were found in questions 4, 5, 6, 9 and 10, revealing a greater degree of user satisfaction with the technology after 17 months of implementation (p = 0.03, p = 0.00, p = 0.013, p = 0.001 and p = 0.047, respectively).
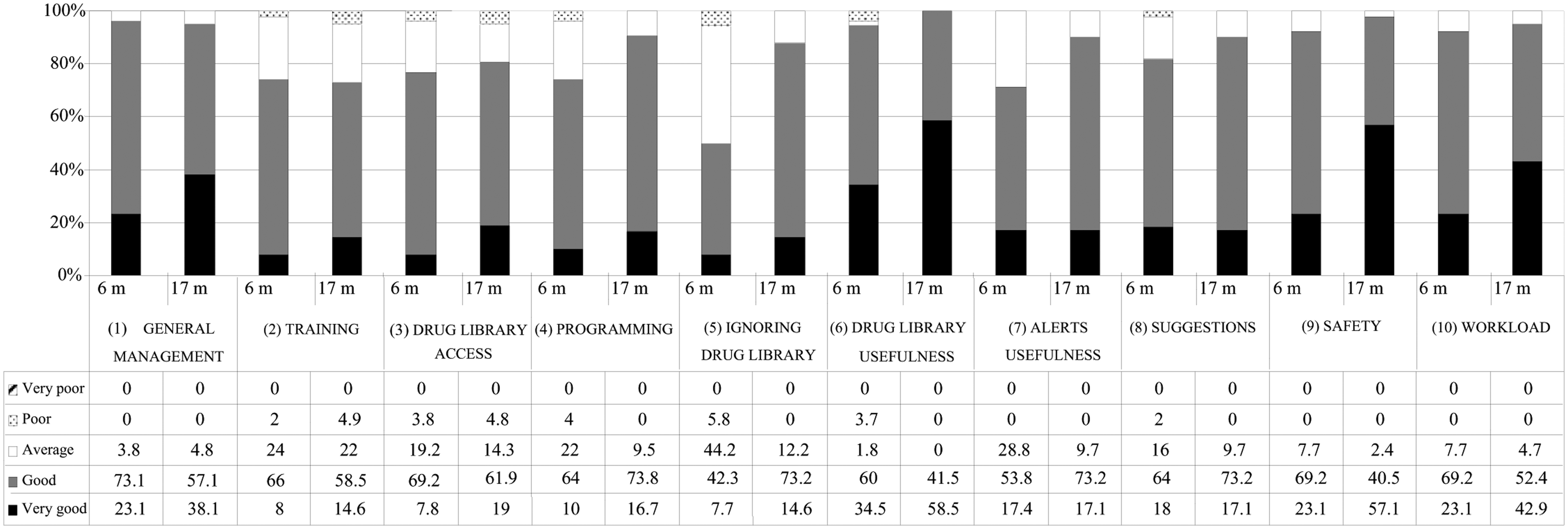
Answers to questions 1–10 from 6 to 17 months of system use questionnaires.
Analysis of stored information
Technology use quality analysis
The drug library compliance rate was 92 per cent, which means that 574,313 of all programmed infusions in the PICU (624,252) were performed using the drug library and therefore within the safety network.
Of the total soft limit warnings, 71.5 per cent (1543) were ignored compared to 28.5 per cent that entailed reprogramming. The total number of alerts was 2500.
Description of health-care setting
Most alarms were distributed between 10:00–13:00 and 19:00–21:00. Tuesday was the day with the highest number of warnings (411 alerts out of 69,638 infusions started with the drug library), with statistically significant differences as regards other days of the week, such as Monday (347 alerts out of 72,453 infusions started with the drug library) and Thursday (313 alerts out of 68,261 infusions started with the drug library). Figure 4 shows the alert distribution for the different months of the study period.
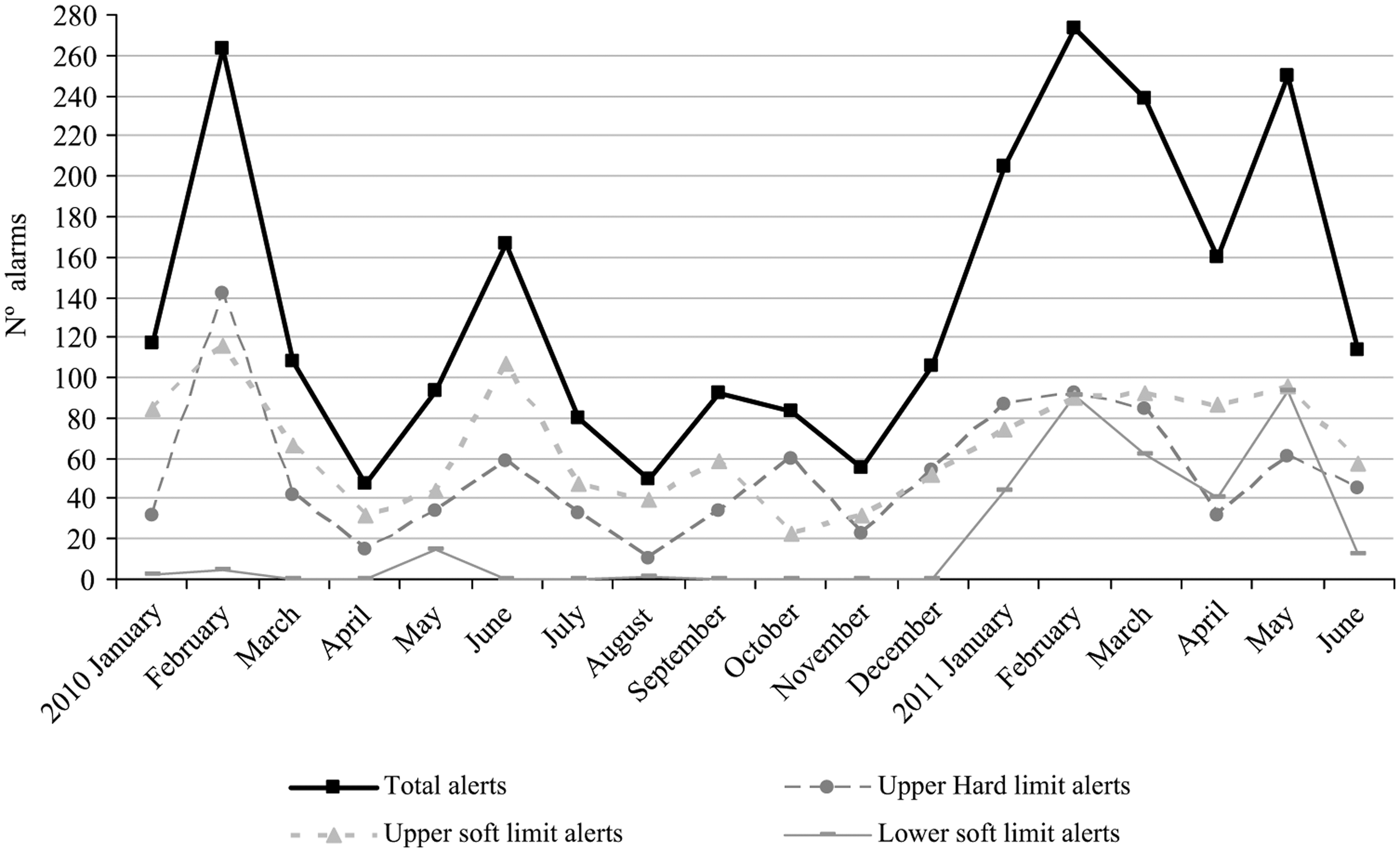
Detailed alert distribution for every month of study period (taken from data treatment Guardrails CQI v4.1 Event Reporter programme).
Chi-square test was used for percentage analysis showing statistically significant differences for the months of January and February as compared to other months (<0.001) and for June and December as compared to the preceding months (p = 0.0278 and p = 0.006, respectively).
Technology efficacy analysis
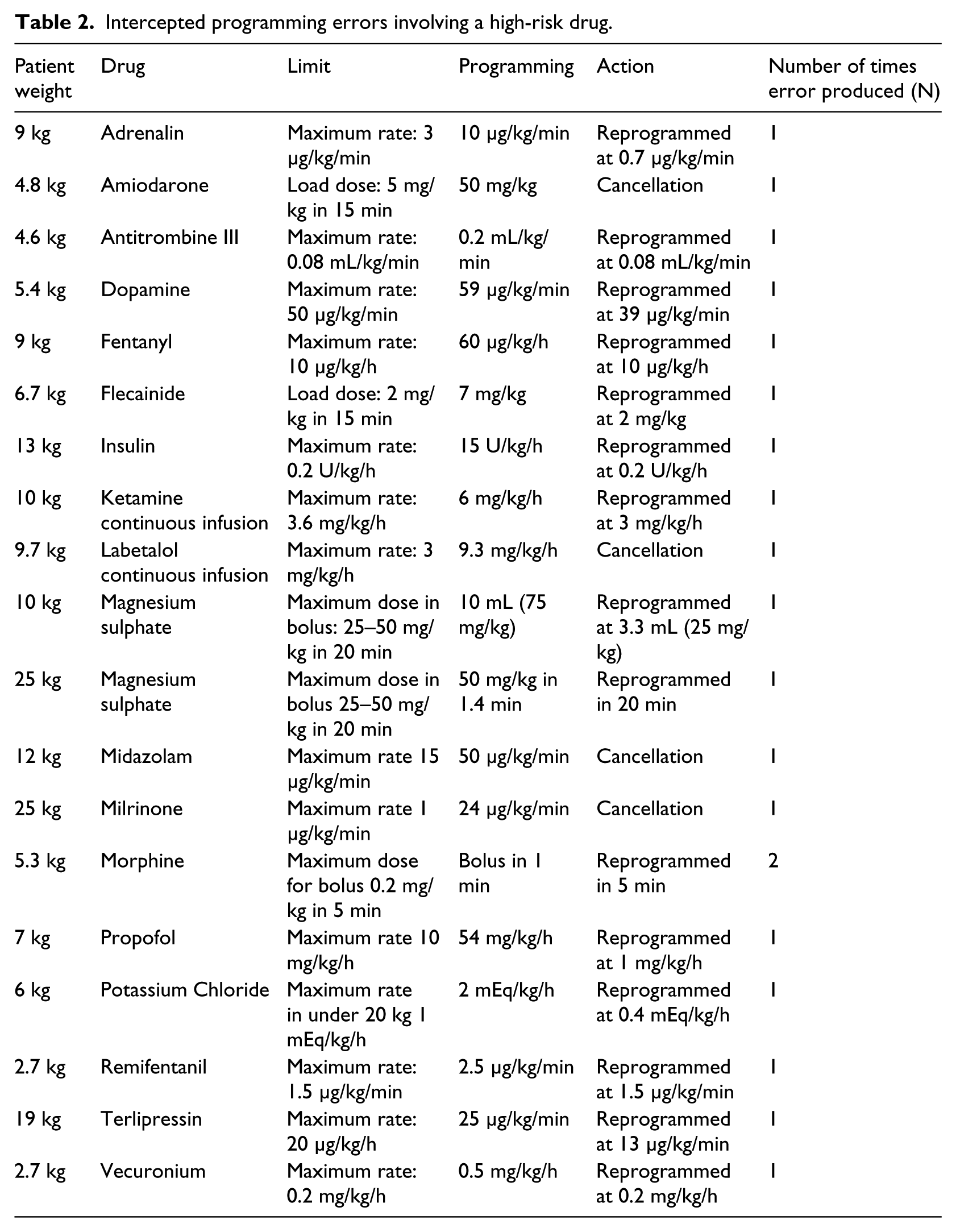
In total, 92 errors were intercepted, 44 of which involved a high-risk drug defined by ISMP. 20 Table 2 shows some examples.
Intercepted programming errors involving a high-risk drug.
According to our self-reporting system, none of the infusion-related programming errors had actually reached a patient. All changes made to the database were informed in writing to unit staff as they were made, and the information analysed along with conclusions was discussed at two meetings with the multidisciplinary team, medical and nursing staff from all shifts at 6 and 17 months of implementation in the unit.
Discussion
The incorporation of a drug library in an infusion system is the first step to migrating a conventional infusion pump into a smart pump. The availability of back-up documents, as guides for administering and compatibility charts, when migrating to intelligent technology is of great use in promoting understanding and commitment in the technology users.
The implementation of the intelligent technology in the unit was carried out with CareFusion systems. It should be noted that Hospira pumps are volumetric systems, therefore less accurate than a syringe pump when administering drugs at low rates. This fact probably caused the infusion flow discontinuity and not the use of the Hospira system as such, despite the technical specifications stating the aptitude of the system for administering small volumes at low flows in paediatrics.
Other authors have described the role of health organisations in the selection of the best infusion system from those available on the market; 21 wireless technology was one of the aspects that most conditioned their choice. Wireless technology can speed up the processes of uploading and downloading information from the pumps, which might be extremely disruptive with wired systems, and may also make smart pump integration with other computer devices available in the organisation easier. 22
The drafting of an administering guide stemmed from the nursing staff request, as most queries posed to the pharmacy department involved preparation of intravenous solutions, as reported by Da Silva et al. 23
The lack of viable lines in critical patients may require several drugs administered through the same infusion line, 24 and this in turn may cause incompatibility problems not detected by smart pump technology. Therefore, the availability of related reference documents may also contribute to increasing safety during the administering process.
As experience with the system increased, so did the positive evaluation on the utility of the drug library, similar to experience reported by Carayon et al. 25
We found statistically significant differences regarding the users’ perception of the increased level of safety provided and scarce repercussions on workload after 17 months of implementation even though users’ satisfaction was already high after the first 6 months of implementation (92% of users thought it was good or very good). No improvements were observed as regards the perception of user training for the technology. Training sessions could be reinforced periodically to cover training requirements for new or cover staff.
Technology use quality analysis
The drug library compliance rate (92%) was far superior to data published by other authors reporting percentages of around 30–40 per cent.22,26 Reinforcing training and encouraging nursing staff awareness on the use of these smart systems is key for increasing adherence to the technology,18,21 as the most important limitation is the possibility of programming infusions outside the safety network despite being incorporated in the system.
In our study, the percentage of reprogrammed infusions due to soft limit alarms was extremely high when compared to the 4–6 per cent published by Breland, 22 yet lower than 43.2 per cent reported by Fanikos et al. 27
In any case, the data obtained question the usefulness of the soft limits as nursing staff tended to ignore them, contrary to the hard limits which do oblige an infusion reprogramming, and therefore are directly involved in error interceptions. 28 In our study, overriding soft limits did not imply a significant error; nevertheless, periodic revision permits the elimination of those which, with clinical practice, are not considered to increase safety in programming and to keep those which suppose an added value to safe programming of certain drugs.
Description of health-care setting
Most warnings were distributed between 10:00–13:00 and 19:00–21:00, coinciding with drug administering times in the unit. However, in the study by Fanikos et al., 27 most warnings occurred during the shift change in the unit from 14:00 to 16:00.
It should be noticed that Guardrails CQI v4.1 Event Reporter software cannot provide the total number of infusions started in every hour interval but the total number of infusions started in a 24-h period. This is the reason why we should be cautious when interpreting the results related to the distribution of alerts by time of day. It seems that most warnings occurred during drug administering times in the unit, but we cannot draw conclusions about the significance of this association as we only present absolute numbers.
Tuesdays were the days with most warnings, with statistically significant differences as regards other days of the week. The multidisciplinary team was not able to find a reasonable explanation to justify this result. In the study published by Fanikos et al., 27 Sundays were the days with most warnings.
Regarding warning distribution in the different months of the study period (Figure 4), the greater part of alerts triggered during January and February 2010 was due to progressive user adaptation to the technology, this number decreased throughout the study period. Likewise, the trend towards increase observed in June and December, as regards the directly preceding month, can be justified by the incorporation of new staff unfamiliar with PICU work and the new technology and more susceptible to errors. Therefore, implementing this technology is key to increasing safety during holiday periods when less trained staff are involved.
Technology efficacy analysis
Nearly all identified real programming errors corresponded to alarms for exceeding UHLs, contrary to those reported by Breland, 22 where the examples described correspond to programming errors exceeding USLs.
Of the intercepted programming errors, 47.8 per cent involved a high-risk drug, proving the technology capacity in preventing potentially serious errors from reaching the patient.
Although there are many studies describing the potential of smart pumps for reducing the incidence of administering errors due to incorrect system programming, some authors doubt the usefulness, 26 as the factor limiting success of this technology is the very acceptance of this technology by nursing staff who are involved in its daily use. In our unit, the degree of acceptance and compliance with the technology was high.
Different studies coincide in stating that smart pumps are limited in detecting potentially serious medication errors when functioning in an isolated manner in a hospital setting, as it would only avoid those derived from incorrect programming. 19 One of the conclusions drawn from this work was that some of the errors were caused by a lack of technological integration among the infusion pumps and the computer tools, like CPOE, so that of the five steps that must be guaranteed during the administering phase (patient, medicament, dose, pathway and correct time), only the third step was guaranteed. 28 This fact, along with the possibility of programming infusions without using the drug library, constituted one of the fundamental limitations of this technology.29,30
Therefore, the efficacy of this technology depends not only on the system design itself but also on the environment in which the technology is implemented, as it is highly dependent on the percentage of users’ compliance.
Until hospitals develop interfaces allowing connection among the different technologies implemented, the drug use circuit cannot be closed nor the totality of medications errors occurring at different stages intercepted. Future studies comparing systems with a higher level of connectivity might determine to what extent maximising technology integration increases smart pumps’ capability to intercept infusion-related programming errors.
Conclusion
Smart pump technology implementation in a PICU has proven able to intercept potentially serious medication errors. Pharmacists’ leadership was key to the implementation process.
Performing an adequate selection of the provider and developing a specific drug library for each unit is key for efficacy.
One of the principal limitations is the possibility of programming infusions without the drug library. For this reason, the availability of back-up documents, communication among health professionals, constant result evaluation and future technology integration may contribute to adherence to this technology.
The results presented in this study may be the basis for the extension of this technology to other units which could benefit as the PICU.
Footnotes
Acknowledgements
The authors would like to thank all PICU staff from Gregorio Marañón General and Teaching Hospital for their collaboration and active participation in the development of this project.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
