Abstract
Background and Aims:
Arteriovenous access ischemic steal (AVAIS) is a rare complication that causes morbidity and threatens hemodialysis access usability in patients with end-stage kidney disease (ESKD). This study aimed to determine the incidence of AVAIS over a 15-year period and assess the access usability after surgical correction.
Methods:
Access operations between January 2007 and August 2022 at Helsinki University Hospital were reviewed. Demographics, clinical, duplex, and contrast examinations with brachial artery volume flows, finger pressures, and delays to surgery were evaluated. Symptom relief, complications, access closures/re-interventions, primary, secondary, and functional patencies were assessed. Endpoints were permanent cessation of access use due to complication(s), transplantation, closure, definitive occlusion, or death.
Results:
Among 2914 access-related operations, the overall incidence of AVAIS was 2.2%. At the first vascular consultation 52% had ulcer(s) or gangrene(s) resulting in 28 direct closures, and 30 corrections as follows: 20 proximalization of arterial inflow (PAI), 6 flow reduction procedures, 2 distal revascularization with interval ligation (DRIL), 1 distal radial artery ligation (DRAL), and 1 venous bypass. The median time from consultation to surgery was 18 days (range: 0–348 days) for direct closures and 43 days (0–170 days) for corrective surgery. The functional patencies after correction were 60% at 1 year and 55% at 2 years; the primary patencies were 45% and 28% and secondary patencies 61% and 57%, respectively. The functional patencies after PAI were 41% and 31%, respectively.
Conclusion:
In AVAIS, access preservation is challenging in ESKD patients with multiple diseases. Attention should be paid to the original choice of vascular access by considering each patient’s risks. Delay to vascular consultation and intervention should be minimized. PAI should be reserved for selected patients when no other option is preferable.
Context and Relevance
Vascular access ischemia is rare after access reconstruction. We reviewed the results of a 15-year period among 2914 access operations when most new accesses have been native distal fistulas. The incidence of ischemia was 2.2%. As many of these patients were multi-diseased having poor peripheral circulation, almost half had a direct closure. Among interventions in preserving function, proximalization of arterial inflow (PAI) was frequently performed but showed less effectiveness with advanced trophic lesions. Multidisciplinary work, patient education, and risk assessment before original access reconstruction are underlined because of high proportion of advanced lesions at first vascular consultation. Survival and access preservation were compromised, emphasizing the importance of preoperative decision-making with reconsideration of patient’s prognosis and other options.
Introduction
Arteriovenous access ischemic steal (AVAIS), a rare complication after arteriovenous access creation, occurs in 1%–10% in upper extremity accesses.1–7 Especially, females, patients with diabetes, atherosclerotic disease, and patients with upper arm fistulas or highly compliant outflow veins are prone to higher risks, but precise definition of “a high-risk patient” may remain unclear in an individual case.3,4 As fit patients receive renal transplants more evidently than individuals with multiple comorbidities do, the incidence AVAIS may increase in future.
Ischemic symptoms can appear after the initial creation or in matured accesses leading to increased morbidity, further interventions, and hospitalizations. 8 Several types of corrections for AVAIS have been established depending on the site of access location, its vascular status, and its function.1,2,4,6
Our aim was to determine the incidence of ischemic symptoms after access reconstruction over a 15-year period and assess the access usability for hemodialysis (HD) after surgical correction. Next, the delays from preceding vascular access reconstruction to vascular consultation and surgery for AVAIS, symptom relief, surgical complications, re-interventions, primary, secondary, and functional patencies were assessed.
Methods
The Review Board of the Helsinki University Hospital approved the study. Access operations performed between January 2007 and August 2022 at Helsinki University Hospital were identified in our prospectively collected vascular database. Patients treated for AVAIS were reviewed, with exclusion of ischemic symptoms resulting only from venous outflow obstruction. The follow-up ended at 31 January 2023. All demographic data were collected including age, sex, the date of preceding access creation, the etiology of renal disease, and the presence of other chronic diseases. The symptoms were graded from stage II to IV (defining stage I as asymptomatic steal), stage II as pain during HD/ exercise, stage III as rest pain, and stage IV as a patient with ulcer(s)/and/or gangrene(s).1,4,9 Pre-, per-, and postoperative brachial artery volume flows (BAVF), finger pressures, and clinical and contrast medium examinations were reviewed. As a potential parameter of access function, all fistulas were divided into three categories according to BAVF: (1) low flow: ≤500 mL/min, (2) normal flow > 500 up to 1300 mL/min, and (3) high flow > 1300 mL/min. For grafts, the limits for low flow were ≤800 mL/min, for normal >800 mL/min up to 1400 mL/min, and for elevated flow >1400 mL/min.6,9
Delay from vascular consultation to surgery and details of perioperative evaluations were established. Symptom relief, complications, finger amputations, access closures after correction, and re-interventions were collected with the data of the continuation of access use postoperatively in HD. Primary patency was defined as a patent access without any early or late occlusion, secondary patency as patency achieved either by percutaneous pharmaco-mechanical thrombolysis (combined with PTA) at the angio suite or by surgery without changing the inflow artery or the outflow vein, and functional patency as verified continuous function of the access in HD. The permanent cessation of access use/follow-up due to postoperative complication, renal transplantation, persistent neuropathy, closure, definitive occlusion, or death were established as endpoints. The collected data were processed by using the SPSS statistical analysis (SPSS version 29 for Windows; IBM, Armonk, 14 NY, USA). The statistical significance of any difference in Kaplan–Meier analysis was evaluated using the log-rank test (univariate analysis). Differences between groups were considered statistically significant for s5.
Protocol for AVAIS
Our clinical decision-making is based on the functional status of the access, the clinical, and duplex ultrasound (DU) findings including BAVF and detection of possible arterial/venous abnormalities (from inflow artery to outflow veins), finger pressure measurements, and contrast examinations, considering also the patient’s cardiovascular status, stage of ischemic lesion(s), and acuity of the onset of symptoms. In accordance with the guidelines of European Society for Vascular Surgery for AVAIS, we prefer a suitable flow reduction method in cases of elevated BAVF: revision using distal inflow (RUDI), proximal radial artery ligation (PRAL), or banding. Distal revascularization with interval ligation (DRIL) is considered in normally functioning (proximal) fistulas. Proximalization of arterial inflow (PAI) is used in accesses with lower to normal flow, and distal radial artery ligation (DRAL) in distal fistulas with normal flow and patent palmar circulation. 9 Pre-/postoperative BAVF are measured by standard DU and peroperatively with a standard transit time flow measurement system. All patients have at least one postoperative control visit at 2–4 weeks with further visits scheduled when needed.
Results
Between January 2007 and August 2022, there were 2914 access operations: 2053 surgical access creations, 575 corrections or re-reconstructions, and 314 closures. Among creations, the yearly proportion of native fistulas was ≥90% compared to grafts. The overall proportion of distal/forearm native fistulas (originating from either radial or ulnar artery) compared to proximal accesses (i.e. fistulas or grafts originating from brachial or axillar artery) was 80%/20%. Hemodialysis catheters increased from 13% to 16% during the study along with the national tendency. 10
Altogether 56 patients had 58 surgical interventions leading to correction or closure for ischemic symptoms, resulting in incidence of 2.2% (yearly variation 1.1%–4.7%), corresponding mean incidences of 1.9% for distal fistulas and 2.5% for proximal accesses, when excluding direct access closures for other reasons than ischemia (Fig. 1). The main difference between the direct closures/surgical corrections in demographics was the former having more males and type II diabetics and the latter having more equal male/female ratio and more type I diabetic patients (Table 1). In total, 52% of the patients had finger ulcers and/or gangrenes at the time of the first vascular consultation. In 28 cases, the access was closed directly, and in 30 cases, a surgical correction was attempted. Deaths during the follow-up were non-related to surgery.
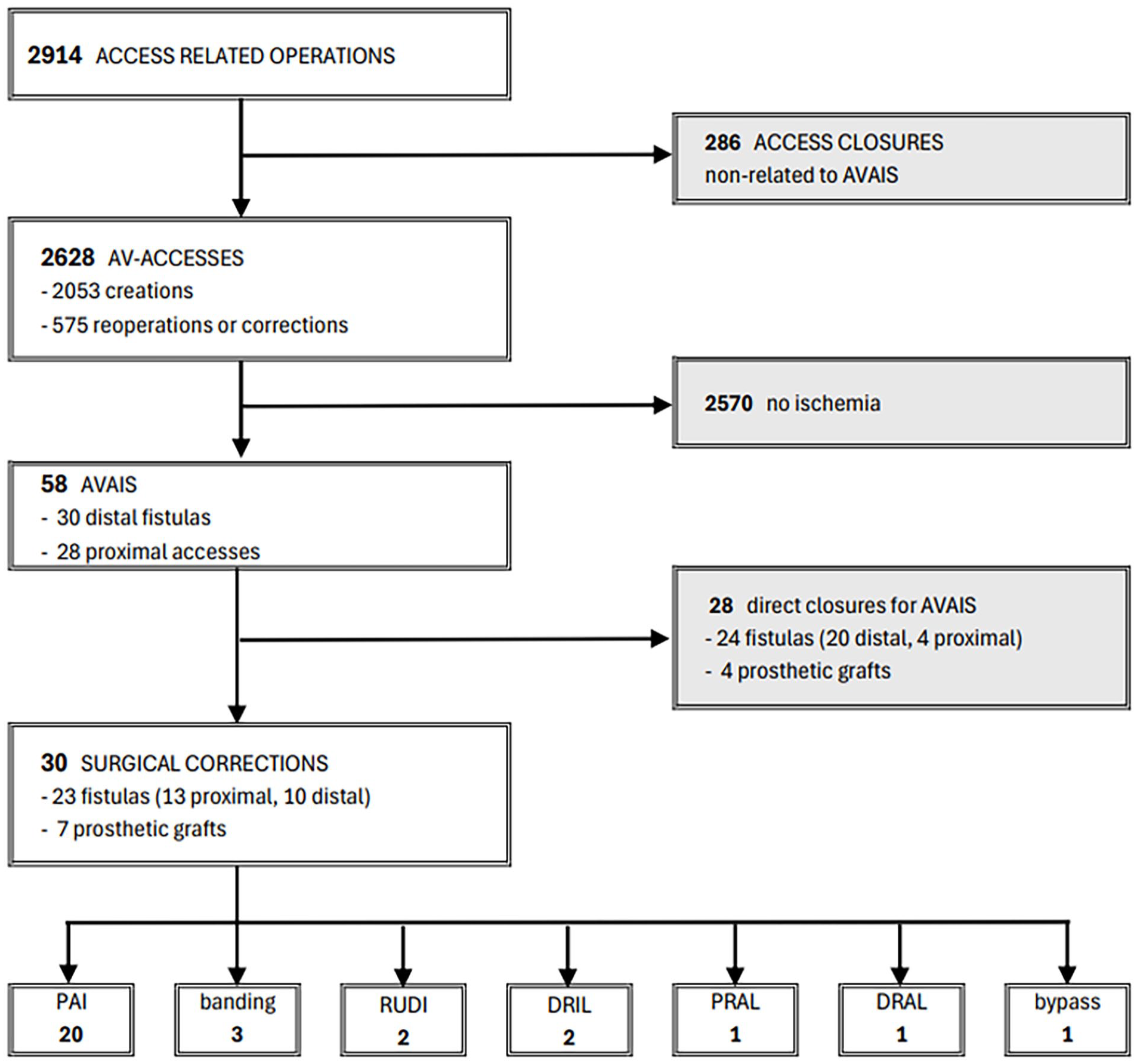
Access-related surgery between January 2007 and August 2021 at Helsinki University Hospital with postoperative AVAIS leading to interventions (58) in 56 patients. All corrected prosthetic grafts had their preoperative inflow from brachial artery.
Demographics, etiology of renal dysfunction, comorbidities, and treatment delays in patients with arteriovenous access ischemic steal treated with direct closure or surgical correction.
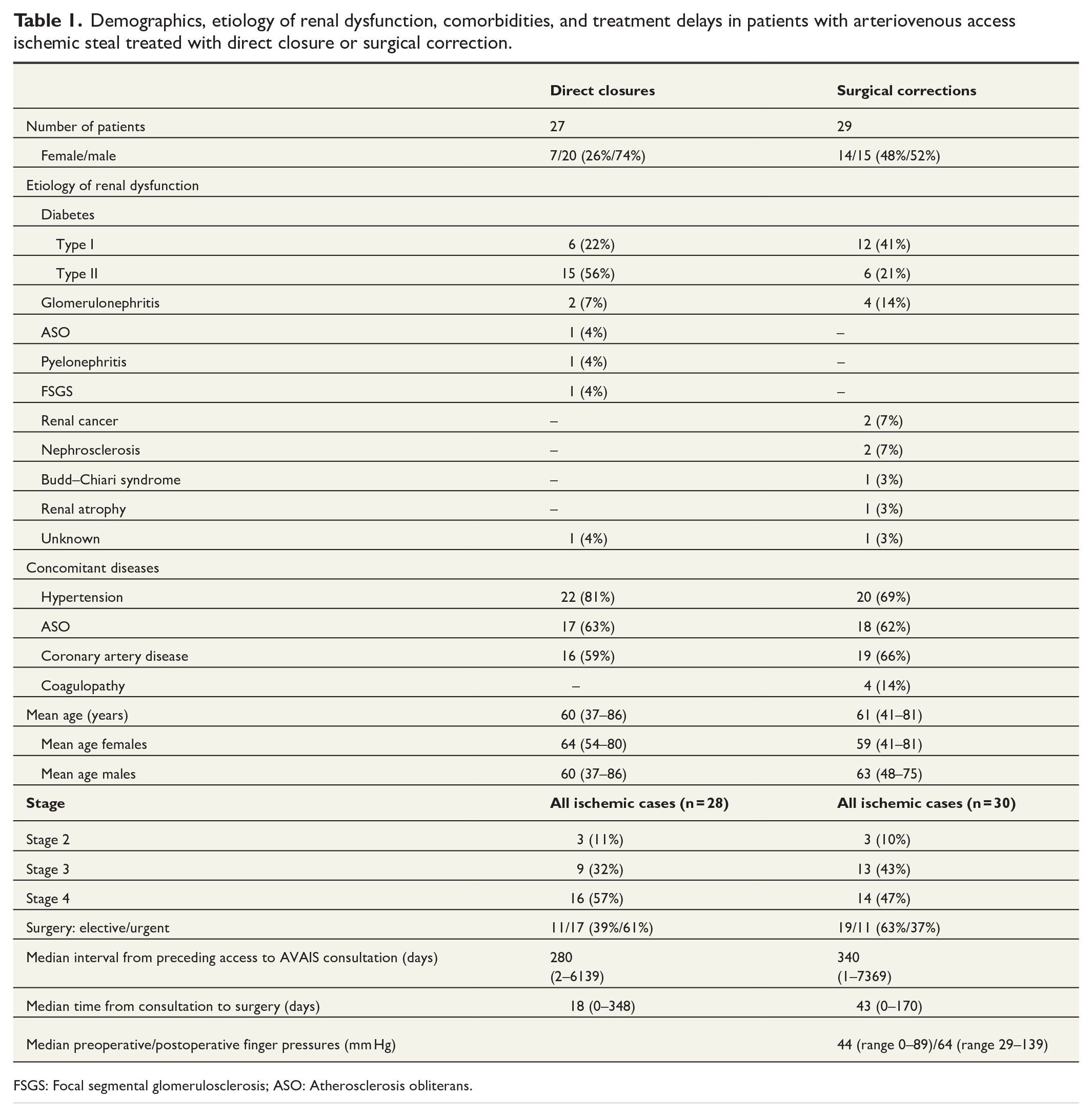
FSGS: Focal segmental glomerulosclerosis; ASO: Atherosclerosis obliterans.
Direct closures
There were 24 native fistulas (20 distal/4 proximal) and 4 prosthetic grafts originating from brachial artery in 27 patients who underwent direct closures (Fig. 1). Of the 27 patients, 20 were continuously on HD, 6 patients had a working transplant, and 1 patient was in the pre-dialysis phase. The demographics of these patients are summarized in Table 1. The reasons to perform direct closure were one of the following or their combination: sufficiently working transplant, severe ischemic symptoms with advanced trophic lesions, unsupportable pain, surgical correction considered impossible, the patient not willing any corrections, and/or pre-dialysis phase of end-stage kidney disease (ESKD). Preoperative percutaneous arterial dilatations were technically feasible only in one patient without subsequent symptom relief. After closure, 19 patients remained in HD, 6 with working transplant, 1 changed to peritoneal dialysis and 1 patient remained in pre-dialysis treatment. The overall survival of these patients is presented in Fig. 2.
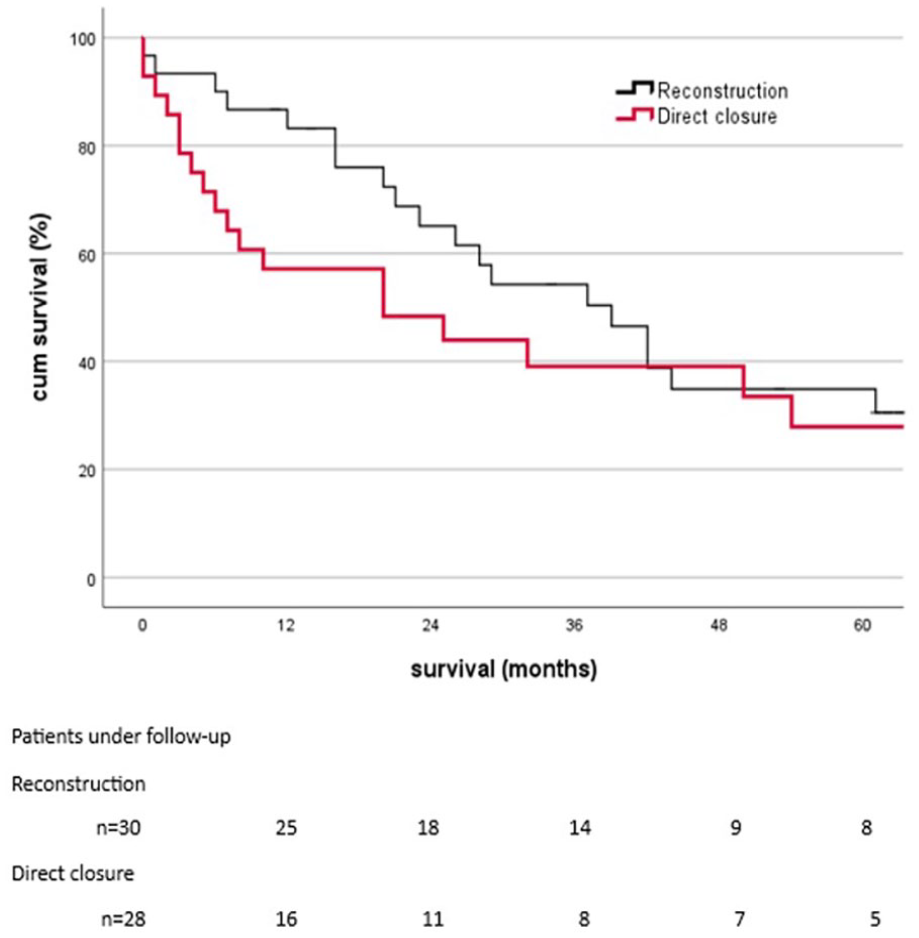
Overall survival in 27 patients after access closure for hand ischemia (red curve) and in 29 patients after surgical correction for ischemic symptoms (grey curve).
Surgical corrections
Twenty-nine patients had 30 attempts of surgical correction of the access. There were 13 proximal/10 distal native fistulas and 7 grafts originating from brachial artery. Of the corrective procedures, 20 underwent PAI, 3 banding, 2 RUDI, 2 DRIL, 1 DRAL, and 1 PRAL and 1 brachioradial bypass (Fig. 1). The demographics of these patients are presented in Table 1. Preoperative duplex was performed in all cases, finger pressures in 87%, angiography (combined with phlebography when needed) in 77% (Fig. 3), and contrast-enhanced magnetic resonance imaging in 7% of the cases. Preoperative PTA was technically feasible only in 7% without sufficient symptom relief. The median time from consultation to surgery was 43 days, but in severe stage III–IV cases, the delay from the decision to operate to surgery was shorter (median 15 days, range: 0–49 days). In 23% of all interventions, the ischemia appeared in combination with preoperatively measured high BAVF. Immediate symptom relief was detected in 93% of all surgically corrected cases with elevation of postoperative finger pressures in 92%. The functional patency rates for 1 year and 2 years were 60% (SE ± 9.5)/and 55 % (SE ± 10.1) (Fig. 4). The primary and secondary patencies were at 1 year 45% (±9.6 SE) and 61% (±9.8 SE) and at 2 years 28% (±9.9 SE) and 57% (±10.0 SE). In univariate analysis, we could not show any differences in access survival in patients with or without diabetes (p = .217), ASO (p = .251) or severity of symptoms, that is, with or without trophic lesions (p = .485). Two of 29 patients were lost from follow-up (after 3 months/after more than 3 years) with well-functioning corrected accesses.
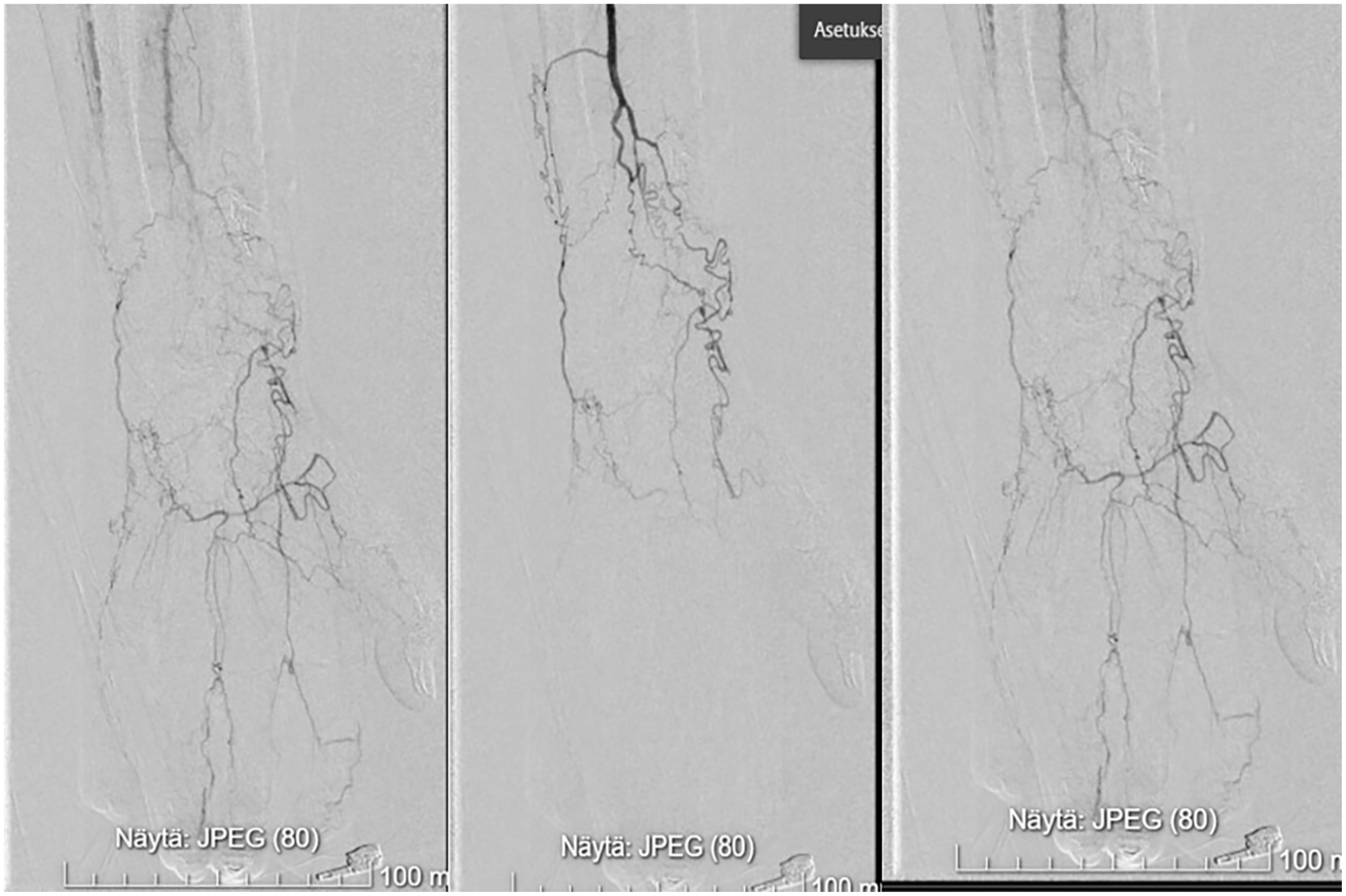
Preoperative angiography in a patient with brachiobasilic fistula with brachial artery volume flow (BAVF) of 1600 mL/min. After compression of the fistula, only interossei artery is patent and collaterals are present toward digital arterial supply. Proximalization of arterial inflow was performed with 4-mm ePTFE prosthesis using axillar artery as inflow. The postoperative BAVF was 1100 mL/min.
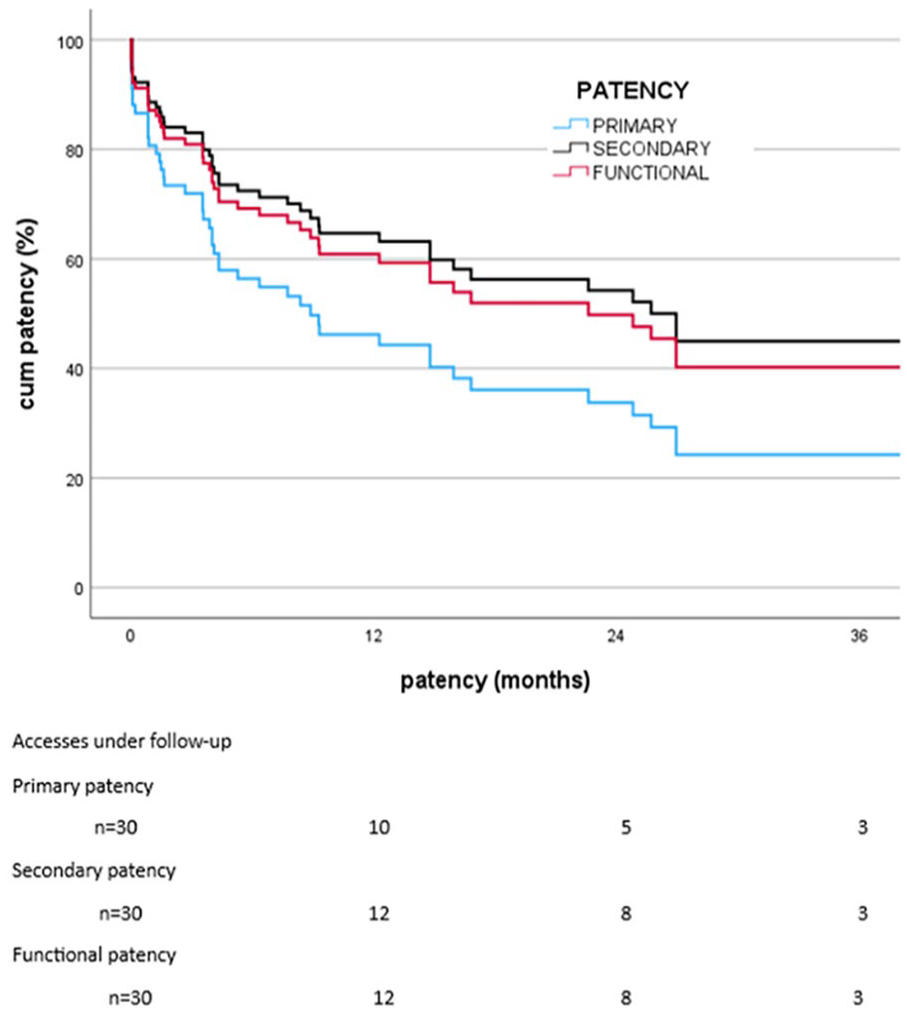
Primary, secondary, and functional patencies after surgical correction in 30 accesses (29 patients) operated on from January 2007 to August 2022 at the Helsinki University Hospital, Finland.
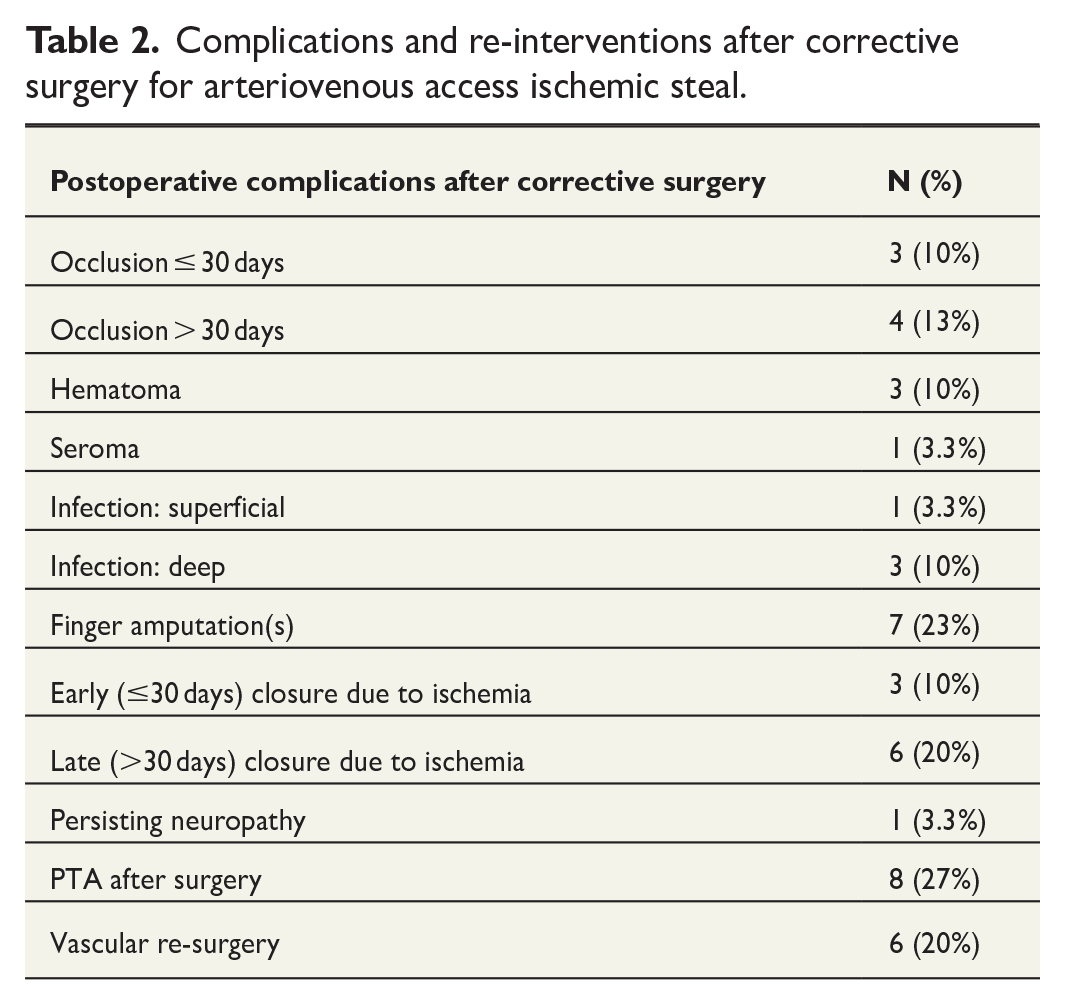
Complications and re-interventions after corrective surgery are summarized in Table 2. All finger amputations (7/30 cases) had stage IV lesions already preoperatively. The three early closures were performed due to continued symptoms, all in PAI group with poor preoperatively diagnosed palmar circulation, and further six patients due to persistent/recurrent symptoms. The overall survival of these patients is presented in Fig. 2.
Complications and re-interventions after corrective surgery for arteriovenous access ischemic steal.
PAI
PAI was our most frequent corrective procedure (67% of all cases) consisting of seven proximal native fistulas, seven proximal prosthetic accesses, and six native distal fistulas. The female/male ratio was equal. In total, 95% of these patients were at stage III or IV already at the first vascular consultation (10 at stage III, 9 at stage IV, and 1 at stage II). Out of the 20 patients who underwent PAI, 11 had type I and 4 type II diabetes. Among all 20 patients, preoperative arterial disease was diagnosed preoperatively in 95% (peripheral artery disease and/or coronary artery disease). The patients in this group were younger (median age 59 years, range: 41–81 years) than the non-PAI patients in surgically corrected group (median 67 years, range: 54–76 years) (p = .038). In total, 75% of all PAI cases had preoperatively normal BAVF up to 1000 mL/min (Fig. 5), 20% had low flow (<500 ml/min), and 1 patient with brachiobasilic fistula had high flow (1600 mL/min) before PAI (Fig. 3). At the arterial inflow site, 4-mm ePTFE prosthesis was chosen in 15/20 cases, 6-mm ePTFE in 4, and a basilic vein in 1 patient. The functional patencies of PAI were 41% (SE ± 12.7)/31% (SE ± 13.0) at 1 year/2 years.
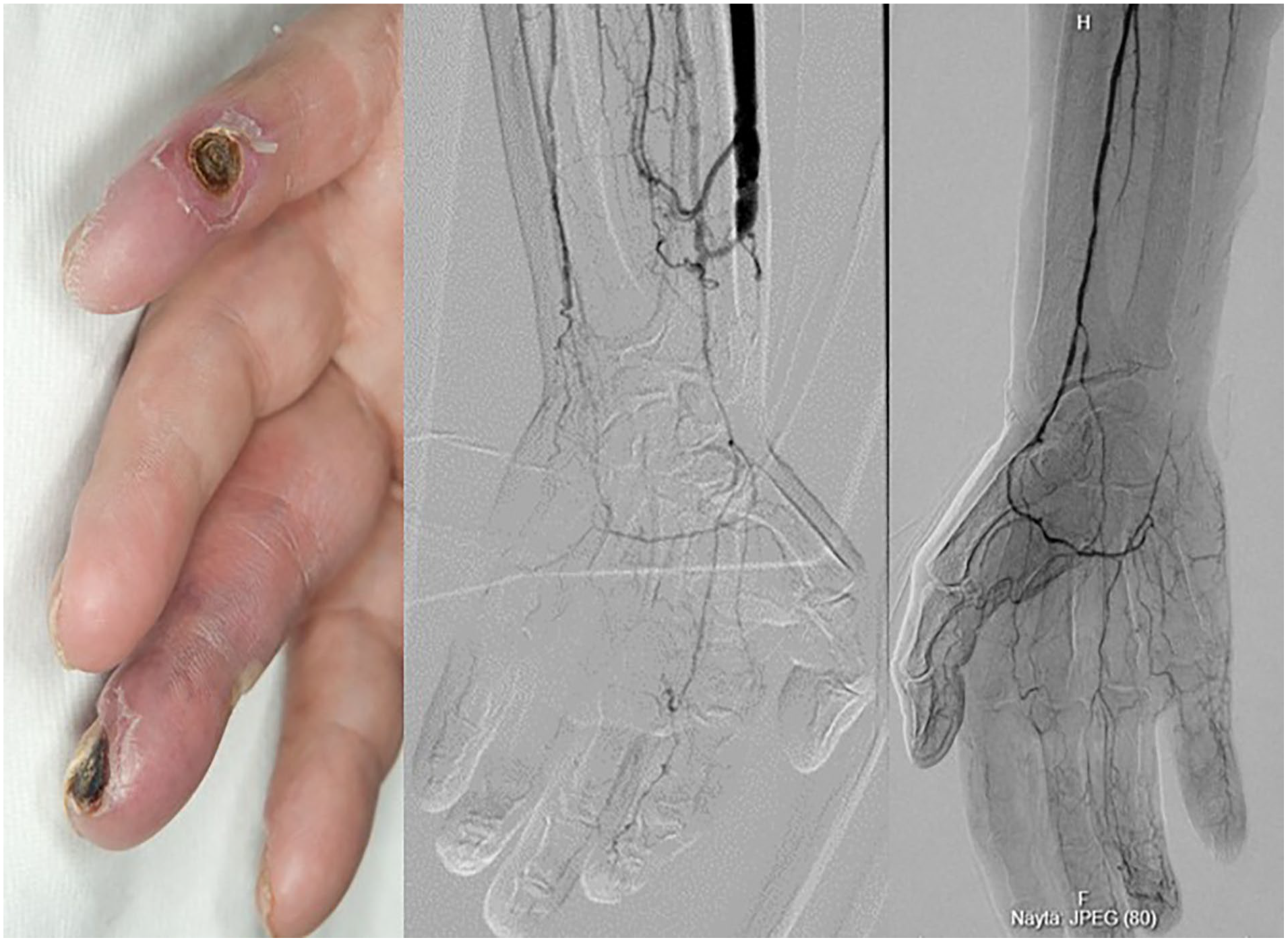
Gangrenated multiple ulcers in a patient with type I diabetes and left radiocephalic fistula with normal brachial artery volume flow (on the left). Distal arteries are heavily calcified and digital arteries at the third and fifth fingers barely detectable in the preoperative angiography (in the middle). PAI was initially successful for the symptom relief, but due to advanced gangrenes/insufficient digital circulation combined with infection, the fistula was closed after being used for 6.5 months. Later, the same patient developed finger gangrenes in his right hand without any preceding vascular surgery with similar distal angiographic findings (on the right).
DRIL, bypass, and DRAL
In normal-functioning accesses, DRIL was selected in two cases with good symptom relief and functional patencies of 808 days/466 days. Disseminated malignancy with central outflow obstruction led to closure in the former and recurrent ischemia in the latter (reoperation with banding). A distal brachioradial venous bypass to distal radial artery was needed in one patient with a well-functioning brachiocephalic fistula, with 1216 days functional patency until lost from follow-up. Another patient with well-working distal radiocephalic fistula with a patent palmar arcade had DRAL functioning until the end of the study (180 days).
Banding, RUDI, and PRAL
For high BAVF, banding was performed in three cases, of which two were functional for 797 days/899 days (endpoint: death). The third case (with previous DRIL) worked up to 130 days (closure for persisting ischemia). Among other flow reduction procedures, RUDI was performed in two patients and PRAL in one patient with good functional patencies of 625 days (until death)/249 days (until end of the study) and 2685 days (no access follow-up at our center after transplantation).
Discussion
Ischemic problems may appear early or delayed after vascular access reconstruction. The early onset can result from either flow diversion toward the venous vasculature, with or without arteriopathy, or it can be related to a very rare complication of ischemic monomelic neuropathy due to acute ischemia in vasa nervorum of the peripheral nerves.1,4 Although arterial inflow stenosis is rare in AVAIS, it must be addressed as a crucial part of the treatment. In the functional point of view, AVAIS may occur under normal, high, or even low volume flow of the access, and poor and/or reversed arterial collateral flow may contribute to ischemic symptoms.4,6 Percutaneous angioplasty can be an option before any other operative interventions are performed 11 but it may not necessarily resolve ischemic symptoms, especially when multiple atherosclerotic lesions, diabetic microangiopathy, mediasclerosis, and/or dysfunction of the access are involved.
Among our patients, the incidence of ischemia after access creation was low (2.2%). The patients selected to surgical corrections mainly presented with a functioning access with normal BAVF. The most frequent corrective procedure was PAI as it can be considered for both distal and proximal vascular accesses, also in cases with severe distal arteriopathies when no other option for correction is possible. As most of these patients had either diabetes or atherosclerosis/coagulopathy with no proper outflow artery for DRIL, and another type of correction was considered unavailable, PAI was attempted. This may explain the lower functional patency among our corrected accesses compared with previously reported series at least over 30 days follow-up.5,12–14 On the other hand, in some cases, a different solution, such as access reconstruction on the contralateral side, could have been an alternative or, according to the patient’s risk profile, other options for kidney replacement therapy could have been considered. 15 As expected, the proportion of distal fistulas was only one-third among the surgically corrected cases, but among the patients with direct closures, the proportion was 71% reflecting our high proportion for distal native fistulas (80%) among our vascular accesses and the complexity of involved diseases with compromised prognosis. The survival, especially among non-transplanted patients with direct closures, was poor, emphasizing the challenge in decision-making in these multi-diseased patients.
In the prospective study by Zanow et al., the most frequently performed procedures were flow reductions (54%), and due to inclusion criteria, PAI was considered possible in 23% of their cases (30/133). In total, 37% of them had ulcers or gangrenes, all of them having normal preoperative angiographies, access function, and BAVF. In this setting, PAI was successful also on long term. 13 Later, Thermann et al. 14 noticed that PAI may not work in patients with severe tissue loss, although elevation of finger pressures with symptom relief may be evident, which was our finding as well. In this study, all flow reduction procedures (banding, RUDI, and PRAL) succeeded in preserving function of the access, although in one of six patients (with severe ASO, type II diabetes, and previous ipsilateral DRIL), the fistula was closed after banding due to persistent symptoms after 4.3 months. Furthermore, as expected, in our two DRIL/one DRAL cases, the BAVF remained unchanged in pre-/postoperative measurements. Thus, in accordance with previous publications, BAVF can be a valuable parameter among preoperative evaluations in the treatment of AVAIS to guide clinical decision-making.6,9,12,13 As reported earlier, complications can be numerous after PAI in real-life practice, 5 but when poor distal arteries are detected already preoperatively, the principle of PAI in the prevention of ischemic events is useful when planning the patient’s next vascular access.16,17
A retrospective study by Leake et al. addressed to the incidence of ischemia in 3000 consecutive access operations with 30 days follow-up. The incidence of ischemia raised from 4% to 6% during the 10-year period, a phenomenon also noticed in our study. Most patients were female (62%) and/or diabetic (68%) with upper arm fistulas (80%). One-third was directly ligated, and symptom relief was best after ligation or DRIL. Their recommendation was DRIL among surgically fit patients. However, their experience with PAI was limited (12 cases) and only one-third of them had ulcers or gangrenes. The symptom relief and access preservation were good in their patient population, but their report included neither long-term follow-ups nor BAVF measurements. 5 In our PAI population, the patients were younger than in these previous series, and possibly due to undeveloped collateral circulation, the healing capacity of our patients may have been impaired. In many of our patients, preoperative angiographies showed impaired/heavily calcified distal arteries, or occluded distal ulnar artery not suitable for PTA, and/or poor digital circulation. Therefore, it may be difficult to compare these earlier series to the present because of the differences in patient populations.
Delays may potentially threaten the preservation of the vascular access and increase morbidity. Recently, delay to surgical intervention among 21 grafts (either correction or closure) concerning ischemic complications after vascular access surgery patients was focused on. 17 The proportion of forearm fistulas was only 26% the majority being upper arm fistulas and various grafts. Among their 21 grafts, 57% had a surgical intervention (either closure or correction) by 1 month and 71% by 3 months. In our study, among patients with direct closures, the median delay from consultation to surgery was 18 days, and in severe stage III–IV cases, the delay from clinical decision to a corrective procedure was acceptable (median 15 days). The delays can be explained by intentions to continue with observation in selected cases and subsequent investigations for procedural planning. In our experience, many delays often depend on various non-access-related issues (severe other clinical health disturbances, compliance, etc.) and not only on delays in health care services. However, all unnecessary delays should be avoided by active patient and HD staff education, by dedicated multidisciplinary team working and by educating specialized vascular access nurses/consultants, 18 which is our ongoing focus as well. As previously emphasized by Lok et al., 15 (re-)considering the patient’s individual needs in kidney replacement therapy for her or his lifetime could help us in avoiding undesired complications, especially in fragile patients.
Recently, Bachmann et al. 19 suggested that PAI would be possible in any BAVF. They performed 71 PAI cases for access-related ischemia between 2017 and 2023. After excluding patients with less than 30 days follow-up, 64 patients were analyzed. In total, 95% of the patients had fistulas and 5% grafts before PAI. The proportion of clinically mild ischemia was high as 25% of the PAI cases were at stage I (blue hand, no pain), 16% at stage II, 33% at stage III, and only 27% at stage IV before intervention. Preoperatively, DU was performed only selectively, no BAVF were measured, and their report did not include other types of interventions performed for ischemia during their study period. The proportion of type I/II diabetes was 3 versus 48 patients, and the number of patients having other arterial diseases than coronary artery disease (50%) was not reported. Not surprisingly, compared to our more diseased population, their results with PAI were better with primary/secondary patencies (after first month) being 81%/87% at 1 year and 71%/84% at 2 years.
There are some limitations in this study. First, the number of patients with ischemic problems was limited reflecting the low incidence of ESKD in Finland. Second, the data were collected during a longer period, thus patient populations in the beginning of the study were not comparable to the patients in the last study period. Furthermore, occasional patient among surgical corrections may have had ischemic monomelic neuropathy combined with lowered finger pressures when a direct closure is preferable. Finally, our interventional databases did not include data on patients treated only conservatively/only with percutaneous angioplasty, and thus, the incidence of AVAIS may remain underestimated.
In conclusion, in our study, the incidence of AVAIS was acceptably low. As renal transplantation is more available for fit patients, those remaining in HD are increasingly frail. Attention should be paid to the original choice of vascular access by evaluating each patient’s individual risks. The delays from consultation to interventions should be minimized by improving cooperation with members of multidisciplinary teams. After correction, the patients should be carefully observed. Finally, we consider sparing PAI for selected cases when other options are not preferable and before presenting with advanced stage IV lesions. By recognizing those patients who will benefit from corrective procedures, we will be able to diminish patients’ morbidity and our health economic burden.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A grant from Finnish Kidney and Liver Foundation (Suomen munuaissäätiö) received by Dr Eeva-Maija Weselius in 2022.
Clinical trial registration and ethical approval
Received/registered 27.12.2021 by the number HUS 6865, under the name “Functional maintenance of surgically created native vascular accesses for hemodialysis. Benchmarking at Helsinki University Hospital before the era of endovascular creation of vascular accesses.” (In Finnish: ”Kirurgisesti operoitujen hemodialyysissä käytettävien natiiviveriteiden toiminnan ylläpito. Esikuva-analyysi (benchmarking) Helsingin ja Uudenmaan sairaanhoitopiirissä ennen endovaskulaarisesti tehtävien natiiviveriteiden aikakautta”)/Eeva-Maija Weselius. Patient confidentiality and informed consent statements: As this was a retrospective study performed solely by the research material derived from registries and patient data with ethical approval of Helsinki University Ethical Committee, no informed consent of the patient was needed. This project licensed to continue up to 31 December 2024.
