Abstract
Background:
Tailoring surgical treatment is mandatory to optimize outcomes in chronic pancreatitis. Total pancreatectomy (TP) offers pain relief in a subset of patients. TP with islet autotransplantation (IAT) has the potential to reduce the burden of postsurgical diabetes. We present the first Scandinavian prospective study assessing outcomes following total pancreatectomy and islet autotransplantation (TPIAT) in chronic pancreatitis. Our aim was to assess short- and long-term outcomes following implementation of a nationwide program of TPIAT at a tertiary reference center for pancreatic surgery in Norway.
Methods:
A prospective, observational single-center study enrolling consecutive patients undergoing TPIAT for chronic pancreatitis at Oslo University Hospital. The selection of potential candidates for TPIAT was based on discussions at multidisciplinary team (MDT) meetings, focusing on tailored surgery in chronic pancreatitis. Patients were finally evaluated in a dedicated TPIAT team. The outcome measures included pain relief, quality of life (QoL) assessed by EORTC QLQ-C30, complications, and glycemic control.
Results:
Between August 2017 and November 2022, 15 patients underwent TPIAT. The follow-up rate was 87% with a median follow-up of 26 months (range = 14–65). Pain relief was achieved in 92%. EORTC QLQ-C30 analysis revealed clinically significant improvements in 28 of 30 domains, particularly in pain and role- and social-functioning. The Clavien-Dindo ≥IIIa complications occurred in one patient. There was no 90 days mortality. All patients maintained C-peptide positivity, although none of the patients reached insulin independence.
Conclusion:
TPIAT was as a safe and effective treatment for a selected group of patients with chronic pancreatitis, providing substantial pain relief and enhanced QoL. Islet autotransplantation prevented complete insulin deficiency, reducing diabetes severity postpancreatectomy. Dedicated chronic pancreatitis MDT meetings were key factor in the success of the program.
Context and relevance
This is the first prospective Scandinavian study focusing on total pancreatectomy and islet autotransplantation (TPIAT) in treatment of chronic pancreatitis. The aim was to assess outcomes in the first 15 consecutive patients following implementation of a nationwide TPIAT program and to outline the logistics and experience in building up this procedure at a Hepato-Pancreato-Biliary (HPB) center.
After more than 2 years follow-up, pain relief was achieved in 92% of patients treated with TPIAT and all patients maintained endogenous insulin production with a significant improvement in quality of life (QOL) and few complications. Regular chronic pancreatitis multidisciplinary team meetings and dedicated TPIAT teams were key factors in the success of the program.
Introduction
In surgical treatment of painful chronic pancreatitis, most patients benefit from parenchymal-preserving approaches. 1 In selected patients, total pancreatectomy (TP) might be the only surgical option.2,3 The primary objective of TP is to alleviate pain and improve quality of life (QoL), but the potential severe metabolic consequences following this procedure warrant careful consideration. 4 By combining TP with islet autotransplantation (IAT), the burden of postsurgical diabetes may be reduced. 5
Since the first report of total pancreatectomy and islet autotransplantation (TPIAT) in chronic pancreatitis in 1977, 6 significant progress has been made regarding postoperative health-related QoL, pain relief, and preservation of metabolic function.5,7,8 Despite its potential benefits, TPIAT is performed at only a few centers worldwide, with particular interest and development of the procedure in North America.7,9,10 During the last decades there has been a growing utilization of TPIAT also in Europe.11 –13 This study is the first Scandinavian prospective study focused on TPIAT in chronic pancreatitis. The aim was to assess the outcomes following the implementation of a nationwide treatment program of TPIAT in tailored surgery for chronic pancreatitis. In addition, the study details the logistics and our experience in establishing a TPIAT team at a tertiary reference center for pancreatic surgery in Norway.
Methods
Study design and study population
Consecutive patients undergoing TPIAT for chronic pancreatitis or recurrent acute pancreatitis from August 2017 to November 2022 at the Department of Hepato-Pancreato-Biliary (HPB) Surgery, Oslo University Hospital (OUH), were enrolled in this prospective observational study.
Norway has a universal healthcare system and a population of 5.4 million. OUH is the largest HPB center, and the only national center for organ transplant. Potential TPIAT candidates from all four Norwegian health regions were referred to OUH and evaluated at the chronic pancreatitis-MDT meeting where eligibility for TPIAT was determined. This article was completed in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14
Ethical approvals and informed consent
This study was approved by the Regional Ethical Committee (2019/1037) and the institutional Data Protection Officer for Research (2017/2867). All patients signed written, informed consent.
Indication for TPIAT
The decision to perform TPIAT was based on an evaluation of patient-reported symptoms, medical history, and radiological findings, following the Minnesota TPIAT criteria. 7 We were reluctant to recommend surgery in cases of prolonged symptom duration 15 but did not use a rigid, absolute cut-off based solely on the duration of pain.
Surgical decision-making adhered to a standardized treatment algorithm, as previously described. 16 TP was considered in cases of painful small duct disease without focal anatomical changes. For patients with acute recurrent idiopathic pancreatitis or suspicion of autoimmune etiology, a comprehensive evaluation was undertaken to exclude conditions treatable by medication before TP was recommended. In cases where both PRSS1 mutation and pain were present, TP was deemed necessary to prevent malignant transformations in the residual pancreatic tissue over time. Patients requiring insulin were not found eligible for TPIAT. In such cases, TP without IAT was considered.
Preoperative workup
Computed tomography (CT) served as the reference standard for the radiological workup. Magnetic resonance imaging (MRI) was performed in selected cases. Genetic testing to assess mutations in SPINK1, CFTR, and PRSS1 was conducted in patients with an unknown etiology of chronic pancreatitis. Laboratory tests to evaluate metabolic function prior to TPIAT included HbA1c (%), fasting blood glucose (mmol/L), and C-peptide (ng/mL) levels.
All patients were evaluated at the outpatient clinic by a TPIAT-dedicated HPB surgeon, followed by additional preoperative counseling. Once all relevant information had been gathered, patients underwent evaluation at a specialized TPIAT-MDT meeting (Fig. 1). If consensus was reached, the patient was scheduled for surgery. At inclusion, a written informed consent was obtained, baseline patient characteristics were recorded, and EORTC QLQ-C30 questionnaire was completed. The etiology of chronic pancreatitis was classified according to the Mannheim classification system. 17 Preoperative opioid and non-opioid analgesic consumption was registered. Total daily dose of opioids was registered as oral morphine equivalent dose (OMEQ). Patient’s data were prospectively registered into the institutional chronic pancreatitis database.
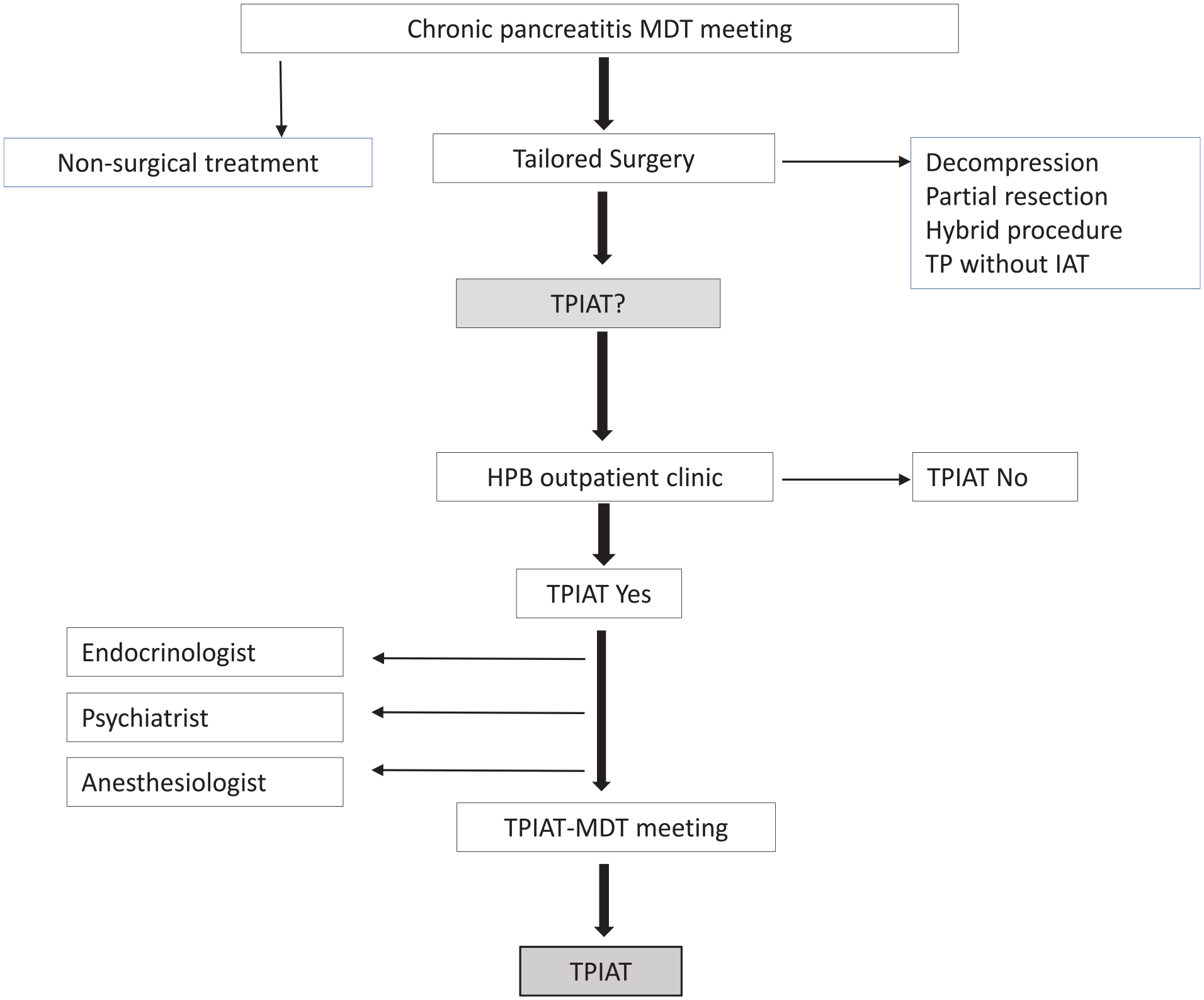
Flow chart TPIAT workup.
TPIAT procedure
Surgery was conducted by a team consisting of both HPB and transplant surgeons. During the implementation period, the same HPB surgeon was in charge of all procedures. TP was performed as previously described. 18 It was crucial to maintain blood supply to the pancreas until immediately before its removal in order to minimize warm ischemia time and improve preservation of the tissue for the islet isolation procedure. Before restoring gastrointestinal and biliary continuity, a dual lumen hemodialysis catheter (Mahurkar®) was introduced into the portal vein (Fig. 2), accessible either from the inferior mesenteric vein or via the umbilical ligament. 19
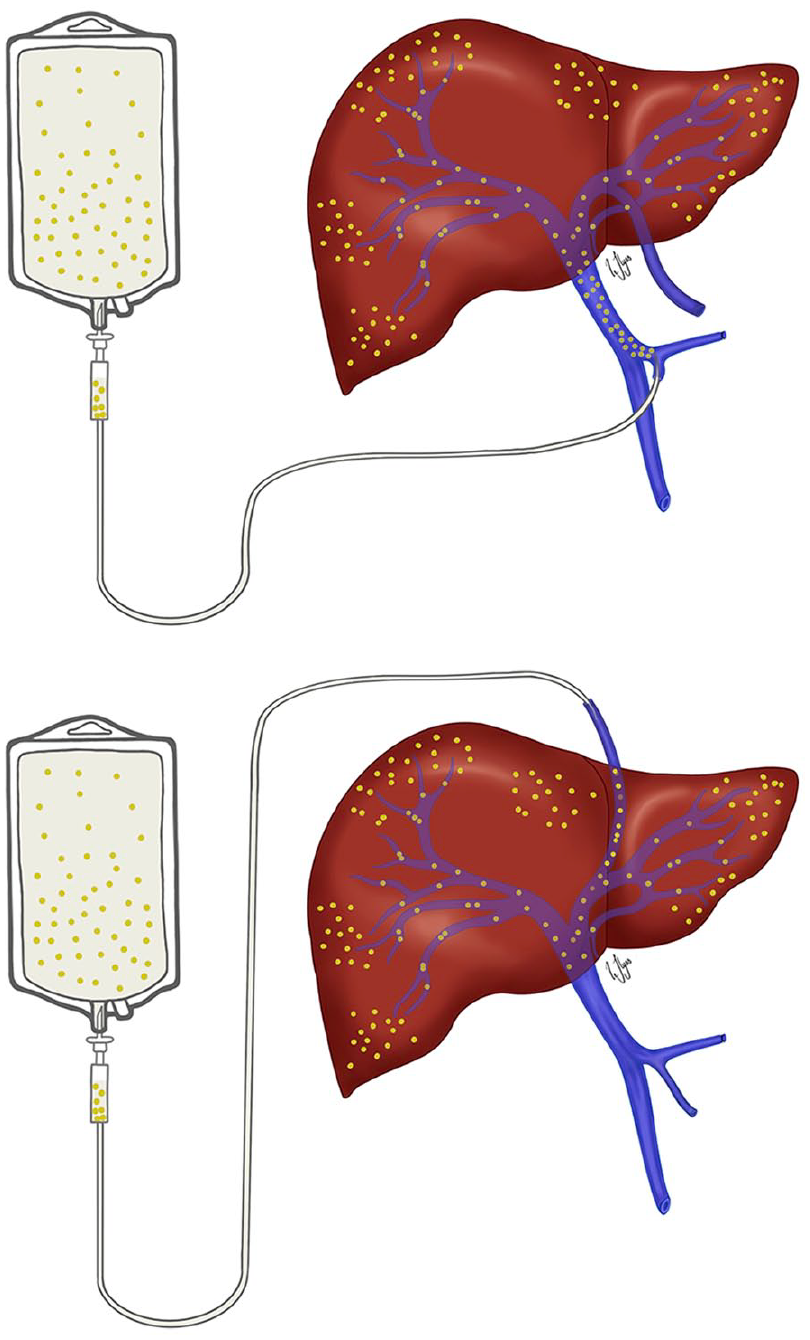
Alternative placements of a 2-lumen catheter for intraoperative islet cells transplantation into the portal vein through. (a) Inferior mesenteric vein and (b) the umbilical vein.
The abdominal cavity remained temporarily exposed, covered by drapes while awaiting islet isolation process.
Islet isolation and transplantation
Immediately following resection, pancreas was perfused with hypoterm organ preservation fluid (IGL-1) and placed in a cooled storage container for transport to the islet isolation laboratory. Islet isolation was performed following the method for allotransplantation. 20 The pancreatic duct was injected with cold collagenase solution for enzymatic digestion, followed by mechanical separation using a modified Ricordi chamber. If the islet preparation exceeded >0.25 mL/kg bodyweight, purification by continuous density gradient on a cell separator was considered. The islet preparation was transplanted into the portal vein (Fig. 2) over a time span of 30–60 min. Low molecular weight heparin (5000E) was added immediately before infusion. Monitoring of the portal venous pressure was conducted and infusion rate was adjusted to prevent values >20 mm Hg. The number of islet equivalents (IEQs) transplanted, purification, and the volume of islet cells were recorded. Once the islet infusion was completed, the abdominal cavity was closed.
Peri- and post-operative procedures
Patients were administered subcutaneous (s.c.) low molecular weight Heparin 5000 IU twice daily starting preoperative and maintained for 4 weeks. Intravenous (i.v.) Methylprednisolone 125 mg and Etanercept 50 mg were given preoperatively, adding 25 mg Etanercept on postoperative day 1 and 2. Doxycycline 400 mg i.v. and Metronidazole 1500 mg i.v. prophylaxis was given. A Doppler ultrasound of the portal vein was conducted on postoperative day 1. All patients received i.v. insulin/glucose infusion to ensure blood glucose levels between 4 and 7 mmol/L immediately postoperative. A strict glycemic control was imperative during the first postoperative week to allow for early islet engraftment. Patients were transitioned to s.c. insulin injections once they tolerated oral food intake.
Epidural analgesia and i.v. opioid analgesics were provided routinely in the immediate post-TPIAT period. Patients were allowed to consume oral diet combined with enzymes starting from postoperative day 1.
Follow-up
Short time follow-up analysis included surgical and medical complications categorized according to the Clavien-Dindo classification system, 21 along with length of hospital stay. Patients underwent clinical examination and assessment of pain level at 3- to 6-month intervals at the HPB outpatient clinic. QoL surveys were completed at least once during follow-up, with a minimum of 6 months postoperative. Endocrinological evaluation was conducted at Oslo University Hospital as needed, otherwise at the patient’s local hospital. C-peptide, insulin use (IE/kg/day), HbA1c (%), and severe hypoglycemic episodes were registered. Islet cell function were categorized as optimal, good, marginal, or failed according to the modified Igls criteria. 22 The time point for glycemic outcome was at least 1 year after TPIAT.
The total daily dose of opioids was converted and recorded as OMEQ. Pain relief assessment was based on self-reported pain levels and analgesic consumption. The date of last follow-up was 1 March 2024.
Statistical analyses
Data are presented as mean ± standard deviation (SD), or median (range) for continuous variables and counts with percentage for categorical variables. Change in QoL was assessed by comparing EORTC QLQ-C30 at baseline and follow-up. Patient scores for the EORTC QLQ-C30 are presented in a radar plot, where each domain is presented on separate axes (scaled from 0 to 100). Delta, defined as difference from baseline, is assessed to estimate clinical significance in the EORTC questionnaires, as recommended by Osoba et al. 23 Difference in health-related QoL score of more than 10% is recognized as a clinically meaningful and significant change. The results are evaluated for statistical significance using Student’s paired t-test for parametric variables and Mann–Whitney U-test for non-parametric variables. A p-value < 0.05 was considered statistically significant. All calculations were made using Stata Statistical Software 17 (College Station, TX, USA).
Results
Patient population
During the study period, tailored surgery for painful chronic pancreatitis was accomplished in 80 consecutive patients. Sixty-one patients (76.2%) underwent parenchyma preserving procedure (28 resections, 16 decompressions, and 17 combined procedures). Fifteen patients (18.8%) fit the criteria for TPIAT and were enrolled in the study. Additional four patients (5.0%) underwent TP, but without islet cell transplantation due to preoperative insufficient endocrine function (n = 2), pancreatitis in native pancreas after prior successful pancreas transplantation (n = 1), or extensive calcification of the pancreatic tissue with unsuccessful islet cell isolation (n = 1).
Baseline characteristics and islet transplant metrics are presented in Table 1.
Baseline characteristics and islet transplant metrics (n = 15).
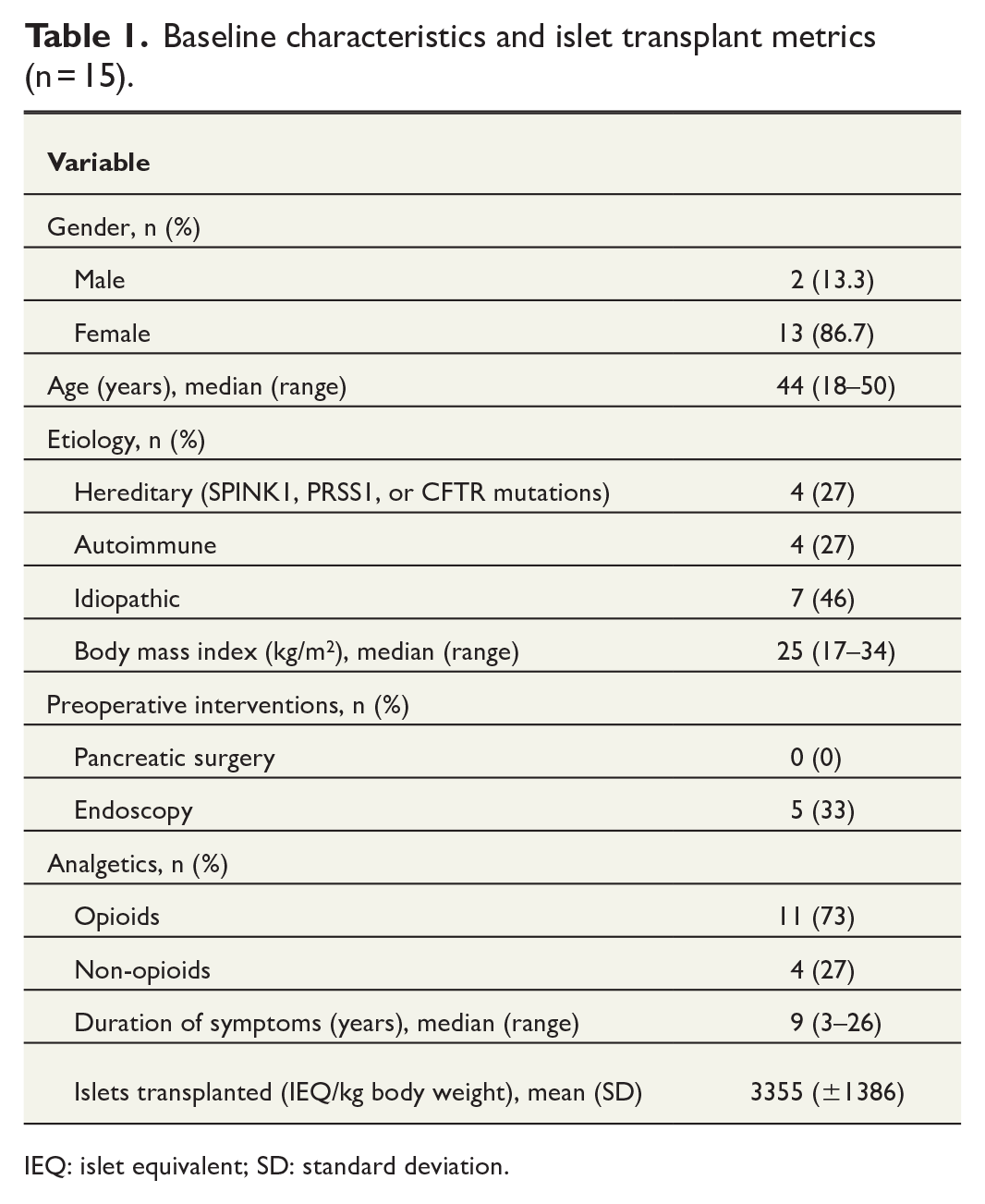
IEQ: islet equivalent; SD: standard deviation.
Median age was 44 years, and the majority were women. All patients denied any current or past alcohol overconsumption. PRSS1 mutation was identified in two patients. In four patients, concomitant autoimmune conditions affecting organs other than pancreas were identified, leading to the consideration that an autoimmune disease might have caused pancreatitis. None of these patients responded to anti-inflammatory medication; hence, TPIAT was indicated.
All patients were on pain medication prior to surgery. 86% received daily opioids with a median OMEQ of 53 mg (range = 15-192). No patients had undergone previous pancreatic surgery. Six patients had performed successful stent placement by endoscopic retrograde cholangiopancreatography. However, TPIAT was deemed necessary due to progression of symptoms. The mean IEQs count was 238,413 IE (SD ± 96.179), which equals 3355 IEQ/kg body weight (SD ± 1386 IE/kg body weight). The mean volume of islet tissue infused was 6 mL (±3 mL) with a 10% (±6%) mean purification.
Surgical outcomes
There were no intraoperative complications. Median blood loss was 100 mL (range = 50–1900). The mean operative time was 367 min (range = 277–517), with an additional 90 min (range = 55–117) for the islet cell transplantation. Intraoperative infusion was carried out through the inferior mesenteric vein in 13 patients and via the umbilical portion of the portal vein in two patients.
Chylous leakage occurred in one patient, necessitating a relaparotomy for the ligation of a lymph vessel 127 days after TPIAT. In two patients, intrahepatic portal vein thrombosis was detected, but dissolved completely on therapeutic doses of low molecular heparin treatment. In one patient, relapsing temporary tachyarrhythmia occurred. The median length of stay in the surgical ward was 12 days (range = 8-27). Two patients were readmitted within 30 days due to recurrent pain. There was no 90 day mortality.
Long-term outcomes
Thirteen of 15 patients (87%) included in the study were followed beyond the immediate postoperative period and were assessed for pain, QoL, and glycemic outcome. The mean follow-up time was 26 months (range = 14–65). In total, 92% reported pain relief without analgetics. In one patient, opioids had been discontinued some months before TPIAT but was reintroduced after the operation.
In total, 92% completed the EORTC QLQ-C30 survey at baseline and once during follow-up. QoL data are presented in Table 2 and Fig. 3. Median time from surgery to postoperative QoL assessment was 13 months (range = 6–44).
EORTC QLQ-C30 responses at baseline and follow-up.
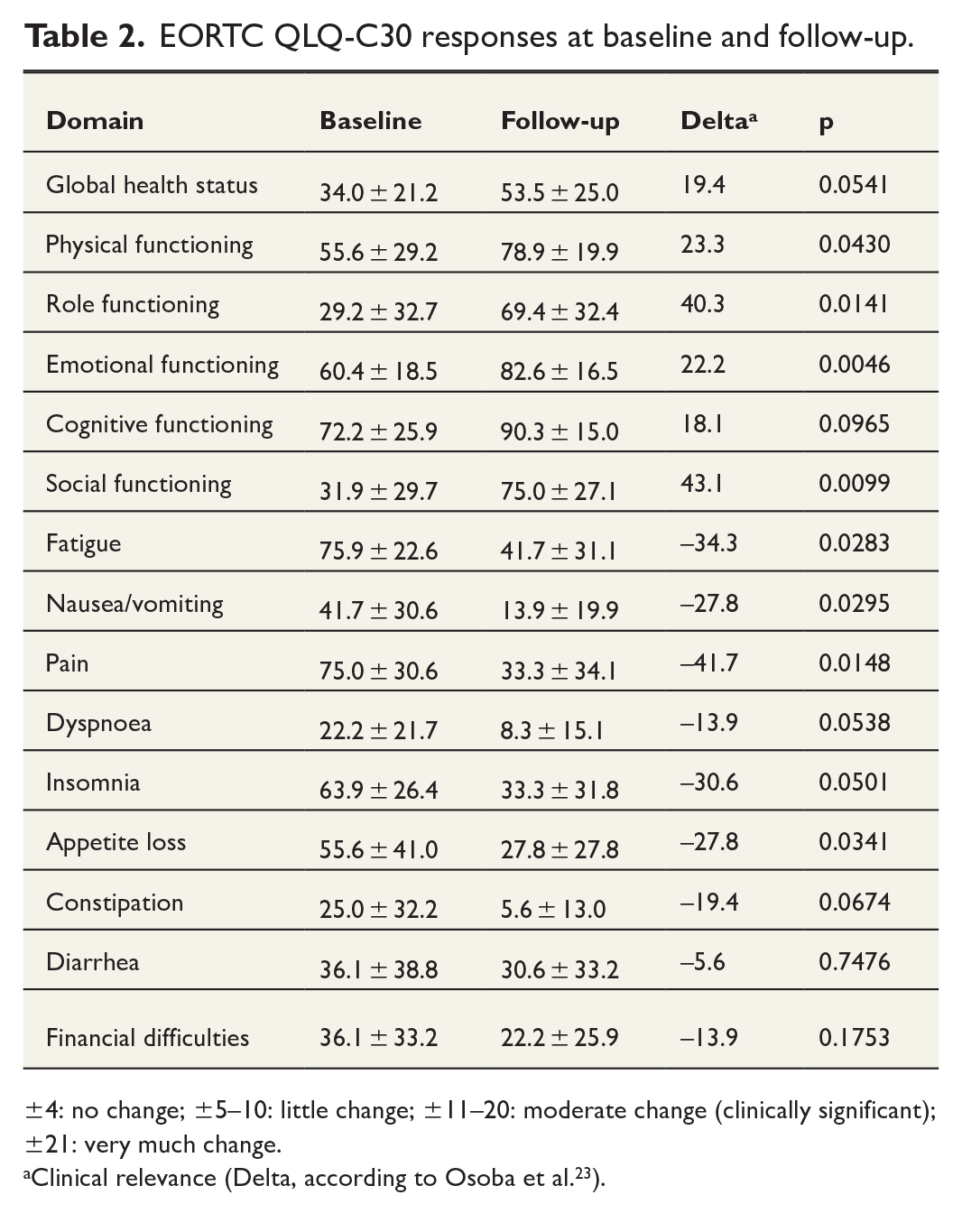
±4: no change; ±5–10: little change; ±11–20: moderate change (clinically significant); ±21: very much change.
Clinical relevance (Delta, according to Osoba et al. 23 ).
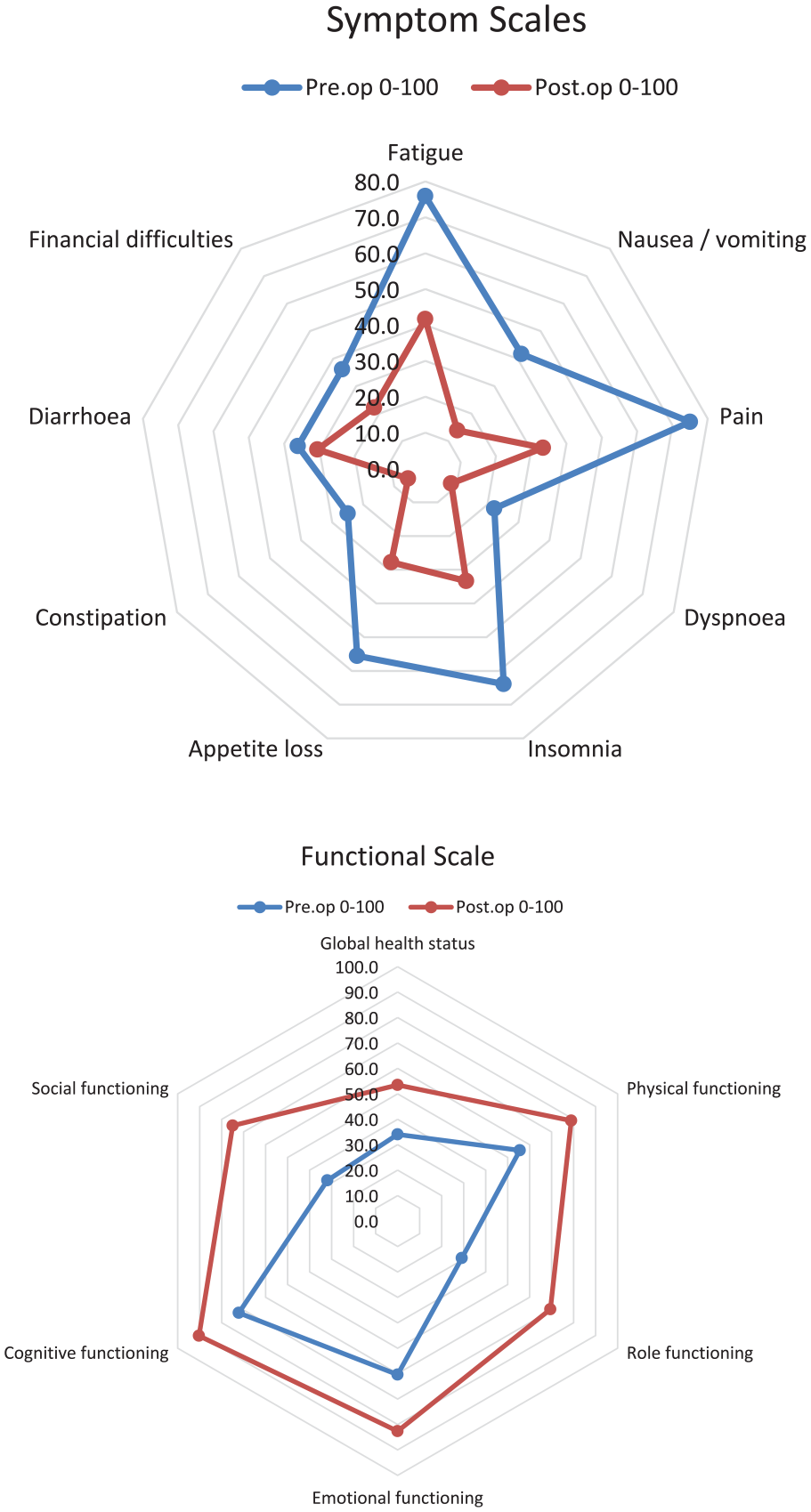
Mean scores at baseline and after total pancreatectomy with islet autotransplantation for. EORTC QOL-C30 symptom and functional scale.
There was a significant improvement in global health. Symptomatic and functional scores demonstrated clinically significant improvements in all domains except for financial difficulties and diarrhea. Across all analyzed domains, the most substantial improvement in scores was observed for social- and role-functioning (assessing a patient’s ability to perform daily activities, leisure time activities, and/or work), fatigue, and pain.
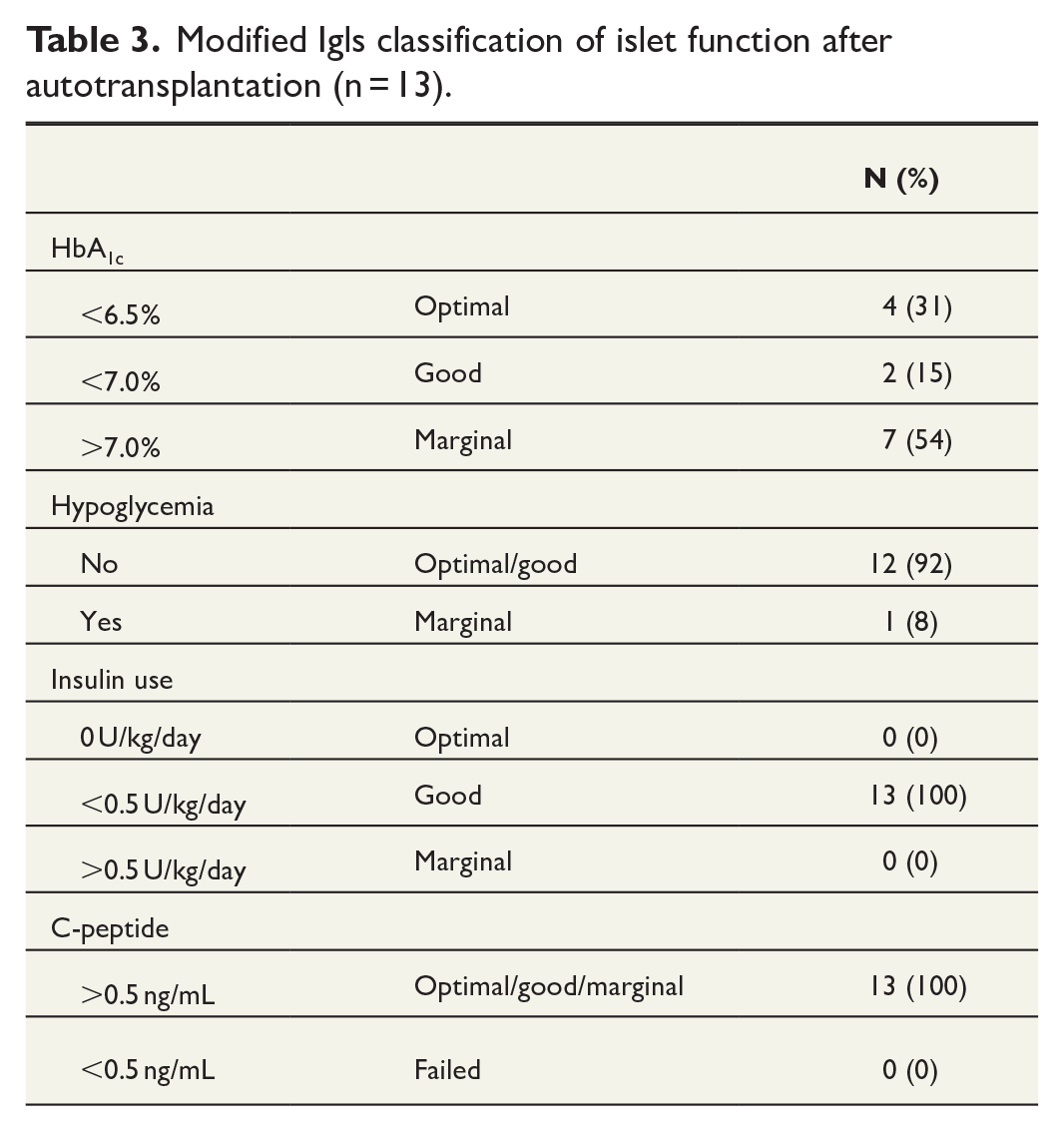
Optimal graft function with insulin independency was not achieved in any patients. However, C-peptide levels indicating functional islet cell grafts were detected in all patients. The mean postoperative C-peptide level was 93 ng/mL (SD ± 47). The total insulin requirements were less than 20IE per day for seven patients. One patient reported severe hypoglycemic episodes. According to the Igls criteria (Table 3), 46% of patients were classified with good graft functions, and no patients experienced complete graft failure.
Modified Igls classification of islet function after autotransplantation (n = 13).
Discussion
Until 2017, patients with diffuse small duct pancreatitis, recurrent acute pancreatitis, or PRSS mutation underwent nonoperative therapies. Following the establishment of a chronic pancreatitis surgery program, 16 TP was introduced as a treatment option due to increased knowledge regarding outcome and promising results reported in the literature.5,7,8 Allogenic islet cell transplantation has been performed in our institution since 2001, 24 facilitating the development of a TPIAT program. Nearly 20% of chronic pancreatitis patients designated to surgery during the study period were selected for TPIAT. The majority of patients experienced pain relief, a clinically significant increase in QoL, and a reduced burden of postpancreatectomy diabetes. In addition, the surgical procedure demonstrated a low rate of complications.
During long-term follow-up, 12 of 13 patients reported pain relief. All patients who required opioids prior to surgery achieved opioid independence at follow-up, which is in line with previous reports. Several institutions are reporting promising outcomes regarding pain relief and QoL.9,25 Chinnakotla et al. 7 have validated the findings based on the largest cohort of TPIAT patients to date, comprising 581 individuals, among whom 92.2% experience pain relief 1 year following surgery.
Timing of intervention is recognized as an important determinant of long-term outcome of surgery in chronic pancreatitis.15,26 Leaving TPIAT as a “last resort” treatment might lead to treatment delay, negatively impacting successful pain relief after TPIAT. The median preoperative duration of pain was 9 years, and the only patient who did not benefit from surgery had a history of pain exceeding 26 years.
No patients achieved insulin independence, but measurable C-peptide levels were identified in all. Glycemic outcomes following TPIAT differ across surgical centers, but in general 22%–38% of patients achieve insulin independent after 1 year.8,9,27 Various predictors for glycemic outcome have been proposed, among which IEQ appears to be the most reliable, supported by Chinnakotla et al. 7 who noted a strong dose–response relationship between the lowest yield category (<2000 IEQ/kg) and the highest (>5000). In 2020, McEachron et al. 22 adapted auto-Igls criteria to assess the outcome of islet auto transplantation. 28 Following this, total islet mass transplanted (IEQ/kg) was the only significant perioperative factor associated with metabolic outcome. 22 Wilson et al. 27 demonstrated a 38% insulin independence rate in 112 TPIAT patients at 1 year follow-up, with a mean islet yield of 6027 IEQ/kg, compared to 3355 IEQ/kg in our study. In a 10-year follow-up study, Turner et al. 8 demonstrated long-term durability regarding glycemic control. In total, 25.7% of 147 patients enrolled were insulin independent at 1 year with an average of 5680 IEQ/kg transplanted. Coluzzi et al. 9 also report promising results regarding insulin independence rate in 116 patients with islet yield exceeding our results. The inferior isled yield in our study may be attributed to the extent of pathology in the pancreatic tissue, 29 with the majority of our patients diagnosed with major calcification and extensive fibrotic changes, influencing the islet yield. This is in accordance with Trout et al. 30 concluding that pancreas volume segmented from pre-TPIAT imaging was a significant predictor of post-TPIAT insulin requirement.
In addition, treatment delay has been associated with lower islet yield following isolation. 31 The median duration of chronic pancreatitis symptoms in our cohort was 9 years, significantly longer than reported by other groups achieving superior metabolic results post-TPIAT.7,9 Remote islet cell isolation might also affect the metabolic outcome. 32 However, in our study, isolation was performed locally and islet cells transplanted intraoperatively, avoiding any delays potentially leading to inferior glycemic outcome.
Strict glycemic control might also impact the metabolic results. Al-Sofiani et al. 33 reviewed the current clinical practice and published the experience of several TPIAT centers, focusing on key aspects in managing patients undergoing TPIAT. The target HbA1c level of 6.5% was found optimal to minimize hyperglycemic stress of the islets. 34 Elevated HbA1c levels in our study might reflect a suboptimal follow-up in terms of glycemic control. Even though no patients achieved insulin independence, the level of insulin requirements, low rates of severe hypoglycemia, and persistent detectable C-peptide levels indicate a lower burden of postpancreatectomy diabetes.
The primary goal of any intervention in painful chronic pancreatitis is to reduce the disabling pain and improve QoL. Although the EORTC QLQ-C30 questionnaire is primarily designed for cancer patients, it is validated for use in benign conditions such as chronic pancreatitis. 35 We found a clinically significant increase in global health and improvements in all but two domains (diarrhea and financial difficulties) postoperatively. Improving QoL after TPIAT aligns with results reported in other studies.5,9,36
Chylous leakage occurred in one patient, and two patients were diagnosed with intrahepatic portal venous thrombosis. There was no 90 days mortality. Reports from other centers have found a higher complication rate for TPIAT. 37 Restricting the surgical procedures to a limited team of experienced surgeons may explain the favorable surgical outcome.
The present study is the first comprehensive Scandinavian prospective study on TPIAT in a cohort of chronic pancreatitis patients. In a Swedish retrospective study of 24 TPIAT, Fröberg et al. 13 have enrolled both malignant patients, rescue pancreatectomies, and elective procedures. However, the heterogeneity of the cohort, varying transplantation locations for islets, and the retrospective study design collectively influence the validity of the study findings.
Strengths and limitations
The small study sample does not permit multivariable testing, limiting the ability to assess predictors of outcome, both regarding pain relief and graft function. The EORTC QOL-C30 questionnaire is not designed specifically for chronic pancreatitis, and a more specific score for pain assessment such as the Visual Analog Scale (VAS) or Izbicki pain score would have been beneficial. However, the domain assessing subjective pain in the EORTC QLQ-C30 questionnaire showed the highest relative rate of improvement after completing TPIAT. In addition, registration of baseline characteristics including both QoL scores and consumption of analgesics converted to OMEQ makes comparison of outcome regarding pain reliable. Standardized intervals from TPIAT to assessments would further strengthen our conclusions, particularly regarding metabolic outcome where results were retrieved at 1 year follow-up or at last visit.
The strength of our study is the prospective design and a high follow-up rate. Since the few HPB surgeons were involved in the TPIAT program, standardization of the preoperative workup, indication, and surgical technique was achieved, further strengthening the results. In addition, in our experience, regular chronic pancreatitis multidisciplinary team meetings and dedicated TPIAT teams were key factors in the success of the program.
Hopefully increased knowledge on TPIAT will draw attention to a subgroup of chronic pancreatitis patients suffering from disabling pain and decreased QoL that until recently had no efficient surgical treatment options in the Scandinavian countries.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
