Abstract
Background and aims:
Chronic pancreatitis may cause intractable abdominal pain, with total pancreatectomy sometimes being the last resort. To mitigate the subsequent diabetes, total pancreatectomy can be followed by islet autotransplantation (TP-IAT). The primary aim of this study was to assess the outcomes in patients undergoing TP-IAT at Karolinska University Hospital with respect to safety, postoperative complications, and islet graft function. A secondary aim was to compare liver to skeletal muscle as autotransplantation sites.
Methods:
Single-center observational cohort study on patients undergoing TP-IAT. Islets were transplanted either into the liver or skeletal muscle. Data on baseline characteristics and pretransplantory conditions were collected. Outcome measures included mortality and major postoperative complications as well as the glycemic measures: insulin use, fasting C-peptide, and HbA1c.
Results:
Between 2004 and 2020, 24 patients underwent TP-IAT. Islets were transplanted into the liver in 9 patients and into skeletal muscle in 15 patients. There was no 90-day mortality, and major complications (Clavien–Dindo ⩾IIIa) occurred in 26.7%, all related to the procedure of total pancreatectomy. Fasting C-peptide could be detected postoperatively, with higher levels in patients receiving islet autotransplantation into the liver (p = 0.006). Insulin independence was not achieved, although insulin doses at last follow-up were significantly lower in patients receiving islet autotransplantation into the liver compared to skeletal muscle (p = 0.036).
Conclusion:
TP-IAT is safe and associated with tolerable risk, the component of islet autotransplantation being seemingly harmless. Although islet grafts maintain some endocrine function, insulin independence should not be expected. Regarding islet autotransplantation sites, the liver seems superior to skeletal muscle.
Clinical trial registration:
Not applicable.
Keywords
Context and relevance
Total pancreatectomy with islet autotransplantation (TP-IAT) aims to mitigate the postoperative diabetes otherwise following total pancreatectomy, currently believed to leave one-third of TP-IAT recipients insulin independent. Islets are generally transplanted into the liver which entails several disadvantages, why alternative transplantation sites are of interest. This was the first study to evaluate the outcome after TP-IAT in Swedish patients, where both the liver and skeletal muscle have been used as autotransplantation sites, enabling a unique comparison between the two. It was confirmed that TP-IAT is safe in terms of morbidity and mortality. Insulin independence was not achieved, despite signs of postoperative endogenous insulin synthesis. Importantly, it was concluded that the liver seems significantly superior to skeletal muscle as islet cell host.
Introduction
Total pancreatectomy (TP) has historically been considered the very last resort to manage chronic pancreatitis (CP)–related pain, as it guarantees an immediate onset of “diabetes of the exocrine pancreas” (DEP). 1 DEP differs from type I and type II diabetes, as it involves a complete loss of all endocrine cells contained in the Langerhans islets, resulting in a “brittle” diabetes associated with swift blood sugar fluctuations and an increased risk for severe hypoglycemia.2,3 To overcome this, the idea of TP with islet autotransplantation (IAT)—TP-IAT, has evolved. This involves a laboratory isolation and subsequent autotransplantation of Langerhans islets from the resected pancreas, aiming to mitigate the effects of DEP without the immunosuppressive treatment required following allotransplantation.
Studies suggest that one-third of patients maintain enough islet graft function to remain insulin independent during the first years after TP-IAT;4–6 another one-third becoming insulin dependent but with partial islet graft function remaining. 6 Several factors associated with islet graft function and the subsequent glycemic outcome have been suggested, whereas the “islet yield” from the resected pancreas is most crucial.7,8
TP-IAT is generally considered safe with minimal mortality and acceptable rates of postoperative morbidity. 9 However, morbidity rates vary between centers performing TP-IAT, ranging from 15% to 65%. 10 Pancreatic neoplasm has long been considered a contraindication for IAT based on the fear of disseminating premalignant or malignant cells. However, emerging evidence suggests that IAT following pancreatic tumor resections might be safe and efficient in selected patients.1,11–15
The most common and well-studied autotransplantation site is the liver. Since a great proportion of islets are lost during portal vein infusion due to an immediate blood-mediated inflammatory reaction, 16 alternative IAT-sites are of great interest. Historically, transplantation sites such as skeletal muscle have been assessed, 17 carrying the potential benefits of rapid revascularization and easy access for graft monitoring or explantation.18,19
Although evidence in favor of TP-IAT is increasing, many uncertainties remain, such as suitable candidates, ideal timing, transplantation sites, and most importantly the efficacy and safety of the procedure. The primary aim of this study was to evaluate the outcome after TP-IAT in a Swedish population, in terms of safety and postoperative complications, as well as islet graft function and glycemic control. A secondary aim was to compare the liver to skeletal muscle as IAT-sites.
Methods
Study design and patient selection
This was a single-center observational retrospective cohort study, comprising all patients selected for TP-IAT at Karolinska University Hospital, Sweden, between September 2004 and December 2020. Notably, TP-IAT was not performed between 2009 and 2016 due to departmental reorganization. Patients were included regardless of indication for surgery and regardless of whether the decision to perform TP-IAT was made electively by a multidisciplinary team or as an acute “rescue” from complications of recent pancreatic surgery. Glycemic preconditions in all patients were normal HbA1c and insulin independence. Patients who underwent TP but not the intended IAT were excluded, although the reason for IAT cancelation was documented. Informed consent for this retrospective study was not obtained in order to avoid the inherent risk for loss of study participants and selection bias, potentially lowering research quality and reliability. Given the small size of the cohort, inclusion of all eligible participants was critical. The study has been approved by the Ethical Review Board in Stockholm (reference no. 2019-00406).
Surgical procedure
The course of TP-IAT was divided into three consecutive steps. The first step was either an elective open TP or a rescue completion pancreatectomy in which techniques to minimize warm ischemia time were utilized, when possible, to preserve the Langerhans islets. The second step was an isolation and purification of Langerhans islets from the resected pancreas at an islet isolation facility, using a previously described technique. 20 Awaiting autotransplantation, islets were held in culture for 2–3 days. The third and last step was an autotransplantation of the isolated islets. IAT was performed only in clinically stable patients, 2–7 days after TP. Patients who underwent TP-IAT before 2009 generally received the autotransplant into the brachioradialis muscle of one forearm, 21 while patients treated more recently received the autotransplant into the liver through portal vein infusion according to modern standards. This distinction defined the two subgroups compared throughout all statistical analyses, labeled “muscle-IAT” and “liver-IAT,” respectively. All patients received perioperative anticoagulant therapy (5000 units low-molecular-weight heparin daily) for a minimum of 4 weeks postoperatively. There was no routine regarding anti-inflammatory drug treatment during the perioperative period.
Data collection and definitions
All data were obtained from patient charts, including preoperative, perioperative, and postoperative variables.
Baseline characteristics
Baseline data included gender, age at day of operation, body mass index (BMI), active tobacco use, indication for TP-IAT, previous surgical pancreatic interventions, and an assessment of comorbidities according to Charlson Comorbidity Index (CCI). 22 Preoperative glycemic function and control was assessed through hemoglobin A1c (HbA1c) in mmol/mol, fasting C-peptide in nmol/L, and preoperative insulin use defined as total amount of insulin units per day (IU/day).
Perioperative data
Perioperative data included date of TP, estimated blood loss in mL, total islet yield defined as islet equivalents (IEQ), total islet yield per kg body weight defined as IEQ/kg, implementation of islet autotransplantation including reason in case of cancelation, autotransplantation site, and the number of islets transplanted, expressed in IEQ/kg.
Outcome variables
Postoperative complications (surgical and non-surgical) were quantified using the Clavien–Dindo Classification. 23 Major complications were defined as Clavien–Dindo ⩾ IIIa. Frequently used definitions and severity scales were used to assess the presence of postoperative delayed gastric emptying, 24 postpancreatectomy hemorrhage, 25 and bile leakage, 26 respectively; only grade B or higher according to consensus guidelines were registered. Additional variables were overall hospital length of stay in days, intensive care unit stay (ICU-stay), 30-day readmission, and 90-day mortality. Post-transplant islet graft function was assessed by obtaining data on fasting C-peptide in nmol/L 1 month postoperatively, and exogenous insulin use expressed in IU/day at discharge, 1, 6, and 12 months postoperatively. Postoperative glycemic control was assessed by obtaining values of HbA1c at 6 and 12 months postoperatively. To reduce the impact of missing data, 6- to 12-month postoperative data were pooled, using the highest registered value of insulin dose and HbA1c to ensure that results were not distorted in favor of TP-IAT.
Statistical analysis
Normally distributed data were presented as mean and standard deviation (SD), while non-normally distributed data were presented as median and interquartile range (IQR). Comparisons of continuous data between patient groups were performed with independent T-test or Mann–Whitney U-test depending on the distribution of data. Normal distributions were studied using the Shapiro–Wilk test. Comparisons of pre- and post-operative data were performed with paired T-test. Categorical data were presented as absolute count and valid percentage. Comparisons of categorical data between patient groups were studied with Fisher’s exact test. p-values < 0.05 were considered significant. Data were analyzed using SPSS (Statistical Package for the Social Sciences, Version 27.0.0; SPSS, Inc., Chicago, IL, USA).
Results
Distribution of TP-IAT recipients
A total of 33 patients were selected whereas TP-IAT was performed in 24 (72.7%), the remaining nine patients (27.3%) receiving TP but not IAT (Fig. 1). In six patients (66.7%), cancelation of IAT was due to failure to isolate enough viable islets. In two patients (22.2%), complications after TP did not allow for further intervention. In one patient (11.1%), the reason for IAT cancelation was not documented. Among the 24 patients who underwent TP-IAT, 16 patients had elective surgery and 8 patients had acute surgery due to complications following previous pancreatic surgery (Whipple procedure). Fifteen patients (62.5%) received the transplant into the brachioradialis muscle and were all treated between 2004 and 2008, while nine patients (37.5%) received the transplant into the liver and were all treated between 2017 and 2020 (except for one patient treated in 2004).
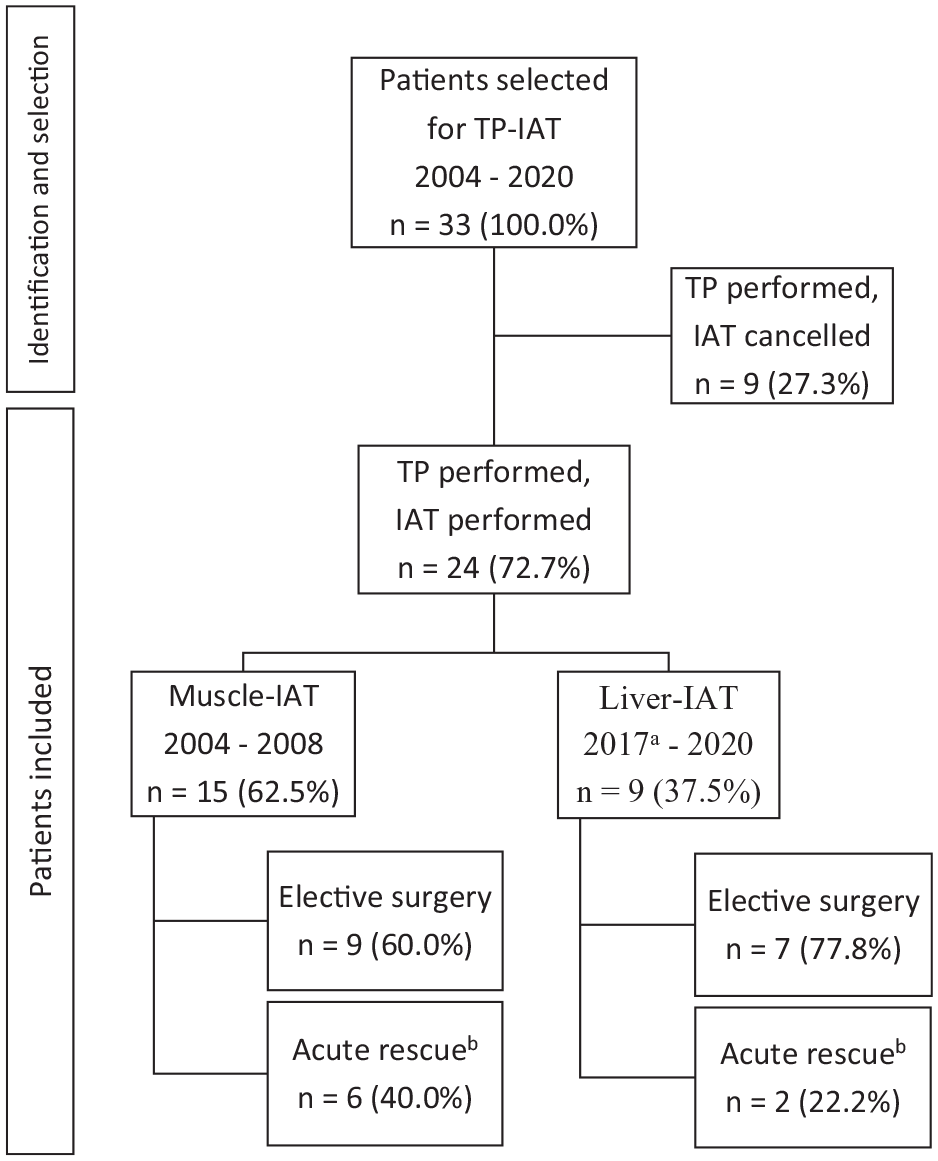
Patient selection and distribution.
Baseline characteristics and pretransplantory conditions are summarized in Table 1. The cohort consisted of 9 females and 15 males with a median age of 63 years and a median BMI of 25.2 kg/m2, the majority being non-smokers. Neoplasia was the most common indication for TP-IAT in the muscle-IAT subgroup (46.7%) and overall (37.5%), while CP was the most common indication in the liver-IAT subgroup (44.4%). Comorbidities tended to be more frequent in the muscle-IAT subgroup than in the liver-IAT subgroup, with median CCI-values of 4 and 0, respectively (p = 0.078). The median blood loss during TP was 800 mL overall, with significantly larger blood loss volumes in the muscle-IAT subgroup than in the liver-IAT subgroup (p = 0.002). The mean transplanted cell mass was 2475.7 IEQ/kg bodyweight and was similar in both IAT-subgroups.
Baseline characteristics and pretransplantation conditions.
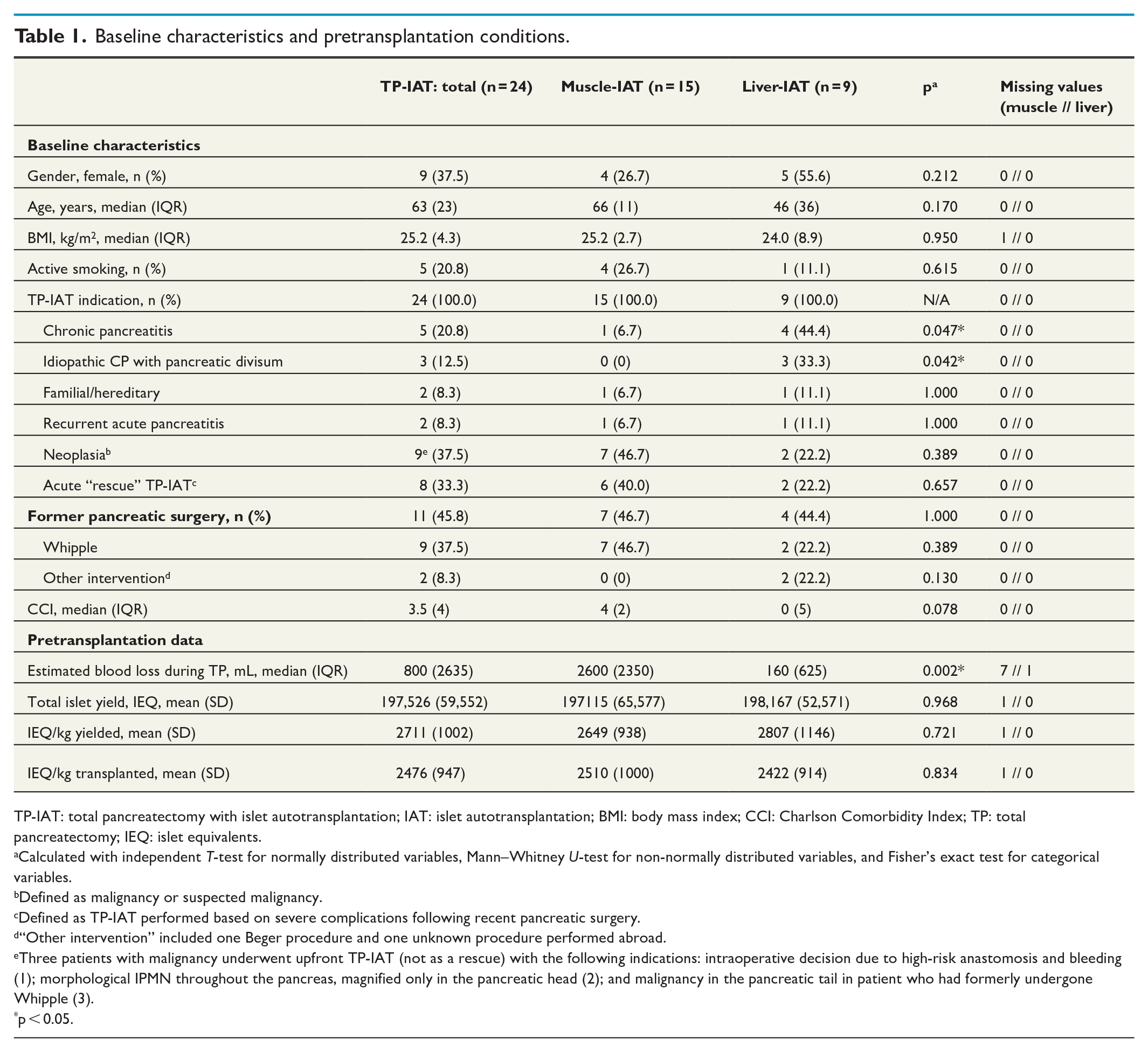
TP-IAT: total pancreatectomy with islet autotransplantation; IAT: islet autotransplantation; BMI: body mass index; CCI: Charlson Comorbidity Index; TP: total pancreatectomy; IEQ: islet equivalents.
Calculated with independent T-test for normally distributed variables, Mann–Whitney U-test for non-normally distributed variables, and Fisher’s exact test for categorical variables.
Defined as malignancy or suspected malignancy.
Defined as TP-IAT performed based on severe complications following recent pancreatic surgery.
“Other intervention” included one Beger procedure and one unknown procedure performed abroad.
Three patients with malignancy underwent upfront TP-IAT (not as a rescue) with the following indications: intraoperative decision due to high-risk anastomosis and bleeding (1); morphological IPMN throughout the pancreas, magnified only in the pancreatic head (2); and malignancy in the pancreatic tail in patient who had formerly undergone Whipple (3).
p < 0.05.
Surgical outcome
Data on safety and postoperative complications are summarized in Table 2. The presented data cover elective cases only (n = 16), as the remaining patients suffered from extensive complications prior to TP-IAT, leaving no other reasonable treatment option than surgical removal of the pancreas as a “rescue.” No in-hospital or 90-day mortality was recorded among the electively operated patients, and the mean hospital length of stay was 15.7 days. The 30-day readmission rate was 6.7%. Within the muscle-IAT subgroup, there was a tendency toward more frequent ICU-admissions (p = 0.088) and longer hospital lengths of stay (p = 0.074). Major complications occurred in four patients (26.7%), one of whom suffered from multiple major complications. The major complications recorded consisted of one relaparotomy due to postoperative abdominal bleeding, one intra-abdominal fluid collection, one gastrointestinal bleeding from multiple stomach ulcers, and two myocardial infarctions. Three patients had delayed gastric emptying (20.0%) and three patients had postpancreatectomy hemorrhage (20.0%), while none of the patients suffered from bile leakage. There were no significant differences between the two IAT-subgroups with respect to any of the postoperative complications recorded and there were no IAT-related major complications (e.g. local bleedings or portal vein thrombosis).
Morbidity and mortality in patients who underwent elective single stage TP-IAT.
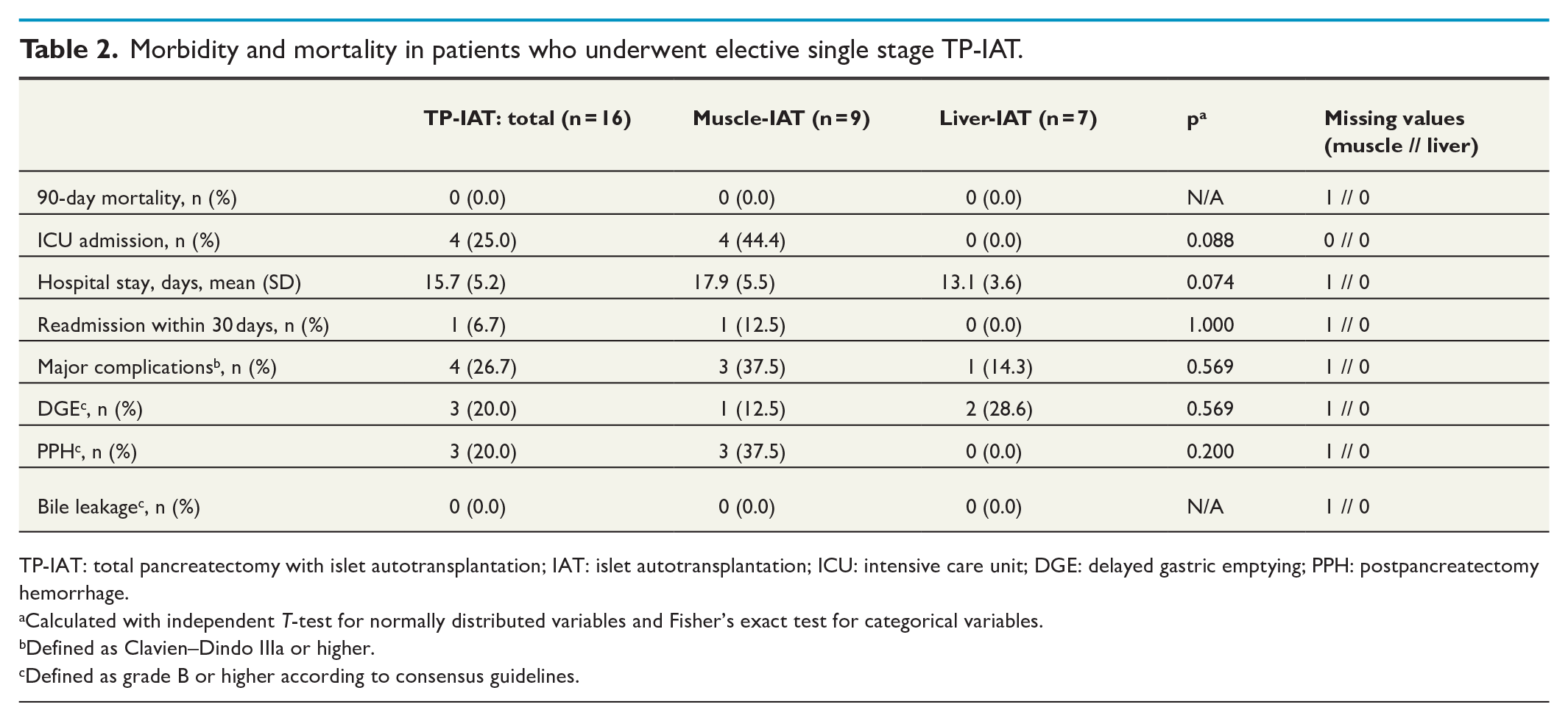
TP-IAT: total pancreatectomy with islet autotransplantation; IAT: islet autotransplantation; ICU: intensive care unit; DGE: delayed gastric emptying; PPH: postpancreatectomy hemorrhage.
Calculated with independent T-test for normally distributed variables and Fisher’s exact test for categorical variables.
Defined as Clavien–Dindo IIIa or higher.
Defined as grade B or higher according to consensus guidelines.
During the study period, three out of nine patients with neoplasia as TP-IAT-indication developed recurrence (one in the muscle-IAT subgroup, two in the liver-IAT subgroup). Only one of these recurrences were located to the IAT-site itself (liver) and occurred almost 2 years after a primary elective Whipple in which a pancreatic ductal adenocarcinoma was resected with a positive surgical margin, later followed by rescue pancreatectomy and IAT.
Glycemic outcome
Prior to surgery, none of the patients were insulin dependent (Table 3). At discharge, all 24 patients were insulin dependent with a mean insulin dose of 28.4 IU/day, without significant difference between the IAT-subgroups. Although all 24 patients remained insulin dependent 6–12 months postoperatively, the mean insulin dose was significantly lower in the liver-IAT subgroup compared to the muscle-IAT subgroup with mean insulin doses of 19.4 and 31.4 IU/day, respectively (p = 0.036). In most patients, C-peptide could be detected 1 month postoperatively. The median fasting C-peptide at this time was 0.08 nmol/L within the muscle-IAT subgroup and 0.53 nmol/L within the liver-IAT subgroup, suggesting a significant difference in insulin secretion when comparing IAT-subgroups (p = 0.006).
Pre- and post-operative glycemic control.
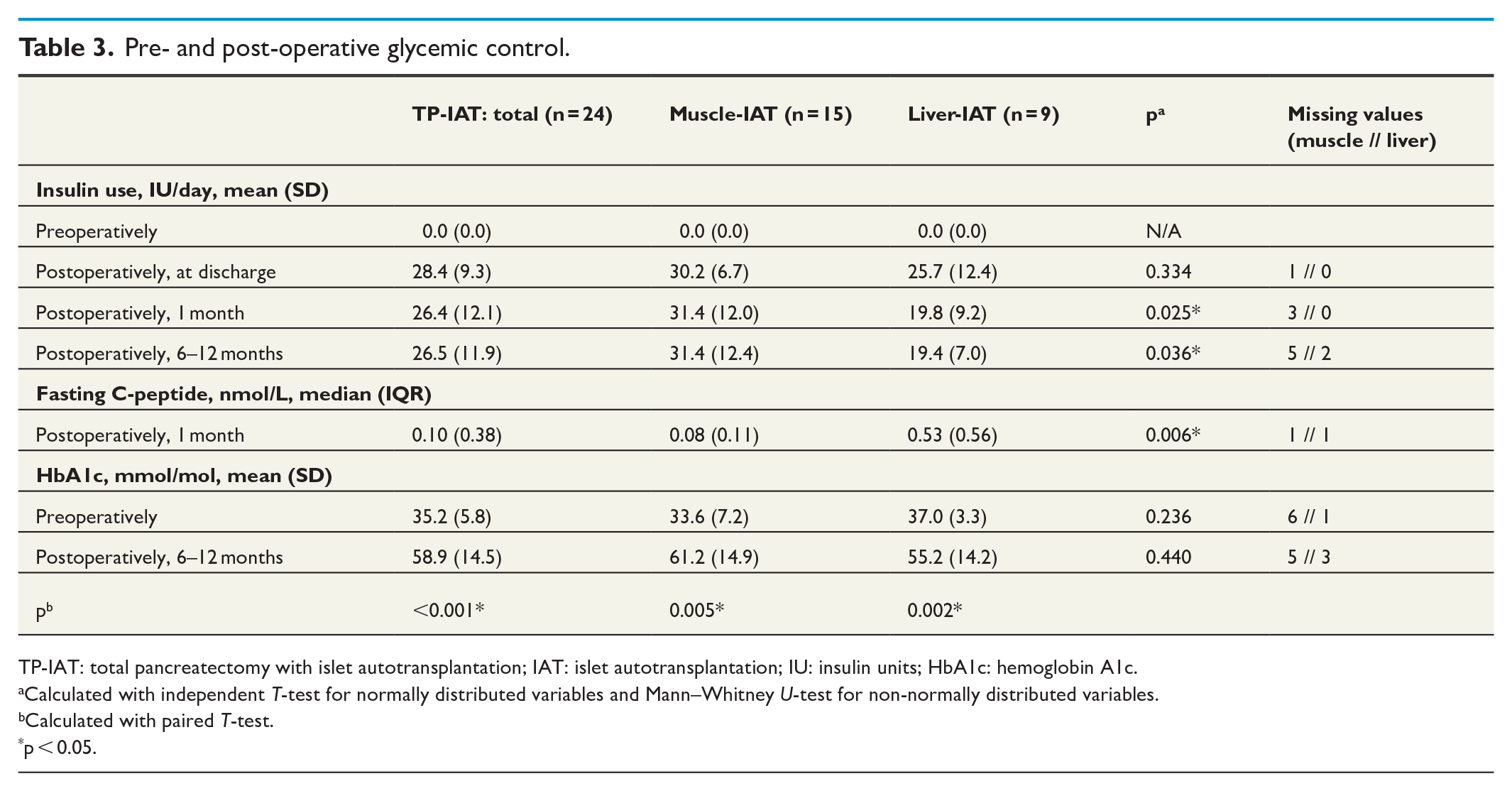
TP-IAT: total pancreatectomy with islet autotransplantation; IAT: islet autotransplantation; IU: insulin units; HbA1c: hemoglobin A1c.
Calculated with independent T-test for normally distributed variables and Mann–Whitney U-test for non-normally distributed variables.
Calculated with paired T-test.
p < 0.05.
Overall, the mean HbA1c-value was 35.2 mmol/mol preoperatively and 58.9 mmol/mol 6–12 months postoperatively (p < 0.001). The significant rise in HbA1c postoperatively was also evident when analyzing each IAT-subgroup separately (p = 0.005 and p = 0.002, respectively). However, when comparing HbA1c-values between the two IAT-subgroups, there were no significant differences neither preoperatively nor 6–12 months postoperatively.
Discussion
This study aimed to examine the outcome after TP-IAT in a Swedish population. The main findings were that all TP-IAT recipients required insulin treatment postoperatively, although there were signs of endogenous insulin synthesis. Patients receiving liver-IAT had significantly higher fasting C-peptide and required less exogenous insulin during the first postoperative year compared to patients receiving muscle-IAT. There was no 90-day mortality, and all major postoperative complications were associated with the TP component.
Our results confirmed, in line with previous studies, that TP-IAT involves minimal risk for death. Although complication rates vary between centers, 10 a pooled major complication incidence of 28.5% has recently been reported, 27 similar to that of our cohort (26.7%). Notably, none of the major complications were directly associated with the IAT component, suggesting that IAT is an innocuous addition to TP. However, there was a noteworthy tendency toward shorter hospital stays and less frequent ICU admissions within the liver-IAT subgroup. This is presumably unrelated to the IAT-site itself as there are other, more plausible explanations. First, it may reflect the different time periods in which patients within each IAT-subgroup were treated, since routine TP-procedures have improved over time, resulting in less complicated postoperative care courses. This reasoning is supported by the significantly smaller blood loss volumes during TP within the liver-IAT subgroup. Second, a greater proportion of muscle-IAT patients underwent TP as a rescue. Third, patients within the muscle-IAT subgroup had a tendency toward higher preoperative CCI, potentially complicating the postoperative care courses. Therefore, the major complication rate of 14.3% in the liver-IAT subgroup may more accurately reflect the true complication rate today. Our results also support the findings of Balzano et al. 15 that IAT may be safe even with neoplasia as indication for surgery, since none of the patients in this study had a recurrence directly linked to the IAT-site.
Despite the general belief that one-third become insulin independent following TP-IAT, 7 insulin independence could not be observed in this study. One possible explanation is the relatively low islet yields in the current population compared to what has been reported.5,6 For instance, Wilson et al. 5 demonstrated a 38% insulin independence rate 1 year postoperatively and a mean islet yield of 6027 IEQ/kg compared to 2711 IEQ/kg in this study. A potential contributing factor to the relatively low islet yields is that islets were cultured prior to autotransplantation, well-known to cause rapid deterioration of isolated cells. 28
However, the success of TP-IAT is not necessarily defined by insulin independence, but the overall increase in quality of life. 27 Life quality improvements may also be influenced by the severity of the insulin-dependent diabetes, as well as pain relief and other gastrointestinal symptoms potentially reduced following the procedure. It has been suggested that life quality following TP-IAT is completely unrelated to postoperative insulin requirements, 29 indicating that the outcome focus should probably be shifted from insulin independence rate toward more comprehensive assessments of life quality. Equally important, postoperative insulin treatment may be required even when transplanted β-cells maintain some function. Evidence suggests that such partial islet graft function generates a milder diabetes easier to control than the classical DEP resulting from TP without IAT. 27 It has also been shown to reduce the risk for the hypoglycemic episodes otherwise characterizing DEP. 27 In this study, there were signs of postoperative β-cell function and endogenous insulin synthesis, demonstrated by the fasting C-peptide detected 1 month postoperatively. Since exogenous insulin requirements at this time were largely unchanged after 6–12 months, engrafted islets reasonably kept synthesizing similar amounts of endogenous insulin during the first postoperative year, potentially reducing the severity of diabetes. IAT should therefore be considered a worthwhile adjunct to TP regardless of whether exogenous insulin treatment is needed or not, since IAT itself is evidently harmless and the partial islet graft function observed may provide a glycemic profit.
The postoperative glycemic control was dissatisfactory both overall and in each IAT-subgroup, with mean HbA1c values well above target values for diabetic patients. Rather than islet graft failure, this could imply an insufficient long-term endocrine follow-up program for TP-IAT patients. Although achieving full glycemic control may be challenging following TP-IAT, acceptable HbA1c values have been reported, even in subgroups with islet yields like those of the current study,4–6 emphasizing the importance of follow-up routines.
The liver appeared as superior to the brachioradialis muscle in terms of successful islet engraftment. Fasting C-peptide levels 1 month postoperatively were significantly higher in the liver-IAT subgroup, representing a significantly higher endogenous insulin synthesis. Conformingly, exogenous insulin requirements within this subgroup were significantly lower both 1 month and 6–12 months postoperatively. The numbers of IEQ/kg yielded and transplanted within each IAT-subgroup were almost identical, implying that prerequisites for glycemic success were comparable in this regard. Although, the 10-year gap between the two IAT-subgroups may have confounded these findings through time-bias. As previously mentioned, TP and the perioperative hospital care have improved during the past decade. As a result, patients within the muscle-IAT subgroup presumably suffered from a greater pretransplantory vulnerability, potentially affecting the glycemic outcome. A greater proportion of muscle-IAT patients also underwent rescue pancreatectomy prior to IAT, further indicating a greater pretransplantory vulnerability. On the contrary, a recent study suggested that postoperative complications do not affect islet graft function, 30 and IAT was performed only in clinically stable patients regardless of whether TP was performed electively or as a rescue. Nevertheless, the muscle-IAT subgroup was probably disadvantaged preoperatively regardless of the surgical aspects linked to TP, considering the tendency toward more comorbidities (higher CCI), and the fact that neoplasia was the most common indication for TP-IAT.
To our knowledge, this was the first clinical study in humans comparing liver and skeletal muscle as IAT-sites. A preclinical study on minipigs comparing the liver to the gracilis muscle detected significantly better islet graft function following liver-IAT, 18 harmonizing with our results and supporting continued usage of the liver as the primary IAT-site.
The main limitations of this study are the inherent weaknesses of the retrospective design, including missing and incomplete data. Considering the general importance of risk for severe hypoglycemia and quality of life following TP-IAT, it is a shortcoming that these data could not be presented. Another main limitation was the relatively small and heterogeneous cohort. The heterogeneity and potential time-bias made it challenging to isolate the effects of the IAT-site, complicating the interpretation of the results. Also, the large timespan entailed a risk for the overall results not reflecting the outcome after TP-IAT the way it is performed today, since both the TP-IAT routines as well as the most common indication have changed over time (CP currently being the main indication). Despite these limitations, our study held a fundamental strength in being the first clinical study comparing the liver and skeletal muscle as IAT-sites in humans.
Conclusion
TP-IAT carries a low risk for mortality and a tolerable risk for major postoperative complications. When present, major complications result from the TP, proving that IAT is a harmless adjunct to TP alone. Although islet grafts appear to maintain some endocrine function, presumably contributing to increased metabolic control and reduced exogenous insulin requirements, complete insulin independence is less likely. Finally, the liver seems superior to the brachioradialis muscle in terms of islet engraftment and subsequent glycemic success, although this finding should be interpreted with caution. Further research is needed to develop and confirm the findings of this study and the knowledge on TP-IAT, preferably through an appropriately designed, large, multi-center prospective study with a control group (TP without IAT), including systematically collected data on glycemic outcomes including hypoglycemia and quality of life.
Footnotes
Acknowledgements
The data analysis was part of the master thesis of Klara Fröberg and the PhD project of Asif Halimi.
Author contributions
P.G. contributed to study conception and design. K.F., A.H., M.V., and P.G. contributed to acquisition of data. K.F. contributed to statistical analysis. All authors contributed to analysis, interpretation, drafting, and or critical revision.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.M.L.: Abbott (lecture fee), Mylan (lecture fee) and M.V.: Abbott (lecture fee), Mylan (lecture fee).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
