Abstract
Background and Aims:
For patients with biliary tract cancer involving the hepatic hilum, major hepatic resection with extrahepatic bile duct resection may be required. In addition to perihilar cholangiocarcinoma (PHCC), the same extent of surgery is used in advanced gallbladder cancer (GBC) and intrahepatic cholangiocarcinoma (IHCC) with hilar involvement. Few studies compare prognostic factors and long-term outcomes across tumor types. This study compared risk characteristics and outcomes after surgery in all subtypes of biliary tract cancer with hilar involvement.
Methods:
Patients with biliary tract cancer with hilar involvement undergoing major liver resection and extrahepatic bile duct resection between 2011 and 2021 at a single center were retrospectively analyzed. The primary postoperative outcome was overall survival. Secondary outcomes were recurrence-free survival and postoperative complications. Survival analysis was performed with Cox regression analysis and Kaplan–Meier method.
Results:
One-hundred and eight patients were included. Seventy-three (67%) had PHCC, 24 (22%) had GBC, and 11 (10%) had IHCC. Hilar-invading IHCC and GBC had more adverse histopathological factors like lymph node positivity (p = 0.021), higher number of positive nodes (p = 0.043), and larger tumor size (p < 0.001) compared with PHCC. Peritoneal invasion and lymph node positivity were significant independent predictors for survival (p = 0.011 and p = 0.004, respectively). Median overall survival was 29 months for PHCC, 22 months for GBC and 21 months for IHCC (p = 0.53). IHCC tended to recur earlier (p = 0.046) than GBC and PHCC (6, 15, and 18 months, respectively).
Conclusion:
Patients with biliary tract cancer with hilar involvement undergoing major liver resection and resection of extrahepatic bile ducts had similar overall survival regardless of subtype, while IHCC recurred earlier. Peritoneal cancer invasion was common in all subtypes, including PHCC, and was an independent prognostic factor. This finding may support routine reporting of peritoneal invasion-status in resected biliary tract cancer.
Keywords
Context and Relevance
For patients with biliary tract cancer involving the hepatic hilum, major hepatic resection with extrahepatic bile duct resection may be required. In addition to perihilar cholangiocarcinoma (PHCC), the same extent of surgery is used in advanced gallbladder cancer (GBC) and intrahepatic cholangiocarcinoma (IHCC) with hilar involvement. Few studies compare prognostic factors and long-term outcomes across tumor types. In this retrospective single-center study including 108 patients, we found a similar overall survival regardless of subtype, while IHCC recurred earlier. Peritoneal cancer invasion was common in all subtypes, including PHCC, and was an independent prognostic factor for survival. This finding may support routine reporting of peritoneal invasion-status in resected biliary tract cancer.
Introduction
Biliary cancer is a rare entity accounting for 1%–3% of all gastrointestinal malignancies with an incidence of 1–2 per 100,000/year. 1 Occurring in the biliary epithelium, it is subdivided into four groups; intrahepatic (IHCC), perihilar (PHCC), distal cholangiocarcinoma, and gallbladder cancer (GBC). Surgical resection is the only treatment offering a chance for long-term survival, but this option is open for just one-fourth of patients due to locally too advanced or metastatic disease at presentation. 2
About 10% of GBC and IHCC are diagnosed at an advanced stage where the tumor infiltrates the hepatic hilum, with reported poor prognosis.3,4 This often requires extended surgery with high postoperative morbidity and mortality questioning this aggressive surgical approach. Surgery involves major liver resection with extrahepatic bile duct resection, and in some cases, additional pancreaticoduodenectomy or portal vein resection and reconstruction to achieve tumor-negative margins.5–7
With no reported difference in overall survival between the anatomical subtypes, most studies have reported separate histopathological results regarding biliary tract cancer but similar recommendations involving selection for surgery and/or oncological management. 8 Two recent studies have compared the histopathological characteristics and outcome of different anatomical subtypes of biliary tract cancer, finding no significant differences in overall survival.8,9 This questions prognosis, interpretation of oncological factors influencing survival and recurrence of biliary tract cancer subtypes. However, prognosis of biliary tract cancer with hilar engagement has only been investigated in one comparative and a few non-comparative studies, none of them including three anatomical subtypes.3,4,10 These studies are also limited by a low number of patients or by their multi-center designs spanning over a long period of time where surgical approach and histopathological interpretation may differ widely.4,3,10,11 Therein, the indications for this aggressive surgical approach and which patients may benefit from it remains an unsolved clinical dilemma.
Thus, this study aimed to investigate differences in prognostic factors and outcomes between patients with different biliary tract cancer subtypes engaging the hepatic hilum, treated by major liver resection including extrahepatic bile duct resection at a single high-volume center, to evaluate which patient-groups stand a greater chance to benefit from extensive surgery.
Methods
Patient selection and variables
All patients with histopathologically proven PHCC, IHCC, and GBC invading the hilum requiring major liver resection with extrahepatic bile duct resection, and if needed pancreaticoduodenectomy and/or portal vein resection, at Karolinska University Hospital (Stockholm, Sweden) between 2011 and 2021 were included. Major liver resection was defined as resection of ⩾3 liver segments. Also, complete hepatoduodenal ligament lymphadenectomy was done as well as sampling of the aortocaval (station 16b) and hepatic artery lymph nodes (8a), regardless of subtype. The retropancreatic lymph nodes (station 13) were sampled at the discretion of the operating surgeon. All data were obtained from medical records. Patients were subclassified and staged according to American Joint Committee on Cancer 8th edition staging system. Resection margins were categorized into margin to invasive cancer ⩾1 mm and margin to invasive cancer >0 mm. The margins reported were the transection margins of the distal bile duct, the proximal bile duct and the liver transection surface, and the circumferential dissection planes of the hepatoduodenal ligament and the hilum. 12 Additional factors analyzed for oncological outcomes included tumor size, grade of differentiation, lymph node positivity, number of positive lymph nodes, perineural invasion, and peritoneal perforation.
Postoperative complications were registered 90 days after surgery and classified according to the Clavien–Dindo (CD) system with major complications defined as CD IIIa–IV. 12 Postoperative mortality was regarded as death within 90 days after surgery (CD V). Overall survival was calculated from the date of resection to death from any cause or end of follow-up, including and excluding patients who died within 90 days of surgery. Recurrence-free survival was calculated from the date of resection to radiological evidence of local recurrence or distal metastasis with or without pathological confirmation.
Statistical analysis
Results were presented as numbers and percentages for categorical data and median with interquartile range (IQR) for continuous variables. Fisher’s exact test was used to compare categorical data and Kruskal–Wallis test was used for continuous data. The Kaplan–Meier method was used to estimate median overall and recurrence-free survival and to calculate survival curves.
Independent variables potentially affecting survival according to tumor and patient characteristics were analyzed using univariable and multivariable Cox proportional hazards regression models. A p value < 0.05 was regarded as statistically significant. Data were analyzed using SPSS 29.0 (IBM Corp., New York, US).
Ethical approval
Approval was obtained from the Regional Ethics Board in Stockholm, Sweden, Dnr 2022-06962-02, waiving the requirement of informed consent.
Results
Patient characteristics
A total of 108 patients with histologically proven biliary tract cancer undergoing major liver resection with extrahepatic bile duct resection were included in the study. The series included PHCC (n = 73), GBC (n = 24), and IHCC (n = 11). Median patient age was 64 years, and most patients were male (55%). No patient with distal cholangiocarcinoma had this operation during the study period. Patient characteristics are presented in Table 1.
Patient characteristics.
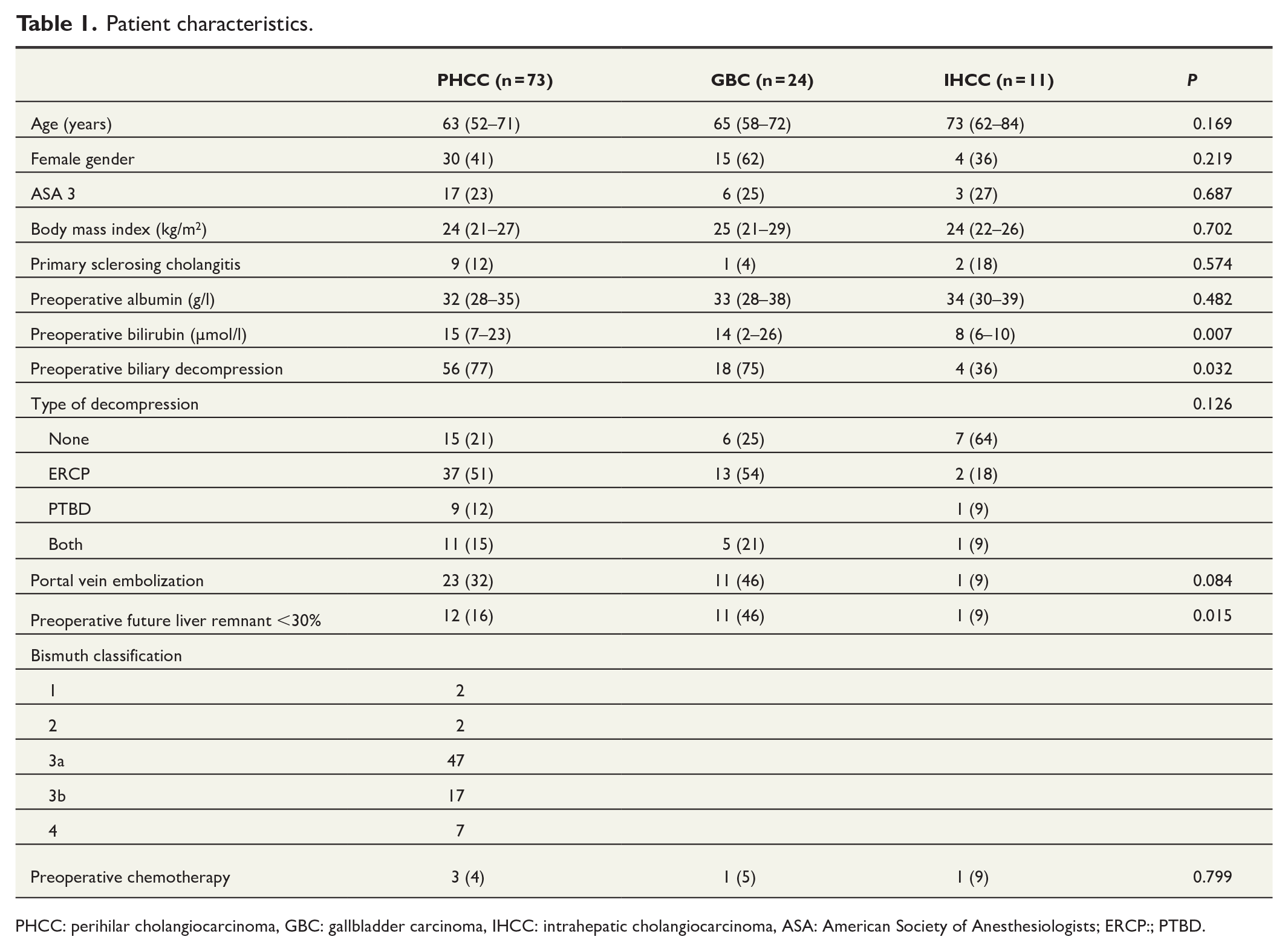
PHCC: perihilar cholangiocarcinoma, GBC: gallbladder carcinoma, IHCC: intrahepatic cholangiocarcinoma, ASA: American Society of Anesthesiologists; ERCP:; PTBD.
Perioperative data
GBC patients underwent more extensive liver resections than the other groups, where almost 80% of patients were operated with an extended right hemihepatectomy. Postoperative mortality in the entire cohort was 13% with a significant difference between biliary tract cancer subtypes (p = 0.023) were the mortality risk in patients with GBC (29%) surpassed that of PHCC (10%) and IHCC (0 %). Perioperative data for the three groups are shown in Table 2.
Perioperative data.
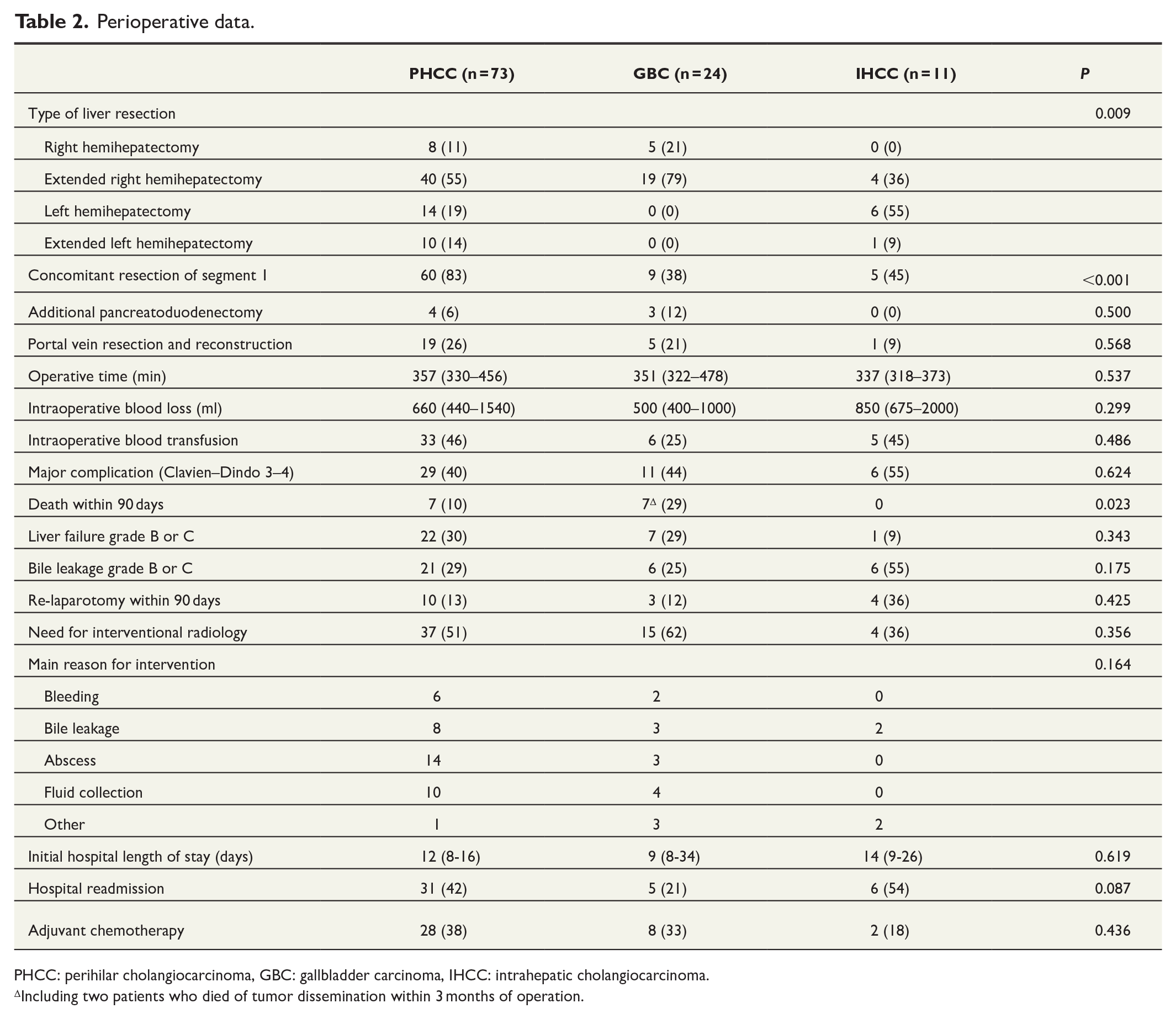
PHCC: perihilar cholangiocarcinoma, GBC: gallbladder carcinoma, IHCC: intrahepatic cholangiocarcinoma.
Including two patients who died of tumor dissemination within 3 months of operation.
Pathology characteristics
Tumor size was significantly larger (p < 0.001) in IHCC (90 mm) than in GBC (54 mm) and PHCC (30 mm), while lymph node positivity was most common (p = 0.021) in GBC (GBC 83%; IHCC 55%; PHCC 52%). Peritoneal cancer invasion occurred frequently in all groups: a majority of patients with GBC (61%), and approximately one third of patients with IHCC (36 %) and PHCC (33 %) (p = 0.061). Detailed pathology characteristics are presented in Table 3.
Pathology characteristics.
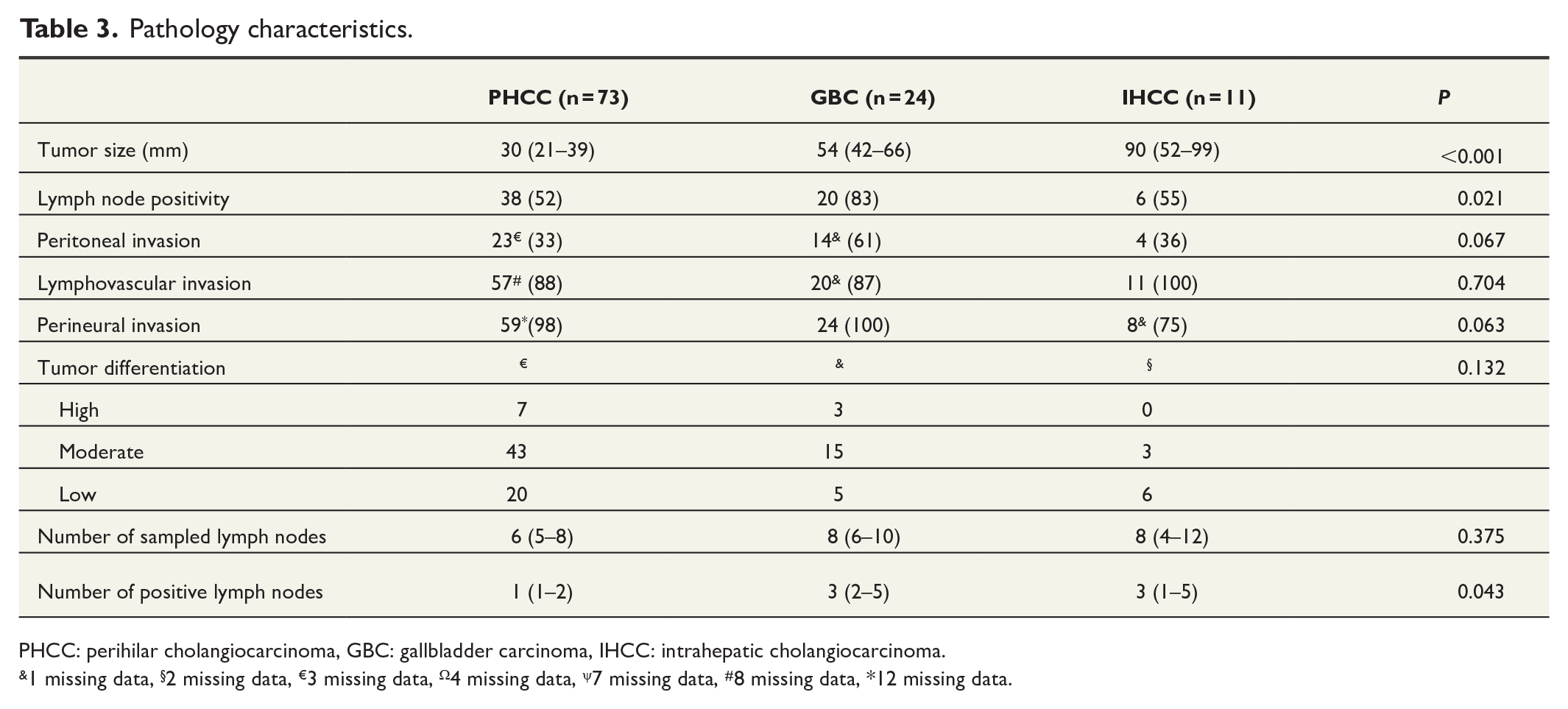
PHCC: perihilar cholangiocarcinoma, GBC: gallbladder carcinoma, IHCC: intrahepatic cholangiocarcinoma.
1 missing data, §2 missing data, €3 missing data, Ω4 missing data, ψ7 missing data, #8 missing data, *12 missing data.
In patients surviving more than 90 days after operation, histopathological analysis showed cancer-negative margins (>1 mm) in 19 patients only (17%), with no difference between groups (p = 0.606). By defining R0 as cancer-free margins (>0 mm), the number of patients with radical resection was 62 (66%) without difference between groups (p = 0.423).
Recurrence and survival
The median overall survival including all patients was 27 (19–56) months, 18 (13–23) months, and 21 (2–40) months for patients with PHCC, GBC, and ICC, respectively (p = 0.326), as shown in Fig. 1. The median overall survival for patients surviving >90 days post-operatively was 29 (23–34) months, 22 (16–28) months, and 21 (3–40) months for patients with PHCC, GBC, and ICC, respectively (p = 0.530), with an estimated 5-year survival rate of 32%, 20%, and 18% (Fig. 2A). Median recurrence-free survival differed significantly between subtypes (p = 0.046) where IHCC tended to recur earlier than GBC and PHCC (6 (1–10), 15 (8–21), and 18 (1–25) months, respectively) (Fig. 2B).
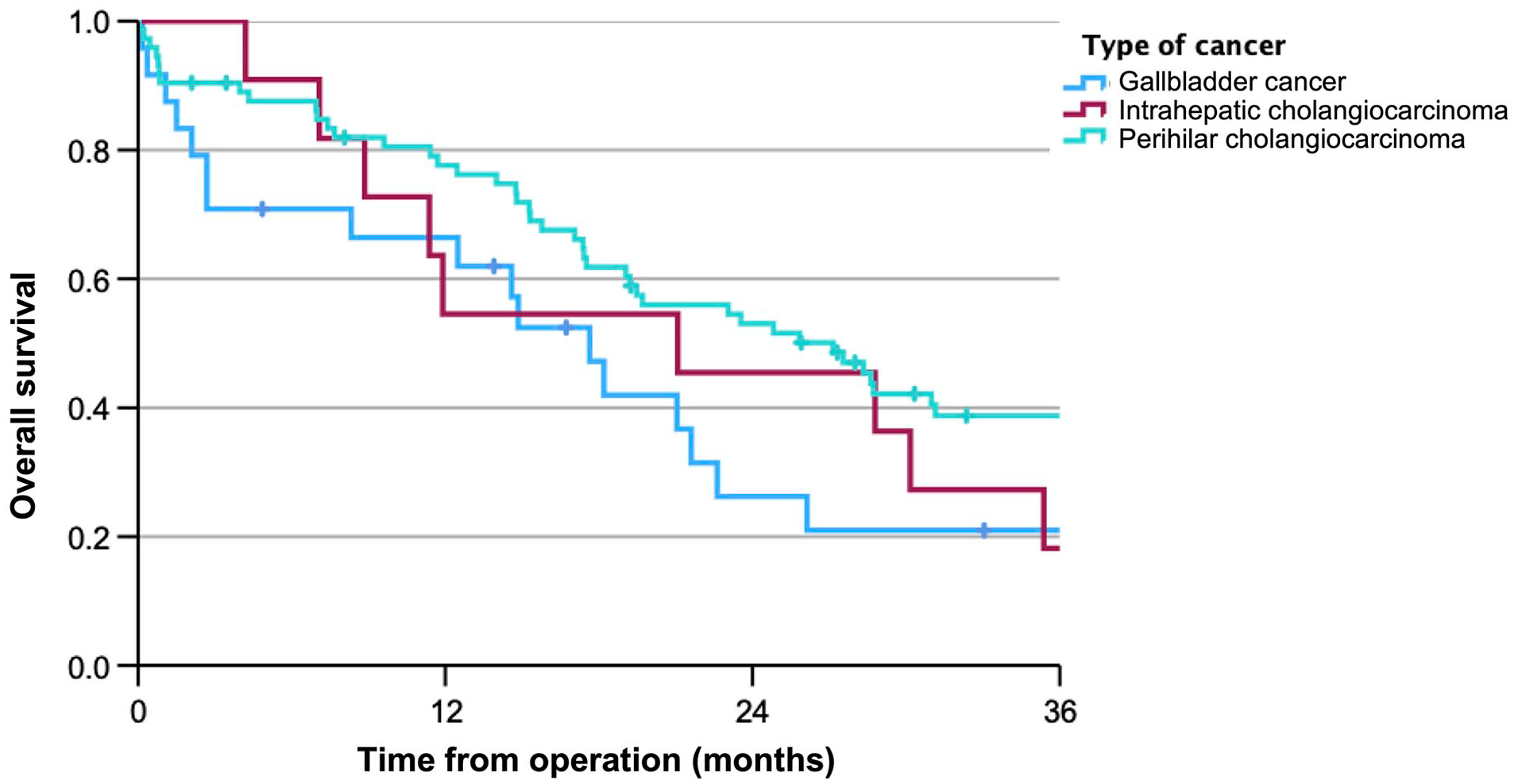
Overall survival in months for all resected patients.
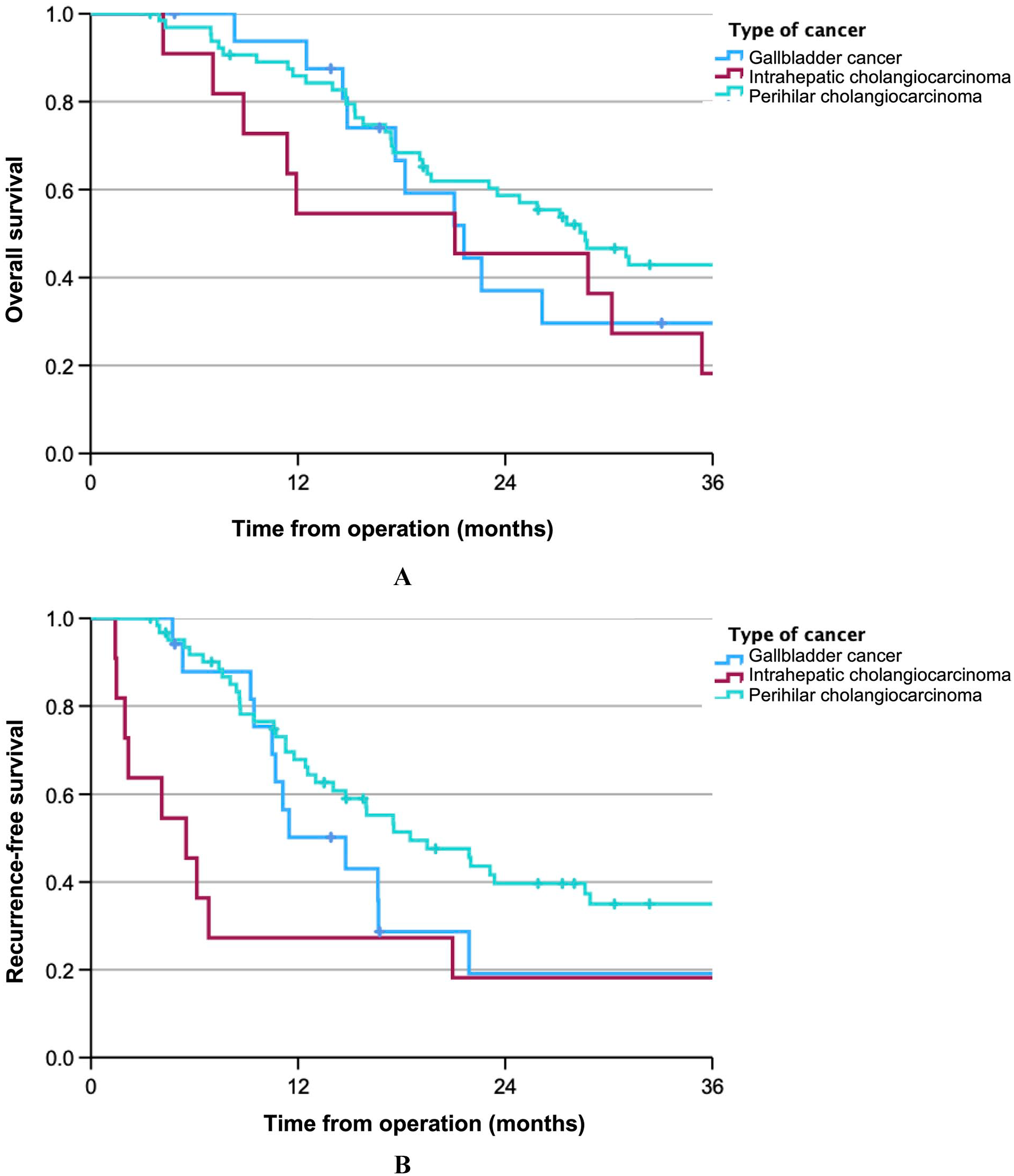
(A) Overall survival in months for patients surviving >90 days after operation and (B) recurrence-free survival in months for patients surviving >90 days after operation.
Among patients surviving >90 days post-operatively, multivariable analysis showed that tumor type did not significantly impact survival. As shown in Table 4, factors with a negative impact on survival included low preoperative albumin, lymph node positivity, peritoneal invasion, and postoperative liver failure.
Factors influencing survival (among survivors > 90 days).
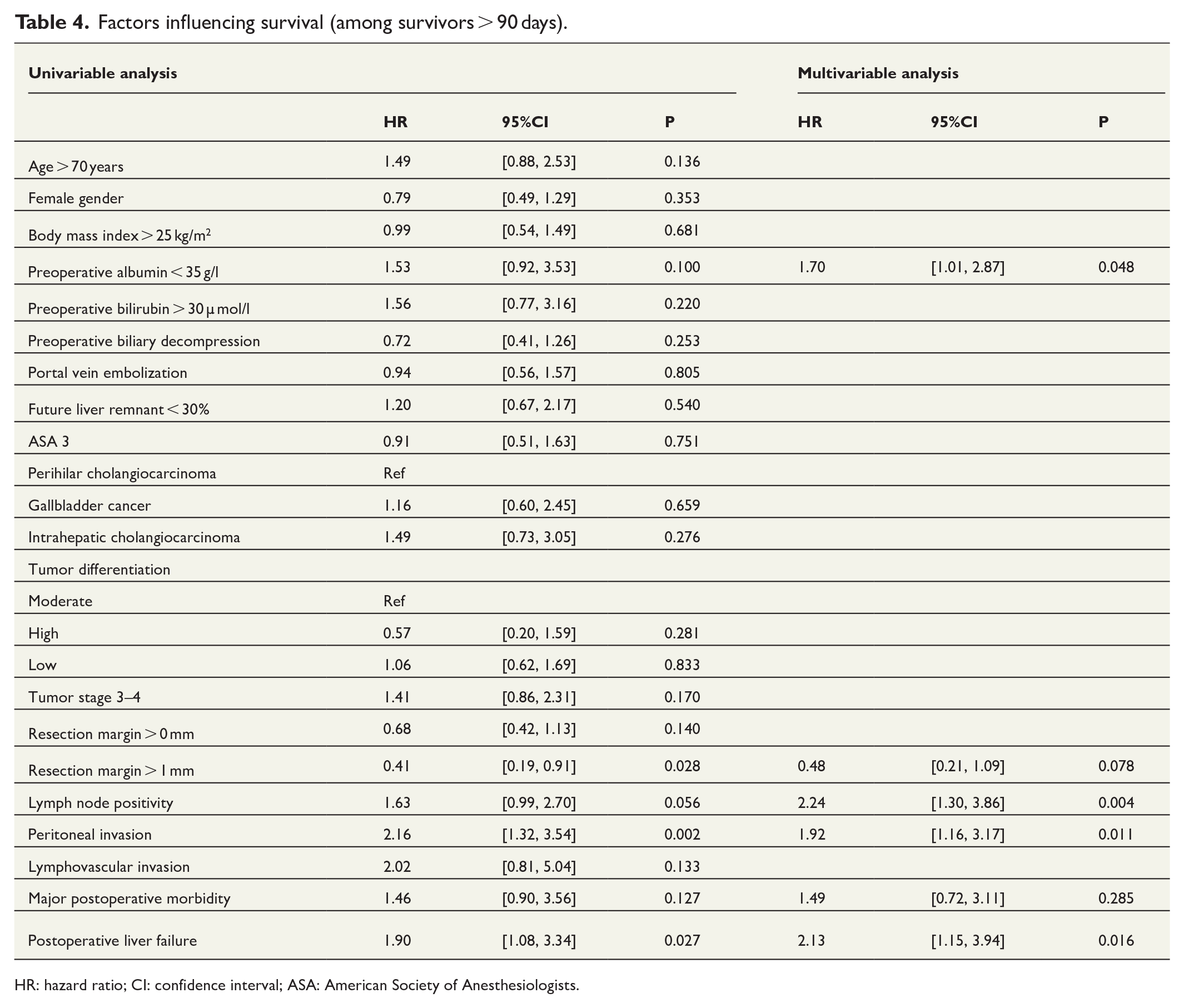
HR: hazard ratio; CI: confidence interval; ASA: American Society of Anesthesiologists.
Discussion
This single-center study compares tumor characteristics and outcomes of the anatomical subtypes of biliary tract cancer with hilar engagement after major liver resection with extrahepatic bile duct resection. Previous studies comparing biliary tract cancer have shown significant differences in postoperative morbidity and mortality that are often attributed to the anatomical location of the subtypes, rather than tumor biology, requiring surgical resection of different extents.4,8,9 Our study analyzed patients undergoing extensive surgical resection techniques for all biliary tract cancer subtypes involving the hepatic hilum and confirmed earlier results, showing no significant difference in median overall survival between subtypes (p = 0.52). Our data on GBC with hilar invasion has an overall survival comparable to that of resected GBC as reported in previous studies. 13 This is in line with a scarce number of publications that have investigated biliary tract cancer with hilar invasion, showing a slightly lower median and 5-year overall survival in GBC, and comparable results for IHCC.3,4,10,11,14–16
Noteworthy, concerning recurrence-free survival, our data revealed significant variation between biliary tract cancer subtypes, showing unusually early recurrence for IHCC, as compared to GBC and PHCC (6, 15, and 18 months, respectively). This is of interest as no study has previously compared recurrence-free survival between all subtypes with hilar invasion. The low recurrence-free survival in IHCC is in line with other western cohorts that recently have shown that about one in four patients with IHCC, regardless of tumor location, experience recurrence within 6 months after surgery.17,18
Our study showed comparable results to previous studies with an overall 90-day mortality of 13% and major morbidity of 42%.8,14,19 In this study, GBC patients had a significantly higher postoperative 90-day mortality (29%). The GBC group had a significantly higher number of patients operated with a future liver remnant (FLR) < 30%, although no difference in liver failure was reported between groups. Of note, two GBC patients died within 3 months due to disease dissemination. Contribution reasons for the high perioperative mortality may be the significantly higher frequency of right/extended right hemihepatectomies that was required to achieve negative margins in hilar-invading GBC, a well-known cause of higher morbidity and mortality compared to left or extended left hemihepatectomy.
To our knowledge, no study has previously compared the incidence of peritoneal invasion between biliary tract cancer subtypes nor studied the presence of peritoneal invasion in PHCC. This is a point of interest as perforation of the visceral peritoneum is known to be an adverse prognostic factor in IHCC as well as GBC and is associated with a higher incidence of lymph node metastasis and recurrence.20,21 In our case, the presence of peritoneal invasion in PHCC was 33%. Results from our multivariable analysis showed that peritoneal invasion is an independent factor (p = 0.011) impacting negatively on survival. This finding may support routine reporting of peritoneal invasion-status in resected cholangiocarcinoma, with benefit from an implementation of standardized histopathological reports.
Another finding in our study is the histopathological results regarding IHCC and GBC with hilar involvement. To this date, very limited eastern series have described or compared either hilar IHCC or GBC with PHCC with none looking at all three subtypes together.3,4,11,15,16 Moreover, few studies have investigated histopathological factors in GBC with hilar invasion influencing survival with only two older reports investigating this entity.3,11 Our results showed that, after resection, hilar-invading IHCC and GBC had significantly more known adverse histopathological factors like lymph node positivity, higher number of positive nodes, and larger tumor size. This is in line with previous studies comparing IHCC with PHCC4,8,9,15,22 and can be attributed to a more advanced tumor stage at presentation in IHCC and GBC when involving the hilum as compared to PHCC, which arises primarily within this region. Our multivariable analysis showed that lymph node positivity was also an independent factor predicting overall survival. As opposed to the extended lymphadenectomy used in this study, results from studies using local lymphadenectomy, which does not involve the whole hepatoduodenal ligament and aortocaval/hepatic artery stations may have understaged the tumor, thus underestimating the prognostic impact of lymph node metastases.23,24
When investigating positive resection margin status, which previously has been shown to be a factor negatively influencing both overall and recurrence-free survival in biliary tract cancer, no significant differences in survival were found in this study, although, by defining a positive margin as a cancer-free margin of >1 mm, we found a close to significant effect (p = 0.078). Our study had an unusually low number of patients with cancer-free margins. This may be attributed to extensive tissue sampling and a higher number of resection margins assessed. Our cohort included the circumferential hepatoduodenal ligament and hilar margins, in contrast to most eastern as well as western studies that reported only radial, proximal, and distal bile duct margins.25,26 Ambiguity and inconsistency in reporting resection margins has previously been described as a controversial subject in biliary tract cancer due to lack of a standardized method of histopathological assessment, which makes comparisons between studies difficult. 27
The main weakness of this study is the relatively small number of patients in each group, making firm conclusions difficult to draw. Major liver resection including resection of extrahepatic bile ducts is standard treatment for localized PHCC. However, few patients with hilar-invading GBC and IHCC are considered for surgery because of advanced disease, introducing a selection bias to our results. With this in mind, conclusively, IHCC and GBC with hilar involvement demonstrated comparable overall survival but worse histopathological characteristics as compared to PHCC. This may be explained by the fact that hilar-invading GBC and IHCC is usually diagnosed at an advanced stage reflecting the difficulty in achieving curative resection despite undergoing the same extent of surgery as PHCC. This may be further supported by our results showing a shorter recurrence-free survival of 6 months in IHCC as compared to GBC (15 months) and PHCC (18 months). Our results may hence question the aggressive surgical approach in hilar-invading IHCC and aid future discussion on which of the biliary tract subgroups with hilar invasion can benefit from surgical resection. Furthermore, the differences displayed in recurrence-free survival in our study motivate further research on complementary and alternative oncological treatments like kinase inhibitors, 28 which only recently have shown promising results in IHCC. The elevated 90-day mortality in patients with GBC similarly motivates consideration of alternative approaches in frail or elderly patients, although preoperative optimization of the FLR (that is, volume modulation) maybe was underused in this group. In addition, two patients with GBC died of tumor dissemination within 90 days of operation after curative resection, addressing the need for a meticulous preoperative radiological workup regarding tumor spread.
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by HAA and CS. The first draft was written by HAA, and all authors commented on the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Region Stockholm and the Center for Innovative Medicine, Stockholm, Sweden.
