Abstract
Introduction:
The best approach for total mesorectal excision (TME) remains controversial. Two recently described approaches are robotic TME (RTME) and transanal TME (TaTME). This systematic review and meta-analysis aimed to compare the outcomes between robotic surgery and TaTME in patients undergoing rectal cancer resection.
Methods:
We structured this systematic review and meta-analysis according to the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) guidelines 2020 and Assessing the Methodological Quality of Systematic Reviews (AMSTAR) guidelines. An electronic search of relevant literature was conducted on 20 May 2023. The protocol was registered in PROSPERO (CRD42023435259).
Results:
Eleven eligible nonrandomised studies were included in this study. The study included 2796 patients (RTME = 1800; TaTME = 996). The RTME group had a higher rate of complete TME. However, no significant differences were observed in mortality, morbidity, severe complications, operative time, conversion rate, anastomotic leak, hospital stay, CRM-positive resection margin, distal resection margin, number of harvested lymph nodes, abdominoperineal resection (APR) rate, or local recurrence between the RTME and TaTME groups.
Conclusion:
The RTME technique may ensure a higher rate of complete TME than TaTME. However, no significant differences were observed in most postoperative outcomes and oncological safety between the RTME and TaTME groups. Evidence does not conclusively favor one technique over the other, highlighting the need for additional randomized controlled trials to better define their roles in rectal cancer surgery.
Keywords
Introduction
Total mesorectal excision (TME) has been established as the gold standard treatment for middle and lower rectal cancers since its initial description by Heald. 1 Over the years, numerous efforts have been made to optimize postoperative results through the advancement of minimally invasive techniques and explore alternatives to TME in patients with favorable response to neoadjuvant therapy. The primary objective of this research was to ensure improved postoperative outcomes while maintaining oncological safety. The ideal surgical approach for rectal cancer remains a topic of debate. TME can be performed using various methods including open surgery, laparoscopy, robot-assisted surgery (RTME), and the transanal approach (TaTME).2,3 Several studies have compared laparoscopic total mesorectal excision (LaTME) and TaTME with RTME.4,5 The conclusions of these studies favor TaTME and RTME in terms of postoperative outcomes and specimen quality.4,5 Robotic surgery provides enhanced comfort and control of the surgeon’s camera. The TaTME is a relatively new approach. It has emerged to address the limitations of LaTME.6,7 It involves laparoscopic rectal dissection through the anal canal in a caudal-to-cephalad direction. 8 Several studies have demonstrated that TaTME provides excellent access to the distal rectum, facilitating better margin control, and theoretically, a higher R0 resection, especially in cases with challenging pelvic anatomy.9,10 Both TaTME and RTME aim to enhance the quality of rectal cancer resection, although few published trials have directly compared the outcomes of these two techniques. The objective of this systematic review and meta-analysis was to offer a comprehensive comparison of the outcomes of RTME and TaTME in patients undergoing rectal cancer resection.
Methods
This systematic review and meta-analysis were performed according to the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) guidelines 2020 11 and Assessing the Methodological Quality of Systematic Reviews (AMSTAR) guidelines. 12 The protocol was registered in PROSPERO (CRD42023435259).
Electronics searches
An electronic search of the relevant literature was conducted on 20 May 2023, using “Cochrane Library’s Controlled Trials Registry and Database of Systematic Review,” “United States National Library of Medicine,” “National Institutes of Health PubMed/MEDLINE,” “Excerpta Medica Database,” “Embase,” “Scopus,” and “Google Scholar” databases. The keywords used were as follows: “total mesorectal excision,” “robot,” “robot-assisted,” “transanal,” “rectal cancer,” “TaTME,” “TME,” “robotic surgery,” “meta-analysis,” “prospective randomized trials,” “controlled clinical trials,” and “mesh placement.” We cross-checked the reference list of relevant reviews for eligible clinical trials.
Included studies
All randomized controlled trials (RCTs) and controlled clinical trials (CCT) comparing RTME with TaTME to treat middle and low rectal cancers, published in peer-reviewed journals, were considered for analysis. Data from non-comparative studies, review articles, and case series (fewer than 10 cases) were excluded from the analysis. No language restrictions were applied.
Participants
Adults (aged >18 years) of either sex who underwent RTME or TaTME were included. We studied two different approaches to rectal cancer surgery in which TME was performed: RTME and TaTME.
Outcomes measures
Primary outcomes were mortality, postoperative morbidity, and severe complications. Mortality and morbidity were considered when they occurred within 30 and 90 days after rectal resection, respectively. Most studies used the Clavien–Dindo classification to report postoperative complications. 13 Severe complications were classified as grade ⩾III.
The secondary outcomes were operative time, conversion rate, anastomotic leak, hospital stay, CRM-positive resection margin, distal resection margin, complete TME, number of lymph nodes retrieved, abdominoperineal resection (APR), and local recurrence.
Study selection
Two authors independently reviewed all abstracts (M.A.C. and A.G.). All full-text studies that met the inclusion criteria were included. Disagreements were resolved through discussions after consulting a third member of the review team.
Assessment of studies quality and risk of bias assessment
All studies that met the selection criteria were appraised independently by two authors (M.A.C. and A.M.) using the CONSORT statement for RCTs 14 or the MINORS scale for CCT. 15 For bias assessment, we used the Newcastle-Ottawa Scale 16 to assess the quality and risk of bias in randomized and non-randomized clinical trials. The scoring system covers eight items related to three major domains (selection of exposed and non-exposed cohorts, comparability, and outcome assessment).
Data extraction
Each author extracted the data independently (M.A.C., B.K., A.C.D.), and disparities were settled with the senior authors after discussion. For the propensity score-matched studies, we used data from a matched cohort. The included studies fully matched most of the information provided in the article.
Missing data
We contacted the authors by e-mail regarding the occurrence of unclear bias domains or missing primary outcome information in our meta-analysis. If the data were not reported numerically, they were extracted from figures.
Handling continuous data
Continuous data were analyzed using Review Manager 5.3.5 a statistical package from the Cochrane Collaboration for Meta-analysis. 17 If the mean and standard deviation (SD) were not reported, they were estimated from the interquartile range (IR) and median based on the formula described by Hozo et al. 18
Assessment of heterogeneity
To assess heterogeneity, the Cochrane chi² test (Q-test), I² statistic, and variance TAU² were used to estimate the degree of heterogeneity. 19 Funnel plots were used to identify the studies responsible for heterogeneity. Subgroup analysis was performed when all the included studies reported outcomes.
Summary of findings
Two authors (M.A.C. and M.A.D.) independently assessed the certainty of evidence using Grading of Recommendations Assessment, Development, and Evaluation (GRADE). 20 We considered the study limitations in terms of the consistency of effect, imprecision, indirectness, and publication bias. The certainty of the evidence was assessed as high, moderate, low, or very low. We considered the following criteria for upgrading the certainty of the evidence, if appropriate: large effect, dose-response gradient, and plausible confounding effect. We used the methods and recommendations described in sections 8.5 and 8.7, and Chapters 11 and 12 of the Cochrane Handbook for Systematic Reviews of Interventions. GRADEpro GDT software was used to prepare the “Summary of findings tables.” Reasons for downgrading or upgrading the included studies were explained by using footnotes and comments.
Evaluation of effect size
RevMan 5.3.5 statistical package from the Cochrane Collaboration was used for meta-analysis. 21 Mean difference (MD) was selected as an effective measure of continuous data. Odds ratios (OR) with 95% confidence intervals (95% CI) were calculated for dichotomous variables. A random-effects model was used. The threshold for significance was set at p < 0.05.
Results
Literature search results
Twelve potentially relevant articles were identified (Fig. 1). We retained 11 eligible studies4,22–31 and three studies were excluded for the following reasons: one international multi-center prospective audit with incomplete data on the subgroups, 32 one network meta-analysis, 5 and one protocol. 33 These articles were published between 2017 and 2023. No RCT has been carried out on this subject. Nine studies involved 2796 patients (RTME, 1800 patients; TaTME, 996 patients) (Table 1). The patients’ demographic data are summarized in Table 2. The mean age was 54–67.28 years in the RTME group and 58–66.6 years in the TaTME group.
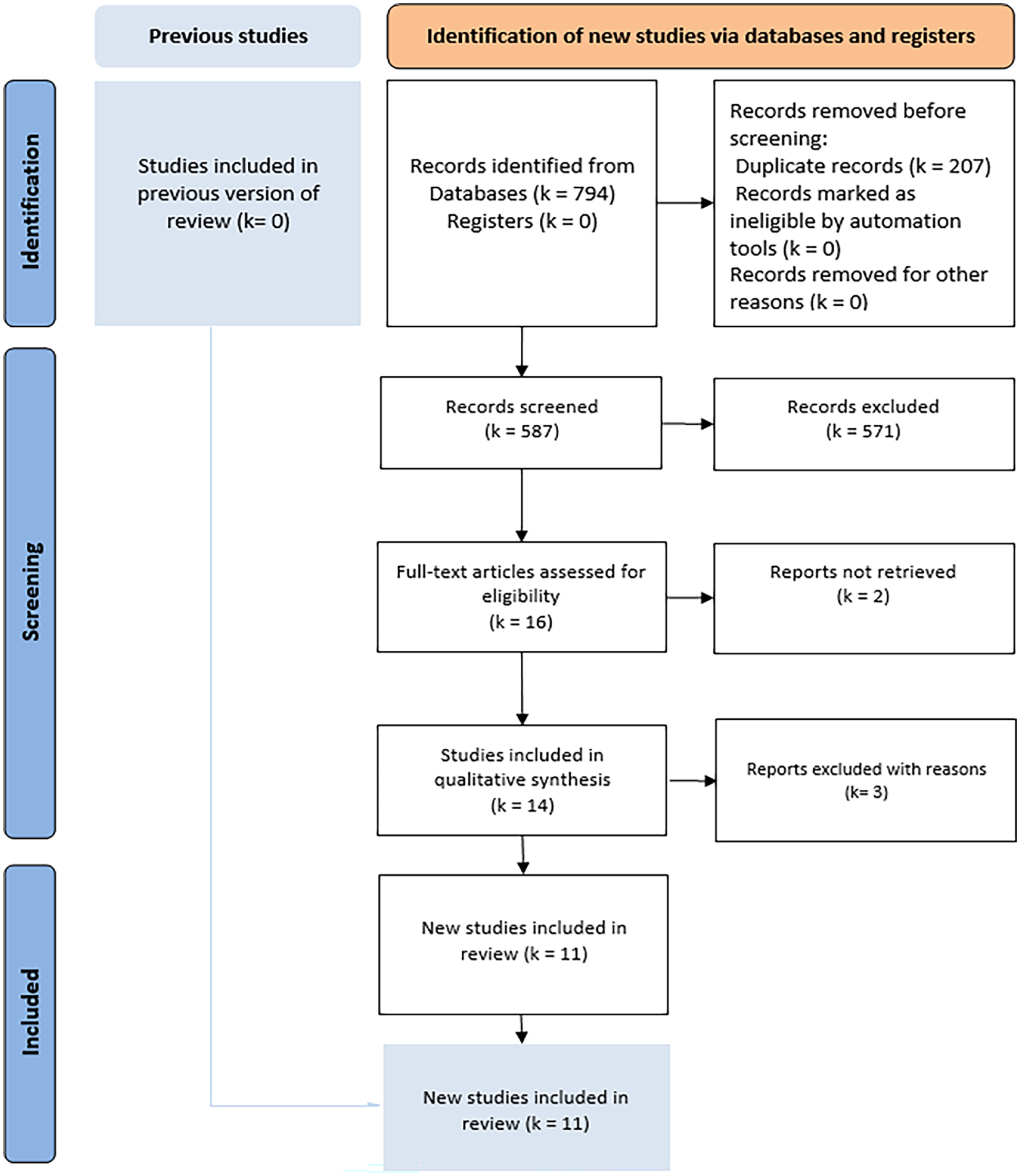
PRISMA flow diagram of the bibliographic research.
List of the retained studies.
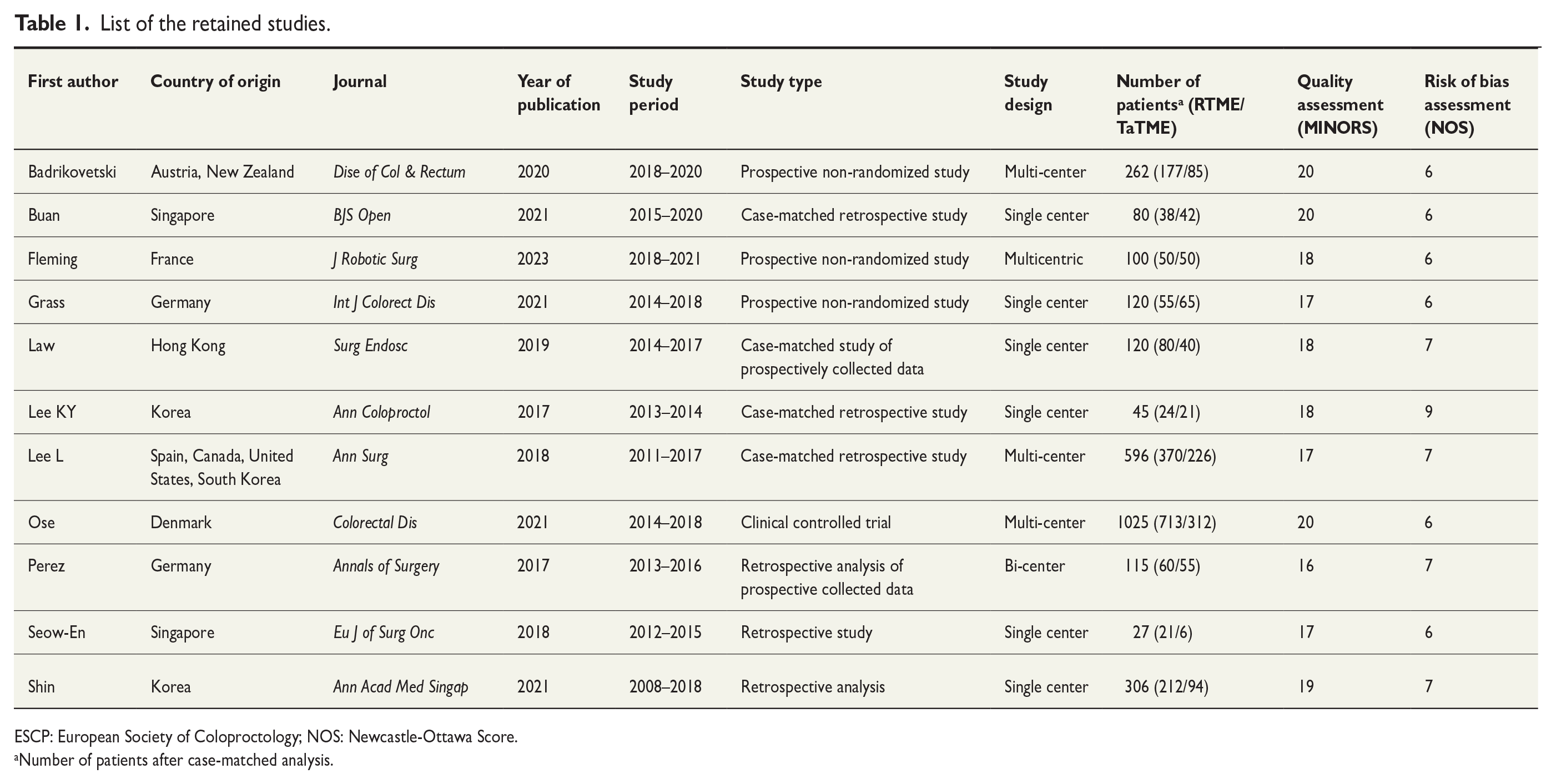
ESCP: European Society of Coloproctology; NOS: Newcastle-Ottawa Score.
Number of patients after case-matched analysis.
Demographic data of the retained studies.
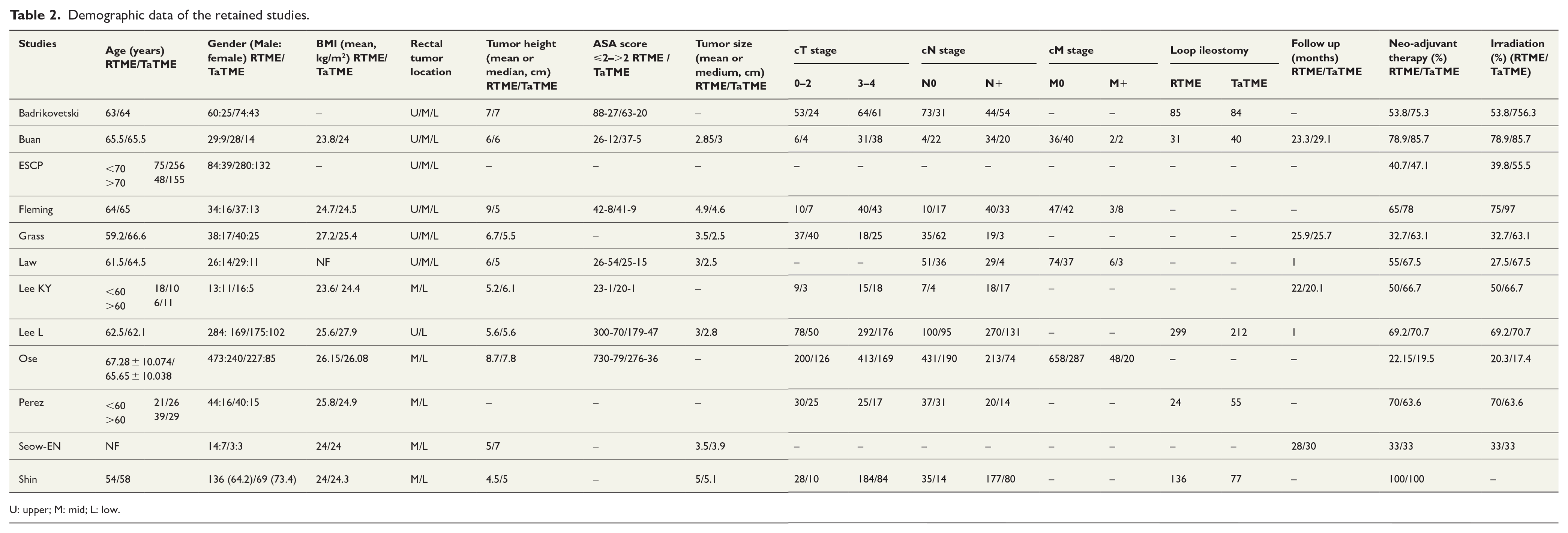
U: upper; M: mid; L: low.
Primary outcomes
Mortality
The mortality rate was reported in six studies22,26–29,31 and occurred in nine of 1345 patients in the RTME group and four of 690 patients in the TaTME group (OR = 0.98, 95% CI: 0.32 to 2.95, p = 0.97) (Fig. 2A).
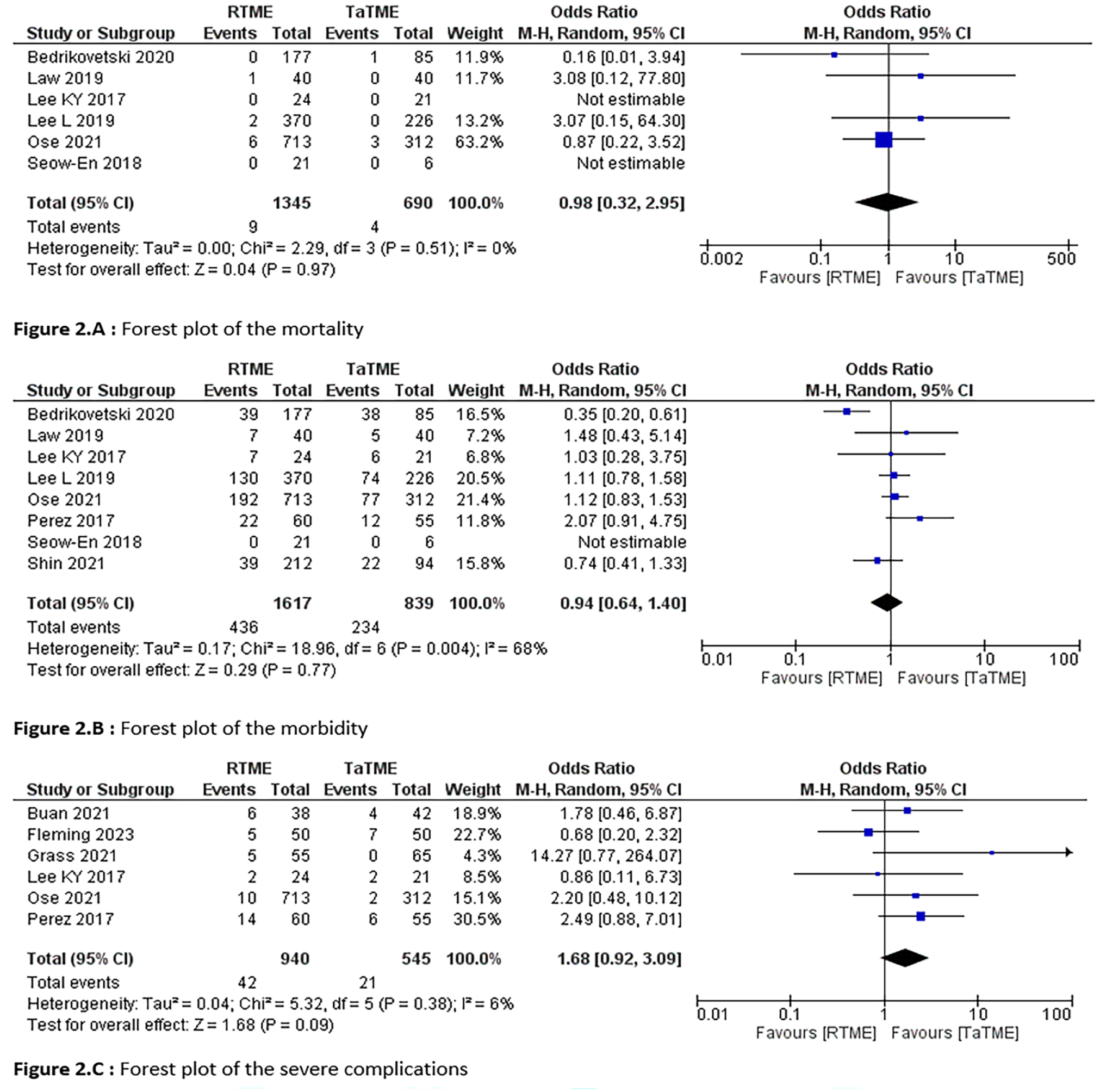
Forest plot of the primary outcomes.
Morbidity
Morbidity rates have been reported in eight studies.4,22,26–31 Postoperative complications were reported in 436 out of the 1617 patients in the RTME group and 234 of the 839 patients in the TaTME group (OR = 0.94, 95% CI: 0.64 to 1.40, p = 0.77; Tau²= 0.17) (Fig. 2B).
Severe complications
A Clavien–Dindo complication ⩾III was reported in six studies,4,23–25,28,31 in 42 of 940 patients in the RTME group and 21 of 545 patients in the TaTME group (OR = 1.68, 95% CI: 0.92 to 3.09, p = 0.09; Tau²= 0.04) (Fig. 2C).
Secondary outcomes
Operative time
Nine studies reported operative time.4,23–30 It was reported in 870 patients in the RTME group and 599 patients in the TaTME group (MD = 4.6, 95% CI: −15.42 to 24.62, p = 0.65, Tau²= 700).
Conversion rate
The conversion rate was reported in eight studies.4,23,26–31 Fifty-six of 1478 patients in the RTME group and 17 of 796 patients in the TaTME group were converted (OR = 1.31, 95% CI: 0.56 to 3.07, p = 0.54).
Anastomotic leak
The anastomotic leak rate was reported in 10 studies.4,22,23,25–31 Anastomotic leak occurred in 120 of 1710 patients in the RTME group and 76 of 946 patients in the TaTME group (OR = 1.00, 95% CI: 0.60 to 1.65, p = 0.99, Tau²= 0.19).
Hospital stay
Eight studies reported data on hospital stays22,22–27,29,30 including 633 and 422 patients in the RTME and TaTME groups, respectively (MD = 0.64, 95% CI: −0.39 to 1.68, p = 0.22, Tau²= 0.87).
Positive CRM
This outcome was reported in nine studies.22,23,25–31 Positive CRM was reported in 49 of 1650 patients in the RTME group and 38 of 891 patients in the TaTME group (OR = 0.90, 95% CI: 0.36 to 2.25, p = 0.83, Tau²= 0.83).
Distal resection margin
Seven studies reported the distal resection margin retrieved in the operative specimen4,23,26–30 in 805 patients in the RTME group and 484 patients in the TaTME group (MD = 0.04, 95% CI: −0.51 to 0.58, p = 0.90, Tau²= 0.47).
Complete TME
Seven studies assessed the complete TME rate.4,23–25,27,28,30 In total, 772 of the 809 patients in the RTME group and 502 of the 553 patients in the TaTME group achieved a complete TME. A significantly higher rate of complete TME was observed in the RTME group (OR = 1.77, 95% CI: 1.13 to 2.78, p = 0.01).
Number of retrieved lymph nodes
Eight studies reported the number of lymph nodes retrieved from the operative specimen.4,22,23,25–29 This was reported for 785 and 540 patients in the RTME and TaTME groups, respectively (MD = 0.28, 95% CI: −0.58 to 1.14, p = 0.53, Tau²= 0.36).
APR rate
The APR rate was reported in four studies22,24,25,31 and 69 of 935 patients in the RTME group and 15 of 512 patients in the TaTME group underwent APR (OR = 4.42, 95% CI: 0.58 to 33.50, p = 0.15, Tau²= 3.10).
Local recurrence
This outcome was reported in three studies.23,27,29 Local recurrence was diagnosed in 8 of 83 patients in the RTME group and 3 of 69 patients in the TaTME group (OR = 1.82, 95% CI: 0.50 to 6.56, p = 0.36).
Quality assessment of the included studies and reporting of the effects of RTME
MINORS and NOS scores are shown in Table 1. A summary of the evidence is presented in Table 3.
Summary of findings table.
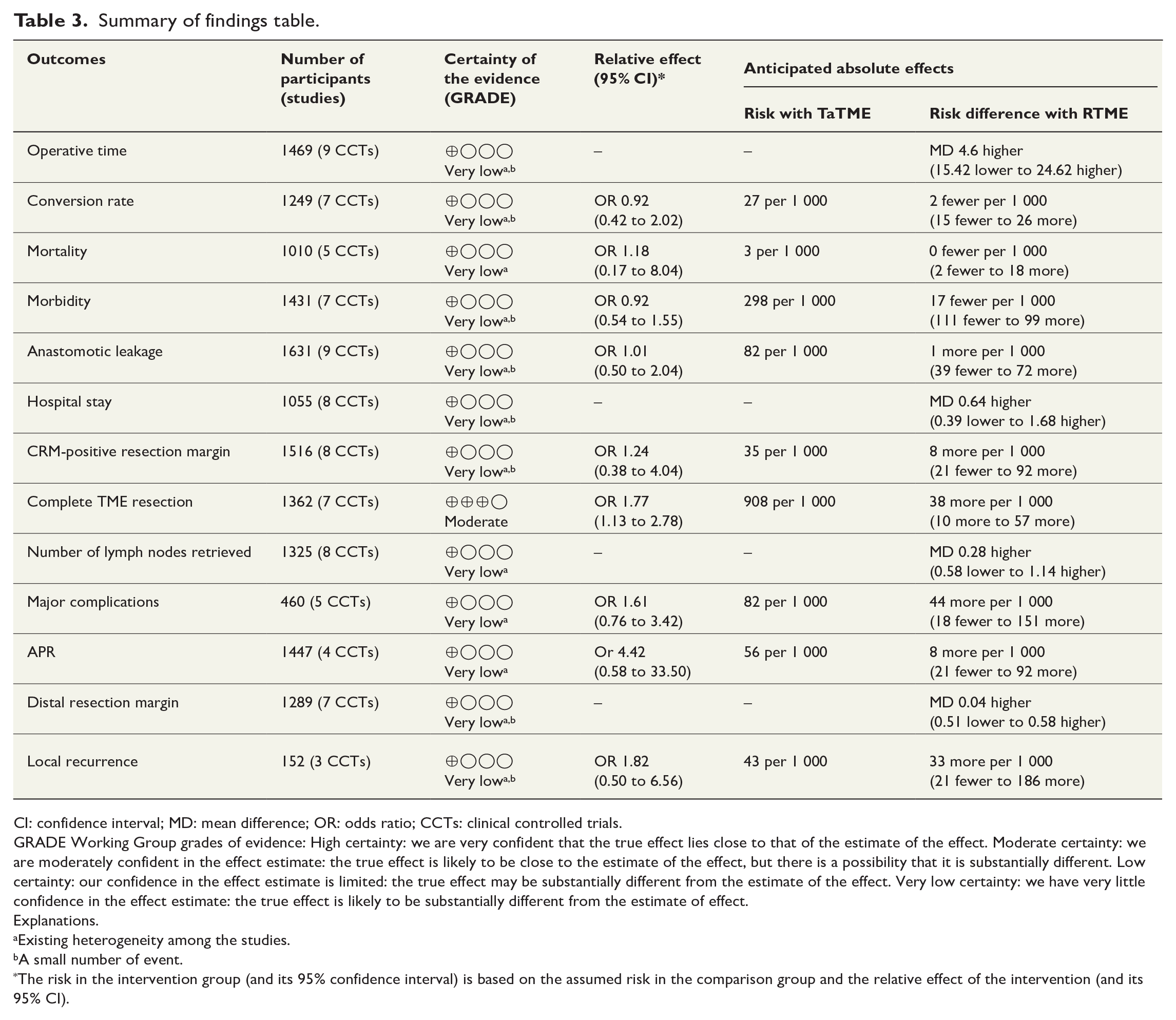
CI: confidence interval; MD: mean difference; OR: odds ratio; CCTs: clinical controlled trials.
GRADE Working Group grades of evidence: High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
Explanations.
Existing heterogeneity among the studies.
A small number of event.
The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
Discussion
This systematic review and meta-analysis indicate that the RTME may exhibit a higher complete TME rate than TaTME. There was no discernible distinction between the two groups in terms of mortality, morbidity, severe complications, operative time, conversion, anastomotic leak, hospital stay, CRM-positive resection margin, distal resection margin, number of harvested lymph nodes, APR rate, and local recurrence.
RTME and TaTME lack standardization due to their ongoing evolution. On one hand, TaTME has faced criticism regarding its oncological safety, 34 while RTME has been criticized for its high cost and steep learning curve. This learning curve may be shorter in the RTME group because the anatomical landmarks resemble those of LaTME and open TME.35,36 In addition, the TaTME technique needs two approaches, perineal and abdominal, often performed with two teams to save time. These disparities may explain some of the heterogeneity observed among the included studies regarding the operative time. Studies suggest that the learning curve for these techniques in rectal cancer ranges from 15 to 43 cases for RTME and from 40 to 71 cases for TaTME.37–39
When new techniques are introduced, safety is paramount. Consequently, postoperative mortality and morbidity were the primary outcomes in our study. In addition, these outcomes can significantly affect the overall survival rate.40,41 Furthermore, anastomotic leakage has been associated with higher rates of local recurrence, which can negatively affect survival. 41 Initial findings from the TaLar trial randomizing rectal cancer patients between TaTME and LaTME indicated comparable mortality. 41 Similarly, the REAL trial suggested that short-term morbidity and mortality is equivalent between RTME and LaTME (REF 49). This aligns with the findings of the ROLARR trial, which reported a 30-day mortality rate of 0.9%.42,43 Our study found no significant difference in severe complications or anastomotic leaks between RTME and TaTME. Several meta-analyses have suggested that RTME is associated with a lower risk of conversion to open surgery, although this was not statistically significant in most studies including ours.44,45 The ROLARR trial showed a reduced conversion risk in RTME in complex patients (high body mass index (BMI), male sex, and low tumors). According to Corrigan et al., 46 the ROLARR trial did not meet the statistical significance criteria for conversion risk, due to a learning effect but multilevel logistic regression analysis revealed that RTME may have less conversion in hands of experienced robotic surgeons. In contrast, the TaLaR trial reported a significantly higher conversion rate in LaTME than that in TaTME. Among 545 patients, only 6 (1.1%) in the LaTME group required transanal surgery due to tumor-related complications, while none of the TaTME group required conversion. 41
Our study assessed the surgical resection quality by examining the circumferential resection margin, distal resection margin, TME completion, lymph node harvest, and local recurrence. Irradiation is an important confounder regarding CRM. Most of the countries using TaTME irradiate over 60% of patients. In the study by Ose and Perdawood, 31 only 16% of TaTME were irradiated versus 20% in the RTME group which could be explained by the higher rate of cT1/cT2 tumors in patients undergoing TaTME. Our findings showed that the integrity of the mesorectum was higher in the RTME group than in the TaTME group. The current literature indicates wide variation in surgical resection quality in TaTMEs. 47 A systematic review and meta-analysis 48 published in 2021 highlights that RTME could provide a larger distal resection margin but a longer operation time than TaTME. A recent network meta-analysis 49 assessing operative and oncological outcomes of open, laparoscopic, RTME, and TaTME concluded that in selected patients, RTME may provide a greater distal resection margin and a shorter length of hospital stay, which is inconsistent with our findings and the literature where sphincter preservation is more likely with TaTME. Numerous studies have attempted to combine the advantages of both the TaTME and RTME. The robotic platform facilitates precise dissection and high-quality TME, whereas the TaTME offers better control of the distal margin. Although Robotic TaTME (RoTaTME) is still in its early stages, initial reports are promising.
The higher rate of complete TME in RTME did not translate into a significant difference in the local recurrence rates. This may seem counterintuitive, especially when considering the emphasis on achieving clear resection margins to reduce the risk of local recurrence. It is important to mention that opening the bowel close to the tumor is a “no-go” in all other oncological surgeries and may be a reason for increased local recurrence rates in some reports, especially in countries with a low irradiation rate. In addition, entering the plane at the most crucial part of the operation, close to the tumor or close to the urethra, where failure to find the plane at once may have severe consequences. Several studies have reported an alarming occurrence of local recurrences especially during the introduction of the procedure and among patients who were not irradiated.34,50,51 The short follow-up and lack of oncological data in included studies make the results difficult to evaluate.
This systematic review and meta-analysis had several limitations. Only 11 studies were included, and none of these trials were randomized, which reduced the quality of evidence and could introduce reporting bias. Certain outcomes, such as sphincter-saving resection and low anterior resection syndrome, could not be assessed due to missing data. Six studies included some cases of upper rectal cancer (>10 cm from the anal verge) where a partial mesorectal excision probably would be sufficient. Furthermore, lack of long-term data is a major obstacle.
In conclusion, based on available data, it is challenging to definitively establish the superiority of RTME or TaTME. The robotic approach may offer a higher rate of complete TME; however, outcomes regarding mortality, morbidity, severe complications, operative time, conversion, anastomotic leak, hospital stay, CRM-positive resection margin, number of harvested lymph nodes, APR rate, and local recurrence are similar. Additional RCTs are imperative for better positioning RTME and TaTME within the repertoire of rectal cancer surgeons.
Footnotes
Author contributions
Mohammad Iqbal Hussain, Mohamed Ali Chaouch, Maissa Jellali, Amine Gouader, Alessandro Mazzotta, Adriano Carneiro da Costa, and Bassem Krimi wrote the draft and performed statistical analysis. Niccolo Petrucciani, Jim Khan, and Hani Oweira supervised the study and verified the final version of the manuscript. All authors reviewed and validated the final version of the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval and patient consent were not required as this was a systematic review and meta-analysis of previously published studies.
Provenance and peer review
Not commissioned, externally peer-reviewed
