Abstract
Aim:
Rectal cancers requiring beyond total mesorectal excision (bTME) are traditionally operated using an open approach, but the use of minimally invasive robot-assisted procedures is increasing. Introduction of minimal invasive surgery for complex cancer cases could be associated with compromised surgical margins or increased complication rates. Therefore, reporting results both clinical and oncological in large series is important. Since bTME procedure reports are heterogeneous, comparing results is often difficult. In this study, a magnetic resonance imaging (MRI) classification system was used to describe the bTME surgery according to pelvic compartments.
Methods:
Consecutive patients with primary rectal cancer operated with laparoscopic robot-assisted bTME were prospectively included for 2 years. All patients had tumors that threatened the mesorectal fascia, invaded adjacent organs, and/or involved metastatic pelvic lateral lymph nodes. Short-term clinical outcomes and oncological specimen quality were registered. Surgery was classified according to pelvic compartments resected.
Results:
Clear resection margins (R0 resection) were achieved in 95 out of 105 patients (90.5%). About 26% had Accordion Severity Grading System of Surgical Complications grade 3–4 complications and 15% required re-operations. About 7% were converted to open surgery. The number of compartments resected ranged from one to the maximum seven, with 83% having two or three compartments resected. All 10 R1 resections occurred in the lateral and posterior compartments.
Conclusions:
The short-term clinical outcomes and oncological specimen quality after robot-assisted bTME surgery were comparable to previously published open bTME surgery. The description of surgical procedures using the Royal Marsden MRI compartment classification was feasible.
Keywords
Context and relevance
Minimally invasive robot-assisted surgery is emerging as an alternative to open procedures in rectal cancers requiring beyond total mesorectal excision. Larger studies reporting outcome is needed. A prospectively included cohort of 105 patients was classified according to a compartment system, and short-term clinical outcomes and oncological specimen quality are presented.
Introduction
Although the introduction of neoadjuvant therapy has resulted in improved local control in the management of advanced rectal cancers, 1 beyond total mesorectal excision (bTME) surgery is still required for complete tumor removal in many cases. Conventional laparoscopy has not been widely adopted for bTME surgery, mainly because the areas anteriorly and laterally in the narrow and confined space of the pelvis are difficult to access with rigid and straight laparoscopic instruments. Therefore, an open surgical approach for bTME surgery is currently standard-of-care in most hospitals. However, the introduction of a robotic platform offers more stable and flexible instrumentation and enhanced visibility in the pelvis, 2 and, therefore, seems well suited for bTME surgery, as shown in recent publications.3–5 Introducing a new surgical approach in complex oncological surgery may have pitfalls, like compromised surgical margins or increased rate of complications. Reporting of short-term outcomes is thus important in an introductory phase.
Studies of rectal cancer cases classified as “locally advanced” may include tumors requiring bTME surgery, 6 but equally often include cases carrying poor prognosis due to high-risk features, such as vascular invasion or lymph node metastases.7,8 Also, while tumors at baseline radiological staging may appear to require bTME surgery, down-staging after neoadjuvant therapy can lend them eligible for TME. 9 This is exemplified in the report from the randomized rectal cancer and preoperative induction therapy followed by dedicated operation (RAPIDO) trial, where two neoadjuvant regimens were compared in 912 cases of high-risk rectal cancer. 10 In this trial, one third of the patients had T4 tumors, but only 69 patients (7.6%) were operated with bTME surgery. In fact, a large proportion of studies focusing on the oncological outcomes of rectal cancer treatment regimens ignore the contribution of the surgical strategy to the treatment outcome. This is not surprising, since no systematic approach has been implemented for surgical reporting of bTME procedures. Such a system should classify the surgical procedures necessary to completely resect tumors threatening the mesorectal fascia or invading adjacent organs and structures, 11 as well as lateral metastatic lymph nodes in the pelvic sidewall. 12
This study reports the short-term clinical results and oncological specimen quality of the first 105 consecutive primary rectal cancer cases operated with robot-assisted bTME surgery at a single tertiary referral center. To accurately report the extent of the bTME resections, surgery was, in addition, classified according to pelvic compartments resected, using the compartmental classification developed for magnetic resonance imaging (MRI) in preoperative planning suggested by the Royal Marsden group.13,14
Methods
Patients and treatment
The Radium Hospital, part of Oslo University Hospital Comprehensive Cancer Centre, is a tertiary referral center specializing in bTME surgery for rectal cancer patients, historically using an open approach. Robotic bTME surgery for rectal cancer was introduced in January 2019, starting directly with the bTME cases. Between January 2019 and December 2020, 210 patients with primary rectal cancer (⩽15 cm from the anal verge by rigid proctoscopy) in need of bTME resections were scheduled for bTME surgery at our institution. Out of these, 105 were selected to laparoscopic robot-assisted bTME surgery. Patient selection was done by the multidisciplinary team, based on the surgical requirement for complete tumor removal and the surgeons’ robotic experience at any given time in the study period. The most advanced cases, for example, cases in need of sacrectomy or recurrent cases were not selected to robotic surgery in this initial phase. Preoperative assessment at baseline comprised chest, abdominal and pelvic computed tomography (CT), pelvic MRI, and proctoscopy with digital examination under anesthesia was repeated 4 weeks after completing neoadjuvant treatment. The patients were operated with either Intuitive daVinci Si or Xi system (Intuitive, Sunnyvale, CA, USA). Two colorectal surgeons started the robotic program, and after 35 procedures, two more colorectal surgeons were added to the surgical team. The multivisceral resections were done by the colorectal surgeons, even for urogenital organs, while reconstruction of the urinary tract including ileal urinary diversions (Bricker conduits) was done intracorporeal robotically by the urologists. Postoperative complications were classified according to the Accordion Severity Grading System of Surgical Complications (Accordion). 15 Severe complications were defined as Accordion grade ⩾3. In the abdominoperineal resection (APR) group, wound healing was defined as complete healing of the perineal incision. In Hartmann’s group, wound healing meant rectal closure without defect, as assessed clinically by digital rectal examination. The study cohort data were registered prospectively, except for data on comorbidity and missing data that were retrospectively collected from patients’ records.
Pathology
Surgical specimens were classified according to the TNM 8 classification. 16 Tumor regression grade (TRG) was classified according to Bouzourene et al. to assess tumor response to preoperative (chemo)radiotherapy ((C)RT), low TRG signifying good response, and high grading poor response. 17 A circumferential resection margin (CRM) >1 mm signified complete resection (R0), margins ⩽1 mm or microscopically involved margins were classified as R1, and macroscopic residual cancer as R2.
Classification of bTME surgery according to pelvic compartments
Surgical procedures were retrospectively classified (by EBT and AMS) according to which pelvic compartments were resected, using the compartmental classification proposed by the Royal Marsden group.13,14 Briefly, this MRI-based classification divides the pelvis into seven compartments along fascia boundaries and anatomical planes amenable to surgical dissection, with each compartment containing specific pelvic structures and organs (Fig. 1). A surgical procedure was defined to include a specific compartment when any anatomical structure or organ in the given compartment was completely or partially resected.
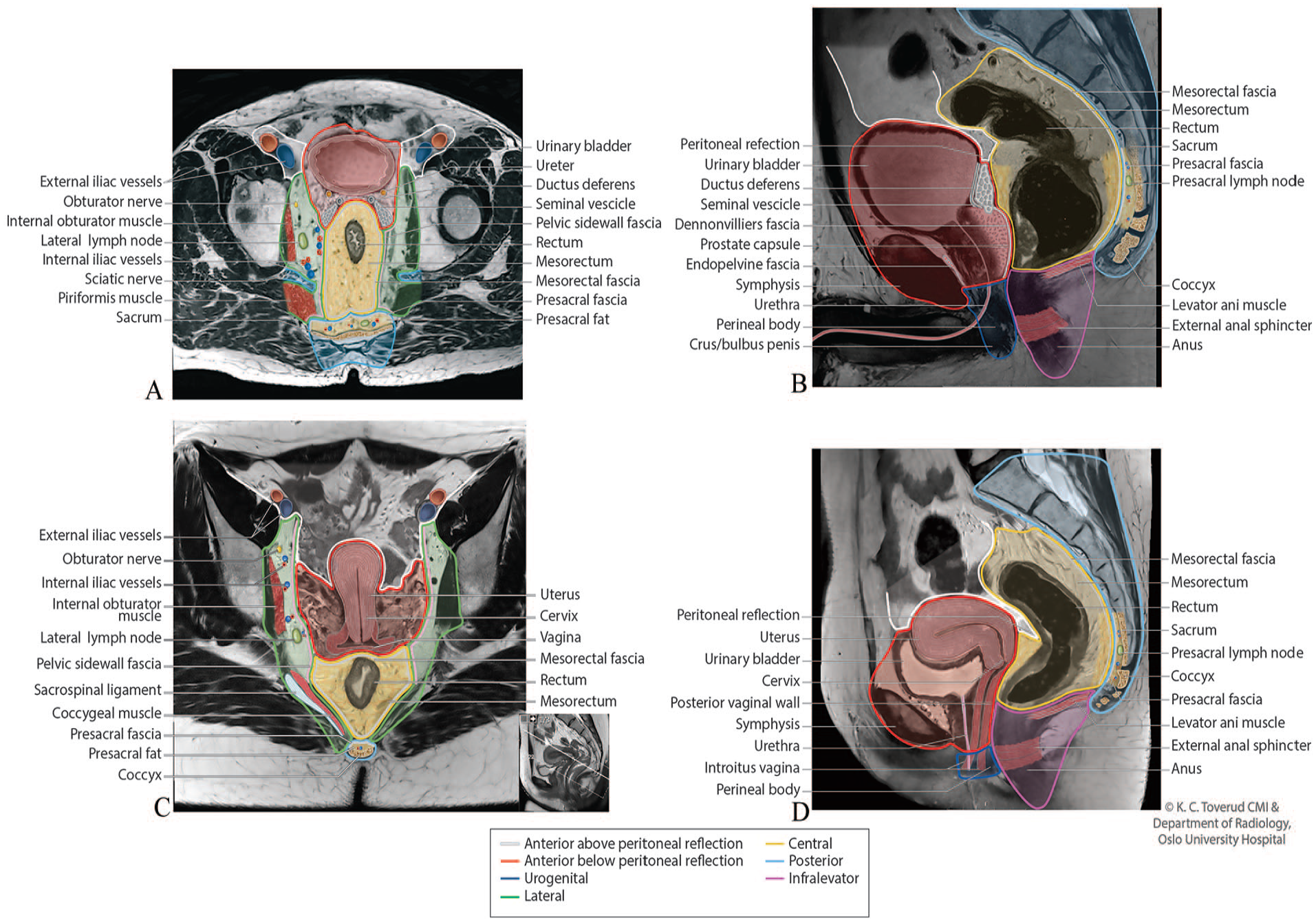
Pelvic compartments with structures and organs resected.
Data presentation and statistical analysis
Clinicopathological data are presented as number and percentage of patients or median and full range. The data were not normally distributed. Mann–Whitney U and the chi-square tests were used depending on data characteristics. The p values < 0.05 were considered statistically significant. Data analyses were performed with SPSS software (version 25; IBM SPSS, Chicago, IL, USA).
Ethics
The project was approved by the Regional Ethics Committee of South-East Norway (2014/1188), and written informed consent was obtained for all patients.
Results
Patient characteristics
Patient characteristics are presented in Table 1. Median patient age was 62 (full range, 38–85) years, and 36% were female. Median body mass index was 25.9 (full range, 12.6–40.8). The Eastern Cooperative Oncology Group (ECOG) performance status was 0 in 79%, 1 in 19%, and 1% were scored 3 and 4, respectively. No patients were scored ECOG 2. The American Society of Anesthesiologists physical status classification systems (ASA) score was 2 in 74%, 3 in 24%, and 4 in 2% of the patients. No patients were scored ASA 1.
Patient characteristics and treatment.
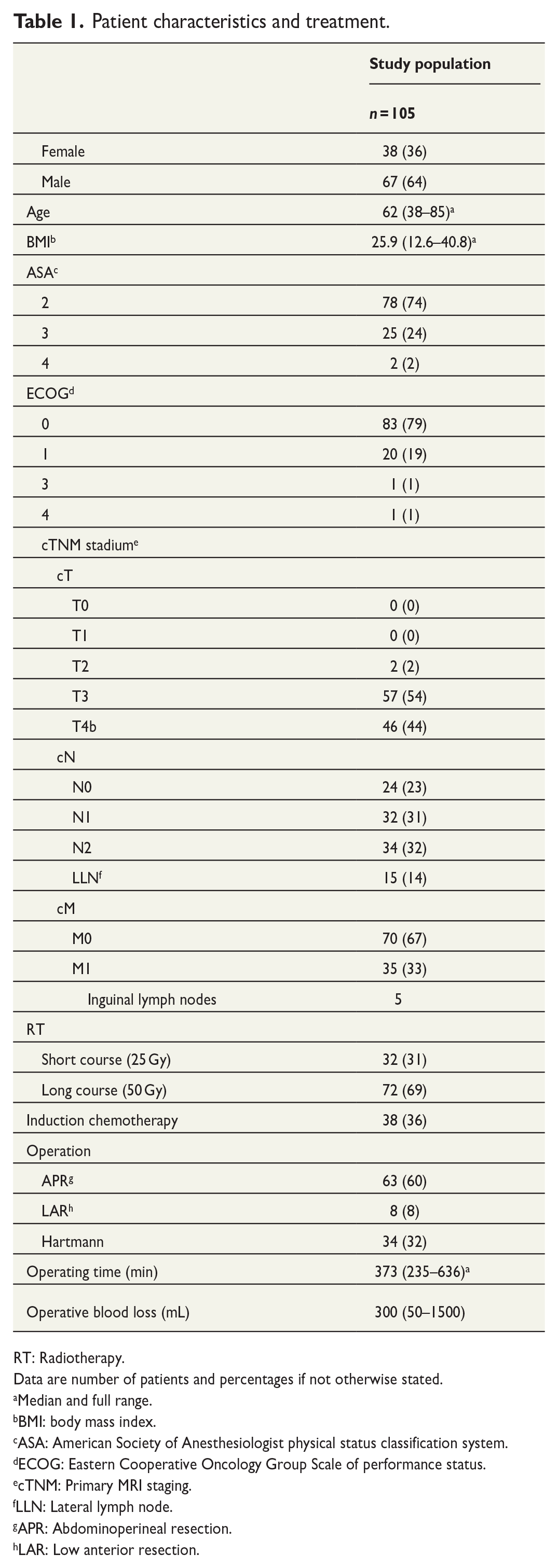
RT: Radiotherapy.
Data are number of patients and percentages if not otherwise stated.
Median and full range.
BMI: body mass index.
ASA: American Society of Anesthesiologist physical status classification system.
ECOG: Eastern Cooperative Oncology Group Scale of performance status.
cTNM: Primary MRI staging.
LLN: Lateral lymph node.
APR: Abdominoperineal resection.
LAR: Low anterior resection.
Baseline staging and neoadjuvant treatment
Based on baseline MRI assessment, two patients had cmrT2 tumors, but were selected for bTME surgery because of a suspected metastatic LLN in one patient and synchronous prostate cancer scheduled for simultaneous radical prostatectomy in one patient (Table 1). About 54% had cmrT3 tumors requiring bTME surgery because the primary tumor, tumor deposits, or mesorectal metastatic lymph nodes threatened or invaded the mesorectal fascia, or because of suspected metastatic LLNs. The remaining 44% patients had cmrT4b tumors with suspected invasion of adjacent structures or organs. Node positive disease within the mesorectum was suspected in 63%, in LLNs in 14%, and in inguinal lymph nodes in 5%. Synchronous distant metastases were detected in 33%, and in 24 cases with the liver as the only metastatic site (23%), all were deemed resectable with curative intention (Table 2). Surgery for synchronous liver metastases was performed prior to the rectal resection in 17 patients, and six of the remaining seven patients had their liver metastases treated within the first year after rectal surgery, either by liver resection (n = 4) or by radiofrequency ablation (n = 2). The last patient had complete response in the liver after neoadjuvant chemotherapy and was never subjected to further liver treatment. No liver metastases were operated simultaneous with the primary tumor.
Synchronous metastases.
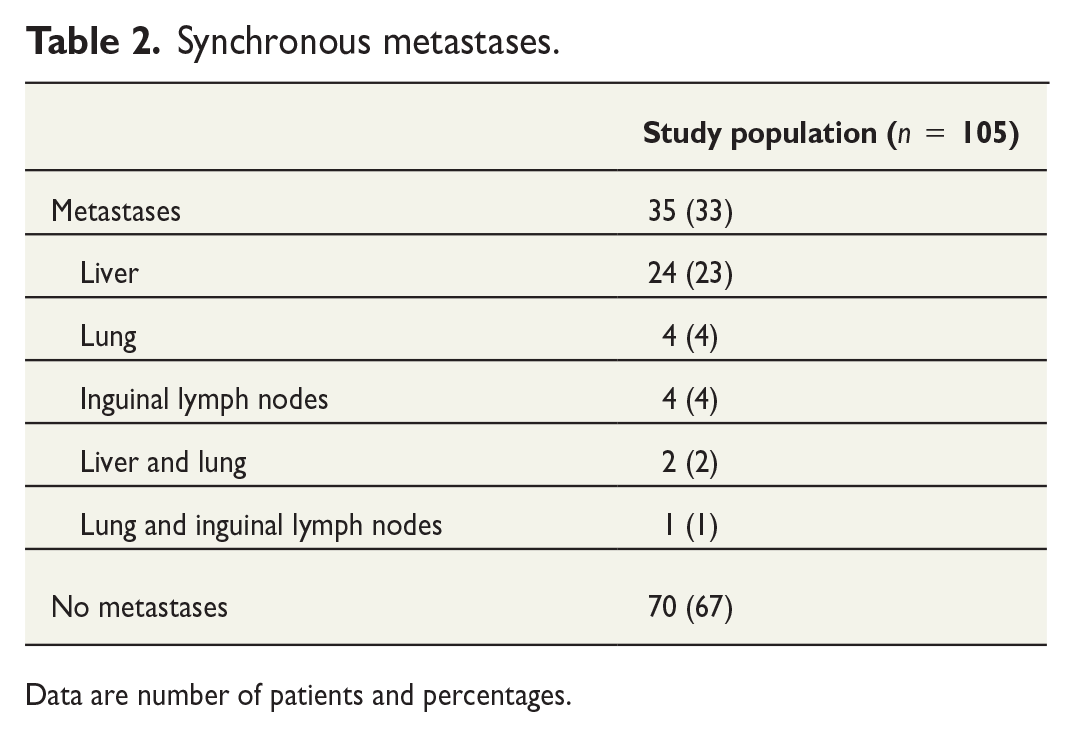
Data are number of patients and percentages.
All except one patient received neoadjuvant (C)RT, 31% had short-course (5 Gy × 5), and 69% had long-course regimens (2 Gy × 25) (Table 1). Two of these patients received a reduced long-course regimen (40 and 48 Gy, respectively) due to toxicity. Concomitant fluoropyrimidine-based chemotherapy was given to patients receiving long-course RT. About 36% received induction chemotherapy prior to (C)RT because of extramural vascular invasion and/or synchronous liver metastases.
Operations and hospital length of stay
Of the total cohort of 105 patients, 60% were operated with APR, 32% underwent rectal resection with end colostomy (Hartmann’s resection), and 8% had low anterior resection (LAR) (Table 1). Median operating time was 373 min (full range, 235–636 min), and median operative blood loss was 300 mL (full range, 50–1500 mL). Four patients had total pelvic exenterations with reconstruction with ileal urinary diversions (Bricker conduits). Beyond the total pelvic exenterations, en bloc resections included posterior prostate (partial prostate resection, n = 15), seminal vesicles (n = 6), seminal vesicles as part of prostatectomy (n = 7), uterus and adnexa (n = 8), the posterior vaginal wall (n = 7), the right ureter (resection and reimplantation, n = 1), pelvic sidewall fascia (n = 29), presacral fascia (n = 26), and the coccyx (n = 1). Fifty-four patients had the levators resected, 21 as the only bTME procedure. Eight patients had LLN resections, one with the lymph node and the internal iliacs en bloc. Seven operations (7%) were converted to open surgery. About 11% had intraoperative events handled as they occurred: 3% tumor perforation, 6% bowel perforation, and 2% urinary bladder perforation. In three patients, the planned bTME surgery was limited to standard TME resection based on intraoperative findings. The hospital length of stay (LOS) was a median 6 days (full range, 3–30 days), with median 6 days (full range, 4–30 days) for APR, 6 days (full range, 3–9 days) for Hartmann’s, and 6 days (full range, 4–11 days) for LAR (Table 3).
Complications.
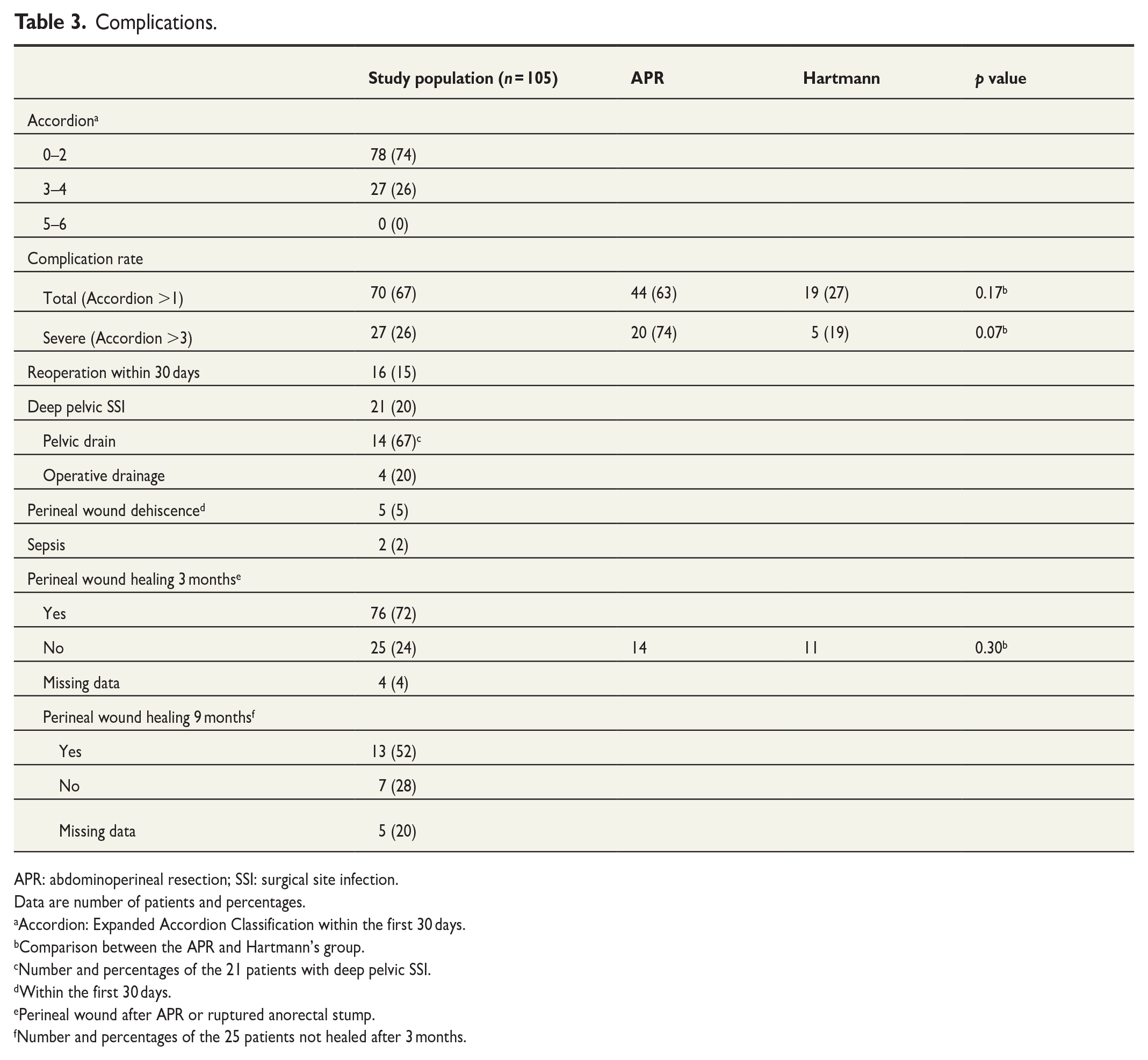
APR: abdominoperineal resection; SSI: surgical site infection.
Data are number of patients and percentages.
Accordion: Expanded Accordion Classification within the first 30 days.
Comparison between the APR and Hartmann’s group.
Number and percentages of the 21 patients with deep pelvic SSI.
Within the first 30 days.
Perineal wound after APR or ruptured anorectal stump.
Number and percentages of the 25 patients not healed after 3 months.
Tumor characteristics
Upon histopathological examination, 15% of the surgical specimens were classified as ypT0-1, 19% as ypT2, 58% as ypT3, and 8% as ypT4 (Table 4). Metastatic mesorectal lymph nodes (ypN1 or N2) were found in 48%, metastatic LLNs in 3%, and metastatic lymph nodes in the inguinal region in 2%. The tumor responses to neoadjuvant therapy were classified as TRG 1 in 24%, 2 in 17%, 3 in 33%, 4 in 8%, 5 in 13%, and not applicable in 5%. R0 resection was obtained in 90.5%, whereas 9.5% had R1 resection. Of the 10 patients with R1 resection, seven had a compromised margin at the primary tumor, two at a tumor deposit, and one due to perineural tumor involvement. Median CRM was 6 mm (full range, 0–30 mm). No patients had macroscopic residual tumor after surgery (R2 resection).
Oncologic results.
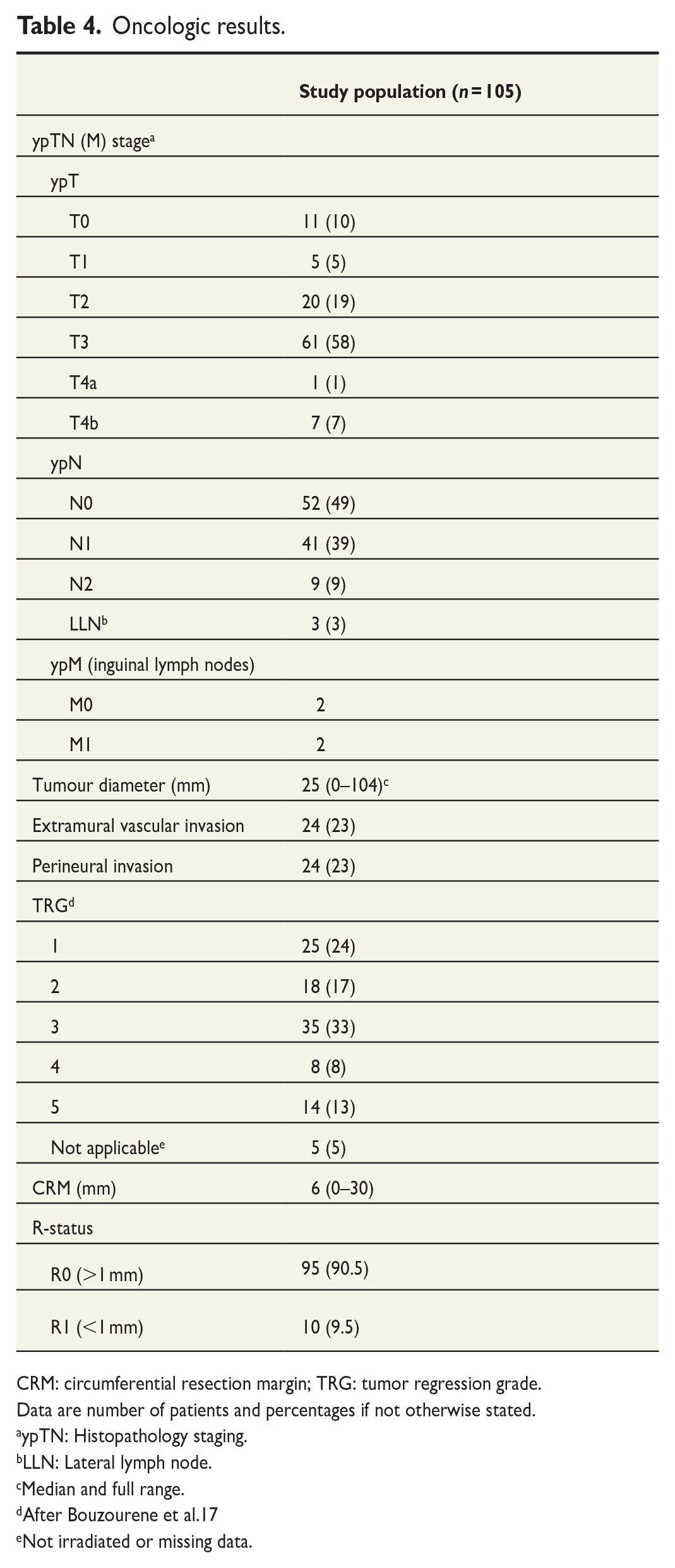
CRM: circumferential resection margin; TRG: tumor regression grade.
Data are number of patients and percentages if not otherwise stated.
ypTN: Histopathology staging.
LLN: Lateral lymph node.
Median and full range.
After Bouzourene et al.17
Not irradiated or missing data.
bTME surgery and risk of involved margins according to pelvic compartments
A median of two pelvic compartments were partially or completely resected (full range, 1–7) (Table 5). The central compartment comprising the rectum and mesorectum was resected in all patients. The infralevator compartment (levator ani muscles or levator ani fascia) was the second most frequent, with resection in 55%, followed by the anterior below peritoneal reflection compartment (bladder, prostate, seminal vesicles, uterus, ovaries, and/or the posterior vaginal wall) in 42%. The lateral compartments (pelvic sidewall fascia, pelvic sidewall lymph nodes, or the internal iliac vessels) were resected in 30% and the posterior compartment (presacral fascia, vessels and lymph nodes and/or the coccyx) in 26%. The anterior above peritoneal reflection compartment (ureters) was resected in 5% and the anterior urogenital triangle compartment (distal urethra/perineal body) in 4%. All of the 10 R1 resections occurred in the lateral and the posterior compartments, five in the lateral, three in posterior, and two in both compartments.
Pelvic compartments resected a .
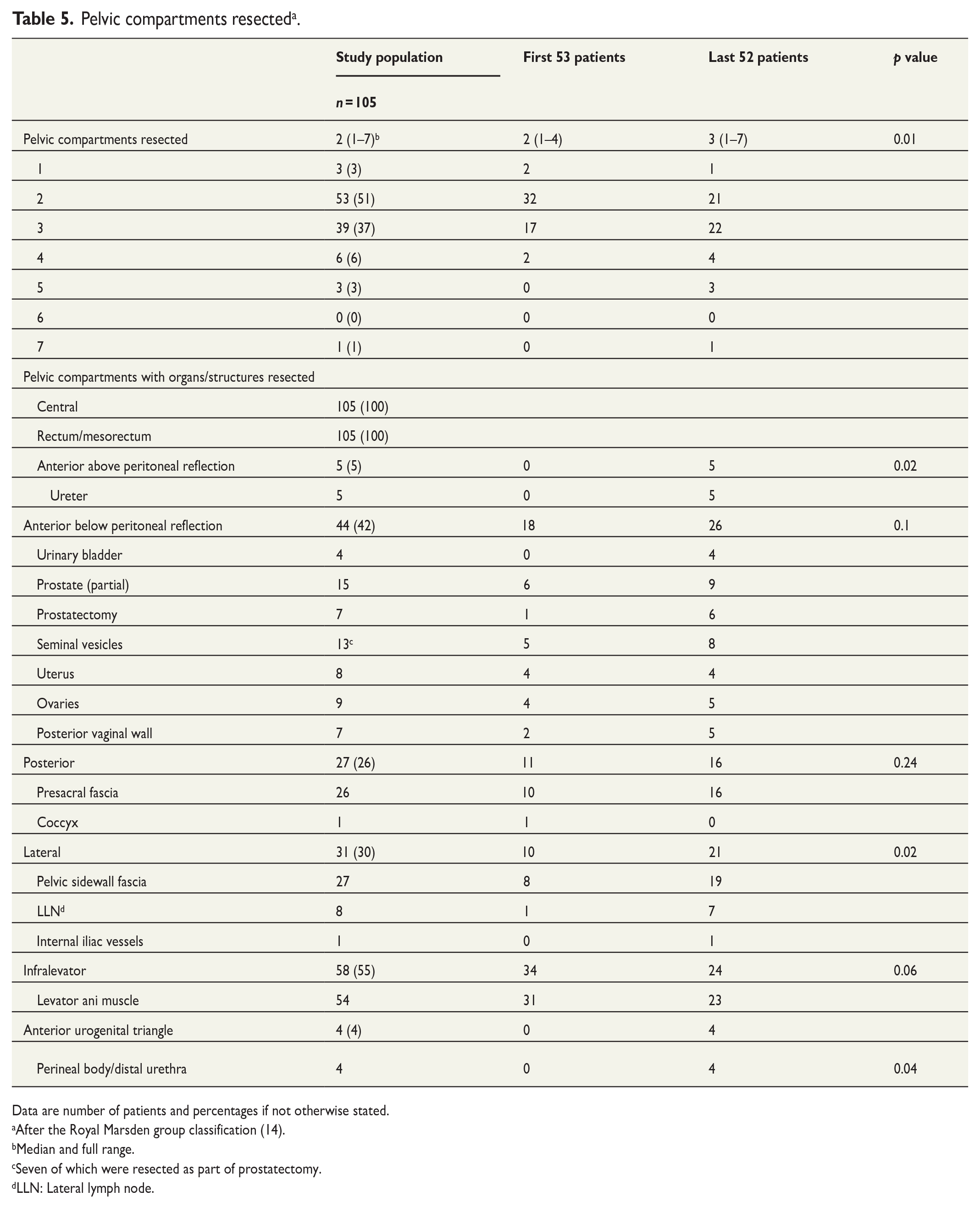
Data are number of patients and percentages if not otherwise stated.
After the Royal Marsden group classification (14).
Median and full range.
Seven of which were resected as part of prostatectomy.
LLN: Lateral lymph node.
When comparing the first 53 operated patients with the latter 52, more patients were selected to bTME surgery with a robotic approach in the latter period (47% in the first period versus 54% in the latter). Also, significantly more compartments were resected per patient in the latter period (median 3 in the latter versus 2 in the first, p = 0.01), with increasing number of resections in the anterior above peritoneal reflection, the lateral, and the anterior urogenital triangle compartment (Table 5). Median blood loss (300 mL) in both periods, operating times (372 versus 375 min), and the number of conversions to open surgery (5 versus 2 (p = 0.25)) remained unchanged in both periods. There was no significant difference in the R1 rate (p = 0.17) between the two periods.
Postoperative complications and perineal wound healing
No or minor to moderate complications were reported in 74%. Severe complications occurred in 26% (Table 3), and of these, 15% needed re-operations due to bowel obstruction, anastomotic leakage, stoma necrosis, combined ileal conduit failure and bowel obstruction, urinary leakage, subcutaneous perineal abscess, or hemorrhage. The most common severe complication was deep pelvic surgical site infection (SSI), which occurred in 20%. No patients experienced organ failure, and there was no mortality. There was a trend toward a higher rate of severe complications in the APR group (p = 0.07). At 3-month follow-up, 24% had incomplete perineal wound healing after APR or ruptured anorectal stump after Hartmann’s procedure. At 9-month follow-up, half of these (13 patients) had healed completely without reconstructive surgery. Seven were still treated conservatively, and five cases had incomplete data/were lost to follow-up, and so far, no patients have been scheduled for secondary reconstructive surgery.
Discussion
This study presents the introduction and initial results of laparoscopic robot-assisted surgery for rectal cancers requiring bTME at a high-volume center, reporting on the first prospectively included 105 procedures that were undertaken. With increasing surgical experience, patients with more complex tumors were selected to robotic surgery in the second part of the study period. Despite this surgical outcomes were not compromised, as no change in blood loss, operating time, conversion, or R1 rates were detected. The obtained R0 resection rate of 90.5% is in line with previous reports from both robotic and open bTME surgery,3,4,18–20 and suggests that adequate short-term oncologic quality of the procedure was preserved in the transition from an open to a minimally invasive approach. Direct comparison with other studies is not straightforward because minimally invasive bTME surgery has been far less investigated than open bTME surgery,21,22 and regardless of strategy, the extent of tumor involvement and surgical complexity are generally poorly described in most reports. In addition, patient selection based on expected resection margins will influence the choice of open or minimally invasive surgical strategy. When open bTME surgery reports up to 25% R1 resections,18–20 this may, therefore, reflect the inclusion of more advanced cases, which in this introductory phase of robotic surgery were selected to undergo open procedures in our institution. Finally, it is important to note that short-term oncological specimen quality does not necessarily guarantee a favorable long-term outcome, 23 and reporting long-term follow-up will be necessary.
Other quality parameters related to short-term outcomes of minimally invasive surgery are conversion rates to open surgery, LOS, and postoperative morbidity. One might have expected a higher conversion rate for surgically more advanced bTME procedures than for standard TME, but the 7% conversion rate in our cohort is in line with other studies reporting minimally invasive techniques in standard TME surgery. 24 The median LOS of 6 days seems to be within the range of previously reported studies, with mean 5 days found after robotic TME surgery, 25 while 12 to 16 days are reported in small series on complex robotic bTME surgery.26,27 The LOS in a previously published cohort operated with open bTME surgery at our institution, was median 10 or 15 days depending on deep SSI or not. 28 Although the cohorts are not case-matched, one could speculate that robotic surgery reduce LOS for these patients. Even though the operating time in this initial phase of the robotic program was longer (median 373 min) compared with the historical cohort (272 min),28 the LOS was lower. Perioperative blood loss in the present cohort was median 300 mL and in the historical cohort median 850 mL. About 26% of the patients in our cohort experienced severe postoperative complications. The most common severe complication was deep pelvic SSI, and the rate in the present cohort did not differ from the rate reported after open bTME surgery in our own institution. 28 Piozzi et al. 3 reported a lower complication rate with 20% Clavien-Dindo III–IV complications after robot-assisted bTME surgery, but the rate of APR was only 9%, compared with 60% in our cohort. Deep pelvic SSI is in particular known to be more frequent after APR, occurring in up to 40% of the patients,28–30 and is often associated with impaired perineal wound healing in irradiated patients.28,31 Interestingly, in the majority of our cases, the perineal wounds healed without additional secondary reconstructive measures even if healing was delayed at initial follow-up. Thus, a conservative approach to perineal dehiscence may succeed within the first year. Because of the high risk of deep pelvic SSI and potentially perineal dehiscence in this group, one might argue that all perineal defects after extensive APR procedures should be primary reconstructed. However, all reconstructions also come with a price as, for instance, the vertical rectus abdominis myocutaneous flaps are reported to have up to 68% complications rate in these patients. 32 An alternative approach aiming to avoid pelvic complications could be omentoplasty. The method is not in routinely use at our center and was not performed in this cohort. In any case, omentoplasty is also associated with complications, with a 40% perineal wound infection rate reported. 33 As for re-operations, a 30-day rate of 15% might seem high compared with other robotic studies reporting rates of 2%–10%.34,35 However, the numbers for comparison are mostly based on standard TME surgery with a majority of LAR, often in radiation-naive patients. In complex pelvic oncologic surgery, the majority of patients experience complications, 36 all the more after modern aggressive neoadjuvant treatment. 29 Although the trauma and inflammatory response after robotic TME is found to be reduced even compared with laparoscopic surgery, 37 it is uncertain if a less traumatic procedure reduces complications. On the contrary, a study like the conventional versus laparoscopic-assisted surgery in patients with colorectal cancer (CLASICC) trial found that complication rates were similar in open and minimally invasive surgery, 38 perhaps indicating that the procedure itself is more important than the surgical method.
In this work, resections performed in the individual surgical procedures were described using the compartment classification system developed for MRI by the Royal Marsden group in 2013, 13 and later slightly modified. 14 To our knowledge, utilizing a classification of advanced pelvic tumors to characterize surgical procedures for complex rectal cancers has not been published before. Variation in the complexity of the bTME procedures was clearly shown, with between one and all seven compartments being partially or completely resected, with the number of compartments resected increasing over time, suggesting that more complex patients were selected for robotic surgery with growing experience. The lateral and posterior compartments were the only compartments where R1 resections occurred and were identified as the areas at highest risk of involved margins. The lateral compartment has previously, by us and others, been identified to have elevated risk of R1 resections.20,39 With specific regions of the pelvis being at increased risk of R1 resection, the absence of a common classification system for bTME procedures makes it difficult to compare the quality of treatment and results reliably. Although several other pelvic MRI-based classification systems have been suggested and have confirmed that resection margins depend on tumor location,3,40,41 none have to date been systematically used to describe bTME procedures. The classifications put forward by the Royal Prince Alfred and the Royal Marsden groups are the most comprehensive, with almost all pelvic organs and structures allocated to specific pelvic compartments. Since the Royal Prince Alfred group has focused on recurrent rectal cancer only, the Royal Marsden classification was chosen to characterize bTME procedures for primary rectal cancers in this study. 41 If validated for bTME procedures and endorsed as a common system, such a classification could facilitate comparison and understanding of the surgical complexity of bTME procedures between studies and centers.
In conclusion, robotic bTME surgery was introduced with short-term clinical outcomes and oncological specimen quality comparable to standard-of-care open bTME surgery. The conversion rate was relatively low and the complication rate acceptable in patients with tumors requiring surgery in multiple pelvic compartments. The description of surgical procedures using the Royal Marsden MRI compartment classification was feasible.
Footnotes
Acknowledgements
The robotic program at the Radium Hospital, Oslo University Hospital is supported by a generous grant from the Radium Hospital Foundation. The funding source had no involvement in study design; in the collection, analysis, and interpretation of data; in the writing of the report or in the decision to submit the article for publication.
Author contributions
All authors made substantial contribution to the concept or design of the work; acquisition, analysis, and interpretation of data, drafted the article, or revised it critically.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ebbe B. Thorgersen is proctor for Intuitive Surgical.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
