Abstract
Background:
The aim of this study was to evaluate the accuracy and added value of specimen tomosynthesis (ST) to specimen ultrasound (SUS) in margin assessment of excised breast specimens in breast-conserving therapy for non-palpable US-visible breast lesions.
Materials:
Between January 2018 and August 2019, all consecutive patients diagnosed with non-palpable breast cancer visible by ultrasound (US), treated with breast-conserving surgery (BCS) and requiring radiological intraoperative breast specimen assessment, were included in this study. Excised breast specimens were examined with SUS by radiologists blinded to the ST results, and margins smaller than 10 mm were recorded. STs were evaluated retrospectively by experienced radiologists.
Results:
A total of 120 specimens were included. SUS showed a statistically significant correlation with pathological margin measurements, while ST did not and provided no additional information. The odds ratios (ORs) for SUS to predict a positive margin was 3.429 (confidence interval (CI) = 0.548–21.432) using a 10-mm cut-off point and 14.182 (CI = 2.134–94.254) using a 5-mm cut-off point, while the OR for ST were 2.528 (CI = 0.400–15.994) and 3.188 (CI = 0.318–31.998), respectively.
Conclusions:
SUS was superior in evaluating intraoperative resection margins of US-visible breast resection specimens when compared to ST. Therefore, ST could be considered redundant in applicable situations.
Context and relevance
Breast-conserving surgery (BCS) is an established approach for early-stage breast cancer management, aiming to preserve cosmesis while ensuring tumor-free margins. Intraoperative assessment of tumor removal and margin status plays a pivotal role in reducing re-operation rates, hospital stay, and psychological impact. This study explores the accuracy of specimen ultrasound (SUS) versus specimen tomosynthesis (ST) in evaluating surgical margins for non-palpable US-visible breast lesions. The unique contribution of this study lies in comparing these techniques, revealing SUS as the superior choice for margin assessment. These findings have significant implications, suggesting that SUS combined with trained surgical staff can optimize intraoperative decision-making and contribute to enhanced patient outcomes in BCS.
Introduction
Breast-conserving surgery (BCS) is regarded an equal treatment modality to mastectomy for the management of early-stage breast cancer.1,2 The current challenge in BCS is improving surgical accuracy by maintaining a smaller excision volume, while achieving an acceptable tumor-free margin, in order to optimize cosmetic outcomes. To reduce the re-operation rates, intraoperative assessment of adequate removal of detected lesions and of margin status is performed. Intraoperative margin assessment can reduce the rate of re-operations, thus affecting total patient hospital stay, with impact on psychological well-being and cosmetic outcome as well as reduce the economic burden.
Specimen ultrasound (SUS) assessment has a greater sensitivity than mammography for the detection of invasive cancer in dense breasts. 3 The use of SUS is especially beneficial in non-palpable lesions not detected by mammography. Studies have also demonstrated the usefulness of intraoperative ultrasound (IOUS)-guided resections in achieving clear margins, but this would require the training of surgical staff in imaging techniques.4,5
There is currently no sufficient comparative data on the value of SUS assessment of resected specimens compared to specimen radiography. Many studies have reported SUS as an effective modality for identifying lesions within specimens and estimating margin size; on the contrary, others report poor performance in the evaluation of surgical specimen margin status and no reduction in re-operation rates after BCS of ductal carcinoma in situ (DCIS). 6 Common specimen radiograph techniques include two-dimensional (2D)-mammography, dedicated digital specimen radiography system and specimen tomosynthesis (ST). ST provides three-dimensional images that eliminates tissue super-imposition and provides a clearer view of the area of interest. A recent comparative study showed the superiority of ST compared to other radiography techniques, not only in the depiction of resected lesions but also in the detection of spiculations and calcifications. 7
The aim of this study was to evaluate the accuracy and added value of ST to SUS in women undergoing BCS for non-palpable US-visible breast lesions. The histological findings were used as the reference gold standard.
Methods
This study was undertaken as part of the continuous improvement, quality control and internal validation of modern surgical specimen imaging technologies at the Breast Unit of Kuopio University Hospital (Kuopio, Finland). During the study period, clinical decisions concerning lesion removal and margin status were made by experienced breast radiologists and surgeons, and decisions were based on all available images.
The more detailed analyses and inter-technique comparisons described in this study were performed retrospectively and thus, did not affect patient management. The chair of the hospital district waived the need to obtain written informed consent from the patients owing to the retrospective nature of the analyses (FinMargins 5063573). All clinical investigations were conducted according to the relevant guidelines and the principles expressed in the Declaration of Helsinki.
Study population
The study population comprised all consecutive breast cancer patients between January 2018 and August 2019, who were referred to the authors’ tertiary University Hospital for further evaluation and treatment. Patients were included in this study if they were: (1) diagnosed with unifocal or multifocal invasive breast cancer or DCIS, (2) had tumors that were preoperatively US-visible, and (3) the tumors were non-palpable or deemed not clearly palpable by experienced breast surgeons and therefore required intraoperative radiological breast specimen assessment. Patients who underwent neoadjuvant chemotherapy were excluded. Some of the patients were included in a previous report that evaluated the accuracy of different specimen radiography techniques. 7
All patients were evaluated preoperatively with a minimum of a two-view mammogram and bilateral breast US. Any suspicious lesions were evaluated using additional lateral and spot-compression views and/or tomosynthesis as needed. All mammograms were re-evaluated upon referral by an experienced senior breast radiologist, and further workup was performed if deemed necessary. Breast magnetic resonance imaging was performed according to national guidelines that are in concordance with the European Society of Breast Cancer Specialists’ recommendations. Patients underwent US-guided core biopsy or stereotactic vacuum-assisted biopsy and were diagnosed with breast cancer or DCIS. In cases with lesions in both breasts, each specimen was evaluated separately.
The mammographic features blindly evaluated by one of the two experienced breast radiologists with 15 and 30 years of experience, according to the fifth edition of the BI-RADS (Breast Imaging Reporting and Data System) lexicon in a retrospective setting. 8
Lesion localization and surgery
The surgical procedure was planned individually according to the patient’s preference, tumor size, tumor location, clinical findings, breast shape, and breast size. All patients were evaluated at multidisciplinary meetings at least twice, that is, pre- and post-operatively.
Non-palpable tumors were localized preoperatively using at least one guidewire (breast localization needle Duo, SOMATEX® Medical Technologies GmbH) under US guidance. Two-view mammography (CC and lateromedial) was routinely performed to confirm the position of each lesion relative to the guidewire, and the location of the lesions was ink-marked on the skin in the surgical position. Tumors were excised en-bloc from the subcutaneous area to the muscle and the overlying skin was removed in patients with superficial lesions to achieve a healthy macroscopic surgical margin of ⩾10 mm, in accordance with national guidelines and hence achieving microscopically negative margins (defined as no “tumor-on-ink”). In DCIS, the need for re-operation was evaluated in a multidisciplinary meeting whenever DCIS margins were less than 2 mm. Intraoperatively, the specimens were placed on a Styrofoam slab, fixed with wooden sticks, and the location of the excision was anatomically marked. Metallic clips were placed directly on the specimen to indicate the orientation. The fascia posterior to the tumor was removed and fixed aside if removed separately. The specimen was then placed in a plastic container and immediately transported to the Breast Radiology Unit.
Specimen imaging protocol
Each specimen was first imaged by full-field digital mammography and tomosynthesis with a protocol described in a previous report 7 that showed ST images having the best tumor visibility and accuracy.
SUS was performed by one of the five specialized breast radiologists using an 18-MHz hockey-stick probe (Logiq E9 class US scanner, GE, Wauwatosa, WI, USA). The observers had access to the preoperative images but not to the specimen radiographs. The specimen was gently scanned with the probe, and tumor removal margins were recorded. The radial distances were measured from the edge of the removed tumor to the edge of the specimen. Removal of target lesion and margins were prospectively recorded whenever resection margins were less than 10 mm.
After that, the specimen radiographs were evaluated. All results were reported to the surgeon in the theater. Additional resections for marginal or cosmetic reasons were not imaged, but they were fully investigated by histopathology.
For the purpose of this study, the ST images were further retrospectively evaluated by radiologists who were blinded to other outcomes. All margins less than 10 mm were recorded. The presence of any additional finding that might change the marginal status was also recorded. Spiculations or their visibility were not evaluated.
Histopathology
Specimen analysis was described previously. 7 Briefly, the specimens were measured, photographed, margins ink-marked, and sliced at 5 mm intervals. The extent of involvement between the invasive and/or in situ cancer in each margin was reported in all six directions. The histopathology data were obtained from structured histopathology reports.
Statistical analysis
All statistical analyses were performed with SPSS for Windows version 27 (IBM Corporation, Armonk, NY, USA). p-values of < 0.05 were considered statistically significant. The margins of the primary resection specimen were regarded positive if there was invasive tumor on-ink or DCIS with resection margins ⩽2 mm. Pearson’s correlation coefficient was used to assess the linear association between the healthy margins measured using US, tomosynthesis, and histopathology. Odds ratios (ORs) and their 95% confidence intervals (CIs) were measured through cross-tabulation and risk analysis.
Results
A total of 119 patients with 120 specimens fulfilled the inclusion criteria and were evaluated. During the study period, a total of 320 specimen radiographs from 307 patients were performed. Of these, six were mastectomy specimens where imaging tests were performed to mark non-palpable lesions for the pathologist. Twenty-eight specimens were benign, discordant, or risk lesions. Thus, a total of 292 specimen radiographs on lesions with a malignant diagnosis were performed during the study period, and specimens included in this study represent 41.1% of all specimens examined (Fig. 1).
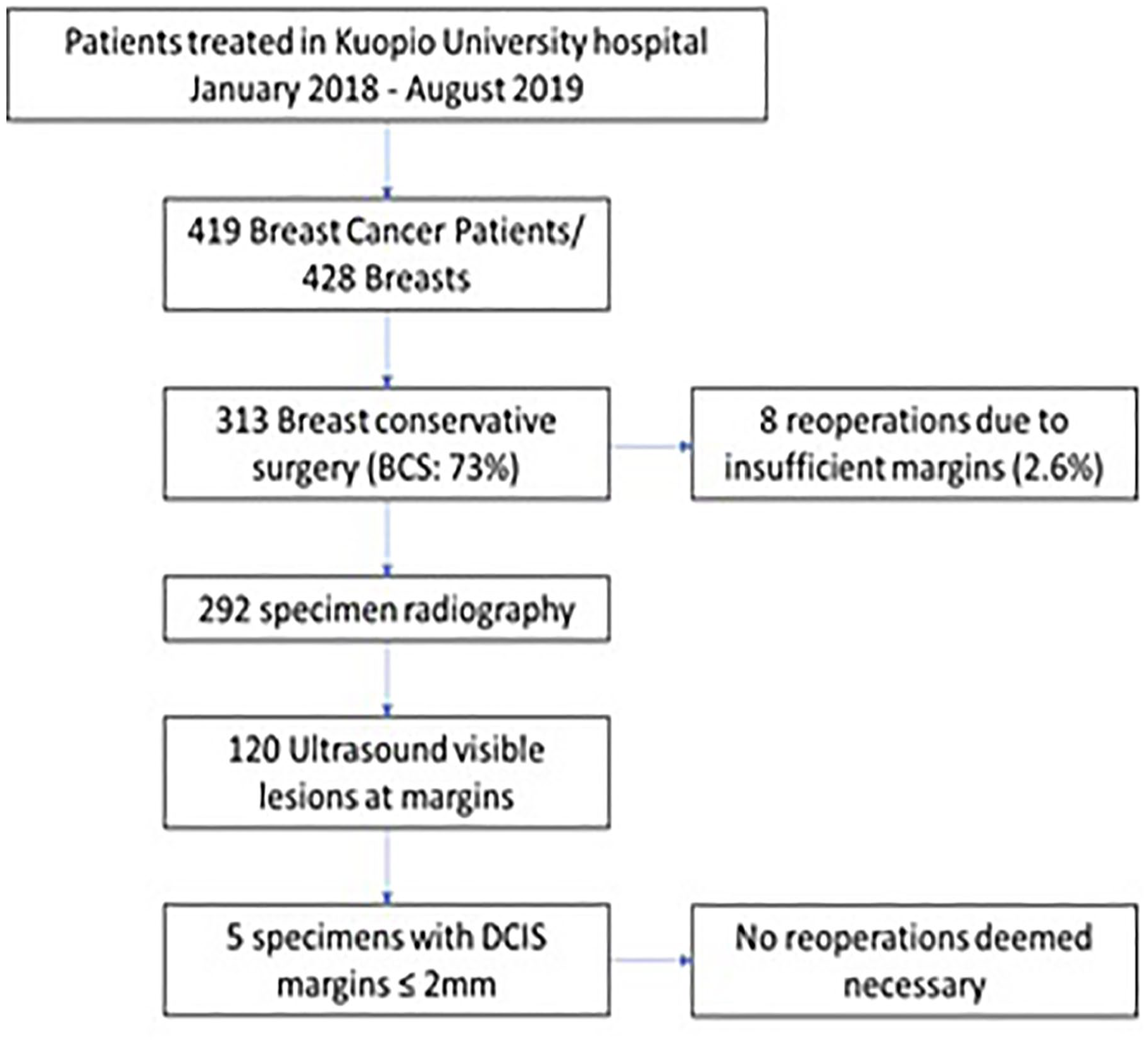
Flowchart of the study.
The mean age of the patients was 63.6 years (range = 33–95 years). Patient characteristics are presented in Table 1, and mammographic characteristics of all patients are presented in Table 2. Four masses were mammographically occult.
Patients, surgical procedure, and tumor characteristics.
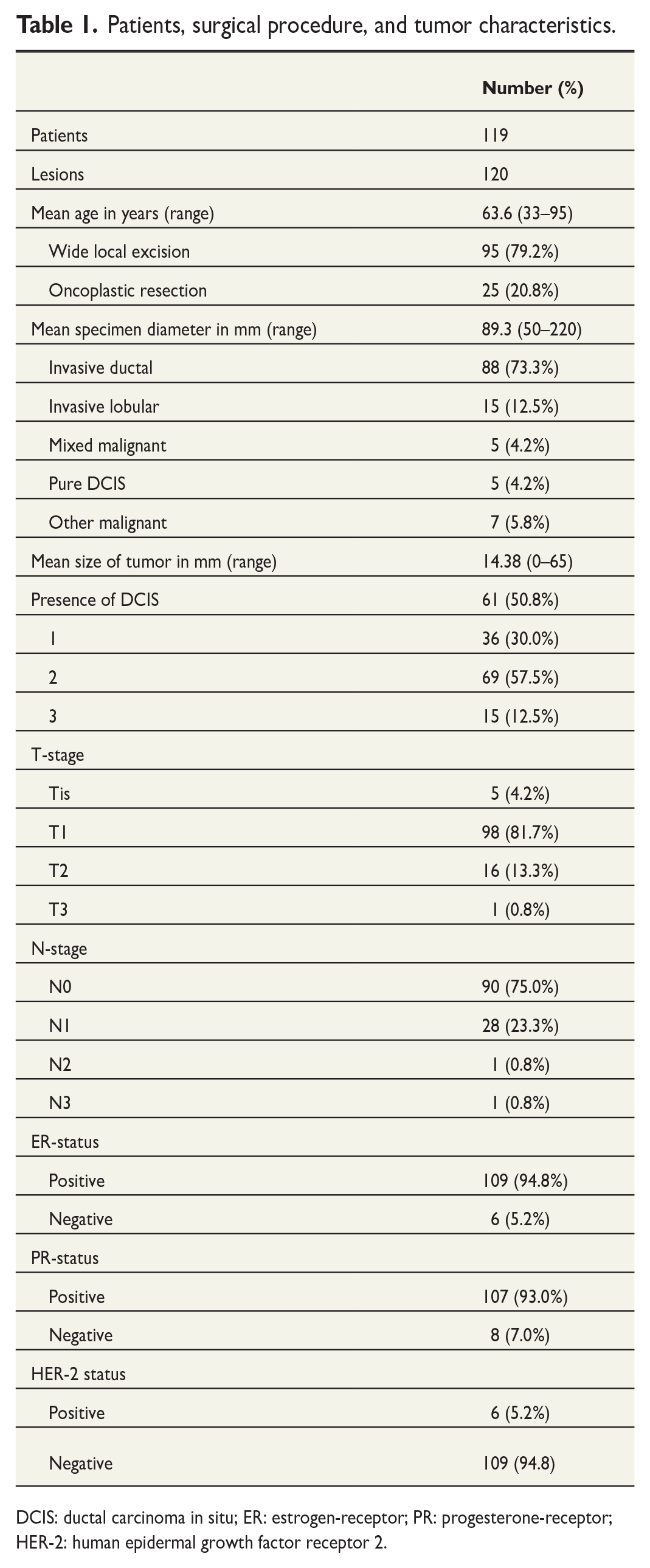
DCIS: ductal carcinoma in situ; ER: estrogen-receptor; PR: progesterone-receptor; HER-2: human epidermal growth factor receptor 2.
Number of the mammographic features and lesion descriptors according to the Breast Imaging Reporting and Data System (BIRADS©, fifth edition).

Number of lesions analyzed 120.
SUS-measured short margins were 5 mm or less in 14 cases, 6–10 mm in 25 cases, and more than 10 mm in 81 cases. ST measured margins were 5 mm or less in 9 cases, 6–10 mm in 17 cases, and more than 10 mm in 89 cases. Margins were not reliably assessable in five cases with ST, while all margins were measurable with SUS. There was a statistically significant correlation between SUS and final pathology margin measurement, while this was not the case for ST (Table 3). ST provided no additional information (Figs 2–4).
Margins measured by specimen tomosynthesis (ST) and specimen ultrasound (SUS) and their correlation (Person’s correlation coefficient) to final histopathology.

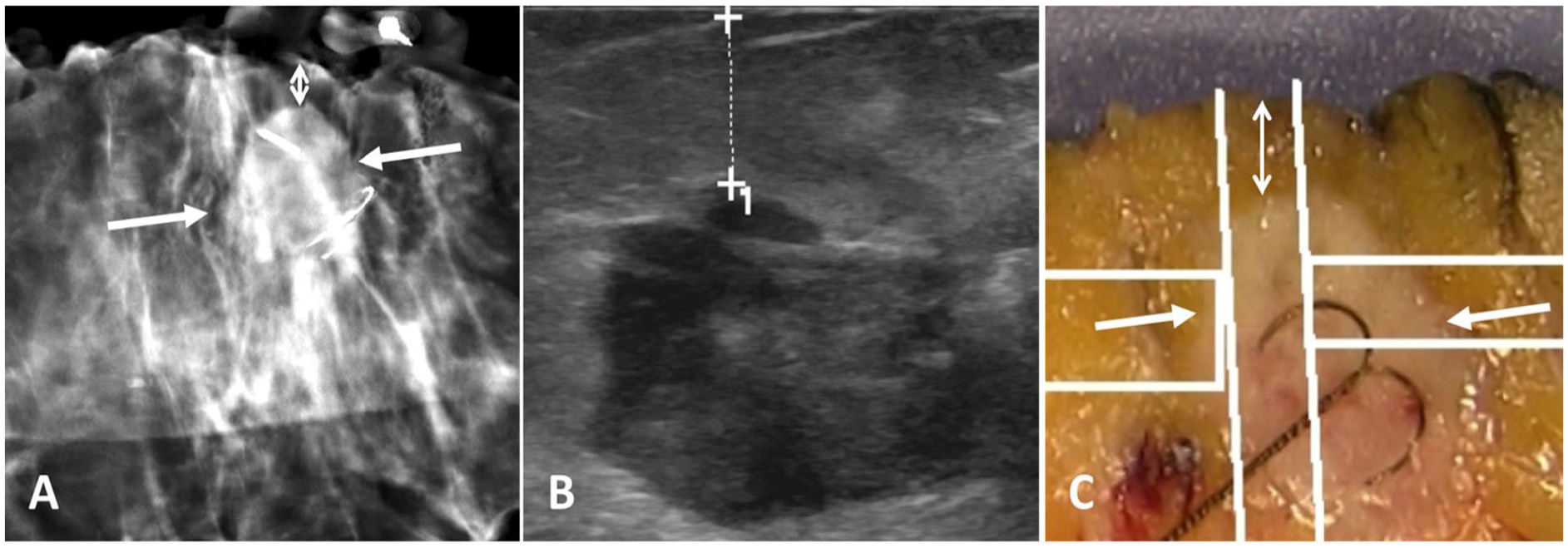
Tomosynthesis, ultrasound, and pathology depiction of an invasive ductal carcinoma treated with guidewire-localized wide local excision. (A) Tomosynthesis at the level of the tumor (lateral long arrows: tumor margins. Vertical two-sided arrows: shortest distance to the resection margin, which was 5 mm). (B) Same short margin was detected by ultrasound. (C) Pathology imaging depicting similar resection margins.
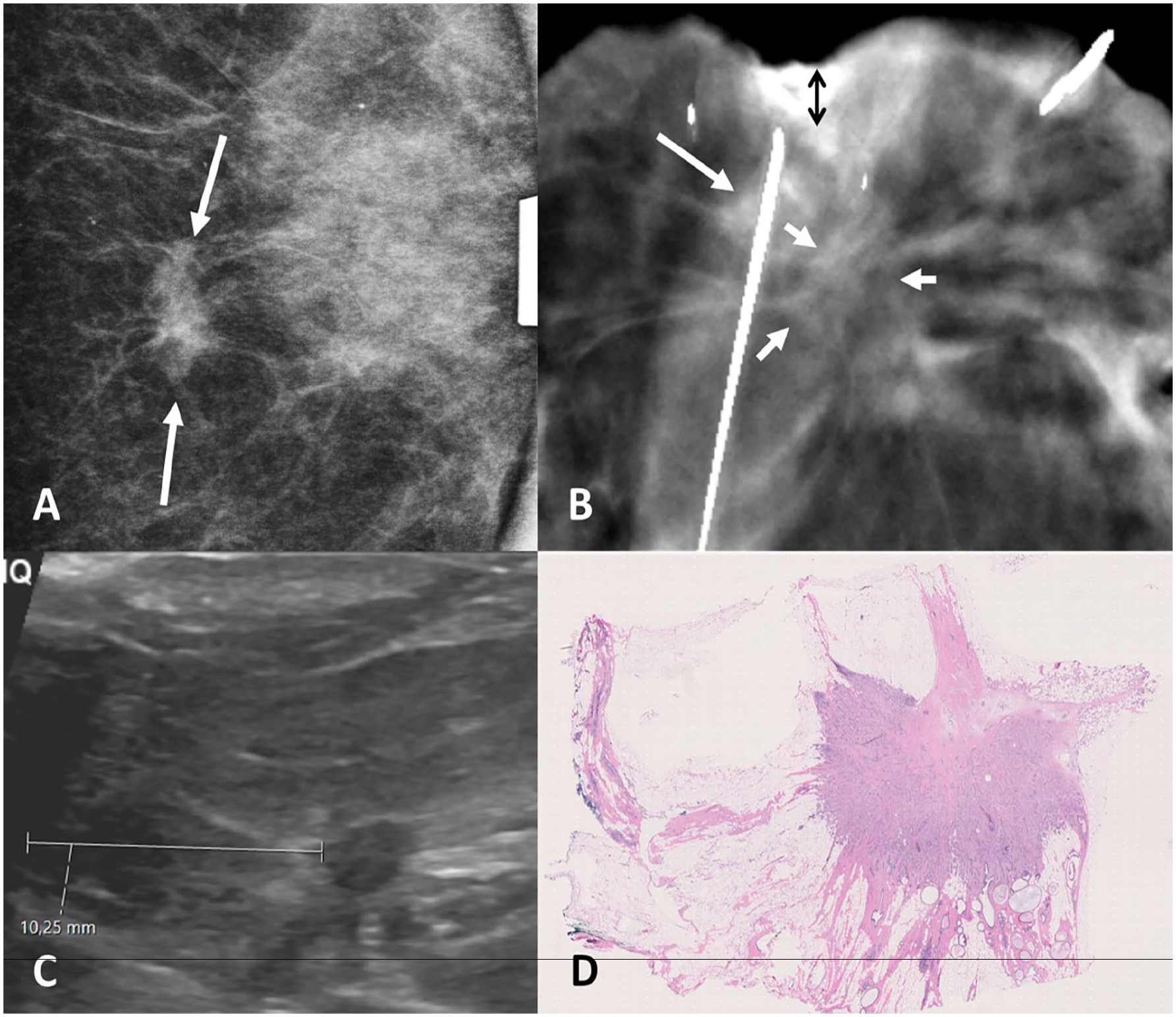
Discordance of the imaging modalities due the improper interpretation of exact tumor extension. (A) Preoperative spot-view mammography of the detected, histologically confirmed invasive ductal carcinoma (arrows). (B) Specimen ultrasound with a 10-mm margin measurement. (C) Specimen digital breast tomosynthesis at the level of spiculated tumor (small arrows) with a margin of 4 mm (vertical two-sided black arrow), measured from the density at the tip of the guidewire, interpreted to be a part of the tumor. (D) Pathological evaluation of the tumor and its margins show only one spiculated tumor corresponding to the ultrasound image.
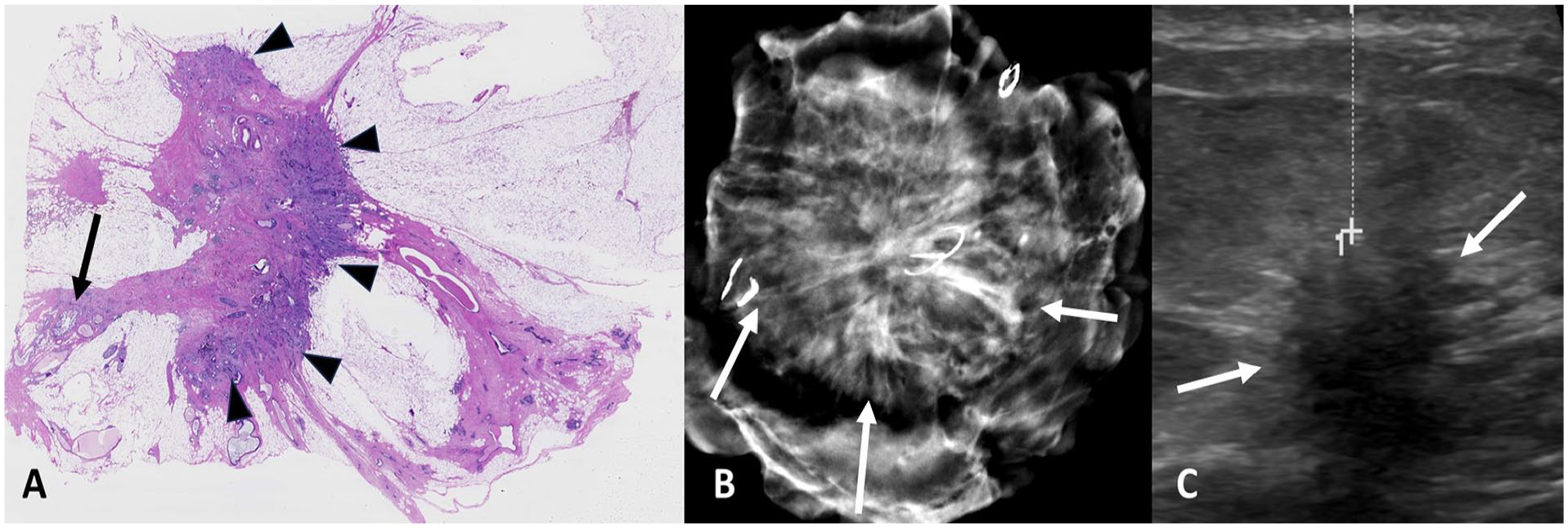
Tumor spiculations are better depicted and appreciated with specimen digital breast tomosynthesis than with specimen ultrasound. (A) Pathologic evaluation of the invasive tumor and spiculations. Tumor is confined to one side of the desmoplastic stroma (arrowheads) with no tumor extension to spicules along the tumor-cell border. On the other side, separate foci of in situ carcinoma are detected close to the resection margin (arrow). (B) Specimen tomosynthesis depicting spiculations close to the resection margin (arrows). (C) Specimen ultrasound showing a large clear margin with no clear visualization of long spiculations (arrows).
Extra resections were performed in 60 of 120 cases (50.0%), both for insufficient margins on imaging or for cosmetic reasons. Of those 60 cases, three (5%) turned out to include additional separate malignant tissue that was not primarily visualized. In those three cases, SUS assessed margins of 4, 8, and over 10 mm for the resected lesions, while ST assessed margins of 8 mm for one lesion and over 10 mm for two lesions. None of these cases had positive margins on histopathology.
Five of the specimens examined had positive margins on histopathology, all for DCIS lesions with a healthy margin of 2 mm or less. Of those five cases, three were multifocal and all had a grade II or III lesions. The OR for SUS to predict a positive margin in histopathology was 3.429 (CI = 0.548–21.432) using a 10-mm cut-off point, while the OR for ST was 2.528 (CI = 0.400–15.994). When the cut-off point was 5 mm, the OR of ST predicting a positive margin in histopathology rose to 3.188 (CI = 0.318–31.998), while SUS reached a markedly higher OR of 14.182 (CI = 2.134–94.254).
When using a cut-off point of 5 mm, SUS managed to successfully detect three out of the five cases, while ST only managed to detect one. This gave SUS a sensitivity of 75.0%, specificity of 90.5%, positive predictive value (PPV) of 21.4%, and negative predictive value (NPV) of 99.1% to detect negative margins, while ST reached values of 25%, 92.8%, 11.1%, and 97.2%, respectively. When increasing the cut-off point to 10 mm, SUS still correctly detected the same three lesions, while the false-positive rate increased considerably to 35 lesions. ST, on the contrary, managed to detect two lesions. This translated into a sensitivity, specificity, PPV and NPV of 60.0%, 69.6%, 7.9%, and 97.6% for US, and of 40%, 79.1%, 7.7%, and 96.8% for ST, respectively (Table 4).
Sensitivity, specificity, PPV, NPV, and OR as measured by specimen ultrasound and tomosynthesis using cut-off points of 5 and 10 mm, compared to histopathology.
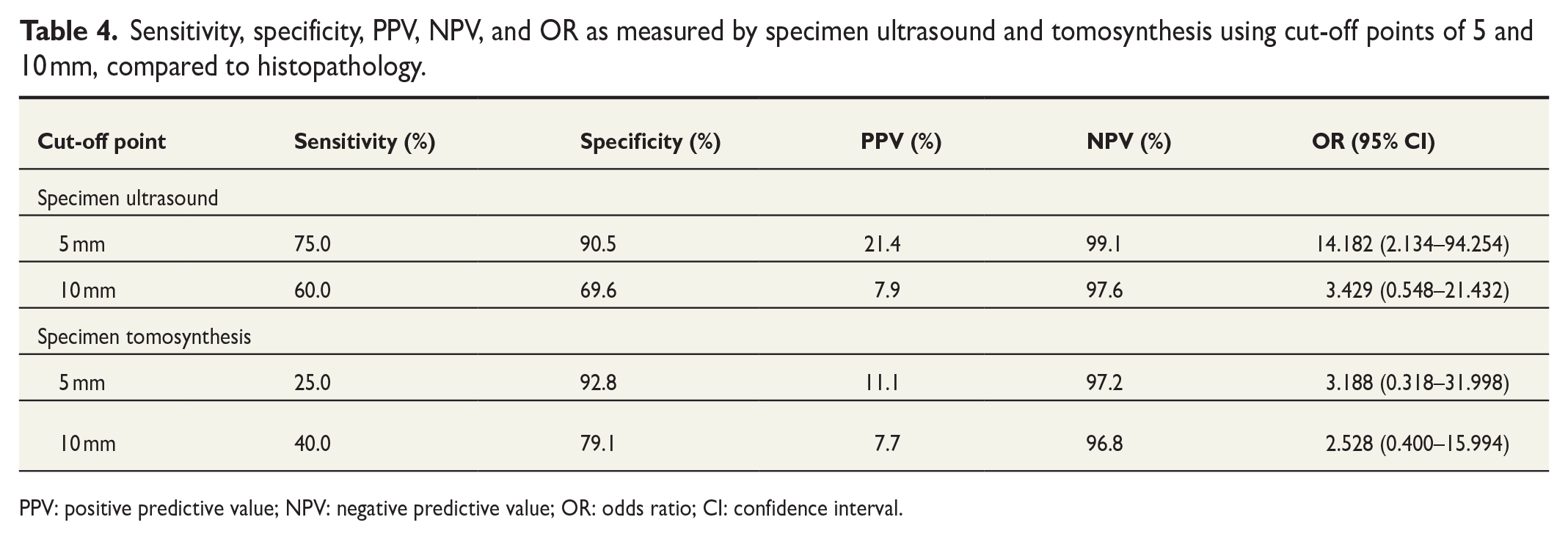
PPV: positive predictive value; NPV: negative predictive value; OR: odds ratio; CI: confidence interval.
Discussion
Results from this study demonstrate that in a defined patient population with non-palpable US-visible breast malignant lesions, SUS is more accurate than ST in surgical margin evaluation. Furthermore, that ST offers no additional benefit. To the best of our knowledge, there are no comparable studies in the published literature.
Previously, in a prospective study on 25 patients, Tan et al. 9 compared intraoperative SUS to specimen mammography and reported that using US alone, higher rates of histologically tumor-free margins were achieved compared to mammography alone. In that study, 88% of lesions were palpable and associated DCIS was found in 76% of tumors. A more recent prospective study by Pop et al. 10 reported higher sensitivity of margin evaluation for SUS compared to mammography (90.91% vs 45.45%), yet with somehow lower specificity (67.21% vs 85.25%). In that study, information on tumor characteristics, localization techniques, and operation guidance was not reported.
Many publications report the use of SUS in the margin assessment of excised breast lesions, yet heterogeneity between studies is high, as was concluded by the meta-analyses.11 –13 This study resembles a more recent study by Perera and Bourke 5 that reported the accuracy of SUS to achieve clear margins. Both this study and the study by Perera have similar inclusion criteria of US-visible lesions, surgical techniques, and SUS protocols, yet contrary to our study, Perera and Bourke 5 did not compare or report ST findings, and furthermore, specimen readers were not blinded to the results.
This study clearly shows a better performance of SUS compared to ST in a selected population. Furthermore, US was previously reported to be implemented in the operating theater to guide tumor resection. This modality has been previously compared to preoperative wire localization and shown to be equally effective or even superior to the latter. 14 A meta-analysis by Mahboobi et al. 13 further reported that SUS has the highest diagnostic performance in intraoperative margin assessment when compared to radiography, digital breast tomosynthesis, and microcomputer tomography.
SUS performed intraoperatively by breast surgeons can be beneficial when compared to ST by reducing operating time through the elimination of specimen transport from the operating room to the radiology department, imaging and reporting by the radiologist. Moreover, an intraoperative analysis, when compared to an analysis in the radiology department, is more accurate through the avoidance of the “Pancake-Phenomenon” brought by gross morphological changes in the resected specimen due to the latent time between surgical excision and the examination in the radiology department. 15 On the contrary, SUS poses certain challenges, in particular flattening of the specimen on the examined surface by the transducer, which could result in a false-positive reading through inadvertently decreasing the length of the margin, specifically, the anterior measured margin. 16 Moreover, US examinations are highly examiner-dependent, which translates to the needed training of breast surgeons to achieve a proper proficiency of US expertise. 17 Esgueva et al. 18 reported that 11 cases are on average sufficient for a surgeon to master the technique. For surgeons already familiar with breast US, which is the case in multiple countries, extra training is not even necessary. 18 Currently, breast cancer treatment is individually tailored in a multidisciplinary setting, and the present results suggest that whenever a lesion is visible on US, intraoperative SUS by properly trained surgeons should be the primary choice for margin assessment. Studies have further shown that US-guided tumor resection is feasible and has at least comparable results to guidewire preoperative localizations.17,19,20 This will not only ease the surgical scheduling and spare patients the additional visits and interventions, but will also save time and resources when combined with SUS. This study suggests that SUS is sufficient and can be easily performed immediately in the operation theater.
It is, however, worth noting that tumor spiculations cannot be fully appreciated with SUS. ST was previously shown to be superior in the visualization of spiculations, 7 and therefore, ST is recommended whenever documentation of the removal of spiculations is required.
No studies are available on the choice of optimal size or shape of transducers for SUS purposes. Different studies report different US platforms and transducers. In the authors’ experience, the state-of-the-art high-frequency, linear array hockey-stick ultrasound transducers are most suitable for this purpose. The slim design allows for easy maneuvering of the specimen without the need to apply pressure and the high frequency allows the clear visualization of the resected lesions and its margins as well as any possible additional lesions.
The risks and reasons for positive margins in BCS are multifactorial and include, among others, variations in the definition of positive margins, the ability to localize impalpable disease, and surgical skills. 11 In our experience, the failure of BCS to achieve negative margins can be attributed to the microscopic invisible tumor burden surrounding or in continuation with the target lesion as well as to an inability to transfer the information from radiological findings in prone or compressed breasts to the surgical supine position of the breasts. Lobular cancers and more often DCIS are specifically associated with higher risk of positive margins.5,21,22 Furthermore, breast cancer is a multifocal disease, 23 and separate microscopic foci of cancerous cells are not infrequent. In this study, three incidental cancers (5%) were detected in additional marginal resections not visualized preoperatively, and this could potentially have resulted in positive margins irrespective of the marginal distance status of the resected lesion.
The limitations of the study are its retrospective nature, being a single-center study, as well as the low number of positive margins in pathology. The specimen readers were, however, blinded primarily to the ST readings. Furthermore, a cohort of consecutive patients was analyzed, consistent with and representative of actual clinical practice.
SUS showed superiority when compared to ST in evaluating intraoperative resection margins of US-visible lesions. Therefore, ST is redundant in US-visible lesions and based on these results not recommended for routine use in applicable situations.
Footnotes
Author contributions
All authors contributed to conceptualization. M.S., A.M., H.O., and S.A. contributed to methodology. H.O., A.M., M.S., and S.J. contributed to validation. H.O., A.M., M.S., and S.A. contributed to formal analysis. S.A., H.O., A.M., and S.J. contributed to investigation. M.S. and R.V. contributed to resources. All authors contributed to data curation management. S.A., A.M., M.S., and H.O. contributed to writing—original draft. All authors contributed to writing—review & editing. All authors contributed to visualization. M.S. and R.V. contributed to supervision. M.S. contributed to project administration. A.M. and R.V. contributed to funding acquisition. All authors contributed to final approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a grant from the Finnish Medical Foundation (S.A.) and from Kuopio University Hospital (A.M. and M.S.) (VTR grant 5063573). The authors declare no relationships with any companies, whose products or services may be related to the subject matter of the article. The funding sources were not involved in study design, data collection or analysis, preparation of the manuscript, or the decision to submit the manuscript.
Ethical approval and informed consent
The chair of the hospital district waived the need to obtain written informed consent from the patients owing to the retrospective nature of the analyses (ethics approval: FinMargins 5063573).
