Abstract
Background and objective:
Defunctioning loop ileostomy (DLI) is frequently used to decrease the consequences of anastomotic leak after ileal pouch-anal anastomosis (IPAA) surgery but is controversial because of stoma-associated morbidity. The aim of this study was to describe stoma-associated morbidity in IPAA–DLI patients compared with terminal ileostomy patients.
Methods:
Patients treated with colectomy for inflammatory bowel disease at Skåne University Hospital, Sweden, between 2005 and 2021 were eligible for inclusion. Terminal stoma-related morbidity was measured until 12 months after colectomy, IPAA surgery, or conversion to ileorectal anastomosis, whichever occurred first. DLI-related morbidity was measured until 12 months after IPAA surgery or stoma closure, whichever occurred first. Laboratory data were reviewed up to 18 months after surgery since patients without complications were rarely subjected to blood sampling. Data on patient characteristics, renal function, surgical complications, and readmissions were collected retrospectively. Primary outcomes were DLI- and terminal ileostomy-related renal morbidity, whereas secondary outcomes focused on stoma-related complications.
Results:
The study cohort consisted of 165 patients with terminal ileostomy after colectomy (median (interquartile range (IQR)): stoma time 30 (15–74) months) and 42 patients with IPAA–DLI (median (IQR): stoma time 4 (3–5) months). One case of anastomotic IPAA leakage was observed. IPAA–DLI patients more often required hospital care due to high-volume stoma output immediately after surgery (0–30 days, 29%) compared with terminal ileostomy patients (4%, p < 0.001). There were no significant differences in acute renal injury (p = 0.073) or chronic renal failure (p = 0.936) incidences between the groups. DLI closure was achieved in 95% of IPAA–DLI patients, with 5% suffering Clavien–Dindo complications > 2.
Conclusions:
IPAA–DLI patients exhibited higher incidence of short-term high-volume stoma output without higher rates of acute renal injury or chronic renal failure compared with terminal ileostomy patients in this small single-center retrospective study suggesting that the risk of renal morbidity in IPAA–DLI patients may have been overestimated.
Keywords
Context and Relevance
A DLI is commonly established during IPAA surgery to reduce the consequences of anastomotic leak but is associated with high morbidity.
A retrospective chart review was performed where complications, especially high-volume stoma output and renal complications, were compared between IPAA–DLI patients and terminal ileostomy patients following colectomy due to IBD.
Out of 42 patients with IPAA–DLI, one patient suffered anastomotic leakage. Despite IPAA–DLI patients more frequently requiring hospital care due to high-volume stoma output than patients with terminal ileostomy, no increased incidence of acute renal injury or chronic renal failure was seen suggesting that the risk of acute renal injury and chronic renal failure in IPAA–DLI patients may have been overestimated.
Introduction
Defunctioning loop ileostomy (DLI) is frequently used to decrease the consequences of anastomotic leak when performing a low anastomosis in rectal cancer surgery or in ileal pouch-anal anastomosis (IPAA) reconstructive surgery after colectomy. 1 IPAA surgery can be performed in a one- or two-stage manner. In a one-stage procedure, the IPAA is created without a DLI, allowing direct restoration of gastrointestinal (GI) continuity. In a two-stage approach, a DLI is established perioperatively in the right lower quadrant to protect the newly created pouch while healing. 2 Typically, DLI closure is performed within 3–6 months.
The evidence for establishing a DLI in conjunction with IPAA reconstruction is to a high extent extrapolated from rectal cancer surgery. Although effective in reducing septic consequences of an anastomotic leak, DLI is associated with considerable morbidity, especially high-volume stoma output and renal failure.3–8 Notably, Vogel et al. 9 showed that high-volume output with subsequent dehydration was the leading cause of readmission for patients with terminal ileostomy or DLI, independent of the stoma indication.
However, IPAA–DLI is anatomically and functionally different compared with a DLI created to protect a colorectal anastomosis in rectal cancer surgery. IPAA–DLI is created more proximally on the small bowel, shortening the functional length of the small bowel which increases the risk of high-volume stoma output and its related complications.2,10,11 The stoma also has lower height since the small bowel mesentery is stretched down toward the anal canal to form the IPAA, which can cause problems with bandaging and stoma leakage. Other stoma-specific complications like parastomal herniation occur frequently.10,11 Only a limited number of studies have been performed on IPAA–DLI, and as of now, no conclusive consensus concerning the risks and advantages of IPAA–DLI has been made.12–14
This study aimed to describe stoma-associated morbidity, especially renal morbidity, with DLI in IPAA reconstructive surgery. We hypothesized that DLI was associated with a higher incidence of high-volume stoma output with subsequent increased frequency of renal complications.
Methods
Patients who underwent colectomy due to inflammatory bowel disease (IBD) at Skåne University Hospital, Sweden, between 2005 and 2021 were assessed for inclusion. ICD-10 codes K50, K51, and K52 and NOMESCO codes JFC40, JFH01, JFH10, JFH11, JGB50, and JGB61 were used to identify eligible patients. Medical records were reviewed retrospectively with data collection being performed up until February 2023. Patients with comorbidity that severely hindered interpretation of complications were excluded on a case-to-case basis after assessment by all authors. All gastrointestinal and surgical events were registered for each patient with subsequent follow-up starting at the date of colectomy or IPAA surgery. Data were registered regarding patient characteristics, diagnosis, renal function, short- and long-term morbidity, readmissions, and stoma-related complications. Surgical complications were classified according to the Clavien–Dindo (CD) grading system, with a CD value > 2 considered severe. 15 Primary outcomes were DLI- and terminal ileostomy-related renal morbidity, whereas secondary outcomes were stoma-related complications.
All patients who received a terminal ileostomy following colectomy were included in the terminal ileostomy group. Patients who underwent reconstructive IPAA surgery with DLI were included in the IPAA–DLI group. Patients who underwent colectomy and, at a later stage, IPAA surgery were included in both groups during their respective time at risk to increase the study population and mitigate selection bias. To balance time at risk for morbidity with DLI and terminal ileostomy, follow-up duration for surgical complications was limited to within 12 months after surgery, until stoma conversion (terminal ileostomy → DLI), stoma reversal (terminal ileostomy → ileorectal anastomosis (IRA)), or stoma closure (IPAA–DLI → IPAA), whichever occurred first.
For renal function, preoperative creatinine for each respective surgery was used as baseline (Fig. 1). There was typically no uniform laboratory follow-up in patients without postoperative complications or renal failure. For both acute renal injury and chronic renal failure assessment, laboratory data were collected up to 18 months after surgery or until stoma conversion or stoma reversal, whichever occurred first. After stoma closure, no additional assessment of acute renal injury was made for IPAA patients. The estimated glomerular filtration rate (eGFR) was calculated with the LMR18 formula, which has been validated in Swedish cohorts and is used routinely in clinical practice in Sweden.16–18
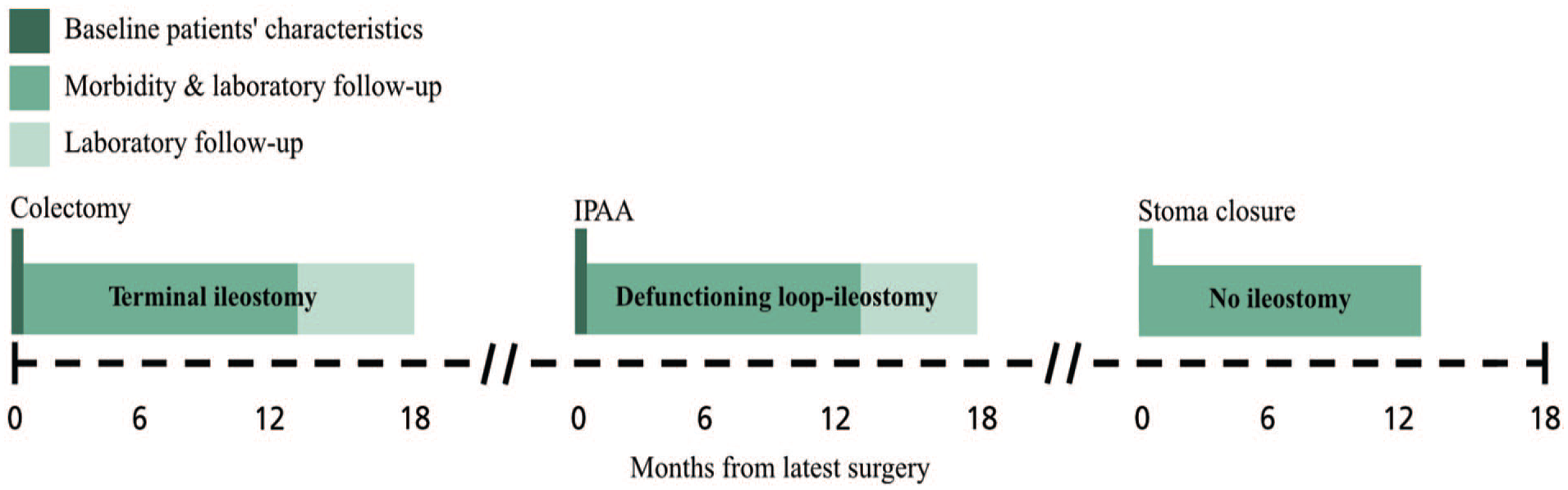
Data collection. Schematic example of data collection for a patient who performed colectomy and at a later point received an IPAA with a DLI. Patients who did not perform colectomy in Skåne Region were included first at the time of IPAA. Patients who did not perform IPAA surgery were only included during the time at risk following colectomy. Morbidity and laboratory follow-up ended prematurely if patients underwent stoma conversion, stoma reversal, or stoma closure. IPAA: ileal pouch-anal anastomosis; DLI: defunctioning loop ileostomy.
Hospital admissions due to high-volume stoma output during follow-up were recorded. Stoma-related morbidity such as small bowel obstruction, stenosis, or stoma obstruction was registered. Clinically evident parastomal hernias were registered based on clinical examination, history of symptoms, or computed tomography (CT) scans. Problems with leakage and/or stoma skin issues mentioned in physicians’ notes during clinical follow-up were registered.
Definitions
IPAA is a type of reconstructive surgery performed after colectomy where a pouch of small bowel is created and anastomosed to the anal canal. The length of bowel used to construct the ileal pouch is typically between 40 and 50 cm.
Anastomotic leakage was defined according to the International Study Group of Rectal Cancer (ISREC) classification and based on physicians’ notes. 19 Symptoms vary from systemic inflammatory or infectious presentations to more subtle symptoms where radiological examination may be required. Anastomotic leakage is associated with an increased risk of complications such as pelvic abscesses and fistulation and may cause severe morbidity if left untreated.
Terminal ileostomy is an end-ileostomy typically placed in the right lower quadrant. Normally, only the distal 5–10 cm of small bowel has been resected during the colectomy and the ileostomy is thereby almost at the end of the entire small bowel.
Acute renal injury was assessed using the highest serum creatinine level during the follow-up period. Staging was performed according to Risk of renal dysfunction, Injury to kidney, Failure or Loss of kidney function, and End-stage kidney disease (RIFLE) classification. 20
Chronic renal failure was measured using the lowest registered serum creatinine during the follow-up period for estimation of baseline renal function. Staging was performed according to Kidney Disease Improving Global Outcomes (KDIGO) 2012 guidelines based on the eGFR result. 21
High-volume stoma output was defined as any instance where the patient required hospital care such as intravenous fluids or hospital admission due to high-volume stoma output.
Statistical analysis
Categorical variables were presented as frequency (%) and continuous data as means with standard deviation (SD) unless stated otherwise. Student’s t-test and Pearson’s chi-square test or two-tailed t-test were used for intergroup comparisons when appropriate. Survival analysis was presented using Kaplan–Meier survival plots, with significant differences assessed with the log-rank test. Missing data were excluded when calculating differences between groups. Statistical analyses were conducted using IBM SPSS Statistics version 29 for Windows (IBM Corp, Armonk, NY, USA). The p value <0.05 was considered significant.
Results
Study population
A total of 166 patients were eligible for inclusion in the study cohort. One patient was excluded due to stage 5 chronic renal failure with ongoing dialysis and liver transplantation before colectomy. Of the remaining 165, 43 (26.1%) patients were subsequently reconstructed with IPAA. One IPAA-reconstructed patient did not receive a DLI and was excluded, leaving 42 IPAA–DLI patients (Fig. 2). Among these, one case of anastomotic leak was observed. In total, 61 (37.0%) patients who underwent colectomy were reconstructed with either IPAA or IRA.
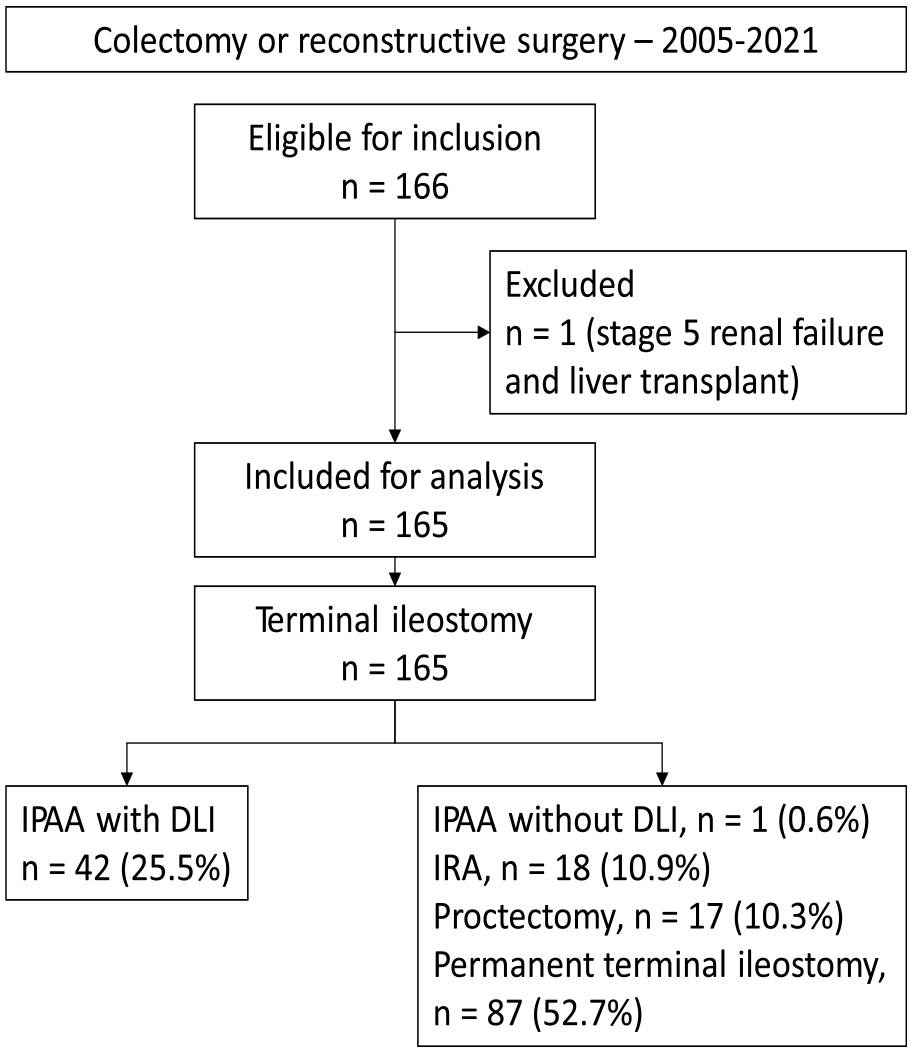
Study flow chart.
Baseline patient characteristics are presented in Table 1. Patients who underwent IPAA reconstruction were younger and had lower American Society of Anesthesiologists (ASA) scores, and all had a history of ulcerative colitis. Before reconstructive surgery, preoperative eGFR was significantly reduced for IPAA patients (p < 0.001) compared with terminal ileostomy patients before colectomy, although no progression in preoperative chronic renal failure rates was seen (Table 1). Laparotomy was the primary approach for both colectomy and IPAA reconstruction during the inclusion period.
Baseline patient characteristics.
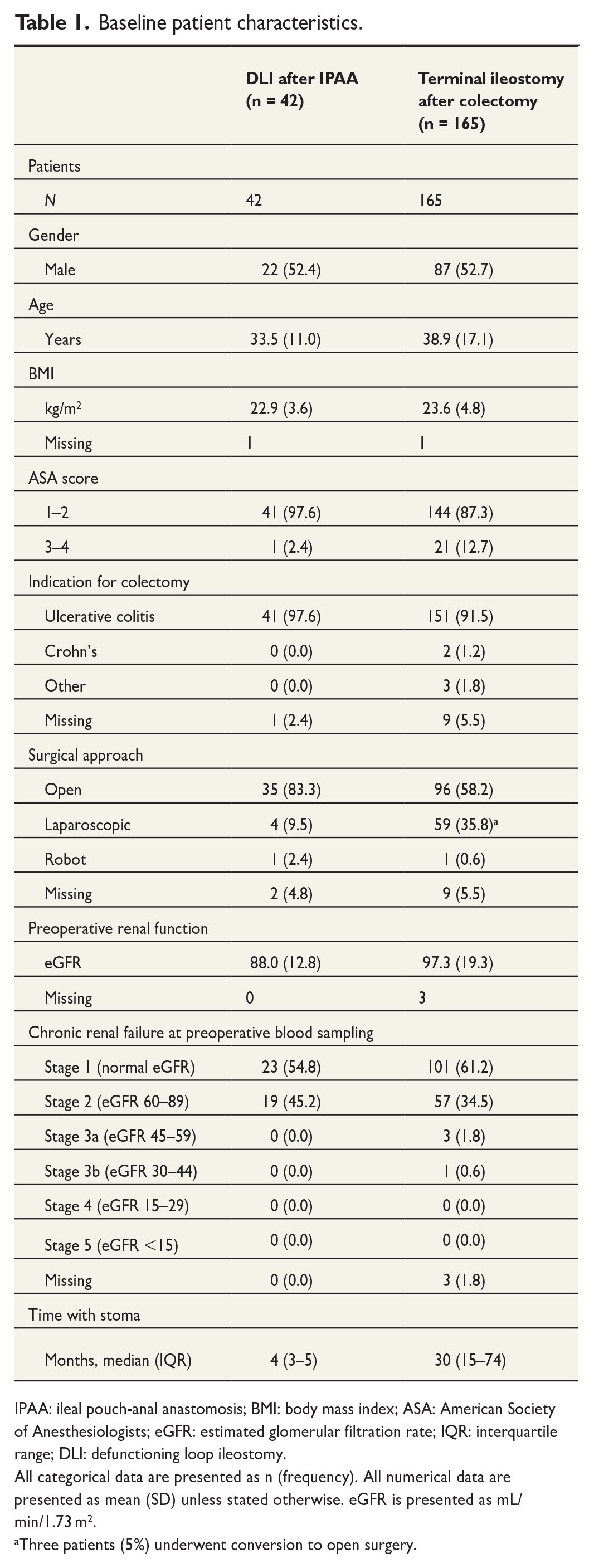
IPAA: ileal pouch-anal anastomosis; BMI: body mass index; ASA: American Society of Anesthesiologists; eGFR: estimated glomerular filtration rate; IQR: interquartile range; DLI: defunctioning loop ileostomy.
All categorical data are presented as n (frequency). All numerical data are presented as mean (SD) unless stated otherwise. eGFR is presented as mL/min/1.73 m2.
Three patients (5%) underwent conversion to open surgery.
Stoma-related morbidity
Stoma-related complications are described in Table 2. In IPAA–DLI patients, hospital admissions and instances of high-volume stoma output events were most frequently reported. IPAA–DLI patients more often suffered from high-volume stoma output within 30 days of surgery (28.6%) compared with terminal ileostomy patients (3.6%, p < 0.001). Treatment with intravenous fluid and electrolyte resuscitation in combination with medications reducing bowel motility and fluid secretion often sufficed, with no interventions reaching a higher CD classification than 2. Stoma leakage and skin issues during the first 30 days were reported by 19.0% of IPAA–DLI patients compared with 1.8% of terminal ileostomy patients (p < 0.001). Overall, IPAA–DLI patients suffered more short-term (0–30 days) complications than patients with terminal ileostomy, whereas long-term (2–12 months) complications were comparable.
Stoma-related complications.
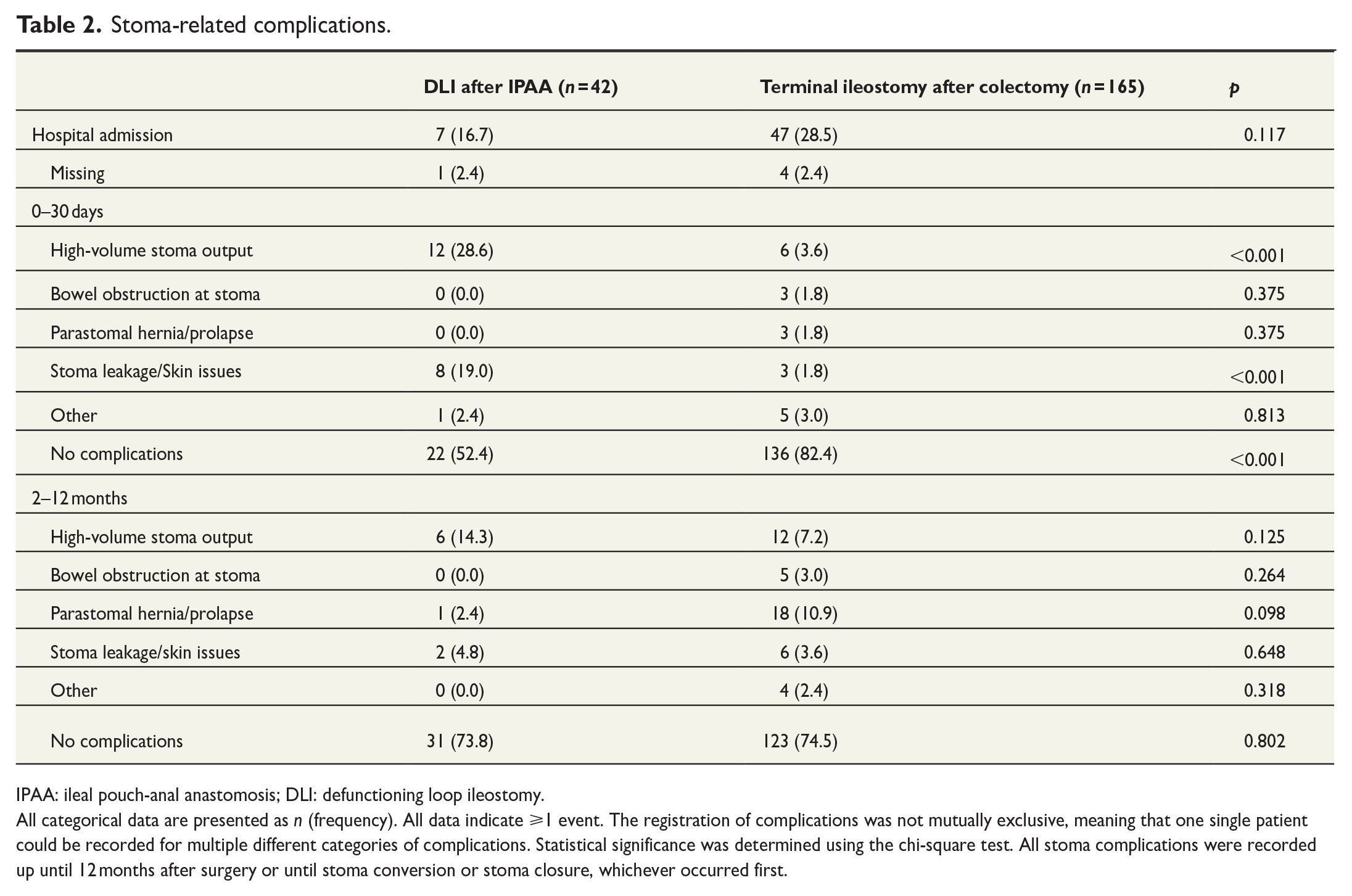
IPAA: ileal pouch-anal anastomosis; DLI: defunctioning loop ileostomy.
All categorical data are presented as n (frequency). All data indicate ⩾1 event. The registration of complications was not mutually exclusive, meaning that one single patient could be recorded for multiple different categories of complications. Statistical significance was determined using the chi-square test. All stoma complications were recorded up until 12 months after surgery or until stoma conversion or stoma closure, whichever occurred first.
Renal morbidity
Acute renal complications were less frequent (9.5% versus 23.0%, p = 0.073) and less severe (no versus two cases of Grade 3 acute renal injury) in IPAA–DLI patients compared with terminal ileostomy patients (Table 3, Fig. 3). Chronic renal failure incidences were comparable between DLI patients and terminal ileostomy patients (33.3% versus 31.5%, p = 0.935; Table 3).
Renal complications.
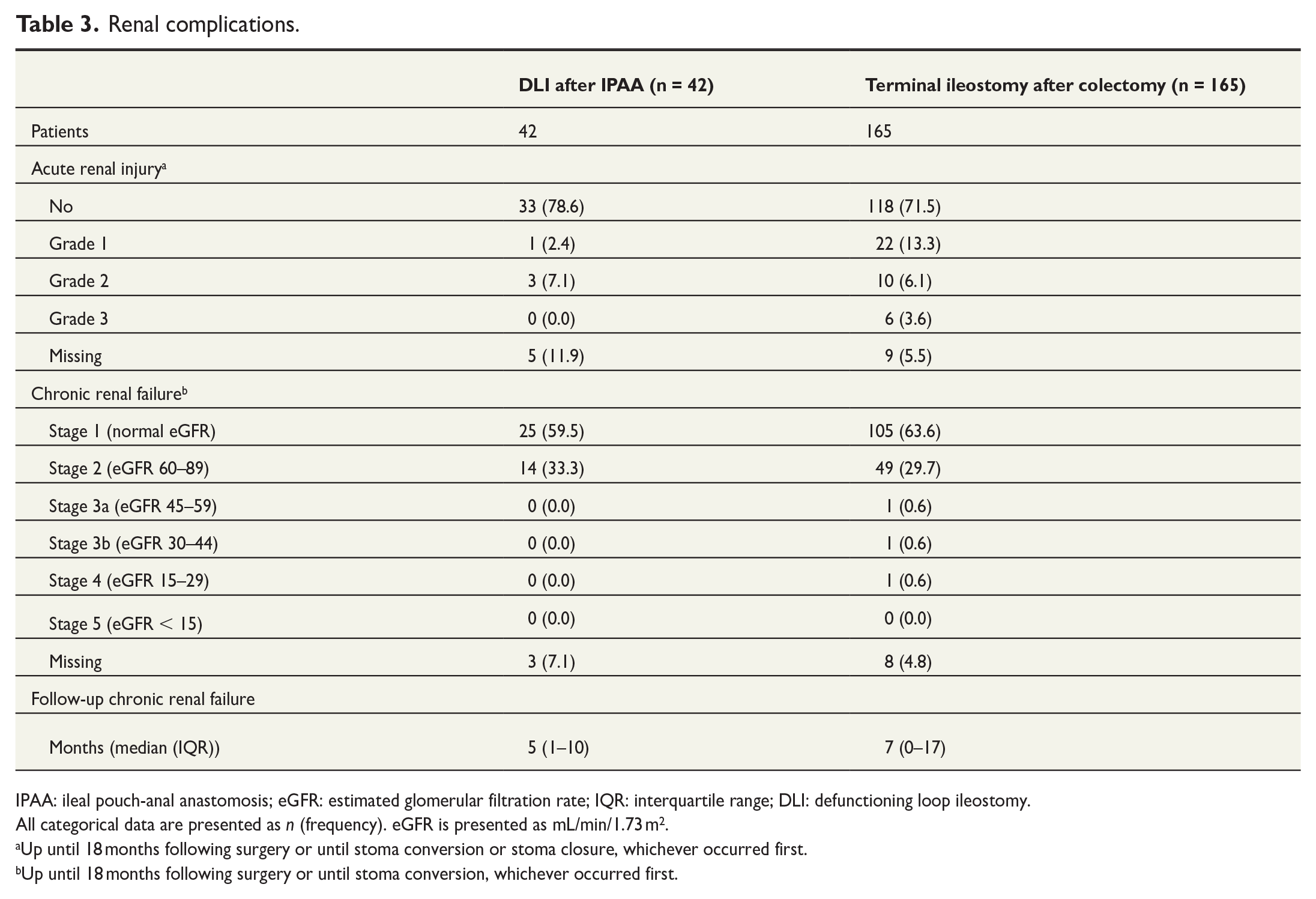
IPAA: ileal pouch-anal anastomosis; eGFR: estimated glomerular filtration rate; IQR: interquartile range; DLI: defunctioning loop ileostomy.
All categorical data are presented as n (frequency). eGFR is presented as mL/min/1.73 m2.
Up until 18 months following surgery or until stoma conversion or stoma closure, whichever occurred first.
Up until 18 months following surgery or until stoma conversion, whichever occurred first.
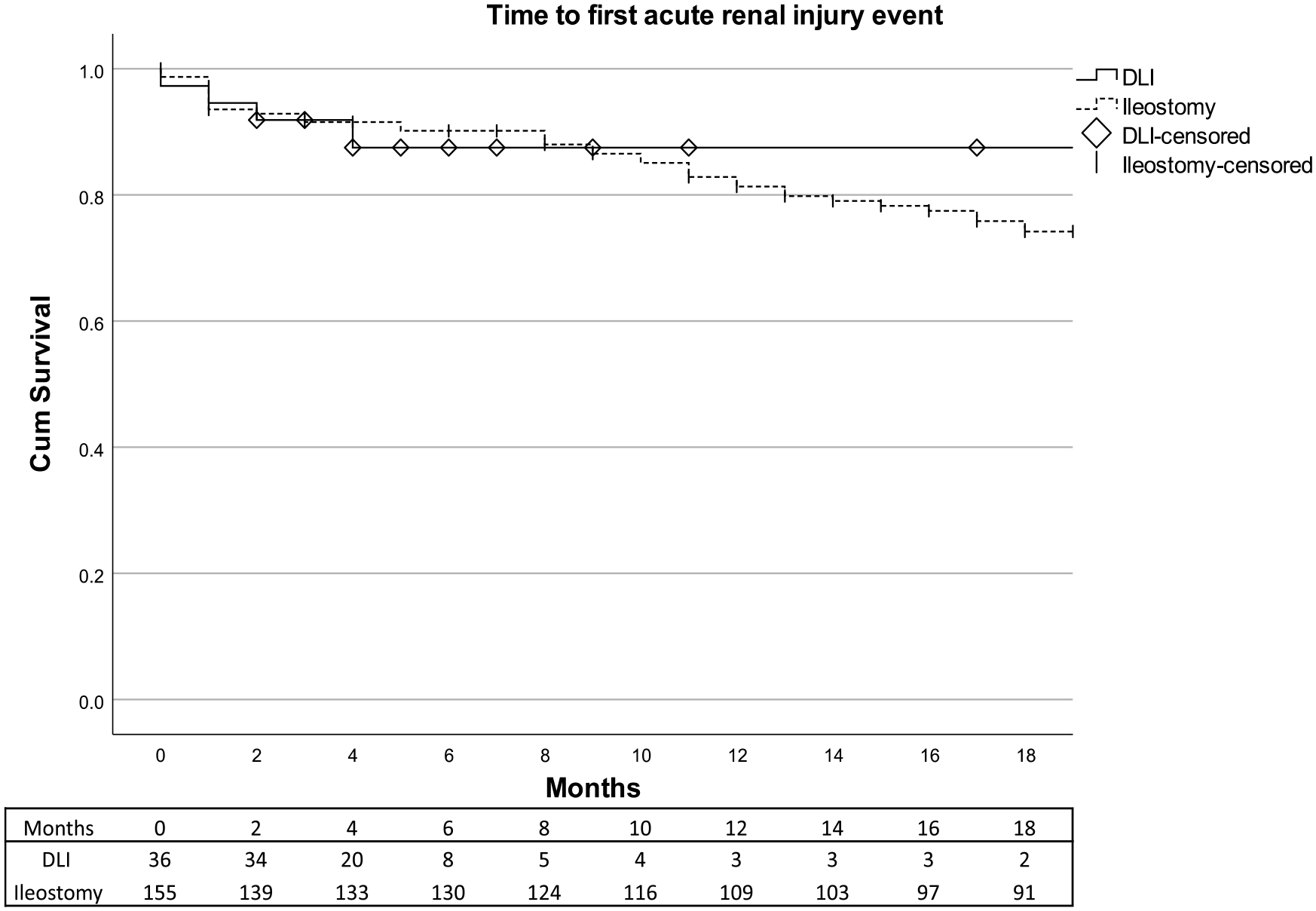
Time to first acute renal injury event for patients with DLI (straight black line) and patients with terminal ileostomy (interrupted black line). Censored patients are marked with a vertical line (terminal ileostomy) or rhombus (DLI). Attached is a corresponding table of the number at risk. No statistical analysis was performed due to crossing curves. DLI: defunctioning loop ileostomy.
Parastomal hernia and prolapse
Patients with IPAA–DLI had numerically lower frequency of parastomal hernia or prolapse within 0–30 days (0% versus 1.8%) and after 2–12 months (2.4% versus 10.9%) compared with patients with terminal ileostomy, although the difference was not significant (Table 2). Furthermore, patients with IPAA–DLI suffered less serious complications (CD value > 2) within 0–30 days (0% versus 6%) and after 2–12 months (2.4% versus 7.9%) compared with patients with terminal ileostomy. However, as illustrated in Fig. 4, IPAA–DLI patients more rapidly underwent stoma closure with only 53% remaining after 4 months, making the time at risk for IPAA–DLI patients significantly shorter than for patients with terminal ileostomy.
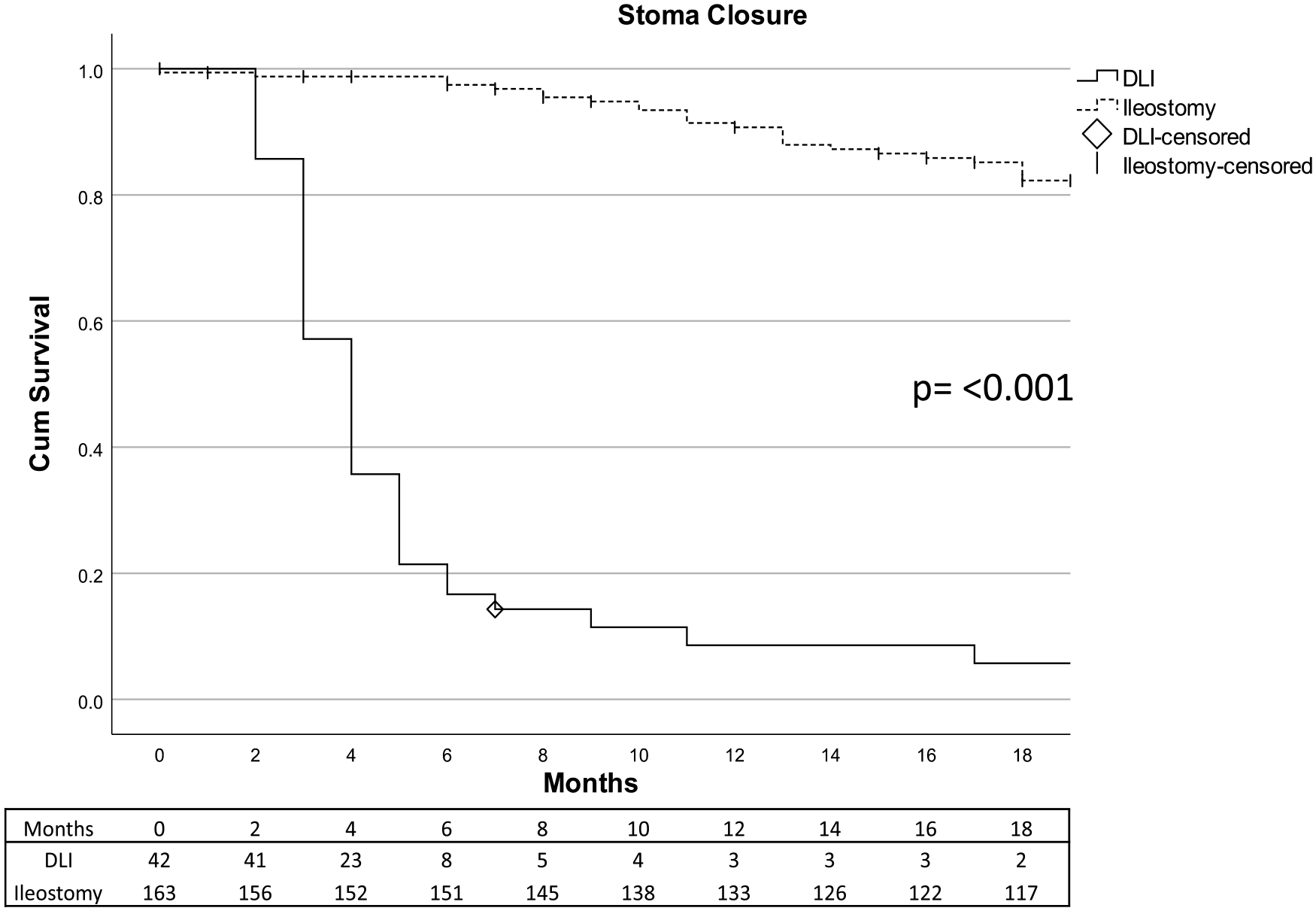
Eighteen months stoma closure or stoma conversion for patients with DLI (straight black line) and patients with terminal ileostomy (interrupted black line). Censored patients are marked with a vertical line (terminal ileostomy) or rhombus (DLI). Attached is a corresponding table of the number at risk. DLI: defunctioning loop ileostomy.
Morbidity related to stoma closure
IPAA–DLI were expediently closed, as shown in Fig. 4. Two stomas had not been closed at the end of follow-up (Table 4). One patient was awaiting stoma closure at the time of data collection, whereas the other patient developed an anastomosis leakage with a rectovaginal fistula whereon no stoma closure attempt was performed. No anastomotic leaks were reported after DLI closure. However, 35.0% of closure patients suffered from other complications, of which two were severe (CD value > 2; Table 4).
Stoma closure.
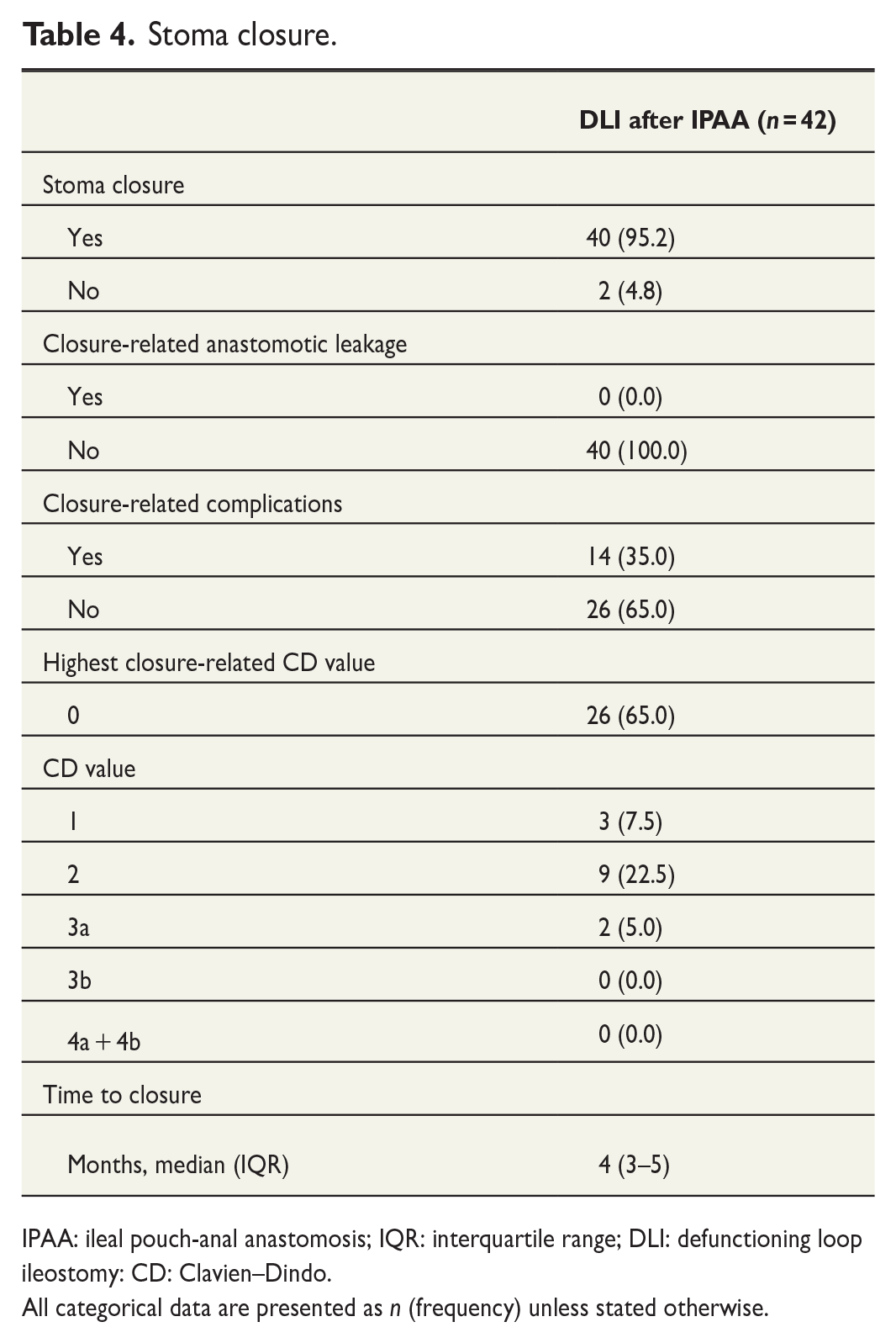
IPAA: ileal pouch-anal anastomosis; IQR: interquartile range; DLI: defunctioning loop ileostomy: CD: Clavien–Dindo.
All categorical data are presented as n (frequency) unless stated otherwise.
Discussion
This study shows that despite an increased risk of high-volume stoma output, DLI for IPAA patients was not associated with an increased incidence of acute renal injury or chronic renal failure compared with patients with terminal ileostomy. Considering the low rate of long-term complications related to DLI in IPAA patients, this study supports the use of DLI.
This study found similar rates of high-volume stoma output to those reported on defunctioning stoma after anterior resection. 4 Unlike these studies, no increase in renal morbidity was observed.22,23 This may be attributed to a more careful patient selection, resulting in a younger, healthier cohort with fewer comorbidities and shorter DLI durations. Indeed, IPAA patients in our study exhibited lower ASA scores and were younger at the time of surgery compared with terminal ileostomy patients, indicating a selection bias.
Moreover, patients with terminal ileostomy experienced more severe complications following colectomy. This may be influenced by the acute setting correlated to colectomy, where patients often received high-dose corticosteroids and biological medication resulting in compromised physiological and nutritional state. 24 IPAA–DLI patients also suffered stoma leakage to a greater extent than terminal ileostomy patients during the initial 30 days after surgery, which is likely due to the anatomical differences between the stoma types which complicates output management and increases the risk of stoma leakage.
Although our study was not designed to compare the risk of anastomosis leakage for IPAA with or without DLI, low incidence of anastomosis leakage in IPAA–DLI patients could be demonstrated. One could argue that a more selective approach for DLI might be warranted, allowing direct IPAA usage and reduced morbidity in patients with a lower risk of anastomotic leakage. However, further studies are required before such risk stratification can be implemented.
Currently, a lack of randomized controlled trials regarding the use of DLI in IPAA patients has left this practice without adequate evidence-based support. Previous attempts have been discontinued due to difficulties including patients. Despite the absence of robust evidence, nearly all IPAA patients in Sweden are subjected to DLI as reflected in this study. In contrast, international practices utilize a more selective approach, as exemplified in a North American study where only 70% of surgeons created a DLI during IPAA surgery. 25 In an attempt to address this gap in knowledge, the randomized IDEAL study was initiated in 2019 and will hopefully be able to answer whether DLI is necessary in IPAA surgery once completed. 26 Until conclusive evidence for IPAA–DLI, our study may aid clinicians in understanding and informing patients about adverse events related to IPAA–DLI.
This study is limited by the single-center retrospective design and small number of IPAA patients. IPAA has become a rare procedure in Sweden, making clinical studies challenging with this study identifying only 43 IPAA patients between 2005 and 2021. Existing studies lack large cohorts with reported renal functional outcomes in these patients.10,11 According to the Swedish Inflammatory Bowel Disease Registry yearly report, roughly 350 IBD-related colectomies were performed annually before the coronavirus disease 2019 (COVID-19) pandemic in Sweden of which 30 patients were reconstructed with either IPAA, IRA, or a Kocks reservoir. 27 This is lower than national data on previously performed IPAA procedures and modern estimation of eligible patients.28,29 Despite the low volume of patients, IPAA may become more common as centralization increases, making research on the risks and benefits of surgical choices such as establishing DLI essential. Important to note is that IPAA overall is a safe reconstructive method with high patient satisfaction. 30
Medical records and doctors’ notes were retrospectively reviewed in this study. Patients were followed by the treating physician more intensively after IPAA surgery than patients after colectomy, which might result in an over-representation of IPAA- and DLI-related morbidity. However, apart from high-volume stoma output which required hospital care and should have been recorded to an equal extent for both groups, IPAA–DLI was associated with less morbidity than terminal ileostomy which is the opposite of the expected result, if IPAA-related morbidity was over-represented. As nurses’ notes were not reviewed, data on complications such as skin irritation, stoma prolapse, and reduced quality of life might have been under-reported.
During the study period, there was no structured laboratory follow-up for terminal ileostomy or DLI patients. To compensate for this, we choose to include the highest and lowest creatinine values during the time at risk for each group. Unfortunately, the time to chronic renal failure assessments was low in both groups. Thus, even though we showed no increased risk of chronic renal failure for IPAA–DLI patients, we cannot exclude that such an effect could have appeared if sampling was performed later. As such, recognition and adequate management of high-volume stoma output remains important to reduce the risk of renal morbidity, regardless of stoma type.
In conclusion, IPAA–DLI patients did not show higher rates of acute renal injury or chronic renal failure despite correlating to higher frequency of high-volume output complications at 30 days compared with terminal ileostomy patients in this small single-center retrospective study, suggesting that the risk of acute renal injury and chronic renal failure in IPAA–DLI patients may have been overestimated.
Footnotes
Author Contribution
The authors all contributed to the conception and design of the study. S.L. conducted the initial data collection and oversaw the inclusion process. S.L. and E.A. performed the statistical calculations, interpreted the results, and contributed to the drafting of the article. All authors participated in the revision of the article and read and approved the final version of the article.
Availability of data and material
Data could be made available upon request to the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Open access funding provided by Lund University. This study was funded by ALF grants.
Ethics approval
The study was approved by the Swedish Ethical Review Authority (grant no. DNR: 2022-00397-01).
Informed consent
Informed consent was not obtained due to the retrospective design and the fact that all interventions, and in many cases of follow-up, were completed at the time of data collection, rendering our findings inconsequential to the outcomes for these patients.
Previous communication
Poster presented at the Swedish Surgical Week 2023 in Örebro, Sweden (21–25 August 2023) and European Society of Coloproctology’s 18th Scientific and Annual Conference in Vilnius, Lithuania (27–29 September 2023).
