Abstract
Background and Aims:
Development of a pouch-related fistula tract is an uncommon but highly morbid complication to restorative proctocolectomy with ileal pouch-anal anastomosis. Pouch failure with permanent ileostomy is reported in 21%–30% of patients, yet the factors contributing to pouch excision remain poorly defined. The aim of this study was to determine the incidence and treatment results of complicated pouch-related fistula, as well as to evaluate factors involved in excision after pouch failure.
Material and Methods:
The study was conducted as a retrospective study. All patients with diagnosed pouch-related fistulas were registered with information related to fistula classification, treatments, and outcome.
Results and Conclusion:
The final analysis included 48 (10.7%) of the 447 total ileal pouch-anal anastomosis patients with complicated pouch-related fistulas. Pouch-vaginal fistulas, pouch-perianal fistulas, and other pouch-related fistulas were observed in 19 (63%), 29 (60%), and 10 (21%) patients, respectively, corresponding to an accumulated risk of 8%, 6%, and 2%, respectively. Time from ileal pouch-anal anastomosis surgery to fistula presentation was 24 (0.2–212) months. Overall pouch failure, defined as pouch excision or a diverting stoma, was seen in 34 (71%) patients, while pouch excision was seen in 23 (48%) of the patients. Patients who developed Crohn’s disease had a significantly higher risk of pouch excision, as did patients with an early onset of the fistula after ileal pouch-anal anastomosis (P = 0.006 and P = 0.007, respectively). In conclusion, the present study demonstrated a high risk of pouch failure in patients with complicated pouch-related fistulas. Furthermore, it showed that Crohn’s disease and the development of early onset fistulas are associated with pouch excision.
Introduction
Ileal pouch-anal anastomosis (IPAA) has become the standard procedure for the preservation of continence after proctocolectomy in patients with ulcerative colitis and familiar adenomatous polyposis (FAP). As long-term outcome data continue to emerge, it seems clear that IPAA has been proven to be a safe and durable procedure that improves the quality of life (QoL). However, it is not without risks and potential complications (1, 2). Development of a pouch-related fistula tract is an uncommon but highly morbid complication.
Pouch fistulas may occur at any time following restorative proctocolectomy, with an incidence of 2.6%–14%, depending on the length of follow-up (1, 3–5). In most cases, the ileal-anal anastomosis is the origin of the fistula. The fistula can become more complicated with involvement of the sphincter.
Several factors have been associated with the development of pouch fistulas, including preoperative colorectal pathology, operative technique, and post-operative pelvic sepsis (6, 7). Patients who develop Crohn’s disease (CD) after IPAA are at particularly high risk. Several operative techniques have been described to control pelvic and perianal sepsis, and ultimately eliminate the fistula tract (8–10), but because of the individual complexity of the fistulas, optimal management continues to be controversial.
Pouch failure with permanent ileostomy is reported in 21%–30% of patients with fistulas (4, 5, 11), yet the factors contributing to pouch excision remain poorly defined. The aim of this study was to determine the incidence and overall results of surgical treatment for pouch-related fistulas, as well as to evaluate the factors involved in pouch excision after pouch failure.
Material and Methods
The study was conducted as a retrospective study. Using administrative data, we identified all adult patients treated with IPAA at a tertiary referral center for inflammatory bowel surgery from January 1983 to August 2013. Follow-up ended 1 August 2014. International Classification of Diseases, Revision 8 (ICD8) and International Classification of Diseases–10th Revision (ICD10) codes were used before and after 1994, respectively. The following diagnosis codes were used: “Ulcerative Colitis” (D56319, DK51), “Crohn’s Disease” (D56301, DK50), “Familiar Adenomatous Polyposis” (D21136, DD126F), and “Unspecified Inflammatory Bowel Disease” (D56101, DK52). In addition to the diagnosis code, a procedure code was required. The data were collected on the following codes: “Proctocolectomy and IPAA” (KJFH), “Proctectomy and IPAA” (43890, 43785/6, KJGB50/1, KJGB60/1), “Ileorectostomy” (43860), “Rectal Resection” (45840, 46100), “Total Colectomia”(45060), and “Occlusion of Enterostomy” (44120, KJFG00/20).
All patients with diagnosed pouch-related fistulas were registered with information related to demographics, preoperative diagnosis, final diagnosis, fistula classification, therapy, and outcome. The fistula classification was determined primarily by the description of the clinical examination by the surgeon. Radiologic intervention was used supplementary in some cases. Simple cryptoglandular anal fistulas and other fistulas without any relation to the pouch were excluded, that is, where the inner opening of the fistula tract was distal to the ileoanal anastomosis and without any secondary tract in relation to the pouch. The staging classification of IPAA was defined as one-stage (total proctocolectomy with IPAA, and no fecal diversion), two-stage (total proctocolectomy with IPAA and diverting loop ileostomy), and three-stage (subtotal colectomy followed by completion proctectomy with IPAA, and a diverting loop ileostomy, followed by ileostomy closure). Finally, an intended-three-stage procedure was defined as a subtotal colectomy followed by completion proctectomy with IPAA without fecal diversion. All patients were treated with the J-pouch design.
Fistula tracts were classified according to clinical and radiological findings (when available) as pouch-vaginal, pouch-perianal, and others, including pouch-entero, pouch-sacral, pouch-labia, and pouch-abdominal wall. Each fistula was defined for each individual patient; thus, a patient could have more than one type of fistula. All treatments were registered; hence, a patient could have several listed treatments. The treatments were divided into seton placement (prophylactic and conservative), collagen plug insertion, fibrin glue, trans-sphincteric fistulectomy with sphincter reconstruction or with advancement flap surgery, simple fistulotomy, laparotomy with abdominal fistula closure, and a conservative approach with a diverting stoma only. Pouch failure was defined as either pouch excision or a permanent ileostomy with the pouch in situ with a loop or an end-ileostomy.
Statistics
Variables were analyzed descriptively according to data type, that is, continuous variables were expressed by median and range, and categorical variables were expressed as frequency counts, including respective percentages. The significance of the differences between the groups was evaluated using a Fisher’s exact test, a Mann–Whitney U test, and a T-test, as appropriate. A two-tailed significance level of 5% was used. Missing data were handled using list-wise deletion. Stata/IC 13 (StataCorp LP, College Station, TX, USA) was used for the statistical analysis.
Results
Of the 447 patients who underwent IPAA, we identified 56 (12.5%) patients who had been surgically treated for fistulas. Eight patients (2%) had a fistula without pouch involvement or had developed perineal fistula after pouch excision, and they were all excluded. In total, 48 (10.7%) patients were included in the final analyses. Pouch-vaginal fistulas, pouch-perianal fistulas, and other fistulas were observed in 19 (63%), 29 (60%), and 10 (21%) of the patients, respectively, corresponding to a total risk in all patients having IPAA of 8% (231 women), 6%, and 2%, respectively. The patient characteristics are given in Table 1.
Patient characteristics.

IPAA: ileal pouch-anal anastomosis.
Values are expressed in medium (range) or number (%).
Pouch-vaginal fistulas are indicated in percentage of women only. Other fistula classifications include pouch-abdominal wall, pouch-sacral, pouch-labia, or pouch-entero.
All patients, except one with FAP, had surgery for ulcerative colitis. The time from IPAA surgery to fistula presentation was 24 (0.2–212) months. The patient with FAP had a mucosectomy and a hand-sewn anastomosis. This patient developed a fistula to vagina during the immediate postoperative period due to an iatrogenic perforation from suturing. CD developed in 11 (23%) patients, which resulted in pouch excision in all 11 cases. Of the 10 patients who presented with “other fistula,” four had a fistula between the blind limp of the pouch and the abdominal wall, and a pouch-sacral fistula developed in three patients. Two patients developed fistulas involving the pouch, the small intestine, the abdominal wall, and the presacral space, and one patient developed a fistula from the J-pouch to the major labia. The number of different treatments is listed in Table 2, including conservative treatment with diverting stoma. It was not possible to classify the treatment in one patient because of missing information.
Fistula treatment.
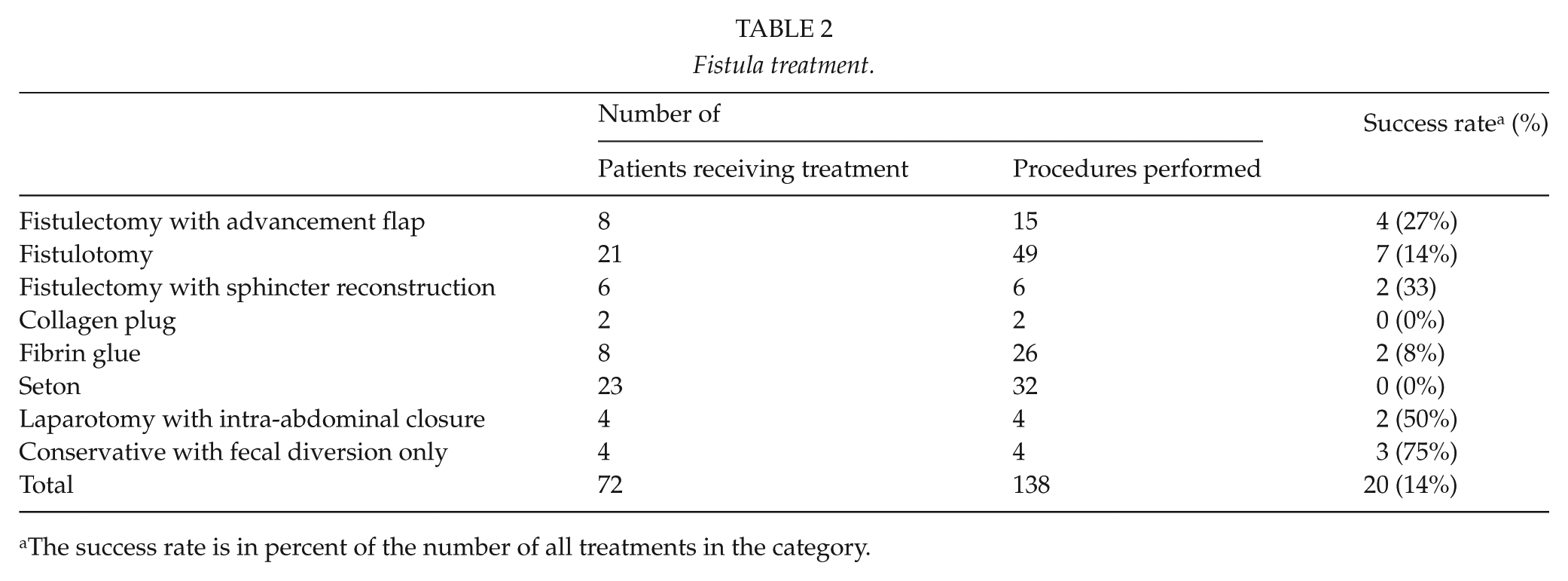
The success rate is in percent of the number of all treatments in the category.
Overall pouch failure was seen in 34 (71%) patients, while 23 (48%) patients had the pouch excised. Patients who developed CD had a significantly higher risk of pouch excision, as did the patients with an early onset of fistula development after IPAA (P = 0.006 and P = 0.007, respectively). No other factors were significantly associated with increased risk of pouch excision (Table 3).
Pouch failure.
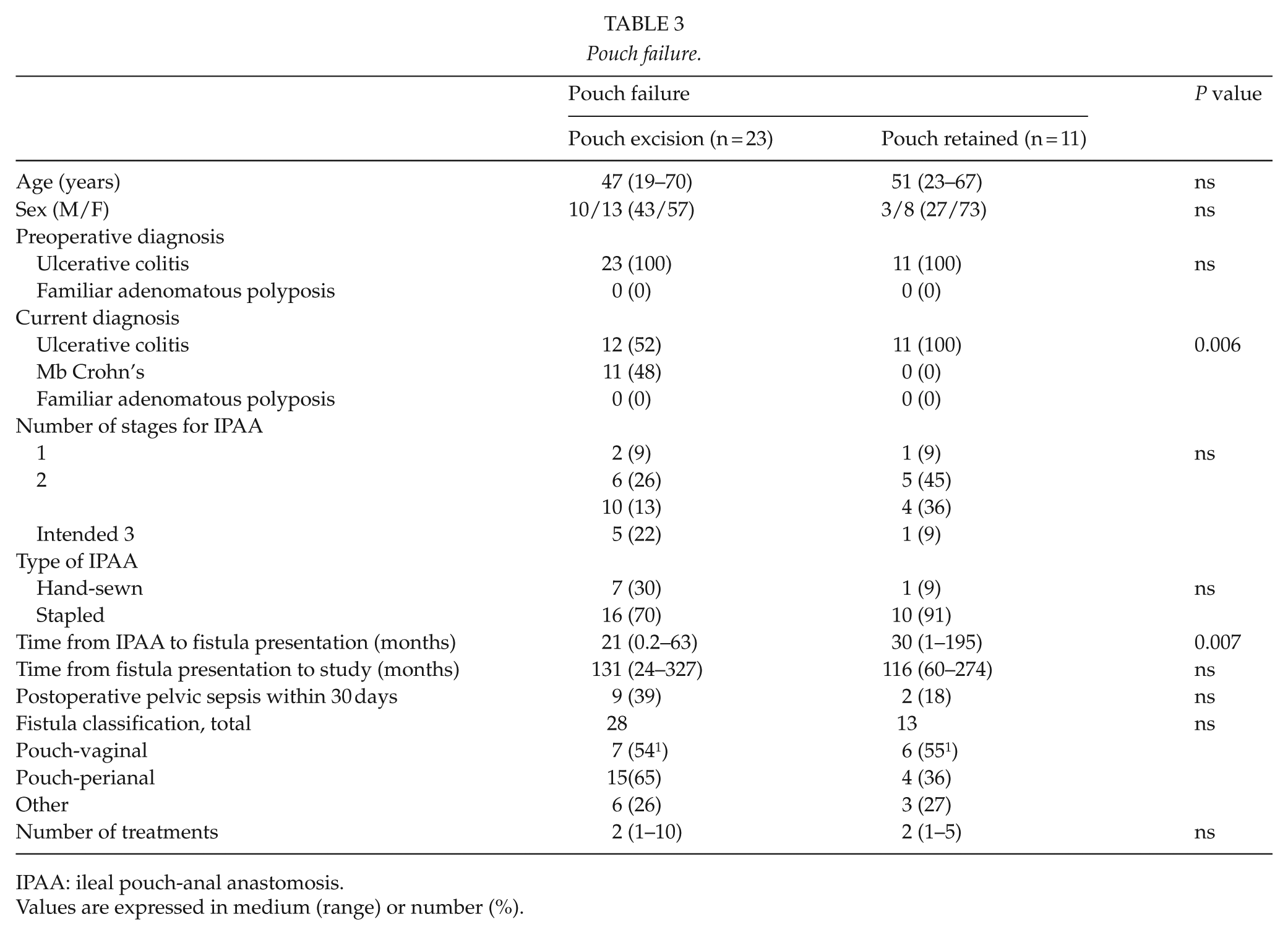
IPAA: ileal pouch-anal anastomosis.
Values are expressed in medium (range) or number (%).
Discussion
The development of pouch-related fistulas following IPAA represents a difficult problem and is an important factor in pouch failure. Pouch failure, defined as a definitive ileostomy with or without pouch excision, was found in 71% of our patients. CD and early fistula development after IPAA were the only significant factors for pouch excision. No relation was found with the anastomotic technique (hand-sewn vs stapled), fecal diversion after IPAA, or septic complications.
The overall prevalence of pouch-related fistulas in the present study was 10.7%, which compared favorably with rates of 2.4%–14% that have been found in other studies (1, 3–5). Mallick et al. (12) reported the development of pouch-vaginal fistula in only 2.9% of 1895 female patients, whereas Wexner et al. (13) found a rate of 6.9% in 304 women and Groom et al. (14) reported a rate of 10.6% in 141 women. In the current study, the fistula rate was 8% among 231 women. Mallick et al. and Groom et al. included both J- and S-shaped pouch designs and reported a hand-sewn anastomosis in 30% and 59%, respectively, in those that developed a fistula. Our study included only patients with a J-shaped pouch design, and a hand-sewn anastomosis was made in 21% of the patients in our study. There are conflicting reports as to whether the type of anastomosis in IPAA predisposes to the development of pouch-vaginal fistulas (12, 14). A prospective randomized trial comparing hand-sewn and stapled anastomosis found no differences (15). The follow-up period was also slightly different; the previously mentioned studies followed patients for 15–27 years, whereas the present study had 31 years of observation.
In the current study, we documented a pouch failure rate of 71% in patients with pouch fistulas. This is higher than the rates reported in previous studies. Gaertner et al. (16) reported a failure rate of 38%, whereas Tekkis et al. (5) only found a failure rate of 21/27% (women/men). There may be several explanations for these differences. Only fistulas arising from the pouch were included in the present study. The uncomplicated fistulas originating from the anal canal without evident involvement of the pouch were excluded, which was not the case in the other studies. In addition, our study has the longest follow-up period, with the latest development of fistulas nearly 18 years after IPAA surgery. Moreover, the definition of pouch failure was different. We included patients with loop ileostomies in the failure group, as the patients had no desire for closure. It is not evident how these cases were handled in the other studies. Finally, our findings may reflect the study location’s role as a referral center for problematic cases.
Numerous methods have been suggested for the repair of pouch-related anal fistulas and no procedure seems to be superior to others, and there are no randomized studies to our knowledge. In the present study, the method used was upon the discretion by the surgeon and the majority of patients underwent two or more different procedure. Our results showed the highest success rate with trans-sphincteric fistulectomy and fistulectomy with advancement flap. It is also worthwhile to mention that fecal diversion alone had a high success rate. However, the numbers of cases are too small to draw any firm conclusions. The present and others’ results call for an international multicenter study.
In the pouch failure group of 34 patients, 23 (68%) of the patients had the pouch excised, implying that 32% still had a non-functioning pouch in situ. The pouch excision is a surgical procedure with a risk of additional complications, such as prolonged wound healing, infection, and the risk of developing new fistulas. In contrast, a maintained reservoir may also cause problems with persistent fistulas, abscesses, and mucus seepage. It is well-established that IPAA patients with CD have a higher risk of pouch failure (17), as do patients who develop pouch-related fistulas (11). This information might be expanded upon by our study, suggesting that the development of CD also increases the risk of pouch excision in IPAA patients with symptomatic fistulas. No fistula patients who had been diagnosed with CD in our cohort retained the pouch, which reflects the complexity of perineal disease in these patients.
In addition, we found the time of fistula onset to be inversely related to pouch excision. This is not supported by Nisar et al.’s (11) findings that found no difference in pouch failure between early- and late-onset fistulas. One explanation might be that many early fistulas develop in patients with postoperative severe and complicated courses involving other intra-abdominal complications; hence, a pouch excision is more favorable in regards to the overall disease improvement. Finally, a pouch excision may appeal more to the patient if the patient develops a fistula prior to the closure of the stoma.
The potential drawbacks of this study relate to the retrospective nature and the sample size. The design is associated with inherent selection and treatment bias, and although the sample size for this study was relatively small, it is consistent with the sample size of other similar studies (8, 16). However, this is the first study that reports the outcome of pouch failure and the factors associated with pouch excision in complicated pouch-related fistulas. In conclusion, the present study demonstrated a high risk of pouch failure in patients with a complicated pouch-related fistula. Furthermore, it showed that CD and early onset fistulas are associated with pouch excision.
Footnotes
Acknowledgements
Secretary assistance from the Department of Surgery, Odense University Hospital, is gratefully acknowledged. The authors’ contributions are as follows: M.D.K. participated in the design, data handling, statistic analysis, and drafting of manuscript. J.K. and N.Q. both participated in the design and data interpretation and critically revised the manuscript. All authors have read and approved the final manuscript. No Ethical Committee approval was needed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
