Abstract
Background and Aims:
Pseudomyxoma peritonei (PMP) is a rare disease characterized by progressive build-up of mucinous deposits inside the abdominal cavity. The aim of this study was to investigate the effect of disease recurrence on overall survival in patients with PMP after cytoreductive surgery (CRS) combined with hyperthermic intraperitoneal chemotherapy (HIPEC).
Methods:
One-hundred thirty-two consecutive PMP patients treated with CRS + HIPEC at Helsinki University Hospital between 2008 and 2017 were included. The impact of clinicopathological and treatment-related characteristics on recurrence and overall survival was evaluated.
Results:
The median follow-up time in the study was 5.04 (range = 0.05–11.60) years. In 121 (91.7%) patients, the disease was classified as low grade and 11 (8.3%) had high-grade disease. In the low-grade group, 26 (21.5%) patients developed a recurrence during follow-up compared to 6 (54.5%) patients in the high-grade group. In the low-grade group, cumulative survival was 98.2%, 91.4%, and 91.4% at 3, 6, and 8 years, respectively. In the high-grade group, cumulative survival was 90.0% and 78.8% at 3 and 6 years, respectively. In patients with recurrent disease, the cumulative survival was 100%, 84.6%, and 84.6% at 3, 6, and 8 years in the low-grade category and 80.0% and 60.0% at 3 and 6 years in the high-grade category, respectively. In the low-grade group, a statistically significant correlation with recurrence but not with overall survival was identified with peritoneal cancer index (PCI), carcinoembryonic antigen (CEA), and the number of affected regions.
Conclusion:
The recurrence of low-grade PMP does not significantly affect overall survival of patients. Disease extent may not be a prognostic indicator after curative CRS and HIPEC in low-grade PMP.
Context and relevance
The effect of recurrence on overall survival of patients with pseudomyxoma peritonei (PMP) has been poorly understood. This article shows that the recurrence does not substantially affect overall survival of patients with low-grade PMP.
Introduction
Pseudomyxoma peritonei (PMP) is a rare condition characterized by progressive build-up of mucinous deposits inside the abdominal cavity. The incidence of the disease is approximately 1–3 per million per year. PMP most frequently originates from a perforated mucinous tumor of the appendix. The clinical presentation is variable and non-specific; many patients are asymptomatic and PMP is found incidentally during cross-sectional imaging or surgery.1,2 Symptoms may include abdominal distension, pain, ascites, suspected acute appendicitis, ovarian mass, or new-onset hernia.3,4
Treatment for PMP previously consisted of repeated surgical debulking operations with a 10-year overall survival (OS) rate of 31%. 5 Standard treatment for PMP currently consists of cytoreductive surgery (CRS) combined with hyperthermic intraperitoneal chemotherapy (HIPEC), a practice pioneered by Sugarbaker. 6 The aim of CRS is to remove visible macroscopic disease from the abdominal cavity and HIPEC is then used to clear any microscopic residual disease. CRS combined with HIPEC for PMP has led to 10-year OS rates of 42%–63%.7–10 There are only a few articles published on PMP recurrence of after curative-intent CRS with HIPEC; the effect of recurrence on OS is not precisely known.
The aim of this study was to determine the risk of PMP recurrence after successful curative-intent CRS with HIPEC and to determine the risk factors for recurrence. In addition, the aim was to determine the outcome of treatment and survival after disease relapse.
Methods
All consecutive 132 patients with PMP that underwent primary CRS combined with HIPEC at Helsinki University Hospital between 2008 and 2017 were included in this retrospective study. In the same period, a total of 55 surgeries were performed for PMP that were either palliative or HIPEC treatment was not given despite intention. The reasons why some patients did not receive HIPEC are shown in Figure 1.
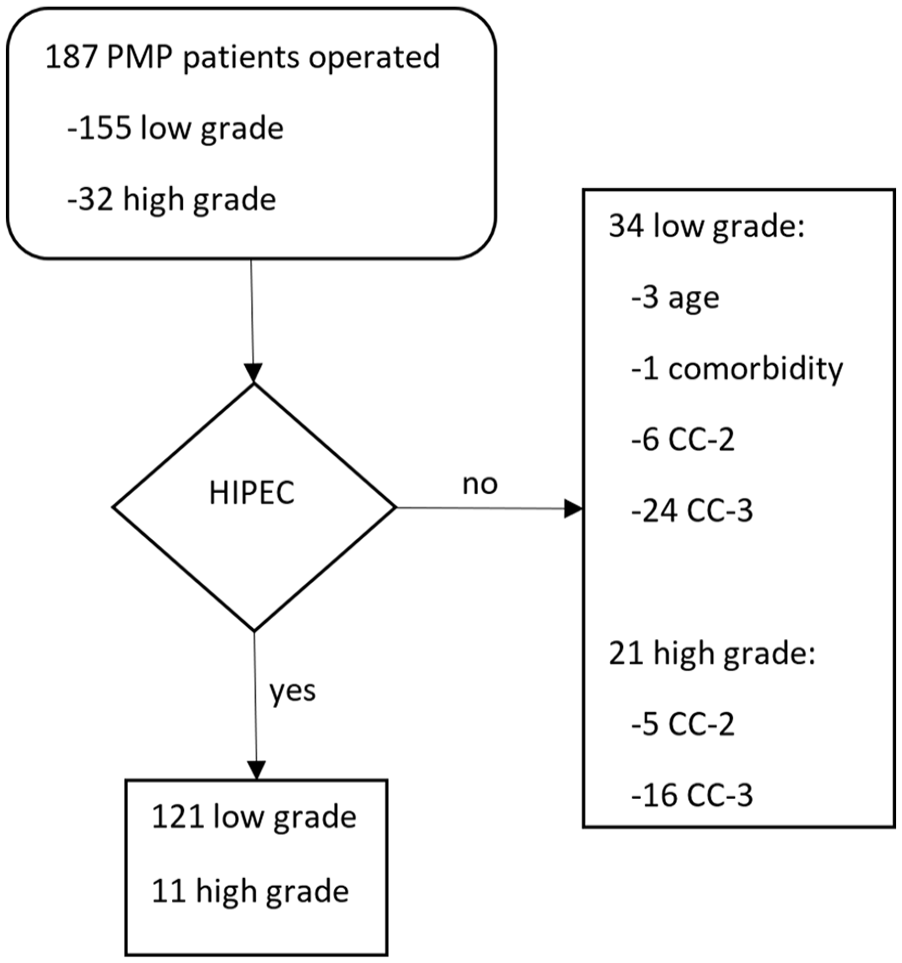
A flowchart showing all patients operated for PMP during the study period. A total of 55 patients were excluded from HIPEC based on the criteria shown on the right.
HIPEC surgeries at our center were started in 2008. The year 2017 was chosen as the end point of inclusion period to insure at least a 5-year follow-up time of all included patients. Patients who received a palliative debulking procedure only were excluded. None of the patients included had undergone a previous debulking or CRS + HIPEC. Patient data were monitored until either patient death or March 2020. A total of seven surgeons acted as main operator, but four of them carried out the majority of the surgeries performed.
CRS with HIPEC (mitomycin C 30 mg/m2 at 40 °C–43 °C for 90 min) was performed as previously described using a semi-closed technique.11,12 The peritoneal cancer index (PCI) score was assessed intraoperatively before cytoreduction. The completeness of cytoreduction (CC) score was determined prior to HIPEC. 13 CC-0 indicates that no visible residual tumor exists, while CC-1 indicates that tumors < 0.25 cm remained; both were considered as successful cytoreduction and macroscopically curative surgery. Scores CC-2 (tumors between 0.25 and 2.5 cm) and CC-3 (tumors > 2.5 cm) were considered as palliative surgery; these patients did not receive HIPEC and were excluded from the study.
Demographic data, preoperative tumor markers (carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9)), American Society of Anesthesiologists (ASA) score, Charlson Comorbidity Index (CCI), 14 surgery duration, intraoperative PCI and CC score, tumor grade according to World Health Organization (WHO) 10 classification, 15 blood transfusion requirement, and length of hospital stay were recorded. All samples were examined by a pathologist familiar with PMP and only patients with mucosal dysplasia of the appendix were included in the study. Patients with acellular mucin only were excluded.
The follow-up protocol included clinical examination, tumor markers, and CT scan (6 and 12 months after the primary operation, then annually up to 5 years, and thereafter every second year). In case of a recurrence, the site of the recurrence and subsequent treatment (surgery, chemotherapy, or palliative treatment) were recorded. In the event of death, the cause of death was categorized as either due to the underlying disease or another cause. This information was retrieved from medical records, or if unavailable, a death certificate was ordered from the Finnish institute for health and welfare. The study was approved by the institutional review board. The requirement for written informed consent was waived due to the retrospective character of this study.
Chi-square and Fisher’s exact tests were used to compare categorical variables. Medians with ranges were used to express continuous parametric variables. Continuous variables with non-normal distribution were compared using the Mann–Whitney U-test. p < 0.05 was considered statistically significant. Patients with missing values were excluded from analyses of that particular variable. For the cumulative survival and recurrence risk analysis, the Kaplan–Meier method was used and comparison was performed with the log-rank test. Cox regression analysis was used to identify factors associated with recurrence. Factors with the p < 0.1 in univariate analysis were included in multivariate analysis. Time to recurrence was computed from the date of first CRS with HIPEC operation to the date when recurrence was detected. OS was calculated from the date of first CRS with HIPEC to the date of the last follow-up or death. Statistical analysis was performed using IBM® SPSS® Statistics version 25.
Results
A total of 132 patients underwent CRS with HIPEC for PMP during the study period. Altogether 98 (74.2%) patients were female. The disease was classified as low grade in 121 (91.7%) patients and high grade in 11 (8.3%) patients. Median ASA score was 2 (1–4), and CCI was 7 (2–11). Median duration of the operation was 497 min (245–885 min). Sixty-six (50.0%) patients required intraoperative blood transfusion. Median postoperative time in intensive care unit (ICU) was 26.5 (16–518) h and length of stay in our HIPEC center was 15 (9–71) days. After this, 112 patients were discharged home, and 20 patients were transferred to another institution for further rehabilitation. Patient characteristics and perioperative features are presented in Tables 1 and 2, respectively. The overview of postoperative complications, their distribution according to the Clavien–Dindo classification, and surgical interventions are shown in Table 3. A total of 18 patients needed at least one surgical reintervention due to a suspected complication.
Baseline characteristics of patients who underwent CRS + HIPEC for PMP.
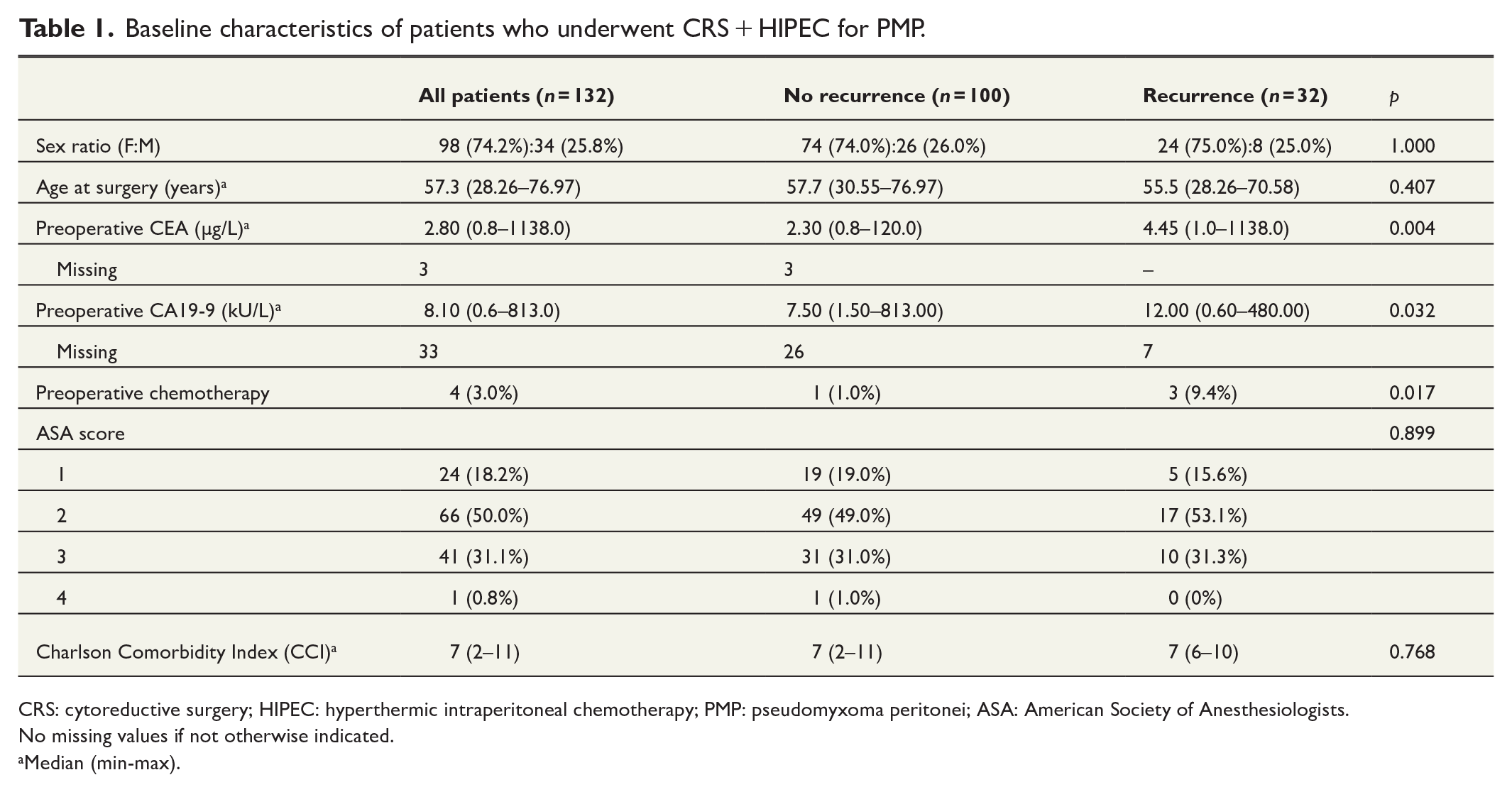
CRS: cytoreductive surgery; HIPEC: hyperthermic intraperitoneal chemotherapy; PMP: pseudomyxoma peritonei; ASA: American Society of Anesthesiologists.
No missing values if not otherwise indicated.
Median (min-max).
Perioperative features of patients who underwent CRS + HIPEC for PMP.
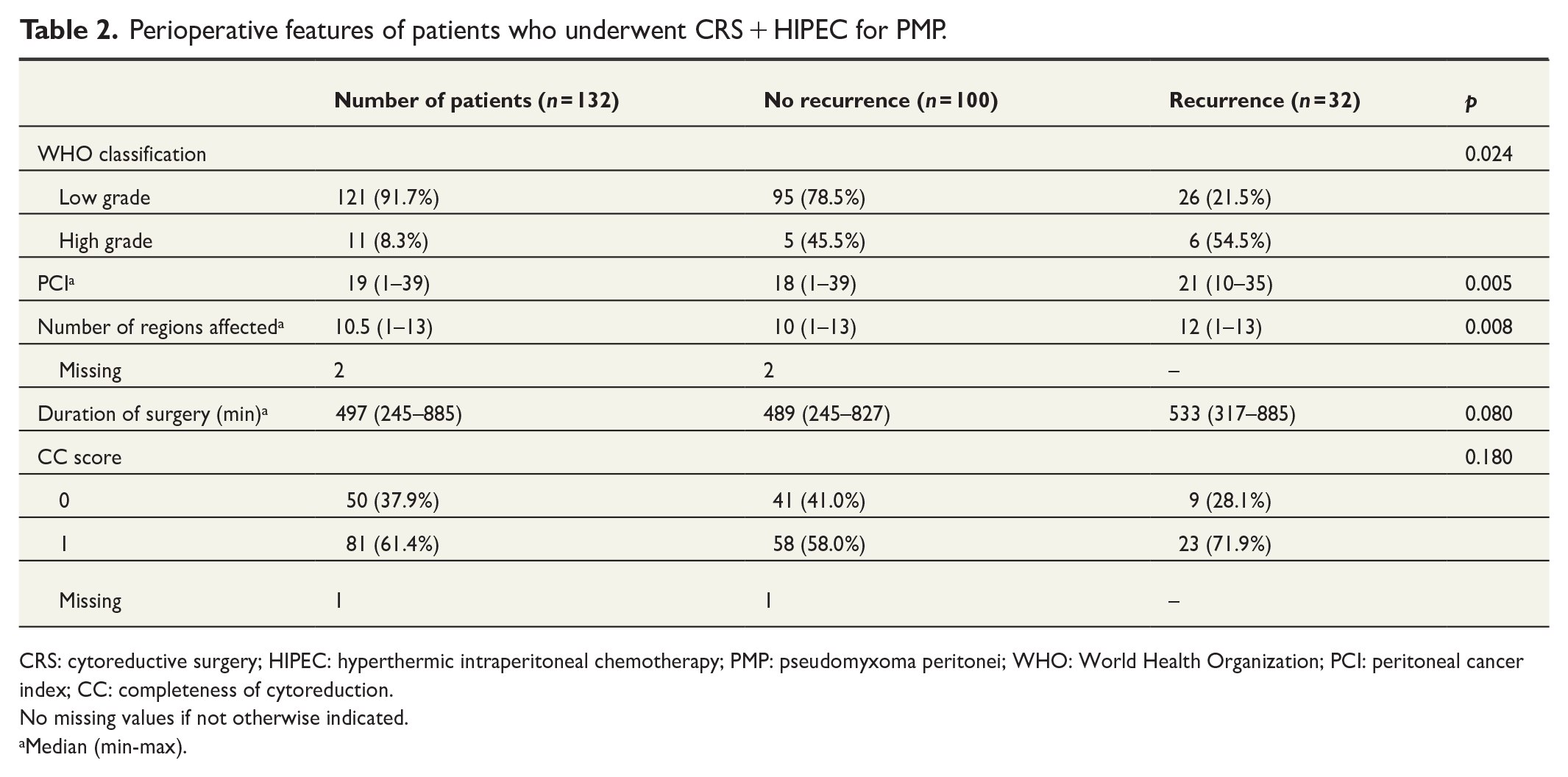
CRS: cytoreductive surgery; HIPEC: hyperthermic intraperitoneal chemotherapy; PMP: pseudomyxoma peritonei; WHO: World Health Organization; PCI: peritoneal cancer index; CC: completeness of cytoreduction.
No missing values if not otherwise indicated.
Median (min-max).
The summary of complications, their distribution according to Clavien–Dindo classification, and surgical reinterventions after CRS + HIPEC for PMP.
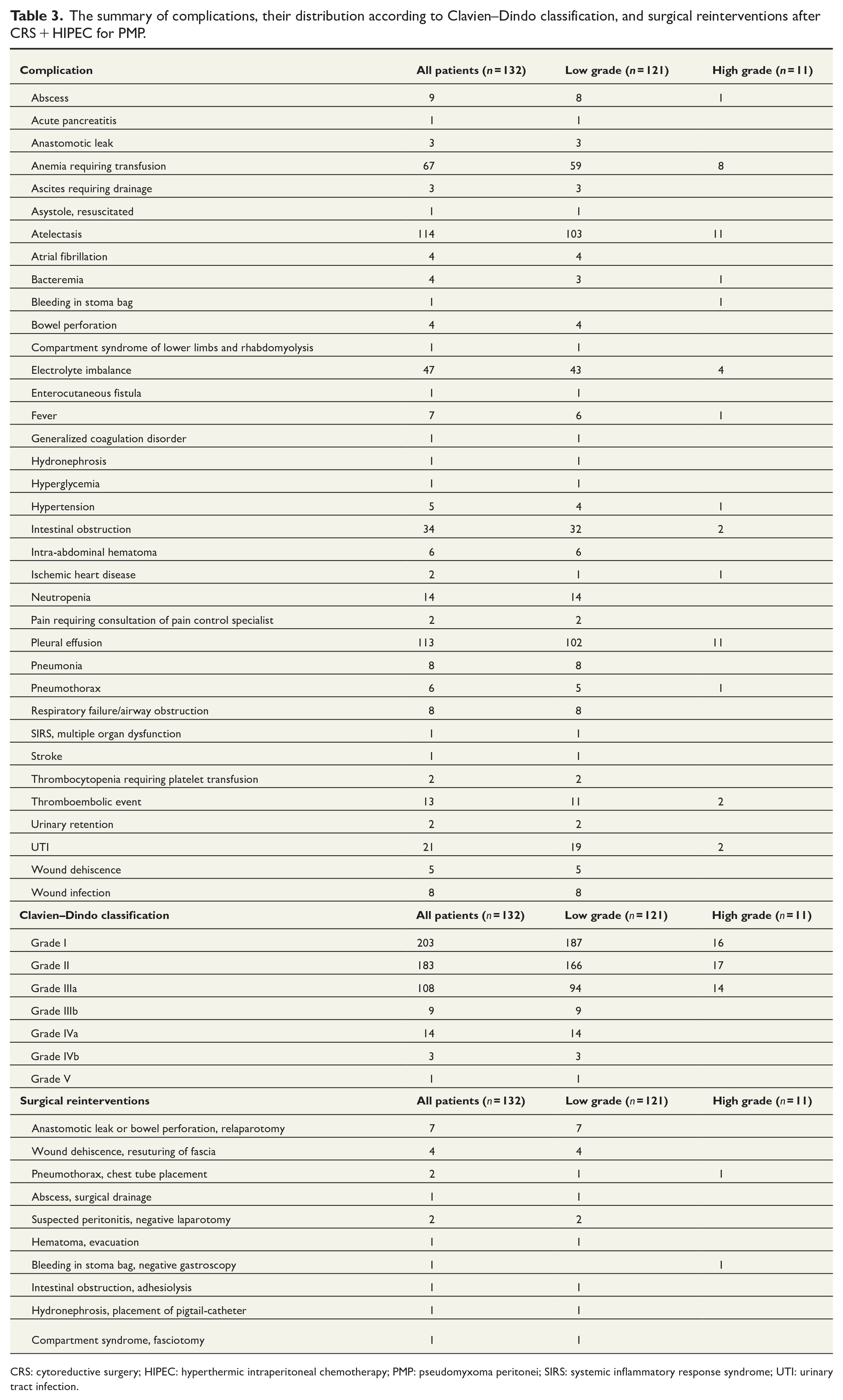
CRS: cytoreductive surgery; HIPEC: hyperthermic intraperitoneal chemotherapy; PMP: pseudomyxoma peritonei; SIRS: systemic inflammatory response syndrome; UTI: urinary tract infection.
Median follow-up time in the study was 5.04 (range = 0.05–11.60) years. A total of 32 (24.2%) patients developed a recurrence during follow-up; median time to recurrence was 2.96 (range = 0.35–9.23) years. Cumulative risk of recurrence was 12.0% and 19.9% in the low-grade group and 18.2% and 48.9% in the high-grade group at 3 and 5 years, respectively (Figure 2).
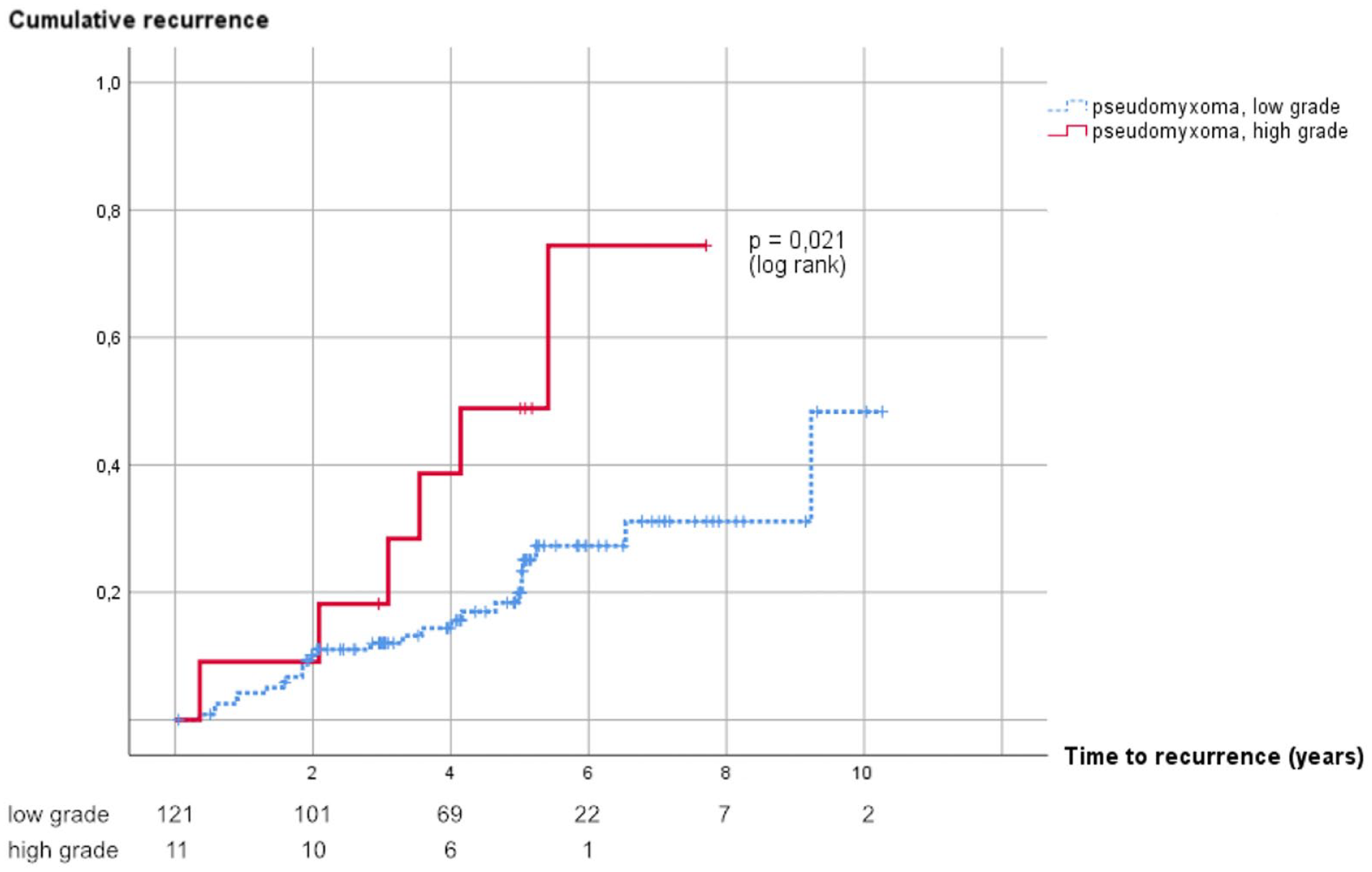
Cumulative risk of recurrence in patients with low- and high-grade pseudomyxoma peritonei.
Recurrence was detected by routine follow-up in 28 (87.5%) patients and in an additional imaging study based on symptoms in 4 (12.5%) patients. Of 121 patients with low-grade disease, 26 (21.5%) had recurrence and of the 11 patients with high-grade disease, 6 (54.5%) had recurrence. The site of 32 recurrences was abdominal cavity in 27 (84.4%) patients (one of which had additional pulmonary metastases), pleural cavity in 2 (6.3%) patients (both of which had a diaphragmatic lesion in the primary operation), and 3 (9.4%) patients had pulmonary metastases. Recurrence of the disease was confirmed by histopathology in all patients operated on for recurrence and in four additional patients. In the remaining patients with recurrence, the radiological finding was very clear. Twenty-two (68.8%) patients received surgery for the recurrence; 5 had re-CRS with HIPEC, 13 re-CRS without HIPEC, and 2 patients received palliative surgery only. For the patients with pleural cavity recurrence, both received hyperthermic intrathoracic chemotherapy (HITOC). Seventeen (77.3%) of these 22 surgeries performed due to disease recurrence were considered macroscopically curative at the time of surgery and 53.1% did not experience a re-recurrence during the follow-up.
Ten (7.6%) patients died during follow-up, median time to death was 4.13 (range = 0.05–11.6) years after the primary HIPEC surgery. Five (3.8%) patients died due to recurrent pseudomyxoma (median time to death was 4.12 years after primary HIPEC surgery; range = 2.12–8.62). Two patients died because of an HIPEC complication; one on the 18th postoperative day due to worsening dyspnoea and generalized coagulation disorder despite intensive care. The other patient died over 8 years postoperatively due to a persistent enterocutaneous fistula leading to long-term parenteral nutrition and eventually to liver failure. Two of the three remaining patients died of myocardial infarction and one of primary gastric cancer.
The cumulative OS was 97.5%, 90.2%, and 90.2% at 3, 6, and 8 years. The cumulative survival was 98.2%, 91.4%, and 91.4% at 3, 6, and 8 years in the low-grade group and 90.0% and 78.8% at 3 and 6 years in the high-grade group, respectively (Figure 3).
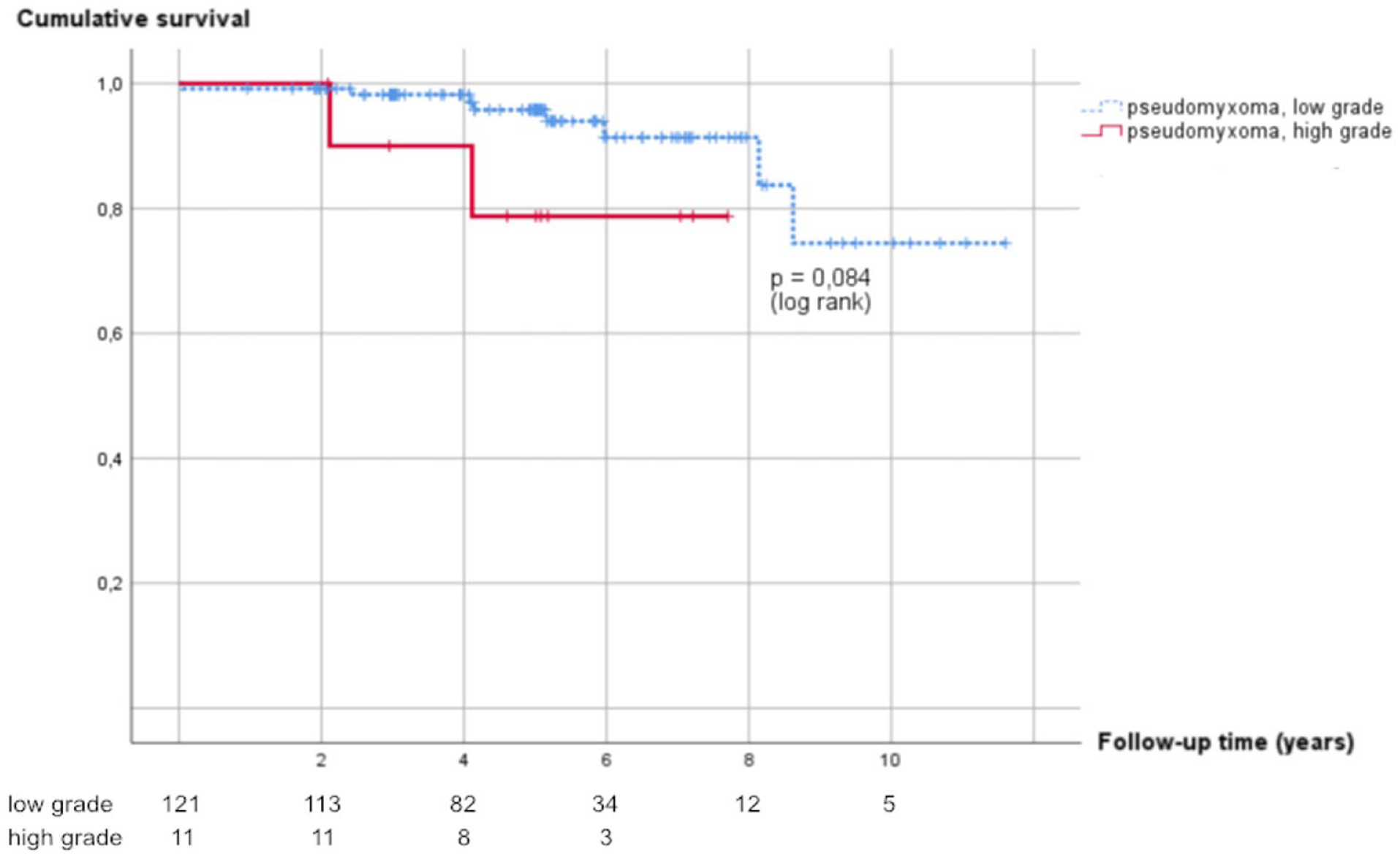
Kaplan–Meier curve for overall survival of patients with low- and high-grade pseudomyxoma peritonei treated with CRS + HIPEC.
Among patients with recurrence, the cumulative OS in the low-grade group was 100%, 84.6%, and 84.6% at 3, 6, and 8 years, respectively (Figure 4), and in the high-grade group, it was 80.0% and 60.0% at 3 and 6 years, respectively.
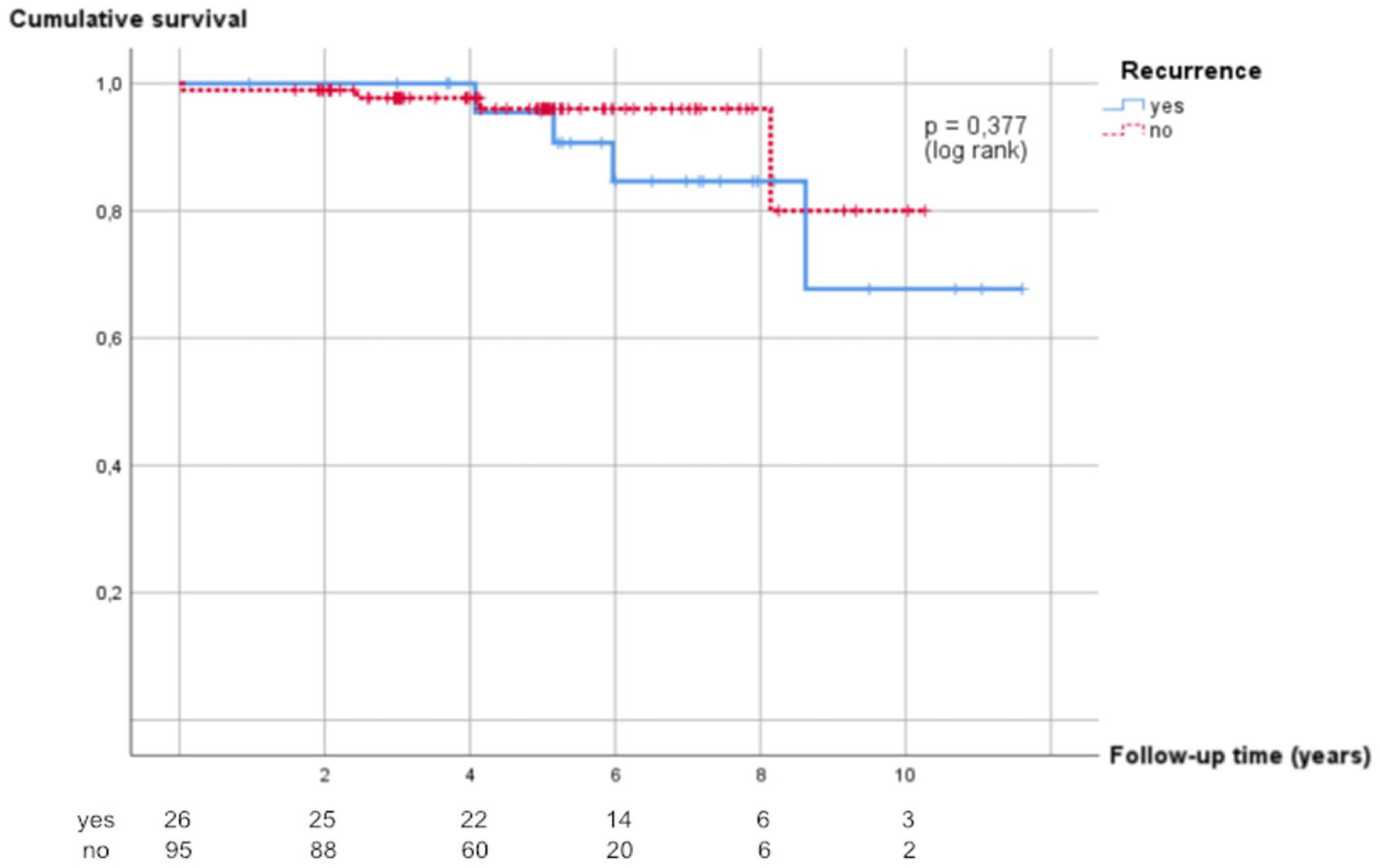
Kaplan-Meier curve for overall survival of patients with recurrent and non-recurrent low-grade pseudomyxoma peritonei.
In univariate analysis, PCI (p = 0.023), number of affected regions (p = 0.023), and CEA (p = 0.000) were identified as statistically significant risk factors for recurrence in the whole cohort of patients (Table 4). These variables were chosen for multivariate analysis. As PCI and number of affected regions are not independent of each other but more or less describe the same thing, we chose to perform multivariate analysis twice; in one analysis, PCI was excluded and in the other the number of affected regions. In the multivariate analysis, all these variables reached statistical significance. In low-grade patients only, PCI (p = 0.029), number of affected regions (p = 0.040), and CEA (p = 0.030) reached statistical significance for disease recurrence. In the high-grade group, none of the tested variables reached statistical significance. In univariate analysis with the same variables, no risk factors for death could be identified, both in the whole cohort of patients and among low-grade patients.
Univariate and multivariate analysis of risk factors for recurrence of pseudomyxoma peritonei.
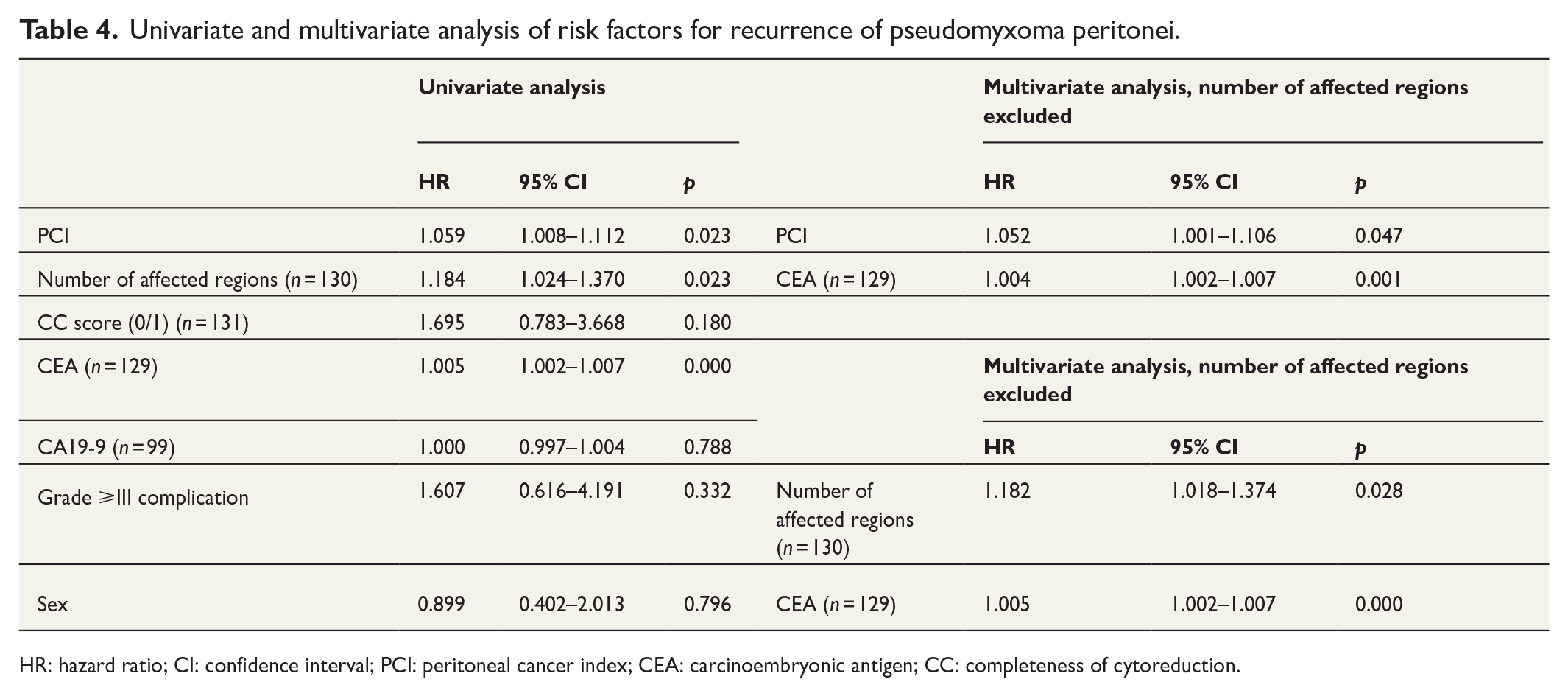
HR: hazard ratio; CI: confidence interval; PCI: peritoneal cancer index; CEA: carcinoembryonic antigen; CC: completeness of cytoreduction.
Discussion
A total of 132 consecutive PMP patients who underwent CRS + HIPEC at Helsinki University hospital between years 2008 and 2017 were included in this retrospective cohort study. The overall risk of PMP recurrence was 24.2% in the study population. The risk of recurrence was more than twice as high in the high-grade group than in the low-grade group. The number of regions affected, PCI score, and serum CEA level correlated with the risk of recurrence. However, in the short term, the survival of patients with recurrence did not differ from those without recurrence.
The total recurrence rate was consistent with previously published results (24.2–37.6%).16,17 The median time from surgery to recurrence was nearly the same as in previous studies (25–26.3 months);18,19 thus, most of the recurrences seem to occur in the 3 first years after surgery. More than half (54.5%) of our patients in the high-grade group developed a recurrence during follow-up. High-grade histology has previously been identified as a significant factor in early disease recurrence. 16 We could not compare the time to recurrence between low- and high-grade groups due to the low number of high-grade patients in our cohort. Recurrences outside the intra-abdominal cavity were extremely rare; over 80% of recurrences were intra-abdominal and mostly peritoneal, as previously reported. 16
PCI and the number of affected areas reflecting disease extent were identified as risk factors for recurrence among low-grade patients. Previously, PCI > 24 was considered as a risk factor for early recurrence. 16 In this study, CEA was also a risk factor for recurrence in the low-grade but not in the high-grade group. Previously, it has been shown that some patients with aggressive high-grade pseudomyxoma have non-polarized immunostaining of CEA and normal serum CEA levels, suggesting that serum CEA is probably not a good indicator for recurrence and death in these patients. 20 High-preoperative CEA predicts early recurrence and poorer survival in PMP.21,22 There was no difference in risk of recurrence between CC-0 and CC-1, supporting the principal idea of HIPEC being effective against PMP deposits of <0.25 cm in diameter.
The overall survival in the whole study group was excellent. The cumulative survival at 8 years was as high as 90%, following the previously published results of 5-year survival rates of 71%–80%.8,10 The exceptionally high survival is partly explained by the low number of high-grade patients in our cohort, as high-grade histology seemed to predict poorer survival even though the difference did not reach statistical significance. The survival of patients with high-grade histology after CRS has been reported in other series to vary from 36 to 48 months with 5-year survival rates of 59%–76%.7,8,10,23 The impact of high-grade histology on survival has also been observed in PMP independent of therapy. 11 Interestingly, neither PCI nor the number of affected regions had an effect on survival after curative CRS and HIPEC in our series. Previously, contradictory results on the effect of PCI on survival have been published; some studies revealed a lower survival with PCI > 24 or >28 and some suggested that high PCI may not be a prognostic sign after successful surgery.7,10,24 At a minimum, curative surgery is worth pursuing in PMP even when extensive disease is present, in contrast to colorectal cancer (CRC) carcinosis.25–28 The recurrence of low-grade PMP after CRS and HIPEC did not affect survival. This could not be evaluated in high-grade patients because of the low number of patients. Considering the previous reports of 55%–67% survival at 5 years and 31%–35% after serial debulking, CRS with HIPEC seems superior to bulkectomy even in cases with later recurrence.5,29 Curative surgery was attempted in 68.8% of patients with recurrence and was successful in 53.1%. High rates of curative re-surgery have also been reported by others. 18 In case of a recurrence, curative-intent treatment combining CRS and intraperitoneal or intrathoracic chemotherapy has been possible in 76% of patients, leading to a 5-year OS of 83% after the treatment of the recurrence. 18 This contrasts with 27% for patients treated with non-curative therapy. 18 In another recently published large study, patients who underwent repeat CRS had 5-year OS of 78.1%. 17
The number of PMP patients operated annually in our center has increased since 2008 which is due to the fact that HIPEC treatments were started in Helsinki first in Finland. Over time, more effort has been put into the radiological diagnostics of the disease. It is also possible that the disease can be suspected more often than before. Patients probably benefit from early treatment, as recurrence is more common in more widespread disease.
Our study has several limitations. The study design was retrospective, and there were a low number of high-grade PMP patients in our cohort. From such a small number of patients, no unambiguous conclusions could be made about the best course of treatment in the event of recurrence. Our results cannot be directly compared to previous results of serial debulking without HIPEC because only patients with CC-0 and CC-1 were included in our study. Also, inclusion of patients on the learning curve in our series may have some influence on the results.
In conclusion, even though approximately 25% of patients with PMP will have a recurrence after CRS with HIPEC, this does not seem to affect survival in patients with low-grade disease. Curative-intent CRS and HIPEC is likely to slow the recurrence of the disease and prolong survival even in patients who eventually relapse. Patients can die from the disease even after long time, so recurrence of the disease may affect survival over a longer follow-up period. Disease extent may not be a prognostic indicator in low-grade PMP after curative CRS and HIPEC.
Footnotes
Author contributions
A.L. contributed to the study concepts. A.L., L.K., and A.Y. contributed to the study design. All authors contributed to the data acquisition. L.K. and A.Y. contributed to the quality control, interpretation, and statistical analysis of data. A.L., L.K., and A.Y. contributed to the preparation of the manuscript. A.L., L.K., and C.H. contributed to the editing and reviewing of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.Y. has nothing to declare. L.K. declares grants from Mary and Georg Ehrnrooth’s Foundation and Cancer Foundation Finland (Syöpäsäätiö), outside the submitted work. C.H. and A.L. have nothing to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
