Abstract
Background & objective:
Major abdominal surgery morbidity can reach 50%. Prehabilitation has shown promising results in decreasing complications. However, it is unknown if prehabilitation can have a positive effect specifically after major abdominal surgery. The goal of this study was to evaluate the feasibility and safety of a prehabilitation program before major abdominal surgery.
Methods:
All patients evaluated for major abdominal surgery between February and April 2018 were eligible. A 4-week trimodal prehabilitation program combining physical therapy, nutritional support and psychological preparation was set up.
Results:
Among 106 patients evaluated for major abdominal surgery during the study period, 60 were included in the prehabilitation program. No cardiovascular events occurred during prehabilitation. The 6-min walking distance increased significantly (+45 m, increase of 9.3%, p = 0.008) after prehabilitation (and before the operation). Anxiety, depression, and several quality of life (QoL) items improved. Postoperative 90-day mortality and morbidity were 3.4% and 48%, respectively. Median hospital length of stay, and intensive care unit length of stay were 14 and 6 days, respectively. For 19 patients readmitted, the treatment was medical, radiological, or surgical, for 11, 5, and 3 patients, respectively.
Conclusions:
Prehabilitation before major abdominal surgery is feasible, safe, and improve patients’ functional reserves, QoL, and psychological status.
Introduction
Abdominal surgery can lead to significant morbidity. Despite the progress in perioperative management and surgical technique leading to a decrease of non-surgical complications, the rate of complications requiring further surgery have remained stable.1,2 Moreover, poor preoperative physical and/or nutritional status have been shown to be associated with increased morbidity, mortality, postoperative recovery time, and care-related costs.3,4 To reduce postoperative complications, better patient selection and better preoperative patient preparation are necessary.
Standardized clinical pathways have improved quality of care. 5 Recently, enhanced recovery after surgery (ERAS), thanks to postoperative management protocols, has reduced the medical complication rate, length of stay and cost of major abdominal procedures, while improving patient satisfaction.6 –8 Another clinical pathway, “prehabilitation” before surgery, was initially developed to reduce the risk of postoperative complications following cardiovascular surgery. 9 This concept combined a trimodal preparation: nutritional, physical, and psychological preparation, and aimed at increasing patients’ functional reserves. 10 The two protocols “prehabilitation” and ERAS are probably synergistic to propose an optimization of the perioperative course of patients. It is currently unknown if prehabilitation can have a positive effect after major abdominal surgery. Overtime, patients considered for major abdominal surgery increased in age and comorbidities. 1 To better prepare such frailly patients we wanted to optimize their preoperative status. The concept of prehabilitation, increasing functional reserve preoperatively, appeared as an interesting way to improve patient’s outcomes. For frailly patients considered for major surgery, prehabilitation program have to demonstrate their safety and feasibility.
This is a pilot study evaluating the feasibility, safety and effectiveness of a trimodal prehabilitation program before major abdominal surgery.
Methods
A prospective single center pilot study was performed to evaluate the feasibility and safety of a trimodal prehabilitation protocol before major abdominal surgery. The Lyon Sud hospital is a multidisciplinary university tertiary center, which takes care of patients of general and oncologic surgery. This study was approved by the local ethics committee (Ref: Rech_FRCH_2018). Of note, the prehabilitation protocol was an addition to our existing ERAS protocol.
Population
All consecutive patients who had a preoperative visit for major abdominal surgery between February 12 and May 25, 2018 were identified. The protocol was started once the operative date had been fixed. Major surgery was defined as:
cytoreductive surgery for peritoneal metastasis,
low anterior resection of the rectum,
resection > 3 liver segments,
giant ventral hernia repair (defined as a hernia larger than 10 cm in diameter with loss of domain), 11
Ivor Lewis esophagectomy,
total or subtotal gastrectomy,
Whipple or distal pancreatectomy.
The inclusion criteria for the prehabilitation program were as follows:
age > 18 years old,
medical clearance for exercise,
the possibility of participating in the prehabilitation program for a minimum of 4 weeks prior to the operation,
major surgery performed during the study period (March 14–July 16, 2018).
Participants provided informed written consent to participate in the study.
Procedures
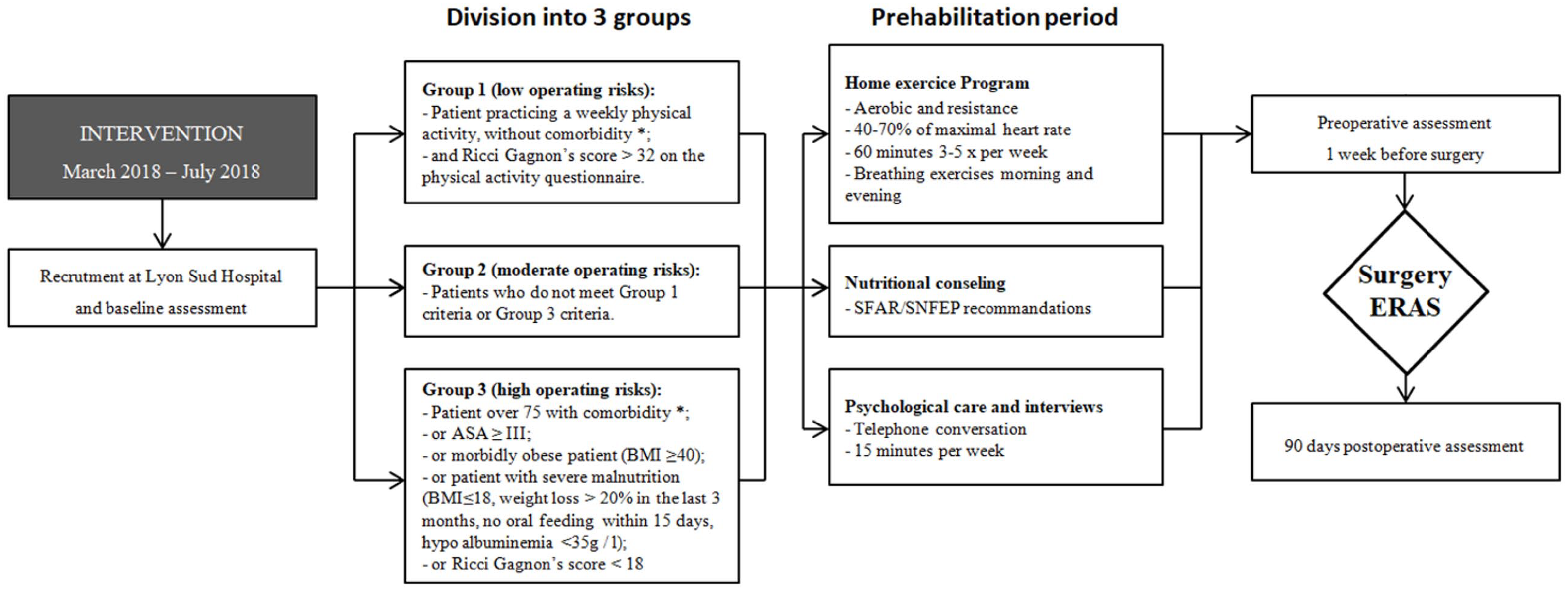
The prehabilitation program included (Fig. 1) the following:
an initial assessment (pre-therapeutic),
trimodal prehabilitation: physical, nutritional and psychological preparation (Fig. 2),
early postoperative rehabilitation according to the principles put forward by GRACE (Groupe de Réhabilitation Améliorée après Chirurgie). 12
Flow diagram showing timing of patient assessments and interventions.

Prehabilitation protocol.
The research coordinator contacted the patients for initial assessment, prior to the preoperative visit. During the initial visit, once the study had been explained and consent had been obtained, a physical activity questionnaire (Ricci Gagnon) was completed with the aid of the coordinator. Patients’ functional reserve was assessed with a 6-min walk test (6MWT) 13 and they were divided into three groups based on their abilities to exercise, during that same visit:
Group 1: physically fit patients;
Group 3: patients with major comorbidities and limited ability to exercise;
Group 2: patients who did not meet group 1 or 3 criteria (Fig. 1).
Following this assessment, each patient participated in a prehabilitation program tailored to his or her reserve for a minimum of 4 weeks. We chose a minimum of 4 weeks of prehabilitation based on a previous study suggesting at least such a period to improve functional capacity. 14
Patients received a teaching booklet with a logbook; the quality of life questionnaire (QLQ-c30) and HADS: Hospital Anxiety and Depression Scale (HADS) questionnaire; physical therapy prescriptions as necessary; and an incentive spirometer (Voldyne®). All oncological patients without dysphagia received immunonutrition of Impact Oral® (Nestle®), a 237 mL brick three times days, 1 week before surgery. A second assessment would take place during the week preceding the operation.
All patients were followed for 90 days after surgery. All complications were recorded and graded according to the National Cancer Institute–Common Terminology Criteria for Adverse Events (NCI–CTCAE) classification 4.0. 15
Outcomes
The primary outcomes of the study were as follows:
feasibility of prehabilitation before major abdominal surgery as evidenced by the proportion of the enrolled patients who completed the program,
efficacy of prehabilitation as evidenced by the change in the 6-min walk test (6MWT),
safety of prehabilitation as evidenced preoperative adverse events, postoperative complications and mortality up to postoperative day 90, readmission rate, treatment at readmission.
Secondary outcomes included the following:
quality of life (QoL) evaluation as per the QLQ-c30 questionnaire,
depression and anxiety evaluation according to the HADS questionnaire,
hospital length of stay and intensive care unit (ICU) length of stay.
Statistical analysis
The sample estimate was based on the inclusion capacity of the project coordinator which was five patients per week, corresponding to approximately 60 patients over 3 months.
The scores for the different items of the European Organization for the Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-c30) questionnaire were calculated according to the EORTC recommendation manual. 16
We did not assume any underlying distribution and treated all continuous data as non-parametric; Mood’s median test was used for independent observations and the Sign test for paired observations. Summative statistics used were of the form “median (median absolute deviation (MAD)).” The only exceptions were the pre- and post-6MWT distance: their means as well as the difference of their means and its standard deviation are also reported in the “Results” section for ease of interpretation.
For nominal categorical variables, we used Fisher’s exact test. For ordinal categorical variables, we used the Cochran–Armitage test for trend. For all categorical variables, the summative statistics used were of the form “number of events (percentage of the corresponding group).”
All analyses were carried out with Rv4.0.5 (R Core Team, Austria, Vienna); the packages used were: arsenal (tableby/paired function) to produce the tables and survminer to perform the time-to-event analysis. For null hypothesis testing, a p-value less than 0.05 was considered statistically significant.
Results
A total of 106 patients had a preoperative visit for major abdominal surgery and met the inclusion criteria. Nine patients were not prospectively screened for prehabilitation, and were not included in the study. Of those, 37 were excluded (specific reasons shown in Fig. 3). We offered inclusion to all remaining 60 patients. No patient refused to participate in the study. Of note, one patient was reluctant to complete the physical preparation due to a lack of motivation. However, that patient quit smoking and followed up with the nutritional evaluation and psychological support. We included that patient in our final analysis.
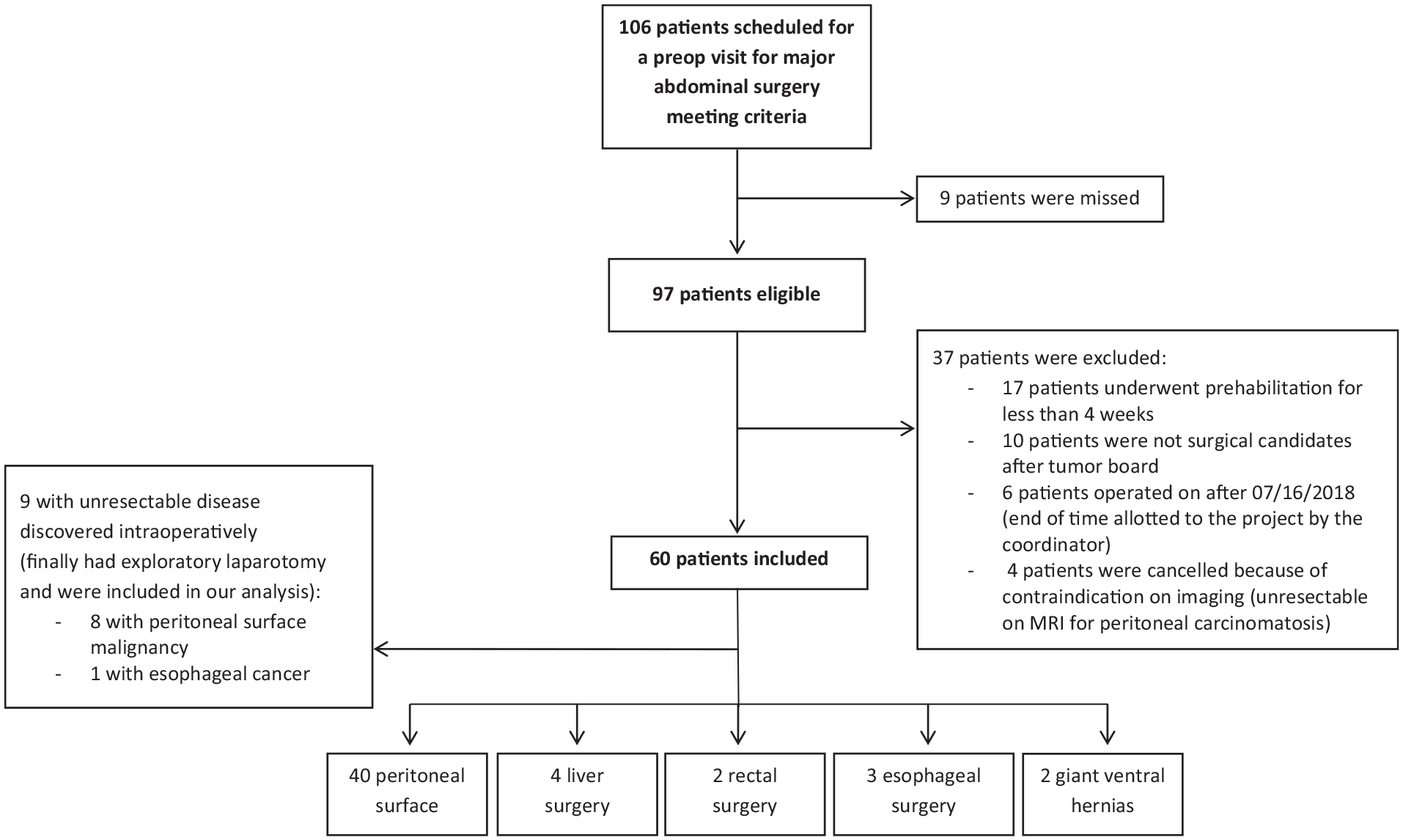
Flow-chart.
Of the 60 patients, 34 were women and 26 men with a median age of 62.5 years (19–81 years). Seven patients (11.7%) were included in group 1, 25 patients (41.7%) in group 2, and 28 (46.6%) in group 3 (Table 1). The median body mass index (BMI) at baseline was 24 kg/m2 (MAD 3). Older patients with higher American Society of Anesthesiology (ASA) scores were included in group 3 when compared to the other two groups and in group 2 compared to group 1. There was no difference between the groups regarding the distance recorded during the initial 6MWT (p = 0.087). There was no significant difference regarding sex (p = 0.118), pathology (p = 0.528), or type of surgery (p = 0.687).
Measures of exercise capacity, quality of life, anxiety, and depression before and after prehabilitation.
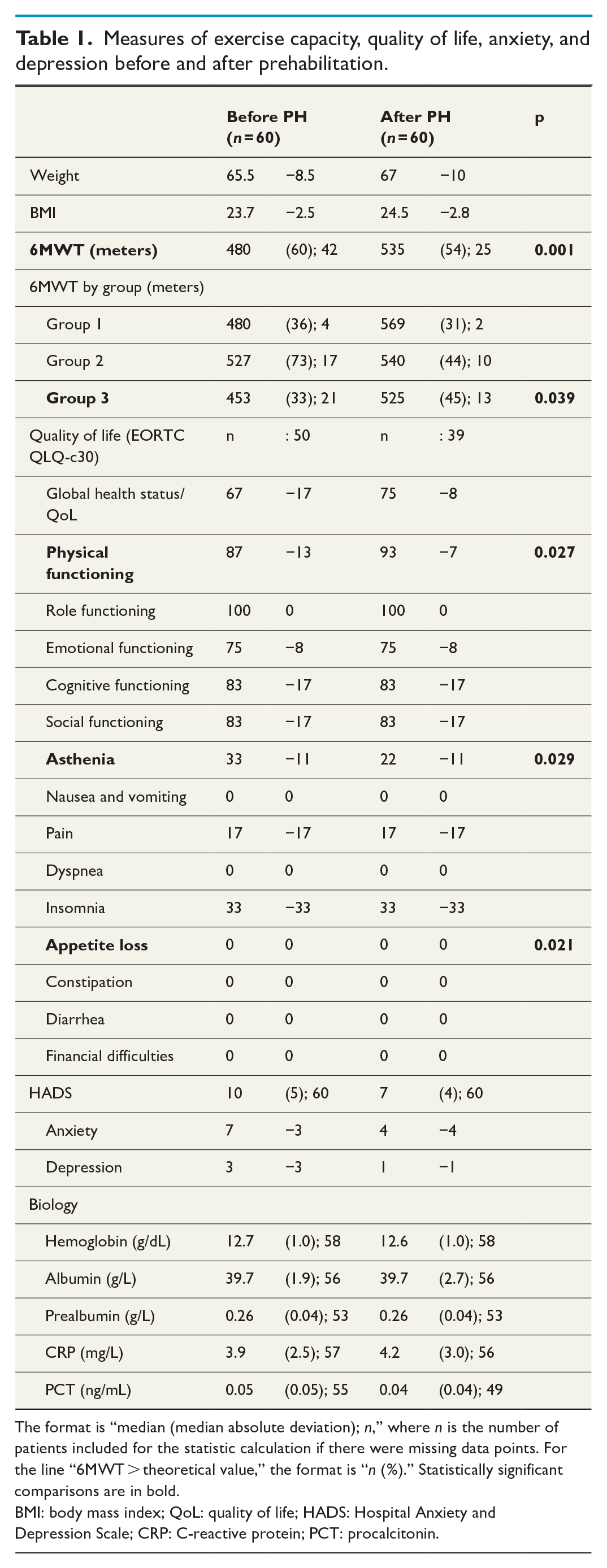
The format is “median (median absolute deviation); n,” where n is the number of patients included for the statistic calculation if there were missing data points. For the line “6MWT > theoretical value,” the format is “n (%).” Statistically significant comparisons are in bold.
BMI: body mass index; QoL: quality of life; HADS: Hospital Anxiety and Depression Scale; CRP: C-reactive protein; PCT: procalcitonin.
Median duration of prehabilitation was 43.5 days (MAD: 11 days, range: 30–119 days). Forty percent of patients underwent prehabilitation for more than 45 days. Seventy percent of patients were followed by a physical therapist. Patients had a median of eight physical therapy sessions during their preparation (MAD: 5.9, range: 5–19).
Regarding nutrition, 87% of the patients received preoperative immunonutrition. Nine patients were malnourished preoperatively and were referred to the nutrition unit for nutritional status optimization. Of those nine patients, seven required oral nutrition only and two required parenteral nutrition. The majority of patients were non-smokers (82%). Among the 11 active smokers, two accepted to make an appointment with a tobacco cessation specialist during the prehabilitation period, but none quit smoking.
During the prehabilitation program, each patient received a median of 3.5 phone calls (MAD 0.7, range: 1–8 times per patient). Ten percent of the calls were not answered (voice messages were left).
No cardiovascular event or trauma was reported during prehabilitation. Five adverse events were reported: bowel obstruction on a peritoneal carcinomatosis nodule, abdominal shingles, port infection, pneumonia, and non-traumatic vasovagal episode. Psychologically, a patient presented an anxiety attack before the intervention for fear of not being sufficiently prepared. No patients withdrew consent or were lost to follow-up during the study.
Table 1 shows the comparison before and after prehabilitation for the 6MWT, QoL, and anxiety score and laboratory values. During prehabilitation, 6MWT increased from an average of 484–529 m (+45 m, +9.3%, SD = 76 m, p = 0.001). In the subgroup analysis, it was only significantly increased in group 3 (451 vs 510 m; +59 m, +13.1%, SD = 89 m, p = 0.039).
Regarding QoL parameters, several QLQ-c30 parameters improved significantly after prehabilitation: physical functioning increased from a median of 87 (MAD = 13) to 93 points (MAD = 7) (p = 0.027) and asthenia decreased from 33 (11) to 22 (11) points (p = 0.029). Even though appetite loss median was 0 (0) before and after prehabilitation signifying that at least 50% of the patients reported absence of appetite loss, nine patients reported a decrease in appetite loss, 24 were stable and one reported increased appetite loss. Given this distribution, appetite loss showed a significant improvement (p = 0.021). The HADS score improved significantly as well (from 10 to 7 points), specifically for anxiety (from 7 to 4 points) and depression (from 3 to 1 points).
There were no significant weight change or laboratory value modifications after prehabilitation in Table 2.
Patient demographics and postoperative outcomes.
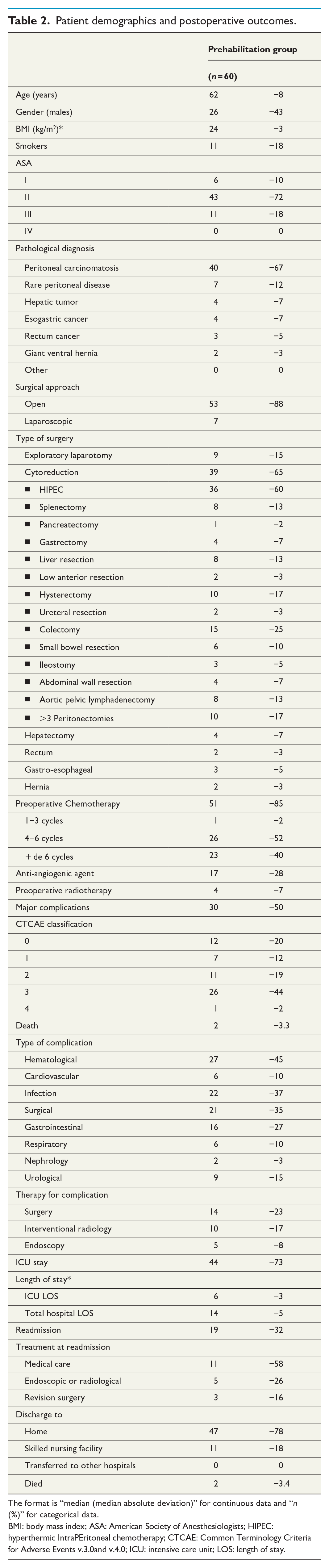
The format is “median (median absolute deviation)” for continuous data and “n (%)” for categorical data.
BMI: body mass index; ASA: American Society of Anesthesiologists; HIPEC: hyperthermic IntraPEritoneal chemotherapy; CTCAE: Common Terminology Criteria for Adverse Events v.3.0and v.4.0; ICU: intensive care unit; LOS: length of stay.
Table 2 reported on the demographics, baseline characteristics, and postoperative clinical data of the prehabilitation groups. Postoperative 90-day mortality and morbidity were 3.4% and 48%, respectively. Median hospital length of stay and intensive care unit length of stay were 14 and 6 days, respectively. For 19 patients readmitted, the treatment was medical, radiological, or surgical, for 11, 5, and 3 patients, respectively. The causes of readmission were classified according to the treatment carried out: medical care, endoscopic or radiological, revision surgery. The majority of readmissions were for medical reasons. Only 16% of readmitted patients required further surgery.
Discussion
This pilot study showed that a prehabilitation protocol for major abdominal surgery is both feasible and safe. Our study also showed that prehabilitation improves physical functioning, patients’ psychology and their QoL.
Patients undergoing major abdominal surgery know that complications (or even death) can occur after the operation and they are probably one of the most willing populations to be as prepared as possible to avoid these adverse events. All patients participated actively in the prehabilitation protocol, and no patient refused psychological support. Several factors might have resulted in the patients willing to participate in/comply with the prehabilitation program, such as fear of surgery, 17 the existence of a medico-surgical team involved in the protocol, 18 and the appearance in recent years of a World Health Organization (WHO) campaign promoting physical activity. Conducting physical exercises at home might have also increased compliance. 19 Furthermore, our experience with this study showed us that coordination is paramount. It is impossible to mount a successful prehabilitation program without being able to coordinate between oncology (chemotherapy and radiation) appointments, surgery and all the necessary appointments for prehabilitation. As most patients underwent preoperative chemotherapy and/or radiation, it was essential to adapt their physical activity to their schedule. The coordinator played a major role to ensure the feasibility of the prehabilitation program. Adapting the prehabilitation protocol to the patients’ needs and abilities seems to be essential to achieve maximum compliance.
In our experience, no surgery was delayed due to prehabilitation program. To be enrolled in prehabilitation program, a planned delay between inclusion and surgery of more than 4 weeks was required. For patients planned to undergo oncologic surgery, neoadjuvant therapy allowed for the completion of the prehabilitation protocol. No cardiovascular or traumatic events were observed during prehabilitation. All the five adverse events that occurred during prehabilitation were secondary to the underlying pathology of the patients and could not be directly attributed to prehabilitation. It should be noted that one patient suffered from “fear of not being sufficiently prepared for surgery” during the program. In cases when patients—such as this one—take a significant psychological toll because of their disease process, its prognosis and the necessary operative management, changes in lifestyle (such as those occurring during prehabilitation) can cause significant anxiety. Patients should therefore be regularly offered psychological counseling. Adapting the protocol to the patient’s abilities could also prevent this phenomenon.
Functional capacity improved clinically with prehabilitation as evidenced by the increase of 45 m walked during the 6MWT. Even though an increase of 45 m in walking distance might seem trivial, it has been demonstrated that an increase of at least 20 m has a positive impact on the outcomes of colorectal surgery. 13 By extrapolation, we therefore believe that 45 m is clinically significant for other major surgeries as well. 13 Even though the average walking distance increased for the entire population, it was significant only for group 3, that is, patients with major comorbidities and limited ability to practice sport. This seems to be an interesting topic for further research, especially in situations where resources are scarce and prehabilitation cannot be offered to all patients undergoing surgery. Li et al. found an increase in the walked distance during the 6MWT that was close to our results (mean: 40 m ± SD = 40) after a median of 33 days of prehabilitation. 20 Conversely, Barberan-Garcia et al. found no change in the distance walked during the 6MWT in 54 patients who received physical and psychological prehabilitation. 21 Those patients did not benefit from any nutritional preparation, most surgeries were laparoscopic, and the physical activity was of high intensity which alone can worsen the results of the physical preparation. 10 However, they did demonstrate a decrease in postoperative complications. This is an important finding highlighting that the 6MWT is probably a surrogate marker for functional capacity and that its improvement (or lack of thereof) is not necessarily related to the postoperative complication rate. It is therefore imperative to look at all the data (6MWT + postoperative complications) to evaluate the efficacy of a prehabilitation protocol.
We found a significant improvement in the physical functioning, asthenia and appetite loss items of the QLQ-c30 questionnaire. The majority of studies evaluating QoL did not report improvement 21 based on the 36-item short form (SF-36) score. Since we used a different questionnaire, our results are not directly comparable to those results. 18 We chose the QLQ-c30 questionnaire because it is certainly less generic by definition, more adapted for abdominal surgery and provides potentially more distinctiveness. Given its higher specificity for abdominal pathology, we used it in hope of being able to detect a difference more easily than with SF-36. Preparation decreased the proportion of anxious patients in half. A minority of patients had a pathological depression score, reported as an independent risk factor for prolonged hospitalization. 22
The main limitation of our study was the absence of comparative control group. However, the reported data will help for creating statistical hypothesis for future randomized multi-institutional controlled trials.
In conclusion, this study demonstrated the feasibility of a prehabilitation program, and its safety. Furthermore, it seems that there might be benefits in improving the physical and psychological condition of the patients. Despite our study’s small size and shortcomings we believe that our encouraging results can serve as a starting point for future studies that will be able to more definitively determine the benefits of prehabilitation.
Footnotes
Acknowledgements
The authors thank the team of Laurent Villeneuve for help with data management and Sonia Krouk and Raquel Azevedo, for their contribution to the program development.
Author contributions
I.E.B. participated in the establishment of the prehabilitation protocol, inclusion and follow-up of patients, collection of data, and the drafting of the manuscript. A.N.F., G.P., and F.M. took part in the writing and editing of the manuscript. A.N.F. is responsible for the statistical analysis of the data. The other authors are the surgeons of the patients included and all participated in the review of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
