Abstract

Keywords
Introduction
Contemporary research shows alarming rates of appendiceal tumors associated with complicated acute appendicitis presenting as periappendicular abscess, and this risk is increased by patient age over 40 years.1–6 The Peri-Appendicitis Acuta Randomized Clinical Trial (Peri-APPAC RCT) 1 comparing interval appendectomy and follow-up with magnetic resonance imaging (MRI) after the initial successful nonoperative treatment of periappendicular abscess was prematurely terminated in 2016 owing to ethical concerns due to high appendiceal neoplasm rate in the appendectomy group with final overall neoplasm prevalence of 20% (12/60). Out of the 30 patients randomized to MRI follow-up, appendectomy was recommended to all patients. At 5 years, this study reports the long-term outcomes of the follow-up group patients declining to undergo appendectomy after study termination and all the patients diagnosed with an appendiceal tumor.
Methods
This is a post hoc follow-up of the Peri-APPAC RCT comparing interval appendectomy with MRI follow-up for periappendicular abscess after initial successful conservative treatment in adults aged 18 to 60 years in five Finnish hospitals enrolling patients from January 2013 to April 2016. This long-term follow-up at a minimum of 5 years was added to the study protocol subsequently targeted to assess the follow-up group patients initially declining to undergo appendectomy after study termination. First, this long-term MRI follow-up was added to ensure patient safety based on the high appendiceal tumor rate at 1-year follow-up. 1 Patients gave a separate written informed consent for this imaging. Second, this observational follow-up assessed the long-term outcomes of the trial patients initially diagnosed with an appendiceal tumor by reviewing their medical records. The detailed methods and primary results have been previously published 1. The difference between groups in age was tested using Mann–Whitney U-test. The study was approved by the Ethical Committee of Turku University Hospital district and conducted in accordance with the Declaration of Helsinki following the relevant portions of the Consolidated Standards of Reporting Trials (CONSORT) reporting guideline.
Results
Out of the 14 patients with an intact appendix, two had undergone appendectomy after the initial follow-up with no appendiceal tumor findings (Fig. 1). Nine out of the remaining 12 patients with an intact appendix underwent MRI with normal findings. The median age of these patients was 35 years (range 22-58 years), and detailed patient characteristics are presented in Table 1.
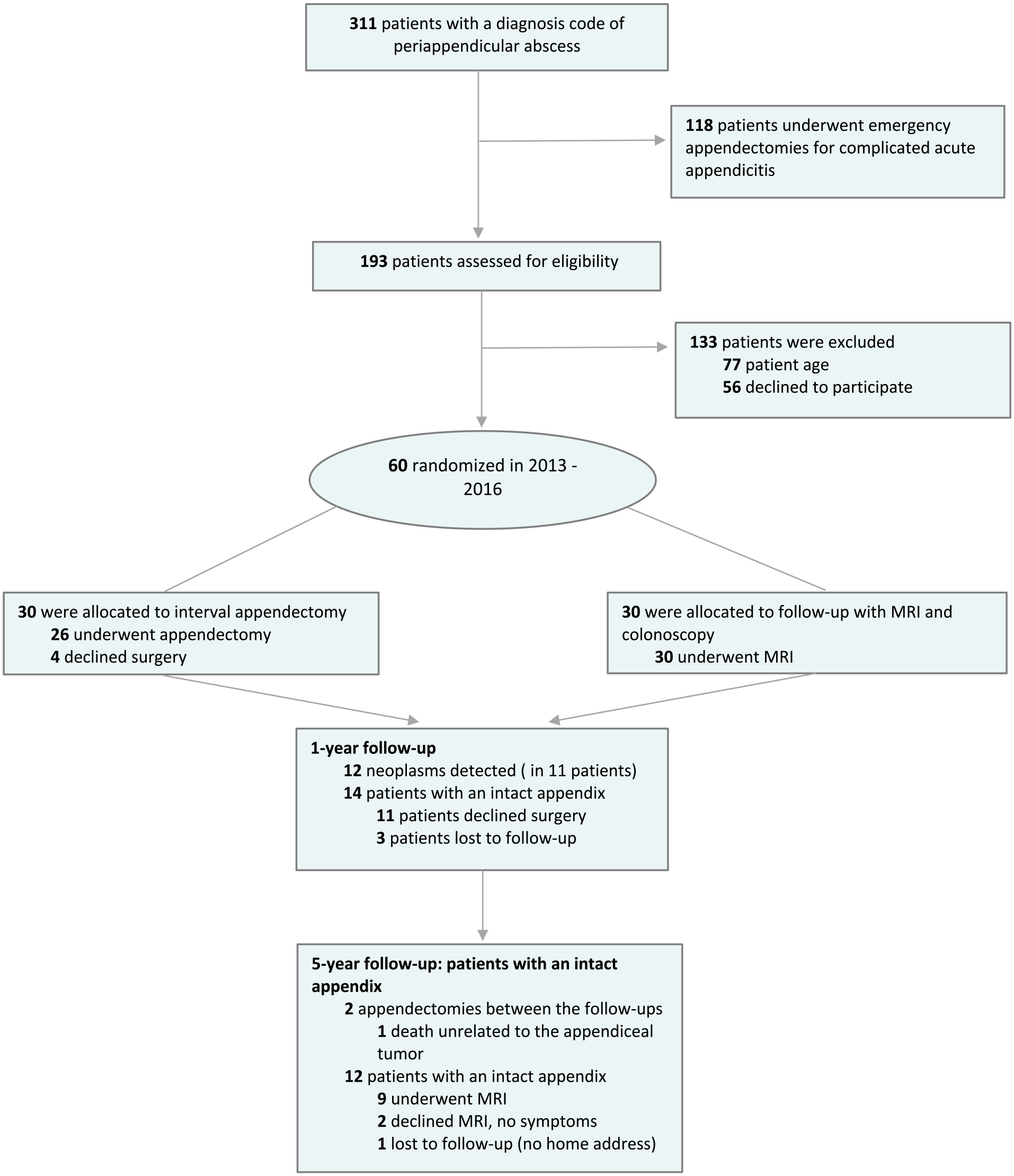
Peri-APPAC trial patient flow-chart at 5-year follow-up.
Characteristics of patients with an intact appendix at 5 years and with an initial diagnosis of an appendiceal neoplasm in 2013 – 2016.
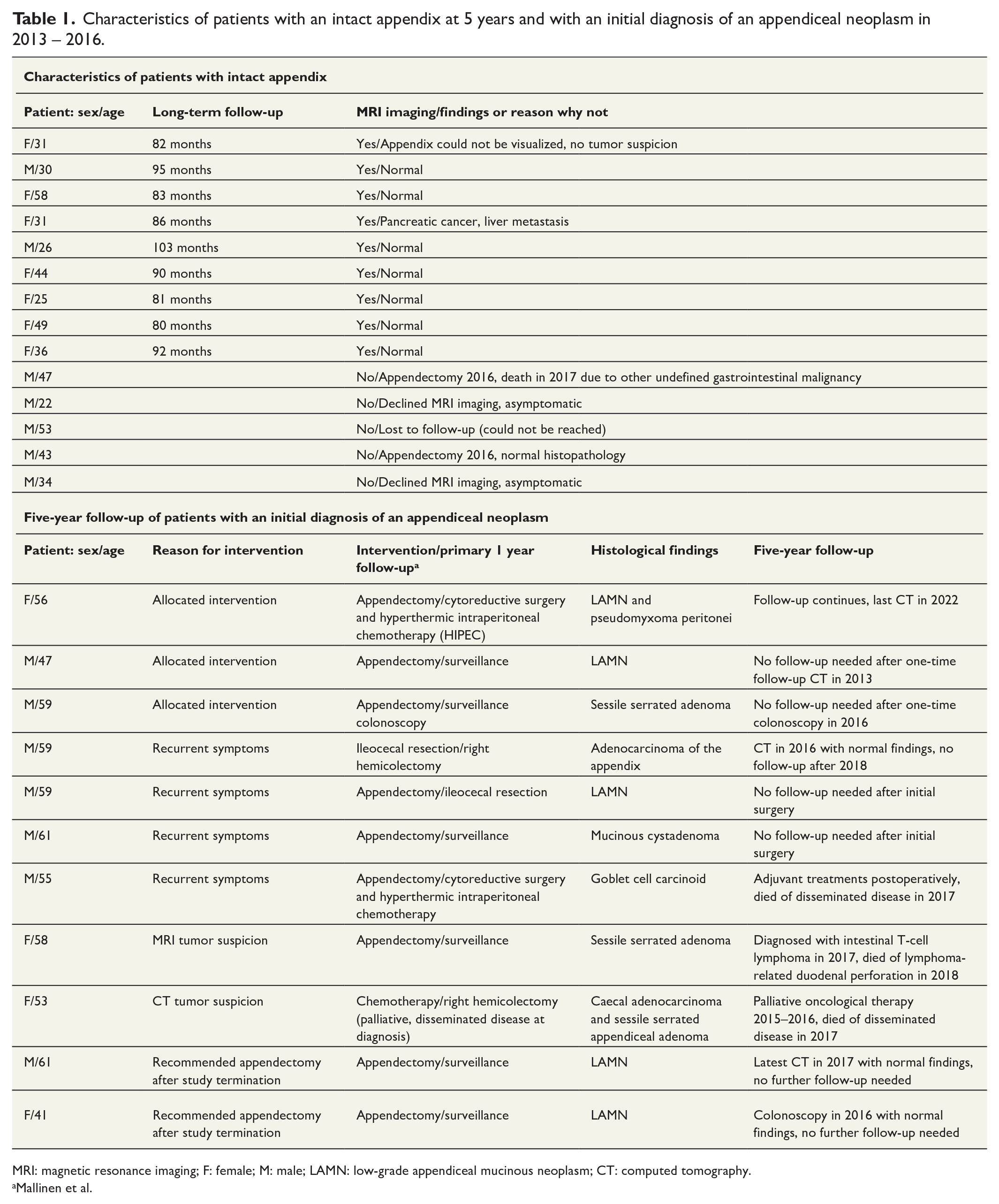
MRI: magnetic resonance imaging; F: female; M: male; LAMN: low-grade appendiceal mucinous neoplasm; CT: computed tomography.
Mallinen et al.
Overall initial appendiceal neoplasm rate after premature termination and surgical treatment of the MRI group patients consenting to appendectomy was 20% (12/60, 12 neoplams in 11 patients). 1 At 5 years, 3 out of these 11 patients had died, with two deaths related to the appendiceal neoplasm. In the remaining eight patients with appendiceal neoplasms, the long-term follow-up at 5 years after the appendectomy was uneventful (Table 1). The median age of the patients with an appendiceal tumor was significantly higher than that of the patients with an intact appendix undergoing MRI imaging (58 years versus 35 years, p < 0.001).
Discussion
Based on the premature termination of the Peri-APPAC RCT in 2016 due to the high rate of appendiceal neoplasms in the interval appendectomy group, the long-term imaging follow-up of the MRI group patients declining to undergo appendectomy was added to the study protocol to ensure patient safety and ethical conduct of clinical trials. At 5 years, there were no additional appendiceal tumors detected at MRI imaging or in the two additional appendectomies performed between the 1- and 5-year follow-up. The patients declining to undergo appendectomy even after a strong recommendation after study termination were statistically significantly younger than the patients diagnosed with an appendiceal tumor. The main limitation of this study is the small number of patients based on the premature study termination.
This long-term follow-up underlines the patient age as a major factor for an appendiceal tumor risk associated with a periappendicular abscess also demonstrated by the initial report1 and other studies.5,6 The rate of appendiceal neoplasms needs to be validated by large prospective cohort studies to further define the necessity of interval appendectomy in all age groups.
Footnotes
Authors’ note
The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The trial is registered on ClinicalTrials.gov, Peri-APPAC (NCT 03013686).
Author contributions
P. Salminen and R. Salminen had full access to all the data in the study and take full responsibility for the integrity of the data and the accuracy of the data analyses. Dr P. Salminen had the final responsibility for the decision to submit the manuscript for publication. Concept and design: P. Salminen, R. Salminen, Rautio; acquisition, analysis, or interpretation of data: P. Salminen, R. Salminen, Rautio; drafting of the manuscript: R. Salminen, P. Salminen; critical revision of the manuscript for important intellectual content: P. Salminen, Rautio, Grönroos, Nordström, Rantanen, Mällinen; statistical analysis: P. Salminen, R. Salminen; funding: P. Salminen; administrative, technical, or material support: P. Salminen, Rautio; supervision: P. Salminen, Rautio.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Paulina Salminen reported lecture fees from Novo Nordisk. No other disclosures were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Sigrid Jusélius Foundation.
Data sharing statement
The data requests can be directed to the primary investigator and will be evaluated by the study group.
