Abstract
Primary sarcomas of bone are rare malignant mesenchymal tumors. The most common bone sarcomas are osteosarcoma, Ewing’s sarcoma, and chondrosarcoma. The prognosis has improved over the years, but bone sarcomas are still life-threatening tumors that need a multidisciplinary approach for diagnosis and treatment. Bone sarcomas arising in the pelvis present a unique challenge to orthopedic oncologists due to the absence of natural anatomical barriers, the close proximity of vital neurovascular structures, and the high mechanical demands placed on any pelvic reconstruction following the excision of the tumor. While radiotherapy has an important role especially in Ewing’s sarcoma and chemotherapy for both Ewing’s sarcoma and osteosarcoma, surgery remains the main choice of treatment for all three entities. While external hemipelvectomy has remained one option, the main aim of surgery is limb salvage. After complete tumor resection, the bone defect needs to be reconstructed. Possibilities to reconstruct the defect include prosthetic or biological reconstruction. The method of reconstruction is dependent on the location of tumor and the surgery required for its removal. The aim of this article is to give an insight into pelvic bone sarcomas, their oncological and surgical outcomes, and the options for treatment based on the authors’ experiences.
Background and aims
Primary malignant sarcomas of bone—which includes chondrosarcoma, osteosarcoma, and Ewing’s sarcoma—are the most common primary malignancies in the pelvis, excluding hematologic malignancies. Approximately 14.5% of primary malignant tumors occur in the pelvic bones, making it one of the most common locations for malignancy, including the proximal and distal femur and proximal humerus. Different histological variants of sarcoma have differing preferences for location. For example, the most common site for osteosarcoma is around the knee and for chondrosarcoma, more than 30% of the tumors are located in the pelvis. 1
The clinical presentation of pelvic bone sarcoma is similar to that of bone sarcomas arising in other anatomical locations, but is often variable and non-specific, at least at the outset. Common features for presentation include persistent pain that gets worse over time and becomes unrelenting, including at night, swelling which makes movement difficult, a noticeable lump, and weakness of the bone leading to pathologic fracture.
Despite considerable advances in treatment options, sarcomas in the pelvic bones are generally associated with poorer outcomes than sarcomas in the extremities or acral locations. This is most likely a result of the size of pelvic tumors at presentation, which can often be extremely large due to the distensible pelvic cavity into which tumors often grow. Pelvic tumors are located in close proximity to vital vessels and nerves, and thereby wide surgical margins are often difficulty of obtain at the time of resection. Depending on the histological variant, disease-specific survival differs between 27% at 5 years for osteosarcoma, 2 59%–75% at 5 years in chondrosarcoma,3,4 and 41%–66% at 5 years in Ewing’s sarcoma.5–7 Tumor grade, stage, size, and surgical margin, as well as patient age and type of surgery, are independent prognostic factors affecting survival.1–7
Treatment of pelvic sarcomas remains one of the most difficult challenges for orthopedic oncologists.8,9 In the absence of reliable adjuvant treatments, hindquarter amputation used to be the only surgical option. 10 With modern and more accurate preoperative imaging and multimodality treatment, limb salvage surgery has become more common and is more often an achievable treatment option for many patients.1,7,8,10–18 Various technical perioperative solutions and reconstruction methods have been described, although not every defect following pelvic resection requires reconstruction. Ideally, pelvic resection should achieve a wide tumor margin and this should be followed by anatomic reconstruction, leading to a restoration of preoperative function and quality of life. Despite these advances, hindquarter amputation is still required, particularly for selected tumors which involve neurovascular structures.9,10,19 Surgery to pelvic sarcomas is associated with high rates of complications and over time, the need for revision surgery. These include intraoperative complications due to neurovascular compromise, a range of mechanical and structural failures after reconstruction, and large soft tissue defects resulting in the need for wound revision, infection, and the use of local flaps or free tissue transfer.1–3,5,13–33
Diagnostics of primary malignant sarcomas of the pelvis
Pain, a palpable mass and difficulties in motion are often the primary symptoms of a tumor of the pelvis. These symptoms are usually investigated with various imaging modalities including plain radiographs, magnetic resonance imaging (MRI), and computed tomography (CT). These modalities play an important role in the detection, diagnosis, characterization, and staging of these lesions.
Radiograph
The plain radiograph is often the first imaging modality to show a pelvic bone tumor. Whether this tumor is then diagnosed as a sarcoma is dependent on its appearance and the interpreter’s knowledge of tumors with a predilection for the pelvis. In addition to tumor location, its ability to produce osteoid, cause osteolysis, the size, and periosteal reaction are important signals which should raise the suspicion of a primary tumor of bone. Benign lesions usually have well-defined margins, whereas malignant lesions have geographic, motheaten, or permeative patterns of destruction with ill-defined margins. While the exact nature of the lesion can be difficult to tell from a plain radiograph, the differentiation between benign and malignant lesions is often made from the radiograph which at the very least, should stimulate further investigation.
MRI
MRI is the most commonly used modality to investigate any tumor of the skeleton but is extremely valuable when investigating bone sarcomas of the pelvis. T1 and fat-suppressed T2-weighted images are required in axial, sagittal, and coronal planes with and without contrast. MRI is useful at assessing the exact extent of the tumor which is sometimes difficult to see from a plain radiograph as the soft tissue component of the tumor often distends into the pelvic cavity. MRI therefore is the modality of choice for local staging. Whole body MRI is often used to assess the presence of skeletal or soft tissue metastases, having largely replaced whole body technetium bone scan.
CT
CT while largely superseded by MRI still has a role in assessing tumors of the pelvis particularly for investigating matrix, periosteal mineralization, and cortical destruction. CT is also required for distant staging to identify the presence of metastases within the lungs.
Positron emission tomography-computed tomography
Positron emission tomography-computed tomography (PET-CT) is often advocated for whole body staging particularly for certain tumor types, for example, Ewing’s sarcoma. The popularity of PET-CT has increased due to its sensitivity at detecting small foci of metastatic disease, although its role as a local imaging technique is less important. Interval PET-CT may be useful in the assessment of response to neo adjuvant treatment and following completion of treatment to differentiate between postoperative changes like fibrosis and local recurrence and may be more sensitive than MRI. 8
Other imaging technologies
Technetium bone scan remains a viable option for whole skeletal imaging to identify osseous metastases, although it has, in recent times, been largely replaced by whole body MRI or PET-CT.
Histology
The gold standard for diagnosis, as for any bone tumor, is the histological diagnosis after biopsy. Biopsy, usually performed with imaging guidance, is essential for the diagnosis and for planning therapeutic options. The biopsy approach should be discussed with the surgeon responsible for the definitive resection, as the tract used for the biopsy should be removed at the time of the final surgery to remove the tumor. The risk of tumor cell seeding in the biopsy tract is unknown, but the development of recurrence within the biopsy tract is not an uncommon experience for tumor surgeons.9,10
Chondrosarcoma
Chondrosarcoma is the most common primary bone malignancy in the pelvis. They are often high-grade tumors, and they are almost twice as likely to present with metastatic disease when compared with high-grade chondrosarcomas of the extremities.
Chondrosarcoma usually affects middle-aged adults, as the mean age at presentation is around 50 years. Chondrosarcomas are further categorized according to their location within the bone as central or secondary (Fig. 1) or peripheral chondrosarcomas (Fig. 2). Secondary peripheral chondrosarcomas are the result of malignant transformation within the cartilage cap of an existing osteochondroma, later named as peripheral chondrosarcoma. Central and peripheral chondrosarcomas are histologically similar. Three different histological grades and dedifferentiation are described for both types. Grading of chondrosarcoma is, however, extremely challenging and prone to inter- and intra-observer variability. Preoperative biopsy is known to be unreliable and furthermore; there is no consensus about the role of preoperative biopsy in pelvic peripheral chondrosarcoma. Determining the tumor grade with small samples of tumor is notoriously difficult with less than 50% of biopsies giving an accurate grade of the tumor. 11
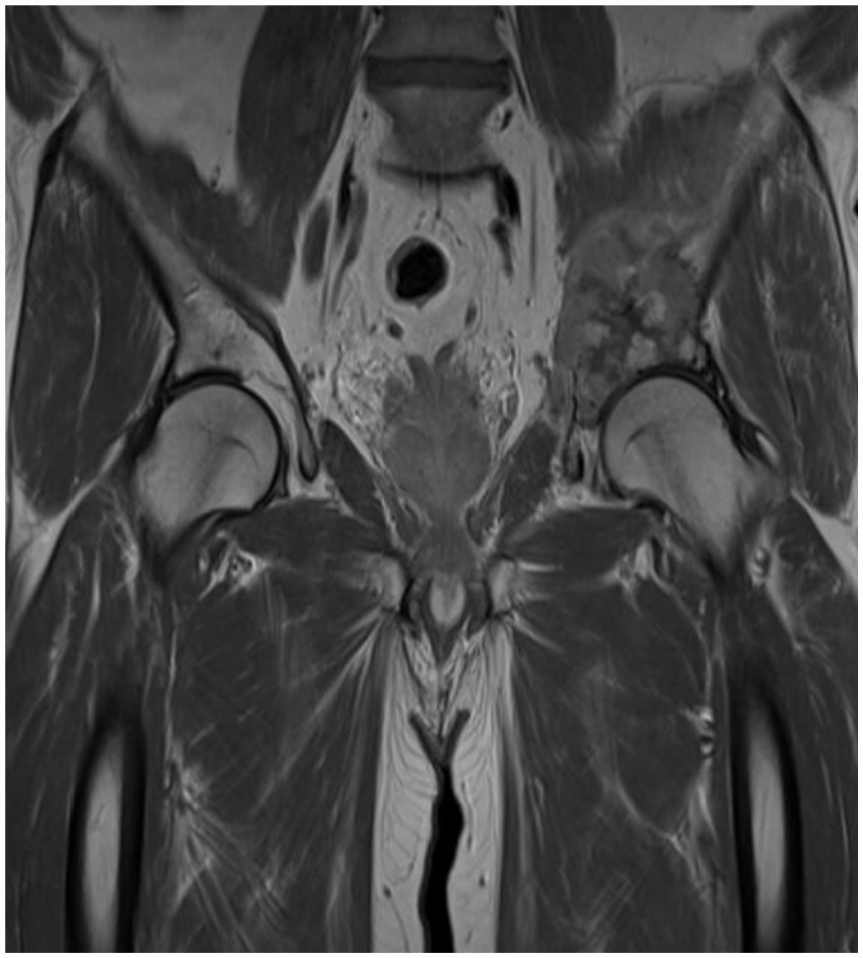
A 52-year-old male with grade 2 central conventional chondrosarcoma of the left acetabulum extending into ilium with an extraosseous component.
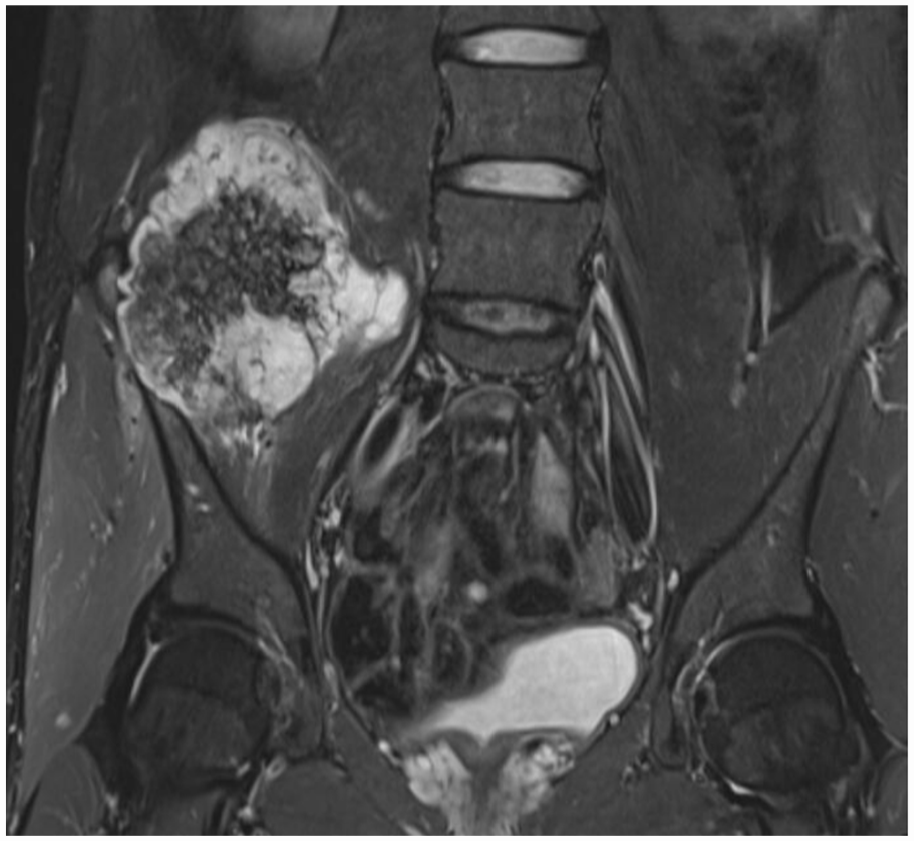
A 29-year-old male with grade 2 peripheral chondrosarcoma of the right ilium. A thick cartilage cap of 5 cm expanding toward spinal vertebrae.
Peripheral chondrosarcoma is more often located in the ilium, whereas central chondrosarcoma more commonly occurs in the periacetabular area. 1 Chondrosarcomas are largely resistant to chemotherapy and radiotherapy making surgery the mainstay of treatment. Current guidelines recommend wide resection of high-grade chondrosarcomas, 12 although there is no specific recommendation for what constitutes an adequate margin. However, estimates of an adequate margin for peripheral pelvic chondrosarcomas, suggests that local recurrence is effectively eliminated by resection margins ⩾1 mm with no patients in one study developing local recurrence, metastasis, or disease-related death. 13 For central, high-grade pelvic chondrosarcomas, estimated margin of 2 mm has been shown to decrease the risk of local recurrence and thereby improve optimal oncological outcome. 14
In the extremities, grade 1 chondrosarcoma has been renamed as atypical chondroid tumor. 15 Although patients with grade I chondrosarcoma of the extremity treated with intralesional surgery experience good local control and long-term disease-free survival, grade I chondrosarcoma of the pelvis treated with intralesional or marginal excision is associated with unacceptably high rates of local recurrence, which tend to be at a higher grade.4,16–20 In addition, as biopsy in the majority of cases underestimates the grade of the tumor as seen at final resection, the aim of resection should be to achieve at least a 1-mm margin in peripheral chondrosarcomas and more than 2 mm in central chondrosarcomas.4,14,21
The role of radiotherapy in the treatment of chondrosarcoma is limited, but may be appropriate in highly selected cases or for palliation. Radiotherapy can be considered in four situations: in the absence of metastatic disease where tumor resection will confer unacceptable morbidity to the patient (e.g. tumors involving the spinal canal or crossing the midline of the sacrum) or where it is deemed that an adequate margin cannot be achieved at the time of resection (curative), in the presence of metastatic disease where resection is not feasible or would cause unacceptable morbidity (palliative), or postoperatively when surgery results in positive margins and reresection is not possible. 12 Radiotherapy may on occasion be considered in the setting of incomplete resection where intralesional resection has been undertaken for symptom control and planned radiotherapy is given to the post resection tumor volume. This is occasionally advocated for chondrosarcomas in anatomically challenging areas such as the skull base and cervical spine. As in all sarcomas, high doses are required. For chondrosarcoma, doses often exceed 60 Gy, which can be difficult to deliver safely using conventional radiation methods. Radiotherapy using particles such as proton and heavy ions (e.g. helium, carbon) is an attractive alternative as it allows delivery of therapeutic doses (>60 Gy) while minimizing the exit dose to adjacent normal structures. The majority of the literature on this topic pertains to chondrosarcoma of the skull base. Preliminary reports show that proton therapy is effective for local control both as an adjuvant after surgical resection and as definitive treatment; however, further follow-up is necessary to better characterize long-term disease control and late toxicities. 22
Osteosarcoma
Osteosarcoma of the pelvis, which accounts for less than 10% of all osteosarcomas, has an expected 5-year survival of between 18% and 38%.2,23 The inferior survival of patients with pelvic osteosarcoma is multifactorial, including large tumor volume at presentation, difficulty in achieving adequate surgical margins, higher likelihood of metastatic disease at presentation, inferior necrosis after preoperative chemotherapy, and the presence of macroscopic tumor emboli in the large regional vessels. The tumor is more often a chondroblastic variant when compared to extremity osteosarcomas, differing from the more common osteoblastic variant seen in the appendicular skeleton, and is often resistant to chemotherapy.2,24–26 Less than 15% of patient with a chondroblastic cell type who went on to resection had a good response to chemotherapy, and the diversion in more than 30% whom the initial intention was surgical resection, to a non-surgical or palliative regimen, raises a treatment dilemma as to how best to treat the primary tumor, particularly when that tumor is a chondroblastic variant: should these patients be treated without preoperative chemotherapy or should they undergo immediate surgery, including hindquarter amputation prior to chemotherapy? 2
Most cases of osteosarcoma (∼75%) occur in patients younger than 25 years. In this young population, 90% occur in the extremities in a de novo fashion. A second peak is seen in those older than 50 years, among which more than 50% of cases arise secondary to pre-existing conditions such as Paget’s disease and following previous radiotherapy. In this older population, up to 40% of tumors present in the pelvis, partly reflecting the natural history of conditions frequently treated with radiotherapy. 2
Within the pelvis, the ilium is the most common location for osteosarcoma (Fig. 3), followed by the acetabulum and the ischium. Tumors frequently involve more than one part of the pelvis, and iliac tumors often infiltrate the sacrum. 2 Resectable, high-grade osteosarcoma is typically treated with multiagent chemotherapy combined with complete surgical resection of the tumor. Complete surgical resection is paramount to cure. Although advances in imaging techniques have played an important role in the surgeon’s ability to plan complete resections, the complexity of pelvic anatomy coupled with the proximity of the genitourinary organs and sacrum makes margin-negative surgical resections exceedingly challenging. Intralesional resections (positive margins) are associated with very high rates of local recurrence. However, rates of local recurrence for pelvic osteosarcoma even following complete resection are significantly higher than that seen in extremity osteosarcoma (34%–50%). 27 The margins achieved at resection are often wider following external hemipelvectomy when compared to limb-salvage surgery, although this has not been shown to translate to an improved overall survival.2,28
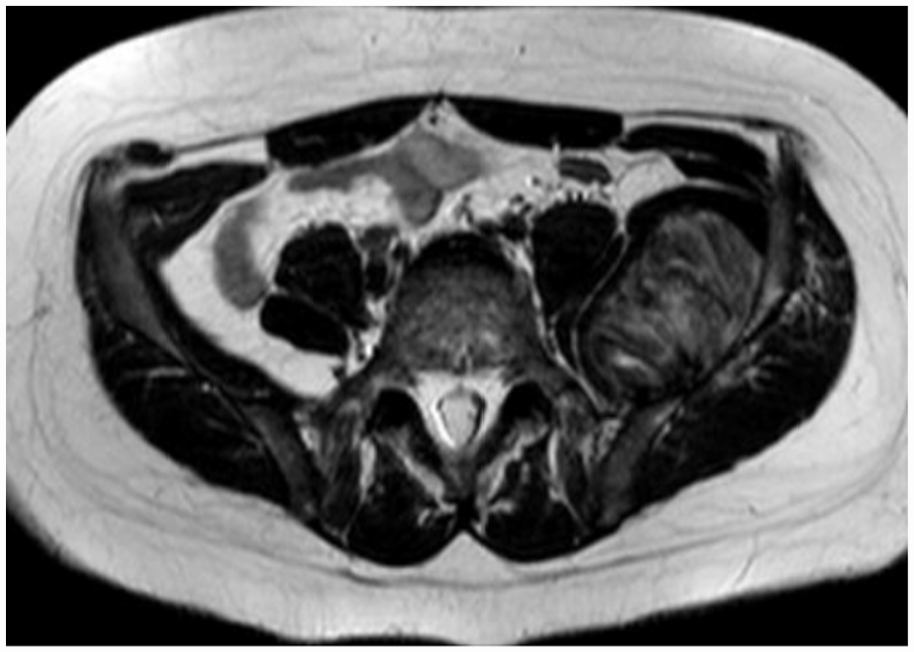
A 12-year-old boy with high-grade osteosarcoma of the left ilium inner table. An adjacent soft tissue mass extending into pelvis.
The high rates of local recurrence for pelvic osteosarcoma demonstrate the need for adjuvant local therapy. Radiotherapy at present is reserved for patients with unresectable tumors or for those with positive surgical margins. 12 Although the optimal doses and delivery methods remain to be defined, radiotherapy has been shown to play a role in treating patients at the highest risk for local recurrence. Clearly, the use of radiation in the management of osteosarcoma should be reinvestigated in light of the evolution of radiotherapy strategies in the past two decades.
Ewing’s sarcoma
Ewing’s sarcoma has a high predilection for the axial skeleton, particularly the pelvis. 29 It occurs most frequently in children and adolescents but is also seen in adults. Approximately 75% of patients present with localized disease, and of these, the 5-year survival is approximately 70%. Patients with metastatic disease at presentation experience an inferior survival rate, estimated as 20% at 5 years. Staging must be oriented to detect lung, bone, and bone marrow metastases since the rate of metastases in lung and in bone marrow is estimated as 10% in both and 5% in combination. Patients with non-pulmonary sites of metastatic disease fare worse than those with lung-only metastases. 29 In addition to the presence of metastatic disease several other prognostic factors have been identified, including tumor size larger than 8 cm, older age (>14 years), response to chemotherapy, translocation type, and detectable fusion transcripts in bone marrow.6,31,32
Ewing’s sarcoma of the pelvis has traditionally been associated with a worse prognosis when compared to non-pelvic tumors. 33 Patients with pelvic tumors often present with larger tumor volumes (Fig. 4) compared with extremity sites, and more often have clinically detectable sites of metastatic disease at presentation. Local control remains problematic. It has been speculated that Ewing’s sarcoma of the pelvis behaves in a biologically different way to Ewing’s sarcoma of the extremities and requires more aggressive local control. 33 Since Ewing’s sarcoma is both chemo- and radio-sensitive, the treatment, especially in pelvic tumors, is often a combination of systemic and local therapies. Optimizing chemotherapy doses and schedules—combined with improved supportive therapy, better radiation techniques, and more aggressive surgery—has led to significant progress overall. 7 However, perhaps the single most controversial topic in the management of pelvic Ewing’s sarcoma is the modality through which to achieve the best local control.
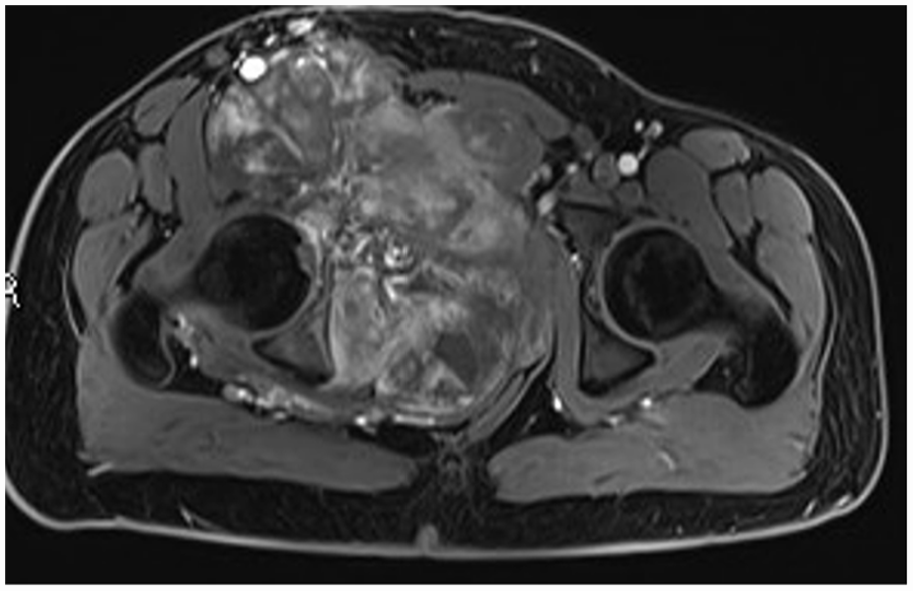
A 15-year-old boy with Ewing’s sarcoma originating from the right pubic bone. A large lobulated soft mass dislocating rectum, bladder, and vessels extending over the midline.
For decades, definitive radiotherapy was the standard local treatment for Ewing’s sarcoma. As improvements in imaging evolved and limb-salvage techniques were refined, surgery became the preferred method of local control when negative margins and acceptable morbidity can be expected. This trend is largely because of reports of higher rates of local failure in patients treated with radiation alone (50%) compared with those treated with either surgery alone or surgery combined with radiation (50%–60%). 7 In Ewing’s sarcoma patients, response to chemotherapy has been shown to have a more important role on local control than surgical margins.7,32 The role of margin is not without importance and with large tumors, clear margins can be difficult to obtain. Taken together, in current European protocols, the option of preoperative radiotherapy is also offered, mainly in the case of expected marginal resections for which postoperative radiotherapy is considered likely. 12 Although therapeutic doses, field size, and fractionation schedules have been established, these parameters and new modalities like proton therapy are worthy of reinvestigation, especially given the long-term side effects of radiotherapy and the potential for secondary malignancies that occur with greater frequency in patients with a history Ewing’s sarcoma.34,35
Surgery
Surgical treatment for sarcomas of the pelvis consists of external hemipelvectomy (Fig. 5) or limb-salvage, internal hemipelvectomy. The term external hemipelvectomy refers to an amputation through the sacroiliac joint. Modifications of the operation exist, including extended hemipelvectomy through the sacrum or amputation distal to the sacroiliac joint, maintaining portions of the ilium.36,37
A 75-year-old male with a high-grade telangiectatic osteosarcoma of the entire right hemipelvis was operated with external hemipelvectomy.
The term internal hemipelvectomy, which refers to limb-salvage pelvic resections, is a misnomer; these operations should actually be called pelvic resections. Pelvic resections are often classified according to the system of Enneking (Fig. 6). 38 Type I resections remove all or part of the ilium with Type I-S, including a portion of the sacrum. Type II resections remove the periacetabular area. Type III resections remove the pubis and/or ischial bone. Combinations such as a Type II + III resection indicate that the acetabulum and pubis and/or ischial bone were removed en bloc. Resection of the entire innominate bone would be a Type I + II + III resection.
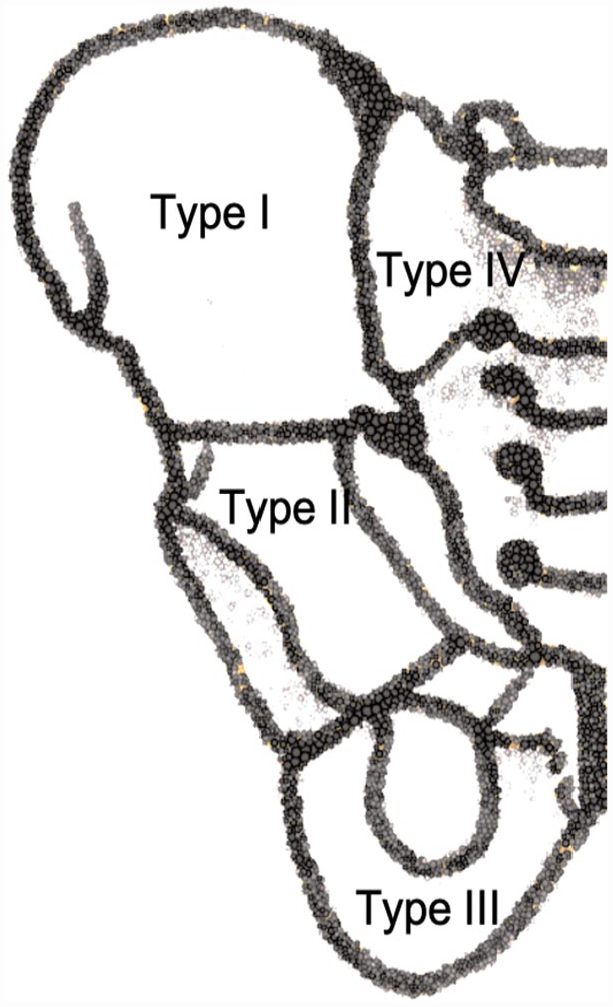
Pelvic resections according to Enneking classification: Type I. Iliac bone, Type II. Periacetabular region, Type III. Pubis or ischium, and Type IV or S. Hemisacrum.
Regarding the optimal method of reconstruction after surgical removal of a pelvic sarcoma, the ideal reconstruction method depends on a host of factors, including the resultant defect, desired functional outcome, use of adjuvant treatment, and age of the patient. Type I resection is the most frequent resection of the pelvis 28 and debate exists as to whether the resultant defect in the pelvic ring should be reconstructed or not. If the pelvic ring discontinuity is not reconstructed, over time an iliosacral arthrodesis or pseudarthrosis can develop. 39 Reconstruction with vascularized or non-vascularized autografts, allografts, or primary arthrodesis of the sacrum and periacetabular area usually restores the normal anatomical position of the native acetabulum (Fig. 7). 40 Complications vary between methods. Non-reconstruction carries a risk of limb-length discrepancy and dislocation of the joint and stress fractures of the contralateral side. Reconstructions have the possibility of mechanical failure and a significantly higher incidence of infection. Both methods report comparable functional outcomes.39,40
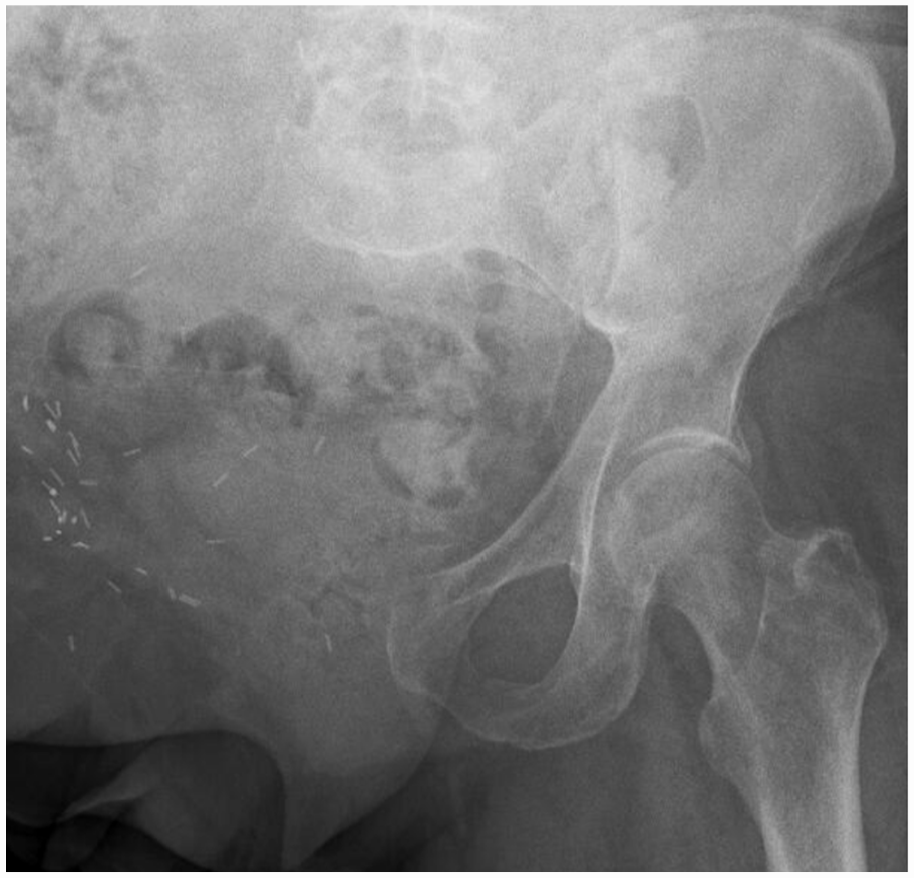
A 29-year-old male with peripheral chondrosarcoma (Fig. 2). After P1 resection, reconstruction was done with non-vascularized fibula and primary healing was guaranteed with sacro-acetabular fixation with pedicle screw instrumentation.
Reconstructions after Type II resections and their associated combinations (Types I + II, II + III, and I + II + III) are the most challenging, complicated and associated with the highest risk of complications. Options for reconstruction include biologic reconstructions with vascularized or non-vascularized autografts, structural allografts, hardware, hip transposition (Fig. 8), and iliofemoral or ischiofemoral arthrodesis. All of these methods require considerable surgical expertise, a good soft tissue envelope, a lengthy recovery period, and acceptance by the patient and surgeon that complications may occur. Early and late infections are common. Mechanical complications, prosthetic failures, and joint instability may occur with moderate frequency. Ultimately, implant failure is inevitable throughout the lifetime of the reconstruction if the patient is cured of their disease. Patients often require multiple surgeries to address the associated complications.2,41–50
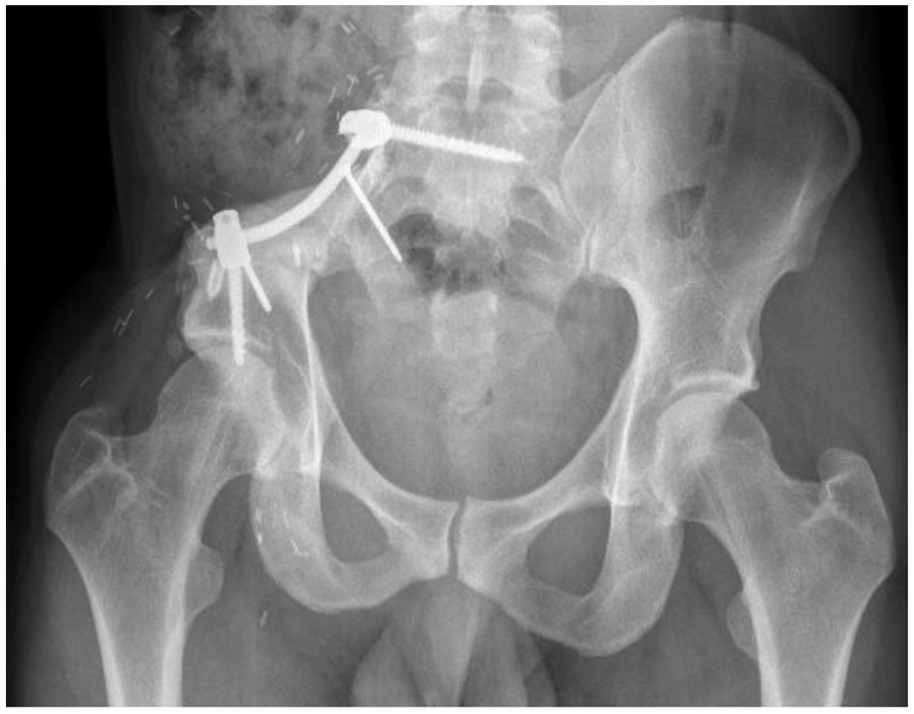
A 51-year-old male with grade 3 central conventional chondrosarcoma of the Enneking classification Types I and II and partially Type III + S treated with internal hemipelvectomy and hip transposition reconstruction.
Type III resection rarely needs any bony reconstruction (Fig. 9). They are well tolerated from the structural perspective as the acetabulum remains in continuity with the axial skeleton. Large resections are associated with herniation of the abdominal and pelvic contents as invariably it is impossible to reconstruct the pelvic floor having removed its bony attachments, and deficiencies in the abdominal wall weaken this structure. 51
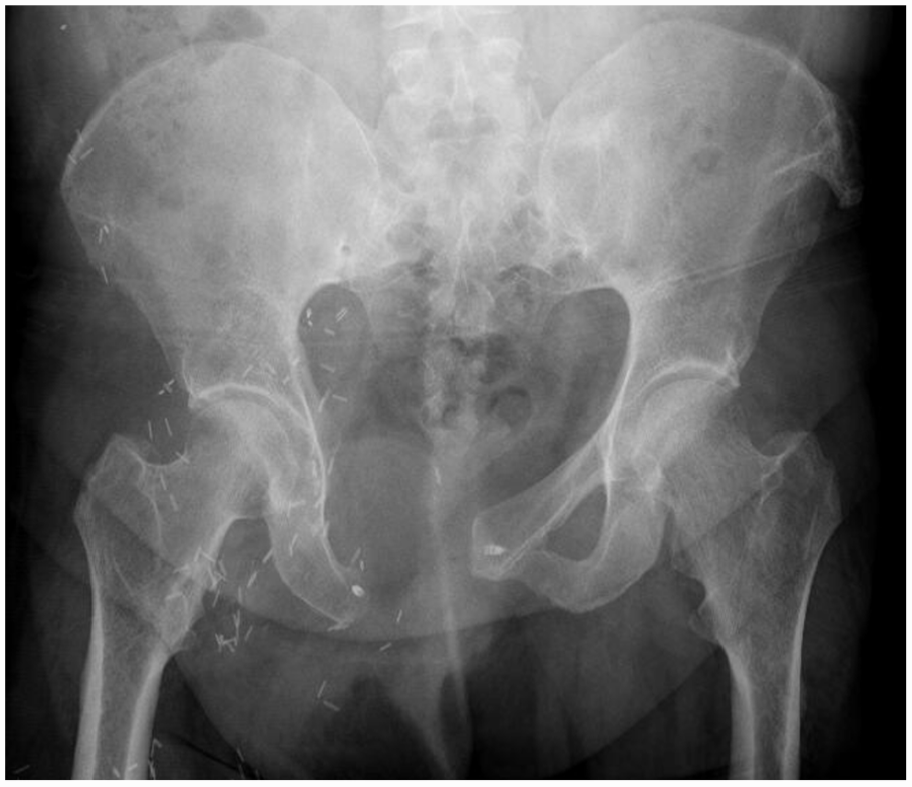
A 59-year-old male with a small dedifferentiated chondrosarcoma of the right pubic bone was resected with Type III without any reconstruction.
All pelvic surgery is associated with short-, medium- and long-term complications, with perioperative mortality rates of approximately 2%–5% and perioperative morbidity rates of 50%.52,53 Common short-term complications include wound necrosis and deep infection. Long-term complications include implant failures and infection. 54 Treatment with external hemipelvectomy versus limb salvage remains controversial. What constitutes a resectable tumor without amputation varies among surgeons, and what constitutes an acceptable functional outcome varies among patients. However, all would likely agree that resections that do not allow for adequate margins are best treated with external hemipelvectomy. Tumors that involve a combination of the surrounding femoral vasculature, lumbosacral plexus, and acetabulum should be strongly considered for amputation. 55 For tumors that can be treated with an effective adjuvant, such as radiotherapy, for example, Ewing’s sarcoma, hemipelvectomy is rarely considered. 7 For tumors with no well-established adjuvant, such as chondrosarcoma, hemipelvectomy is more often considered to ensure complete surgical resection. 56
Endoprosthetic reconstruction
Endoprosthetic reconstruction options after pelvic sarcoma resections, especially resections that include resection of the acetabulum are well established and include saddle prostheses, periacetabular pedestal cup prostheses, custom made prostheses, and total hip type prostheses often with the addition of allograft bone or irradiated and reimplanted autograft bone. Saddle prostheses with its saddle-shaped surface and its axial rotation provide a bearing against the remaining ilium together with unconstrained motion was previously an often-used modality of reconstruction. However, the complication rate was high, as typically the prosthesis would migrate cranially. The prosthesis is now rarely used in pelvic reconstructions. 57 Pedestal cup prostheses based on a modification of the Ring type hip reconstruction prosthesis were introduced to reduce the mechanical complications encountered with periacetabular reconstruction after pelvic tumor resection (Fig. 10). One of the important steps for success is the correct positioning and alignment of the implant stem within the ilium. It provides low rates of aseptic loosening on medium-term follow-up, but infection and dislocation are common complications. The rate of dislocation can be reduced by the use of a mesh or artificial tube.58,59 With the development of computer-aided design (CAD) and computer-aided manufacturing (CAM) techniques, prostheses can be customized to individualized bone defects. Tumor resection and reconstruction with custom made prostheses have unique advantages over traditional techniques in this area with complex spatial geometry. The custom made prosthesis, usually with a porous contact surface, overcomes the shortcomings of traditional manufacturing techniques achieving favorable biomechanical characteristics and promising long-term stability. 45 Allograft or extracorporeally irradiated autografts together with prostheses is also a reasonable option but requires the establishment of a bone bank or the option to irradiate bone during the resection operation. 41 Non-union, fracture or infection are the main complications, resulting in early failure of the reconstruction, but if these are avoided, long-term reconstruction survival is possible. 60
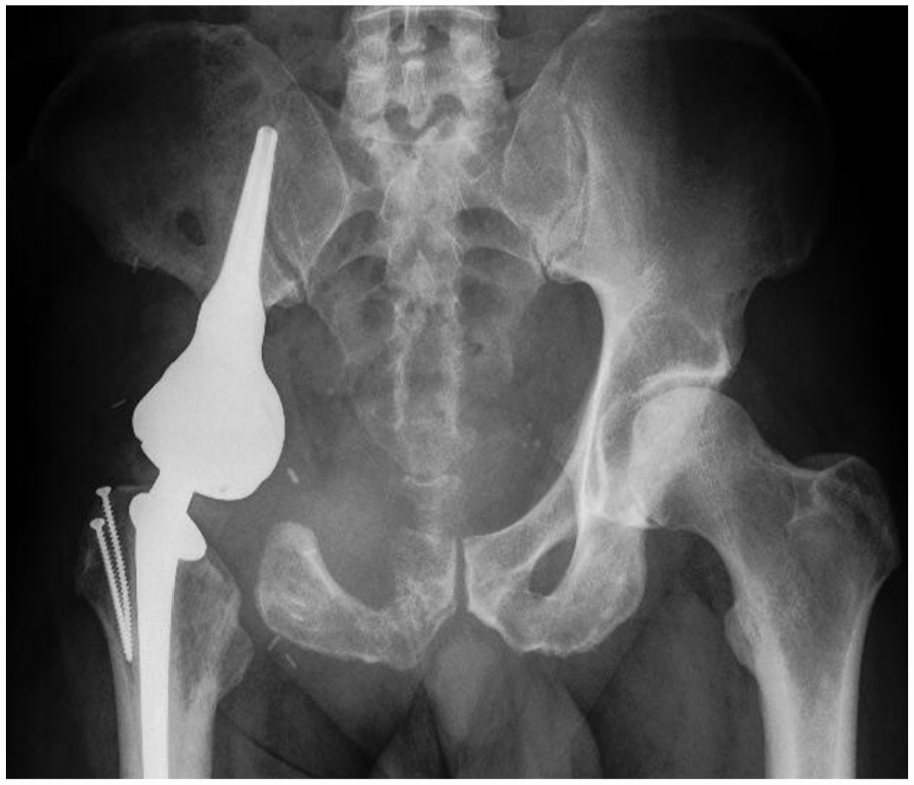
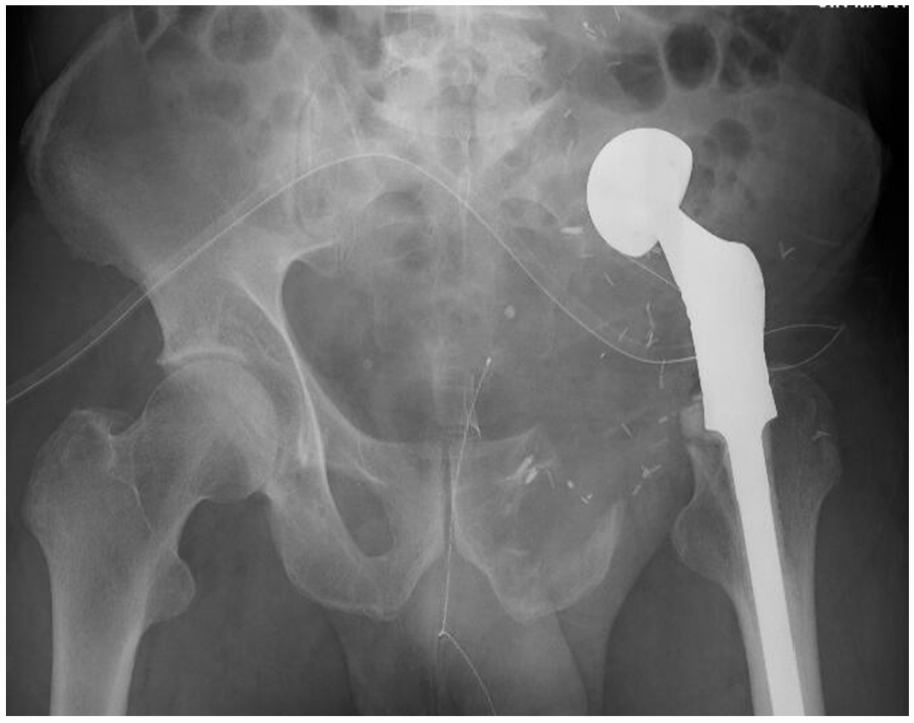
A 35-year-old male with grade 2 central conventional chondrosarcoma of the right periacetabular region, treated with Type II resection and reconstructed with a periacetabular pedestal cup prosthesis.
Navigation
Computer-assisted navigation or computer-assisted tumor surgery (CATS) has been used in orthopedic surgery since the early 2000s, particularly for pelvic sarcoma resection.61,62 The principle is to integrate preoperative and intraoperative imaging and measurements, providing the surgeon with a precise and real-time high-definition model of the tumor and its surrounding anatomy. 63 Without navigation, resection often requires extensive dissection and mobilization of structures to allow control of bleeding and identification of structures around the tumor. The degree of the intraosseous disease cannot be appreciated with the naked eye. The use of navigation is associated with less extensive dissection and improved accuracy in terms of the placement of planned bone resection margins, and therefore, the use of the preoperative scanning coupled with the preoperative resection planning has been shown to reduce the incidence of involved margins at resection.43,46,64
Patient-specific instruments and three-dimensional printing
Patient-specific instrument (PSI) technology has gained favor as an application of three-dimensional (3D) printing having been developed as an alternative to intraoperative navigation. PSI requires preoperative scanning followed by the production of a 3D model of the tumor and the bony anatomy around which a precise cutting jig is designed. The unique anatomy of the patient and the PSI shape allows the surgeon to place the guide only in a predefined position. These contact surfaces must be defined by both surgeon and engineer, considering the surgical approach, the bone exposure, and tumor extension. The jig is pinned to the adjacent bone and acts as a guide for the osteotome or bone saw. Thus, this method reproduces surgical planning with more accuracy than a free-hand technique. 65
The performance and accuracy of PSI have been shown to improve alignment and reduce surgical and operating times, as well as reducing the risk of contamination and waste66,67 The limitations of the use of PSI are governed by the soft tissue extension of the tumor. As this is a static system, there is no real-time imagery for intraoperative referencing as is seen with intraoperative navigation systems. Second, the time lag between the planning scan and the development of the PSIs may mean that the tumor has grown resulting in a mismatch between jigs and the tumor extension, which can result in intralesional resections. The cutting jigs are designed to fit to bony landmarks but can be difficult to fit accurately as a result of the soft tissue extension of the tumor or changes to the bony anatomy between the time of the scan and the time of surgery.68,69
Conclusion
In the modern era, the principle indication for major resections of the pelvis, including hindquarter amputation, is for the local control of malignant disease, most notably primary malignant tumors of bone, but also for metastatic bone disease and hematological malignancies. The most common pelvic primary malignancies are osteosarcoma, chondrosarcoma, and Ewing’s sarcoma. The extent of the resection, including the anatomical structures that must be removed, and those which can remain, is based on the histology of the tumor, the response to neo adjuvant treatment, where indicated, and the preoperative imaging. The method of reconstruction is planned based on the extent of the resection, the risk of recurrence, the use of adjuvant treatments, and the wishes of the patient. The surgical management of tumors arising from the pelvis is technically demanding and invariably results in a reduction in function for the patient. Surgery is not without risk, none more so when considering resection of the pelvis which, in the worst-case scenario, can result in death. Due to the rarity of the disease and the need for a multidisciplinary approach, the treatment should be centralized to units with the skill set and expertise to manage these tumors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval was not required for this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Patient confidentially and informed consent statement is not available.
Trial registration
The presence of clinical trial registration is not available.
