Abstract
Background and objective:
The aim of gender affirmation surgery is to ease gender dysphoria. In transgender men, chest wall masculinization is the most common gender affirmation surgery. The BODY-Q Chest module is currently the only instrument developed to measure health-related quality of life (HRQL) in men undergoing chest wall surgery. Linguistic validation and cultural adaption to Finnish were performed previously. The study aims to validate the BODY-Q Chest module in transgender men who have undergone surgical chest wall masculinization.
Methods:
All transgender patients who underwent chest wall masculinization at Helsinki University Hospital between 2005 and 2018 were invited to the study. The BODY-Q Chest module comprises two scales—chest and nipple. Data were obtained using the BODY-Q Chest module, the 15D questionnaire, and specifically targeted items designed by the authors. The statistical analyses were conducted to exclude selection bias, evaluate validity of the instrument, and compare it to other instruments.
Results:
Of the 220 patients invited, 123 participated in the survey (response rate 56%). Ceiling effects were observed with 18.9% and 20.5% scoring maximum points. Cronbach’s alpha was 0.92 and 0.88 for the chest and nipple scales, respectively. In exploratory factor analysis, both scales loaded to one factor confirming unidimensionality. Correlation with the generic 15D questionnaire was low.
Conclusions:
The BODY-Q Chest module provides valid scores with sufficient consistency and reliability when measuring HRQL in transgender men undergoing chest wall masculinization. Moreover, it offers specificity that existing or generic instruments cannot provide. Ceiling effect was expected due to the postoperative status of participants.
Keywords
Context and relevance
The BODY-Q Chest module is currently the only instrument developed to measure health-related quality of life (HRQL) in men undergoing chest wall surgery. The translation to Finnish has been made. This study aims to provide validation of the BODY-Q Chest module, focusing on postoperative satisfaction after chest wall masculinization in Finnish transgender men.
The instrument obtains sufficient consistency and reliability to provide valid scores in measuring HRQL. BODY-Q Chest module enables specificity in the evaluation and follow-up of transgender men who undergo chest wall masculinization, which generic instruments cannot provide.
Introduction
In transgender people, the incongruence between the sex assigned at birth and the gender gives rise to gender dysphoria and psychosocial distress. As a result, a growing number of patients are seeking gender affirming treatment. In the United States, for example, there was a 597% increase in the number of people undergoing gender affirmation surgery over a 5-year period, with cases increasing from 2740 in 2015 to 16 353 in 2020.1,2 The aim of gender affirming treatment is to decrease the dysphoria and to increase health-related quality of life (HRQL). The World Professional Association for Transgender Health provides guidelines on the standard of care for transgender people. The guidelines include recommendations on hormonal therapy and surgery among others. 3 There are, however, no guidelines on the reporting and evaluating of surgical outcomes in this population, and the practices vary widely. 4
In trans men, the most common surgical procedure is chest wall masculinization. Since the purpose of surgery is to decrease dysphoria and increase HRQL, the measurement of patient-reported outcomes (PROs) is of the utmost importance. Although numerous PRO instruments have been designed to evaluate chest wall surgery when aiming for a feminine result, 5 there is a lack of corresponding instruments for men.
The BODY-Q Chest module is a PRO instrument comprising two scales—chest and nipple. First developed in 2018, the module is designed to measure the outcomes of chest contouring surgery in male patients. 6 Although the instrument is not as yet widely used, it is the only PRO instrument specifically designed for male patients undergoing chest contouring surgery. Furthermore, the module has recently been validated in a Canadian population undergoing chest wall masculinization. 7
In this study, we aim to provide validation of the BODY-Q Chest module, focusing on postoperative satisfaction after chest wall masculinization in Finnish transgender men. This will be a crucial step in guaranteeing validated measurements for assessing the effectiveness of chest wall masculinization among transgender patients.
Methods
Patients
Retrospective patient material, including all patients who underwent surgical chest wall masculinization at Helsinki University Hospital between 1 May 2005 and 31 December 2018, was collected from patient records. A total of 225 patients were identified using the International Statistical Classification of Diseases and Related Health Problems–Tenth Edition (ICD-10) code F64.0 for gender identity disorders combined with procedure codes for chest contouring surgery. From the data, we excluded five patients: two due to having foreign addresses, two due to having an unknown address, and one for non-disclosure. The remaining 220 patients were invited by letter to participate in a cross-sectional survey that comprised an evaluation of the quality of chest wall masculinization surgery at Helsinki University Hospital and the validation of the Finnish BODY-Q Chest module in transgender men.
General information on the participants was obtained from patient records to exclude statistically significant differences between the participants and those who did not respond to the survey. Information on age, height, body weight, cup-size, psychiatric and somatic comorbidities, medication, smoking, occupation, follow-up time, technical details of the surgery performed, complications, re-operations, attendance to genital surgery, and regret was collected. The data were collected from the Helsinki University Hospitals patient records which cover all records from the hospital. Collection of data was finalized in October 2020, and all records available at that time were used.
The invitation letters also contained a general information letter, an informed consent form, the BODY-Q Chest module, the 15D HRQL questionnaire, and a prepaid return envelope. The primary invitations were sent in May 2020 and those who did not reply to the first letter received a second invitation in September 2020. As gender affirmation surgery is not performed on minors in Finland, all participants were more than 18 years old.
Instruments
The BODY-Q Chest module is an HRQL instrument comprising two scales. The chest scale contains 10 items measuring subjective satisfaction with the appearance of the chest wall and one optional item evaluating scars in the case of previous surgery. The nipple scale contains five items evaluating the nipples. All items are completed on a scale from one to four—with one being very dissatisfied, two being somewhat dissatisfied, three being somewhat satisfied, and four being very satisfied. In cases of missing items, the empty items are completed using the mean of the completed items, when the missing data are less than 50%. If the missing data are 50% or more, the questionnaire is excluded from the analyses.
The total score is then converted into equivalent Rasch transformed score, according to the instructions of the instrument. Regarding the chest scale, the conversion included items one to ten, whereas the conversion of the nipple scale included all five items. The score had a range of 1–100 for the chest scale and 0–100 for the nipple scale. We obtained permission to use the Finnish BODY-Q Chest module from the copyright holders. The translation to Finnish and linguistic validation had been previously performed. 8
The 15D is a generic instrument for measuring HRQL. 9 The instrument comprises 15 health-related dimensions: mobility, vision, hearing, breathing, sleeping, eating, speech, elimination, usual activities, mental function, discomfort and symptoms, depression, distress, vitality, and sexual activity. The dimensions are divided into five levels from best to worst. Missing dimensions were excluded from the analyses.
Statistics
The statistical analyses were completed using RStudio 1.4.1106 with packages “ggplot2,” “devtools,” “ggbiplot,” “lavaan,” “semPlot,” “psych,” “pspearman,” and “psychometric.” Reporting of the results follows the STROBE and COnsensus-based Standards for the selection of health status Measurement INstruments (COSMIN) checklists.10,11 The data are reported as medians, inter quartile ranges (IQRs), 95% confidence intervals (95% CI), counts (N), and percentages (%).
Patients were divided into two groups, depending on whether they assessed the questionnaires or not. To exclude selection bias, the sociodemographic data of the groups were compared with Fisher’s exact test for categorical variables and the Mann–Whitney U test for continuous variables.
Ceiling and floor effects were evaluated with proportions of maximum and minimum scores. The ceiling or floor effect was confirmed if >15% answered the maximum and minimum scores, respectively. Internal consistency was determined using Cronbach’s alpha. Values exceeding 0.8 were considered sufficient. 12
Linear regression models were used to evaluate the correlation between the scales of the BODY-Q Chest module and the time from the primary operation. The autocorrelation between the two scales was also evaluated. We reported non-adjusted R-squared, β, 95% CI for β, and the p-value for β. R-squared was determined as very weak (<0.02), weak (0.02–0.12), moderate (0.13–0.26), and substantial (>0.26) in agreement with Cohen’s reference values. β was interpreted as small (<0.1), moderate (0.1–0.5), and strong (>0.5) effect.
The correlation between 15D dimensions and the scores of the two scales was evaluated with Spearman’s correlation coefficient (Spearman’s p), and CIs were computed with the “CIr”-function. The correlation was interpreted as follows: 0.0–0.1 negligible, 0.1–0.4 low, 0.4–0.7 moderate, 0.7–0.9 high, and 0.9–1.0 very high. 13 Exploratory factor analysis (EFA) with varimax rotation was conducted to evaluate internal structure. The factors were chosen according to the Kaiser criteria, including factors with a minimum eigenvalue of one. 14 In loading values, a cutoff of 0.4 was used.
Ethical considerations
The protocol of this study was approved by the Ethics Committee of Helsinki University Hospital, Finland, decision no. HUS/2711/2019.
Results
Of the 220 patients invited to the study, 123 patients responded, giving us a response rate of 56%. The follow-up time from primary operation to replying to the questionnaire had a median of 63 (IQR = 60) months. The sociodemographic features of those patients who responded and those patients who did not are compared in Table 1.
Sociodemographic characteristics.
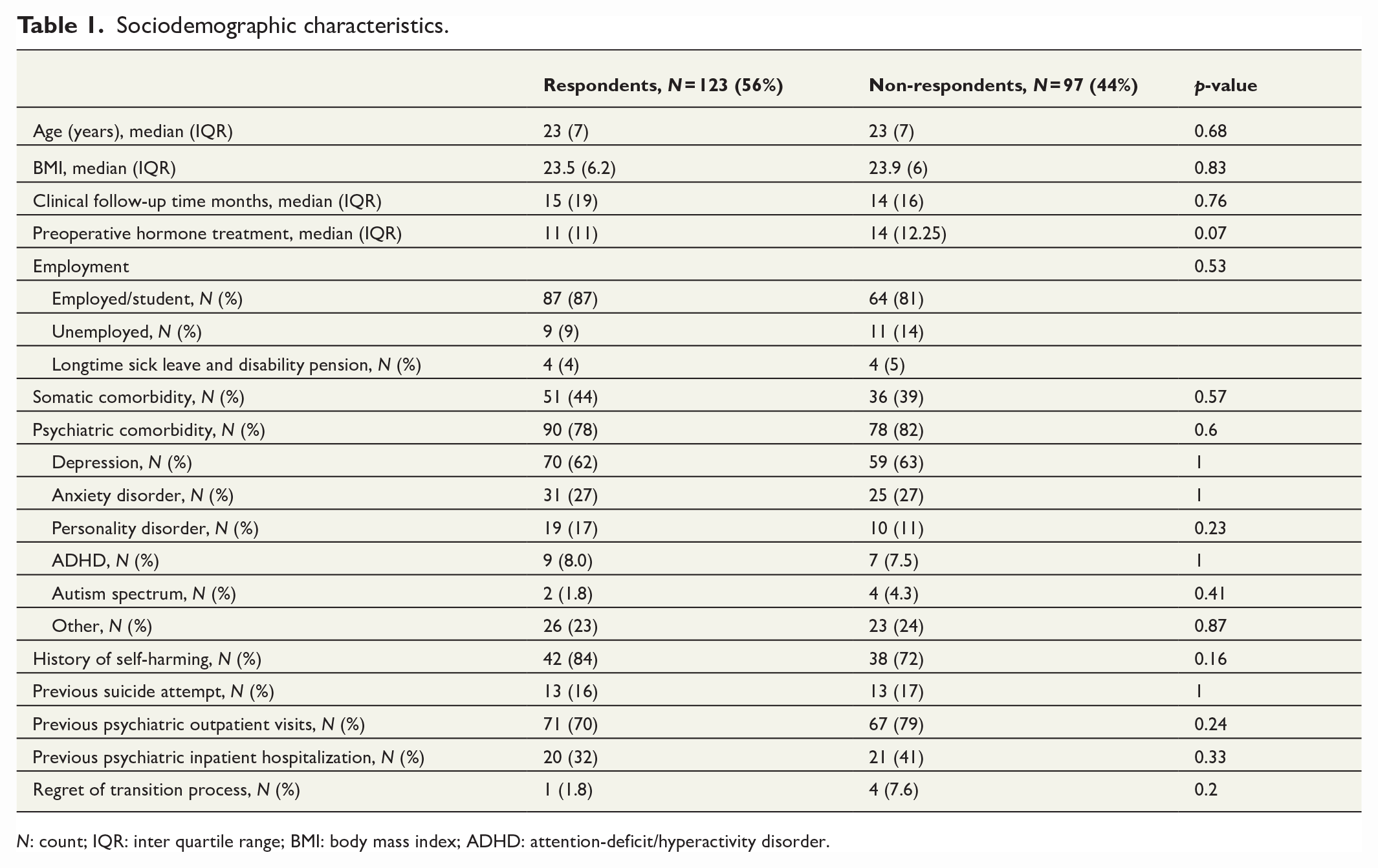
N: count; IQR: inter quartile range; BMI: body mass index; ADHD: attention-deficit/hyperactivity disorder.
Score distributions of the scales are presented in Figures 1 and 2. Regarding the chest scale, six patients had missing data: missing data > 50% (N = 1) and missing data < 50% (N = 5). Concerning the nipple scale, three patients had missing data: missing data > 50% (N = 1) and missing data < 50% (N = 2). On the chest scale, patients scored between 25 and 100 points with a median score of 76 and IQR = 29. The distribution was skewed to the high points and showed a trimodal structure. Maximum points were scored by 23 patients (19%), showing a clear ceiling effect (>15% scoring maximum points). No patients scored minimum points. Thus, no floor effect was observed. On the nipple scale, patients scored between 0 and 100 points. Here, the scores were also skewed toward the high points, leading to non-normal distribution. The median score was 68 and IQR = 34. One patient scored minimum points, indicating no floor effect. Maximum points were scored by 25 patients (20%), showing a clear ceiling effect. The median, IQR, proportion of maximum and minimum scores, and correlation to total score for all individual items are presented in the Supplementary Material. A shift toward high points can be observed here as well. Cronbach’s alpha was 0.92 and 0.88 for the chest and the nipple scales, respectively.
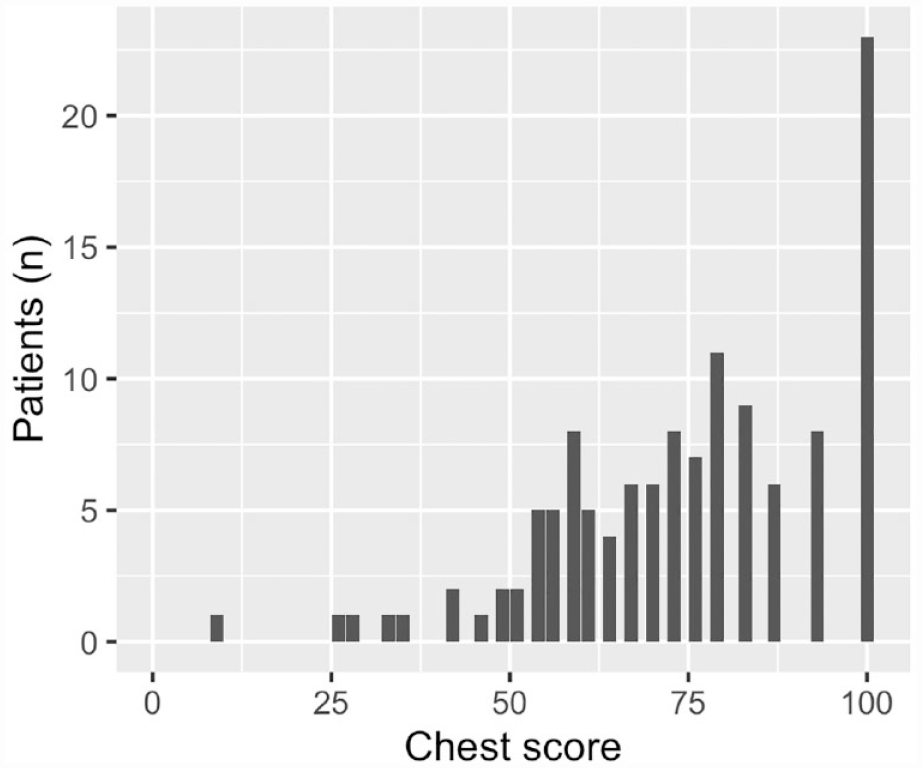
Score distribution of the chest scale.
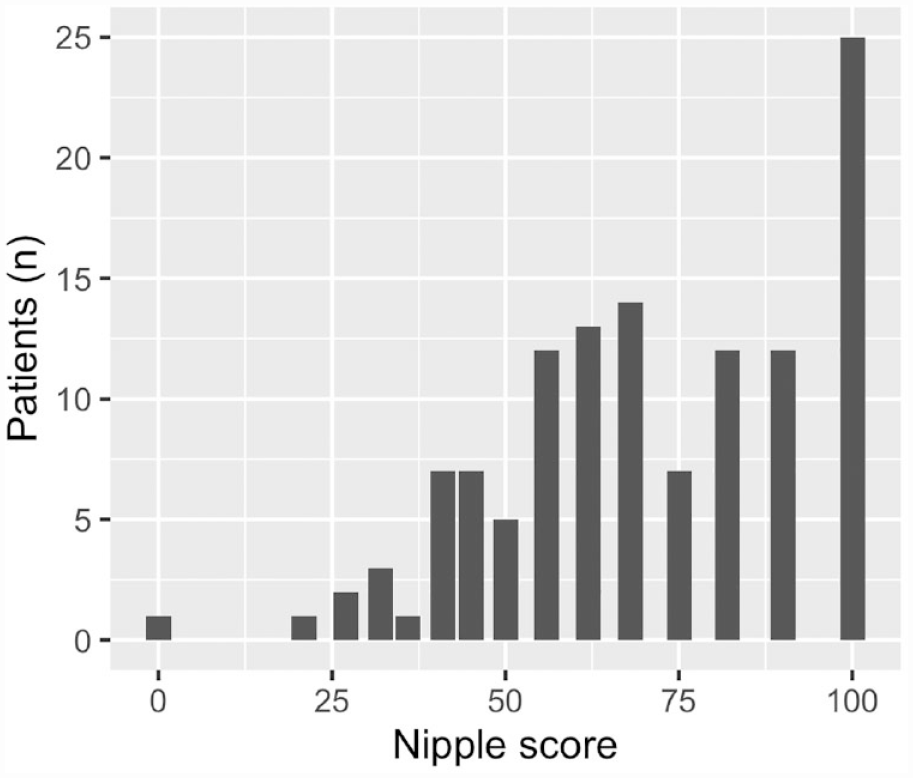
Score distribution of the nipple scale.
Figures 3 and 4 show linear regression between total score and the time from surgery expressed in months. Regarding the chest scale, the regression model is considered statistically significant having a p-value of 0.01. The non-adjusted R-squared shows a weak goodness of fit, and β indicates the effect to be small and negative. On the nipple scale, statistical significance was not achieved.
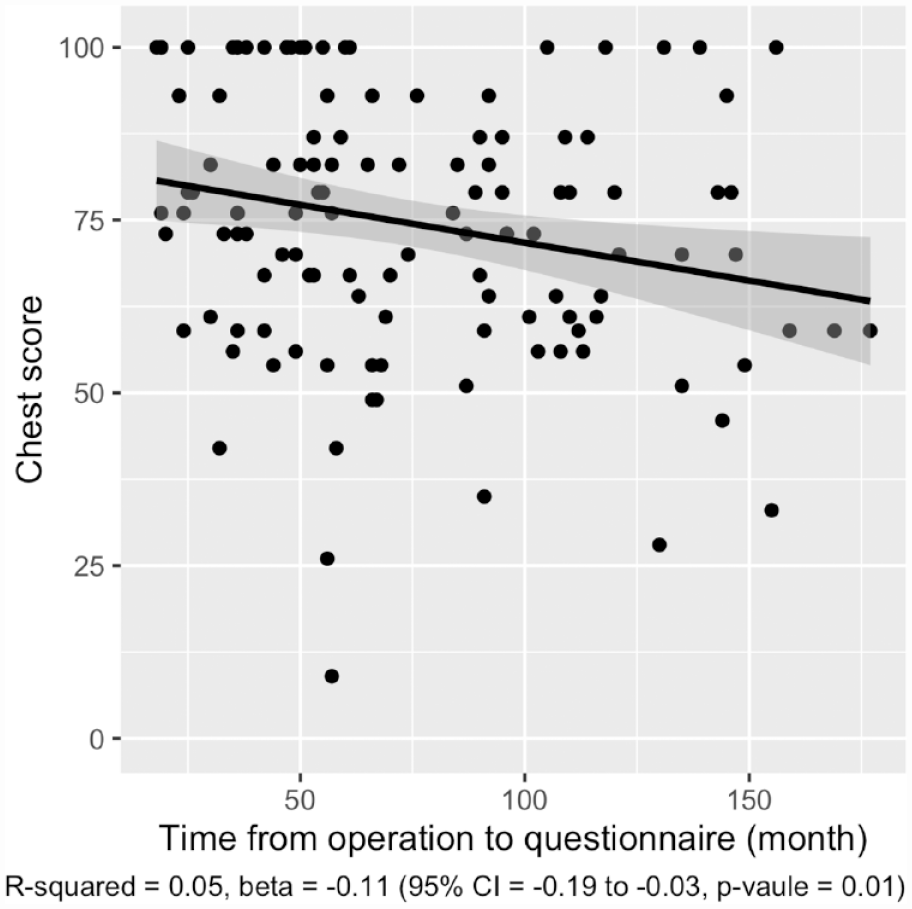
Correlation between chest scale score and time from operation. Gray area showing 95% CI.
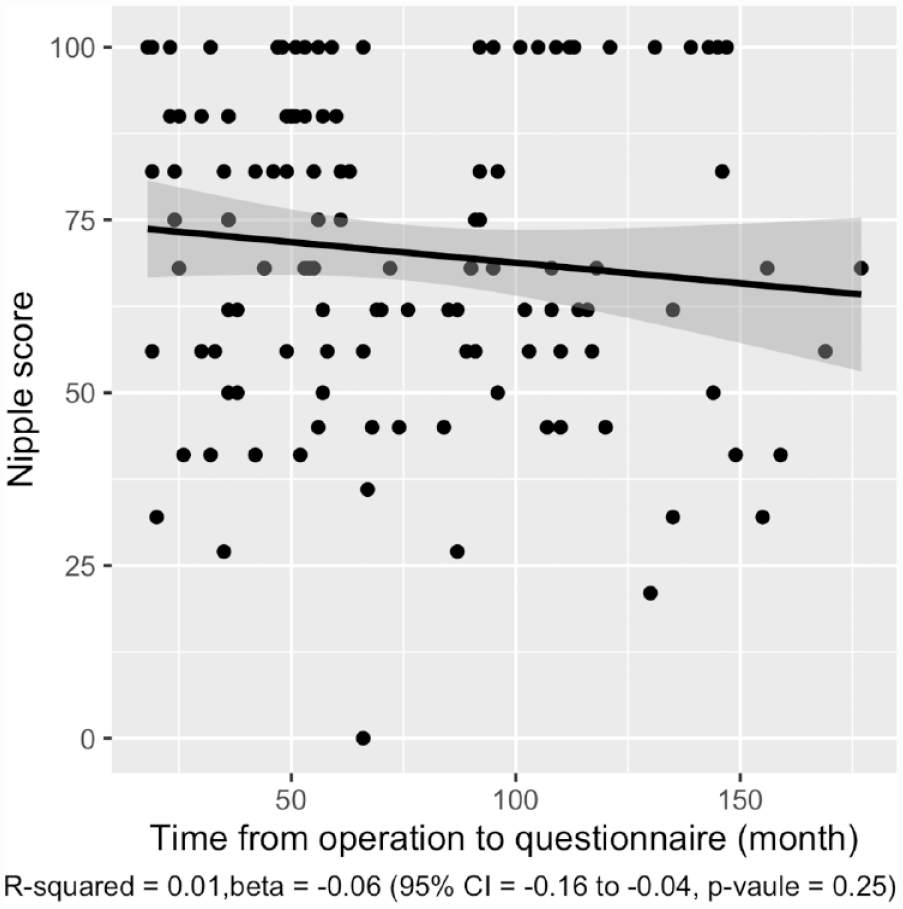
Correlation between nipple scale score and time from operation. Gray area showing 95% CI.
Correlations between the dimensions of the 15D and the scores of each scale are presented in Table 2. Regarding the 15D, one patient had missing dimensions. The table comprises Spearman’s p and 95% CI. Spearman’s p varied from 0.04 to 0.34 and from 0.01 to 0.22 for the chest and nipple scales, respectively. On the chest scale, the strongest correlation was observed with dimension 14. However, on the nipple scale, the correlation was the strongest with dimensions 11, 12, and 14. All patients reported equally on dimension 6, and therefore, correlation could not be measured.
Spearman’s p between 15D dimensions and scores of the chest scale and nipple scale.
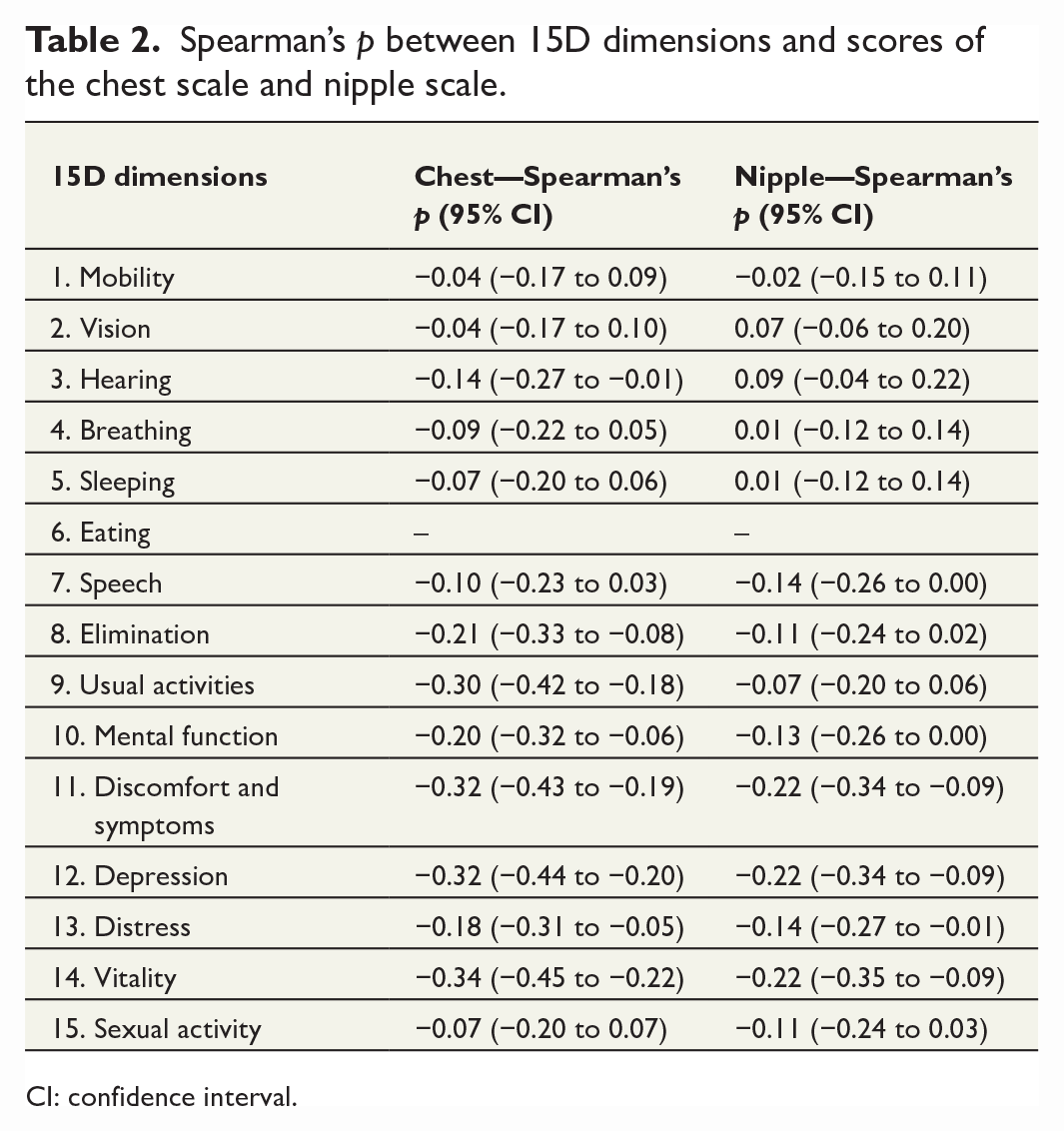
CI: confidence interval.
Figure 5 presents the linear regression between the chest scale and the nipple scale, where high scores in one scale associates with high scores in the other. Statistical significance is confirmed with p = 6.36e−13.
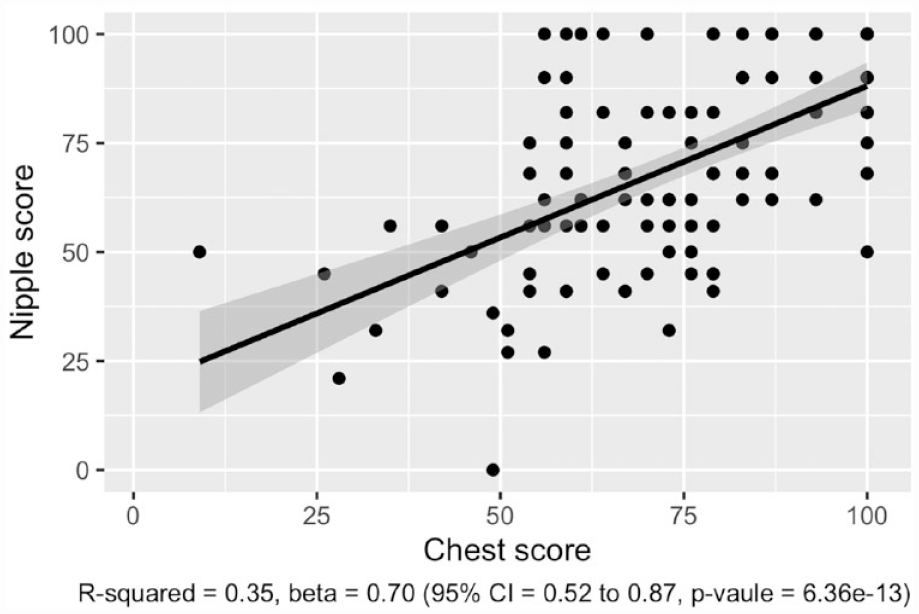
Correlation between scores of the chest and nipple scales. Gray area showing 95% CI.
The EFA found both scales had one component each receiving an eigenvalue over one. Observing the chest scale, component one (eigenvalue = 6.0) caused 55% of the total variance. Component one loaded rather evenly to all items of the chest scale with the lowest loading for item two (0.46). Moreover, the nipple scale loaded to one separate component (eigenvalue = 3.4). On the nipple scale, component one explained 60% of the total variance. Item-specific loading values are presented in the Supplementary Material.
Discussion
In this study, the BODY-Q Chest module showed sufficient consistency and reliability when applied to transgender men after chest wall masculinization. The ceiling effect observed was expected due to the postoperative set up and does not therefore threaten the accuracy of the instrument. The result is in line with the previous study using the BODY-Q Chest module in a transgender population, where ceiling effect was also observed. 7 Moreover, the same phenomenon has also been registered in a population that included gynecomastia and weight loss patients, with 15% and 17% scoring maximum scores. 6 Hence, the results appear to confirm the proper postoperative outcome after chest wall masculinization, and the absence of ceiling effect would have been highly suspicious. However, a change in the long-term rehabilitation could be hard to assess using BODY-Q Chest module due to the skewed scores. Sufficient internal consistency was ensured with Cronbach’s alpha, and EFA confirmed a unidimensional structure. Since there is a scarcity of previous studies on the subject, the comparison to previous findings leans heavily on the development process of the BODY-Q Chest module and the recent validation of the module in the Canadian population.6,7
Correlation between the dimensions of the 15D and the scores of the scales was negligible or low at best. This emphasizes the differences between the 15D, being a comprehensive quality-of-life instrument but not sensitive enough, and the BODY-Q Chest module having a specific aesthetic target. This signifies the demands on a PRO instrument that explicitly measures an aesthetic result and enables the detection of relevant outcomes after chest wall masculinization.
In general, the postoperative aesthetic result is considered to improve with time. Surprisingly, when we analyzed the correlation between the BODY-Q Chest module scores and the time from chest wall masculinization in Figure 3, we found the score to be higher among those patients who had recently been operated. However, due to the cross-sectional nature of this study, conclusions about the development of scores over time cannot be made.
Regarding autocorrelation, this study found substantial goodness of fit and a strong positive correlation between the two scales. Thus, it is justified to include both scales in the BODY-Q Chest module.
Clinical significance
This validation enables the use of the BODY-Q Chest module questionnaire for the study of the development of HRQL in transgender men undergoing chest wall masculinization. As a continuation, we aim to further study the development of HRQL in a similar population in a prospective design. However, the test–retest reliability of the BODY-Q Chest module remains to be studied. Also, after our surveys were performed, the development of a new PRO instrument designed for transgender patients specifically, GENDER-Q, has started. However, GENDER-Q is neither yet in clinical use nor has it been translated to Finnish. For now, BODY-Q chest module is still the only PRO instrument in clinical use and enables us to report and compare HRQL in transgender men and to further improve the quality of treatment in this rapidly increasing patient group. BOQY-Q chest module will also offer a suitable reference instrument for developing new tools to evaluate satisfaction with chest wall masculinization.
Strengths and weaknesses
The response rate of 56% can be considered good. Moreover, we found no statistically significant difference between respondents and non-respondents. Hence, the data are considered to represent the population of interest. Several clinical features were not, however, included. As this study focused on the validation of the BODY-Q Chest module for masculinization, data on surgical techniques as well as complications and re-operations will be the focus of a separate study. Although the response rate can be considered sufficient, face-to-face recruitment would most likely have produced higher participation, but this was not possible in this study setting. A characteristic of our patient group, the operation usually includes the shaping of both the nipple and the areola. Thus, the lack of areolar items is a limitation that should be taken into consideration when applying the BODY-Q Chest module to transgender men. We were also unable to examine test–retest reliability due to the lack of two separate completions of the instrument. This was, however, outside the scope of this study. Also, the validation in non-binary patients remains to be studied.
Conclusion
To conclude, the BODY-Q Chest module provides valid scores in measuring HRQL in transgender men who have undergone chest wall masculinization. The instrument obtains sufficient consistency and reliability. Furthermore, the BODY-Q Chest module approaches the subject with a specificity that existing instruments cannot provide. The BODY-Q Chest module is therefore a reliable PRO instrument that enables the evaluation and follow-up of transgender men who undergo chest wall masculinization. BOQY-Q chest module will also offer a suitable reference instrument for developing new tools to evaluate satisfaction with chest wall masculinization.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969231176111 – Supplemental material for Validation of the BODY-Q Chest module in Finnish trans men undergoing chest wall masculinization
Supplemental material, sj-docx-1-sjs-10.1177_14574969231176111 for Validation of the BODY-Q Chest module in Finnish trans men undergoing chest wall masculinization by Mirjam Saarinen, Kaisu Ojala, Sinikka Suominen and Jussi Repo in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969231176111 – Supplemental material for Validation of the BODY-Q Chest module in Finnish trans men undergoing chest wall masculinization
Supplemental material, sj-docx-2-sjs-10.1177_14574969231176111 for Validation of the BODY-Q Chest module in Finnish trans men undergoing chest wall masculinization by Mirjam Saarinen, Kaisu Ojala, Sinikka Suominen and Jussi Repo in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank Anders Vuorijoki for statistical consulting and Linda Laamanen for contributing to the data collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was received from the Finnish Medical Society (Finska Läkaresällskapet) and the Musculoskeletal and Plastic Surgery Research Center Helsinki, Helsinki University Hospital, and the University of Helsinki, Finland. Open access funded by Helsinki University Library. The funders were not involved in planning or implementing the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
