Abstract
Background and objective:
Consensus on management of pelvic sentinel lymph nodes (PSLNs) has not been reached and thus the extent of sentinel lymph node biopsy (SLNB) of the groin in melanoma patients varies among centers and surgeons. Lymphatic drainage to PSLNs is often identified in, but the diagnostic and clinical relevance of PSLNs has been debated. Our aim was to determine if the presence of PSLNs affected recurrence or survival rates in patients with melanoma in the lower extremities.
Methods:
This retrospective study consisted of 702 patients with cutaneous melanoma operated between 2005 and 2018. Of these, 134 patients with melanoma in the lower extremities were included in the study. Images of lymphoscintigraphy and SPECT-CT studies were thoroughly observed together with surgery reports to define the status of SLNs.
Results:
Overall, 85 of 134 patients went through SLNB and 28 of them had PSLN identified. Two had their PSLN removed, which led 26 patients with PSLN to be compared to the 57 who did not have PSLN identified. We did not find statistically significant differences in overall recurrence (26.9% versus 28.0%, p = 1.00), regional nodal recurrence (11.5% versus 15.8%, p = 0.67), local or in-transit recurrence (19.2% versus 8.8%, p = 0.17), or distant recurrence rates (15.4% versus 19.3%, p = 0.66). Disease-free survival did not differ between the groups (median 23.0 (IQR 15.0–39.0) versus 19.0 (IQR 10.3–61.8) months, p = 0.82). Likewise, there was no statistically significant difference in melanoma-specific 5-year survival (78.6% versus 87.2%, p = 0.42).
Conclusions:
We did not find more frequent recurrence, shorter disease-free survival, or poorer melanoma-specific survival in patients with drainage to PSLN. Our findings strengthen the evidence that PSLNs should not be routinely biopsied if they are not the first-tier nodes.
Keywords
Summary
Having a pelvic sentinel lymph node (PSLN) does not seem to affect survival or recurrence rates in patients with melanoma in lower extremities when PSLNs were left under observation.
Context and relevance
No consensus or guideline of how pelvic sentinel lymph nodes (PSLNs) should be managed in melanoma patients does exist. A few earlier studies have found that having a PSLN does not affect melanoma-specific survival or groin recurrence rates. It has been suggested that in many cases, PSLN biopsy could be omitted. Although the earlier studies mostly had PSLNs routinely biopsied, but in our study, they were left under observation, which provides a new perspective. To standardize the clinical practices of managing PSLNs, coherent scientific evidence is needed, and our study brings us a bit closer to it.
Introduction
Sentinel lymph node biopsy (SLNB) is a standard procedure along with a wide excision for cutaneous melanomas greater than 0.8–1 mm in Breslow’s depth or those with lower thickness showing ulceration of the tumor.1–3 SLNB offers valuable prognostic information by enabling accurate staging, thus leading to more targeted adjuvant therapies and prolonging disease-free survival. 4 The sentinel lymph node (SLN) is defined as the node that first receives the drainage from the site of the tumor, thus being the most favorable site of metastasis. 5 Quite often more than one SLN is identified, and it is recommended that they should be excised if the radioactive count is more than 10% of the “hottest” SLN. 6
Melanomas of the lower extremities have a typical pattern of lymphatic drainage to the ipsilateral groin most commonly first receiving superficial inguinal lymph nodes. 7 However, drainage to pelvic nodes, including deep inguinal and iliac lymph nodes, can be identified remarkably often. However, 23% of cases showed drainage to the pelvic sentinel lymph nodes (PSLNs) when the primary melanoma tumor located in the lower extremities. 8 Studies that have also taken truncal melanomas into account have found percentages of between 8% and 69.8%.9–12
Although the drainage to PSLNs is quite common, it is relatively rare to find metastasis at these sites. According to earlier studies, in which PSLNs were routinely resected 2.9%–7.5% were positive for nodal metastasis in pathological evaluation.9–11,13 Having metastasis only in PSLNs without superficial node involvement is infrequent; 0%–0.6% had metastasis in the PSLNs if the superficial sentinel lymph node (SSLN) was negative, when the tumor drained both to the SSLNs and PSLNs.9,11,14 However, Schuitevoerder et al. 9 demonstrated that, if the drainage goes directly only to the pelvic nodes, 17.3% of the PSLNs examined showed metastasis.
The routine retrieval of PSLNs still varies among centers and surgeons. The challenging anatomical location of PSLNs makes the surgical approach more demanding, and retrieval of PSLNs has been shown to significantly prolong operative time.14,15 However, SLNB of the groin area has the highest incidence of complications (14.9%) compared to other regions. 16
When PSLNs are routinely retrieved, association with more frequent groin recurrence or poorer melanoma-specific survival has not been found.10,11 Karakousis et al. 8 did not perform routine SLNB, but in the cases where this procedure was performed, they did not find a difference in pelvic recurrence rates but found slightly shorter disease-free survival in patients with PSLN but histologically negative SSLN. It has been suggested that PSLNs should be retrieved if there is an isolated PSLN without SSLN, or they are the more or equally radioactive to the SSLNs.9,11,14
Since the indication of SLNB is to define whether patient needs further treatments and adjuvant immunotherapy, should the biopsy of PSLNs impact melanoma staging and bring added value when compared biopsying only SSLNs. PSLNs have been shown to have very little impact on staging, which speak against their routine retrieval.10,11 The main aim of our study was to determine whether there is any effect on recurrence or survival when drainage to PSLNs is identified, but they are not removed.
Methods
The study was approved by the North-Savo institutional review board (ID: 62/2019). The study was retrospective, so ethical approval was not applied, and patient consents were not asked for. Patient identification information was kept separately from the file containing collected data, stored safely, and afterward destroyed so that patient confidentiality was guaranteed.
In this retrospective study, we evaluated treatment outcomes of patients who had a melanoma tumor in the lower extremities. All patients were operated at Kuopio University Hospital between 2005 and 2018. During that time, 702 patients with invasive, biopsy-confirmed cutaneous melanoma were operated, and of these, 138 were in the lower extremities. The inclusion criteria were also that the melanoma had to be operable and a wide excision was made with the intention of curative effect, which excluded four patients. In total, 134 patients fulfilled the criteria and were selected for the final study.
Study material was collected in 2020 from the digital patient record system. Medical reports, surgery reports, pathology reports, and digital images were included. Images of lymphoscintigraphy, and in most cases SPECT-CT, were reviewed by a single senior plastic surgeon (H.N.) and compared with the information from the surgery report to distinguish pelvic and superficial inguinal sentinel lymph nodes. According to these findings, the comparison groups with drainage to PSLNs and without drainage to PSLNs were formed.
During the study period for the identification of SLNs, Technetium-99m nanocolloid was used together with blue dye. In our academic teaching hospital, as a common practice, surgeons did not perform routine retrieval of PSLNs, unless they were particularly suspicious and if an SSLN with higher radioactive count was detected and removed. Two patients, who had their PSLN removed, were excluded from the drainage to PSLNs comparison group.
Follow-up time was determined by the last mention of either clinical examination or treatment procedure performed due to melanoma in the patients’ medical records. Disease-free survival was calculated from the time of re-excision to the first recurrence of melanoma. Recurrence was defined as pathologically confirmed recurring disease in the skin or lymph nodes, or metastasis at any site. In the case of distant metastasis and lymph nodes, highly suspected progressive disease on a CT scan was also counted. The melanoma-specific survival was calculated from the time of re-excision to death caused by melanoma.
Data were analyzed with IBM SPSS Statistics version 27. Parametric variables were analyzed using the Mann–Whitney U test. For nominal variables, Fisher’s exact test was used. The p-values less than or equal 0.05 were interpreted as indicating significant differences.
Results
In our study population, the percentage of lower extremity melanomas was 19.7% (138 of 702 patients), and 134 who fulfilled the study criteria were chosen for further analysis. Overall, 73.9% (99 of 134) of patients were female and the median age of the whole group was 61.5 (interquartile range (IQR) 50.0–72.0) years. Median follow-up time was 26.0 (IQR 22.0–49.0) months. In the group, 45.6% of all tumors were located either in the anterior thigh (29.9%) or shin (15.7%), which were the largest subgroups (Table 1).
Clinicopathologic features and long-term outcomes of patients with melanoma tumor in lower extremities.
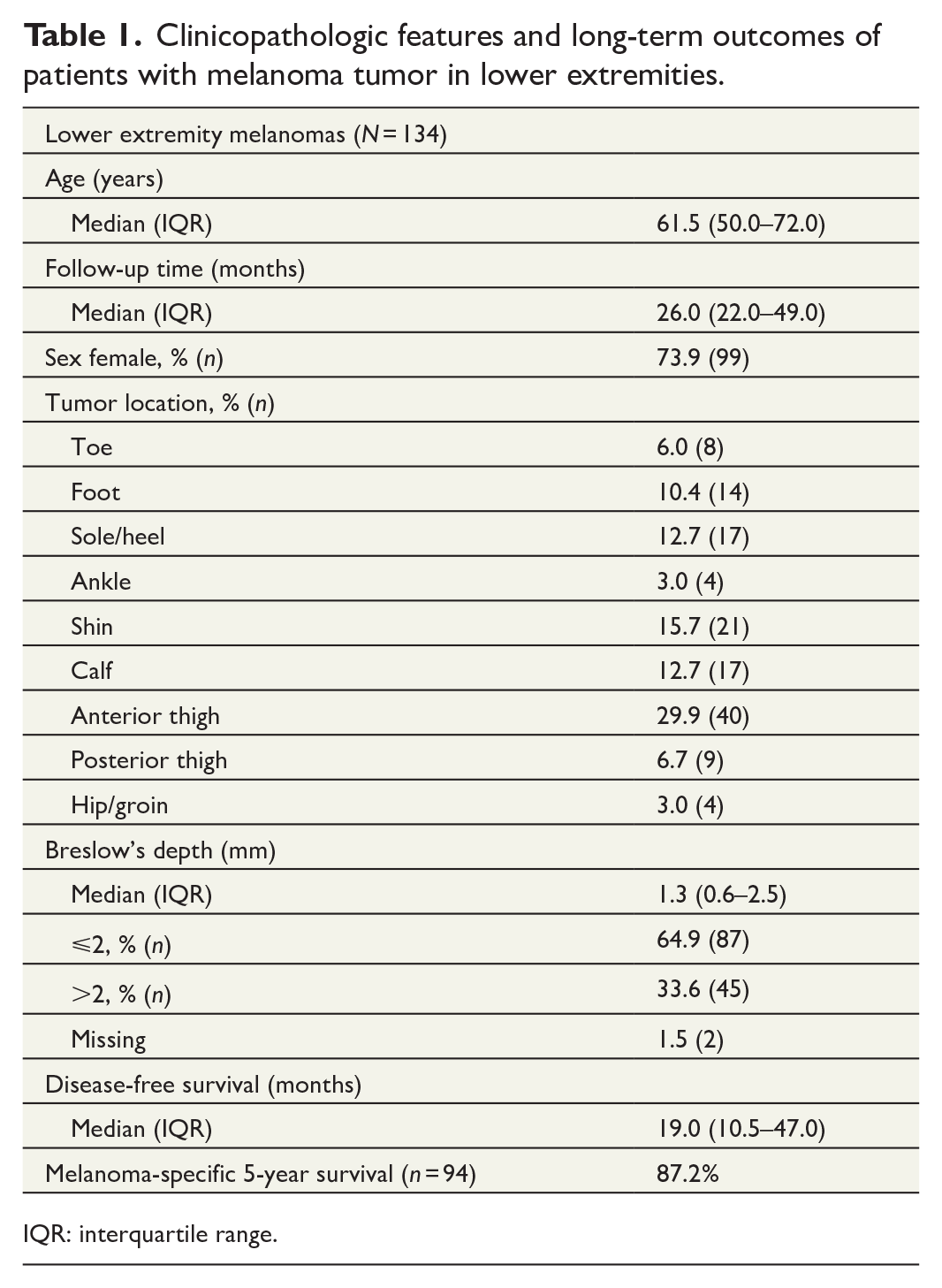
IQR: interquartile range.
Below-knee melanomas
Overall, 81 patients had a tumor located below the knee and 53 above. We did not find a statistically significant difference in age (p = 0.69) or sex (p = 0.14) between these groups. Below-knee melanomas had deeper Breslow’s depth, with a median of 1.5 (IQR 0.8–3.0) mm, than above-knee melanomas with median of 1.0 (IQR 0.6–2.0) mm (p = 0.03). SLNB was also performed more often in the group of below-knee melanomas (71.6%, 58 of 81) compared to 50.9% (27 of 53) in the group of above-knee melanomas (p = 0.02). PSLNs were found in 36.2% (21 of 58) of the below-knee group and 25.9% (7 of 27) in the above-knee group, but this difference was not statistically significant (p = 0.46). Follow-up time was longer for tumors located below the knee: median 29.0 (IQR 21.5–57.0) versus above the knee: 24.0 (IQR 21.0–35.0) months (p = 0.05). Melanoma-specific 5-year survival was 83.9% in below- and 92.1% in above-knee melanomas, including 94 patients who completed at least the 5-year follow-up for the study. The difference between these groups was not statistically significant (p = 0.35) (Table 2).
Clinical and histopathologic factors associated with the location of the melanoma under or below the knee.
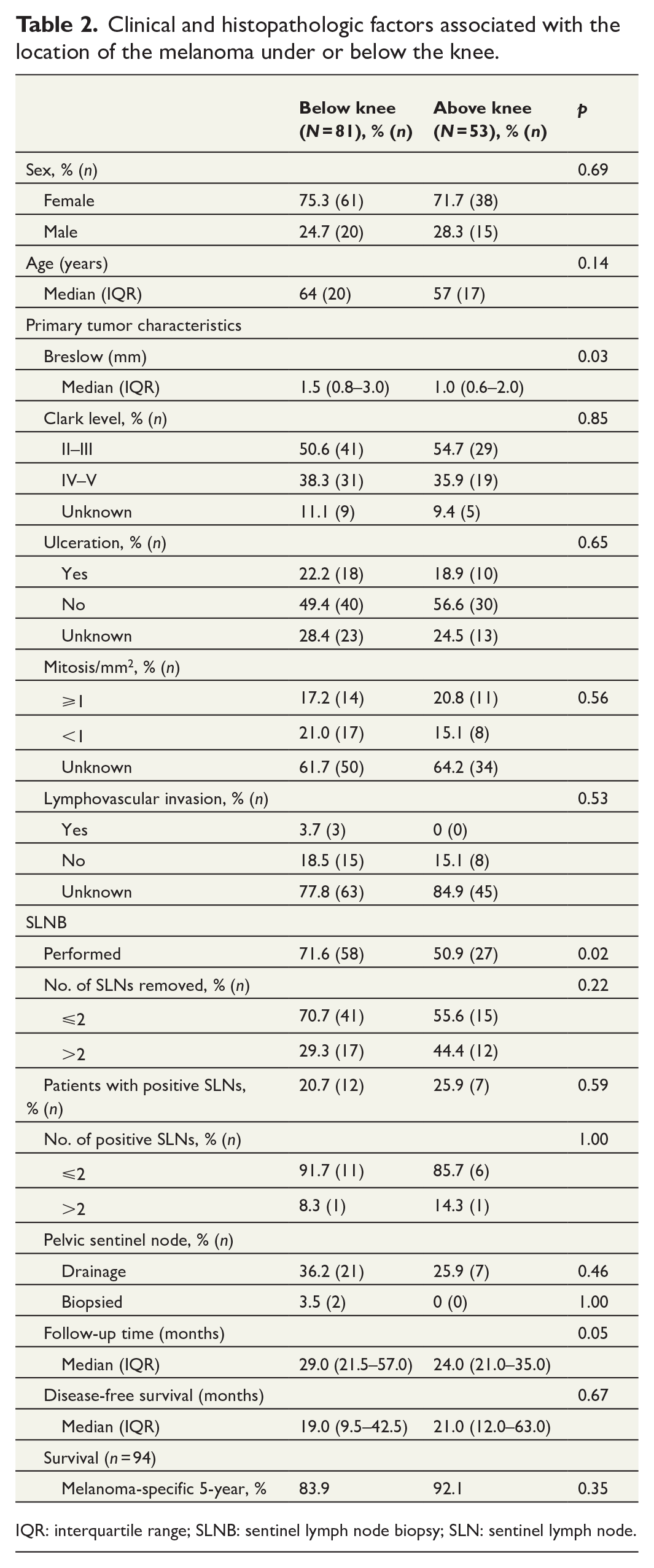
IQR: interquartile range; SLNB: sentinel lymph node biopsy; SLN: sentinel lymph node.
PSLNs
In total, 85 of 134 patients were subjected to SLNB. Of these, 28 (32.9%) had drainage to PSLNs, whereas 57 did not. Of the 28 patients who had PSLNs, two had PSLNs removed and 26 had PSLNs left under observation. Of the two patients who had PSLNs removed, one had histologically melanoma-positive lymph nodes on pathological examination. Of the 26 patients with PSLN, only one had later metastasis found in the pelvic lymph nodes, and the other 25 did not develop metastasis in the pelvic lymph nodes during follow-up (Fig. 1).
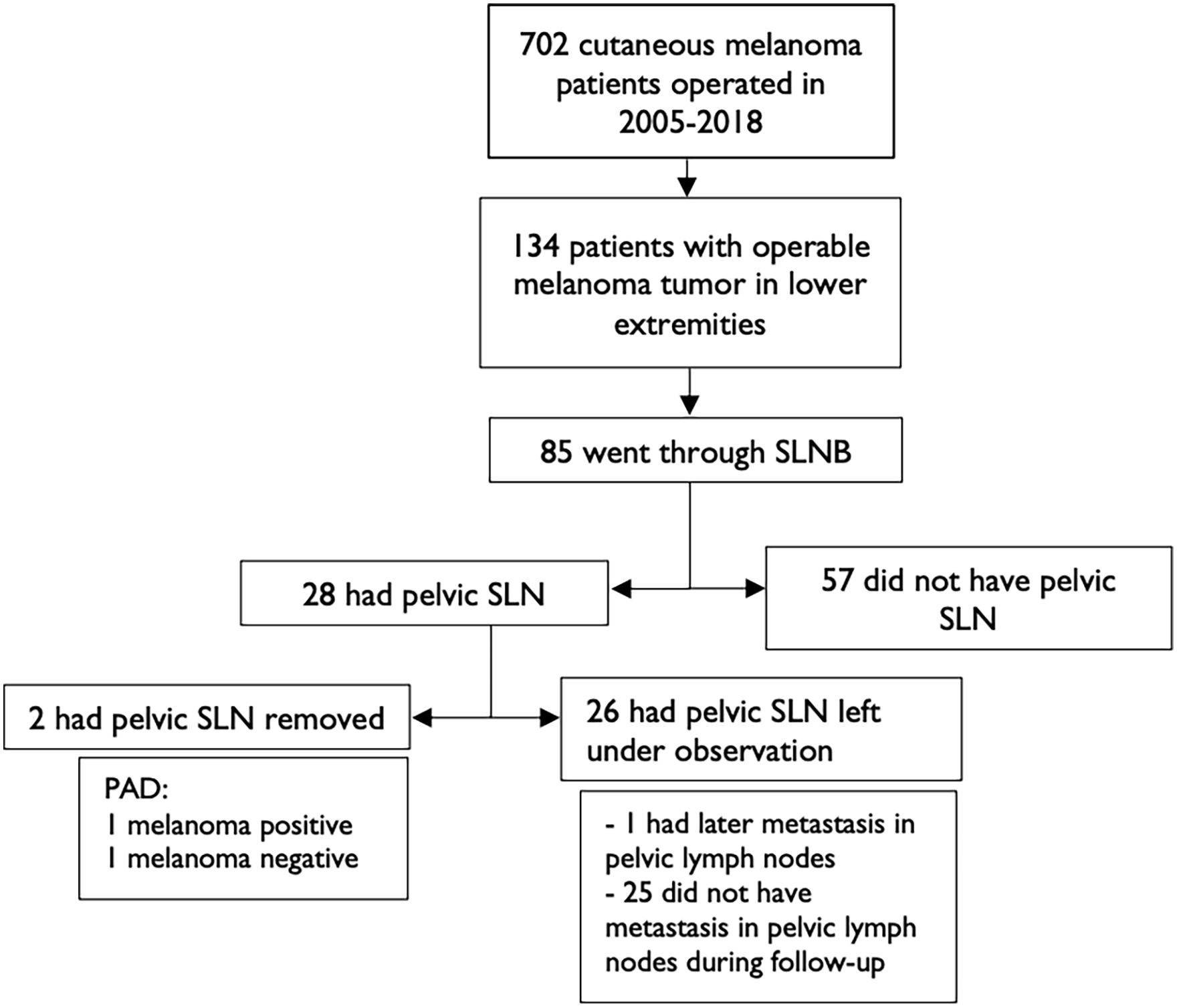
Study flow chart.
A comparison was performed between the following groups: patients with PSLN, which were not removed (n = 26), and patients without PSLN (n = 57). Breslow’s depth did not differ between these two groups: median 2.0 (IQR 1.1–3.6) and 1.9 (IQR 1.3–3.1) mm, respectively (p = 0.94). The tumor was located below the knee in 73.1% (19 of 26) of cases with PSLN and 64.9% (37 of 57) without PSLN, but this difference was not statistically significant (p = 0.61). The median number of removed SSLNs was 2.0 (IQR 1.0–4.0) in patients with PSLN and 2.0 (1.0–3.0) in patients without PSLN (p = 0.18). Overall, 30.8% (8 of 26) of patients had one or more SLNs with metastasis in the removed SSLNs when they had PSLN, but only 17.5% (10 of 57) if they did not show PSLN, but this difference was not significant (p = 0.25). Of the patients who had positive sentinel lymph nodes, 87.5% and 80.0% proceeded to completion lymph node dissection (CLND) in the groups with and without drainage to PSLN, respectively (Table 3).
Clinicopathologic features and long-term outcomes of patients with or without PSLN.
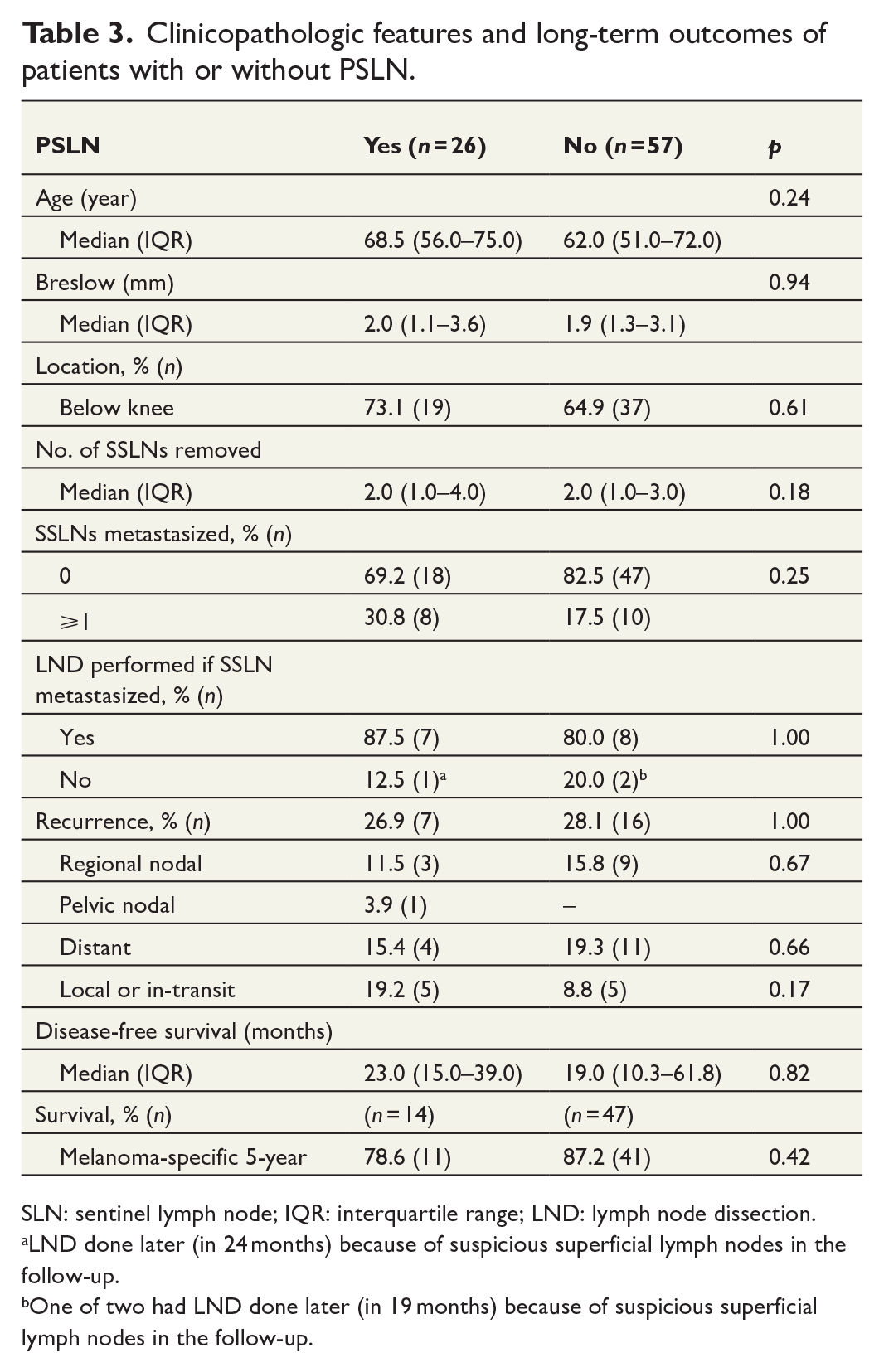
SLN: sentinel lymph node; IQR: interquartile range; LND: lymph node dissection.
LND done later (in 24 months) because of suspicious superficial lymph nodes in the follow-up.
One of two had LND done later (in 19 months) because of suspicious superficial lymph nodes in the follow-up.
The overall recurrence rates were same across the groups: 26.9% (7 of 26) in patients with PSLN and 28.0% (16 of 57) in patients without PSLN (p = 1.00). Regional nodal recurrence rates were 11.5% (3 of 26) in patients with PSLN and 15.8% (9 of 57) in patients without PSLN (p = 0.67), distant recurrence rates were 15.4% (4 of 26) and 19.3% (11 of 57) (p = 0.66), and local or in-transit recurrence of the disease affected 19.2% (5 of 26) and 8.8% (5 of 57) (p = 0.17), respectively. We did not find a difference in disease-free survival between the groups: the median was 23.0 (IQR 15.0–39.0) months in patients with PSLN and 19.0 (IQR 10.3–61.8) months in patients without PSLN (p = 0.82). Of the 85 patients, 61 completed at least 5 years of follow-up, and their melanoma-specific 5-year survival rates were 78.6% (11 of 14) in patients with PSLN and 87.2% (41 of 47) in patients without PSLN. The difference between the groups was not statistically significant (p = 0.42) (Table 3).
Complications
Of the patients who underwent SLNB, 15.3% (13 of 85) were reported to have lymph edema of the operated extremity after the procedure. Other complications were reported in 15.3% (13 of 85), including hematoma (2.4%), seroma (5.9%), wound infection (3.5%), or combined seroma and wound infection (3.5%). In the patients who underwent LND, complication rates were as high as 50.0% (10 of 20), and additionally 40.0% (8 of 20) of patients developed lymph edema after LND. After wide excision, 24.6% of patients were reported to have complications (Table 4).
Complication rates.
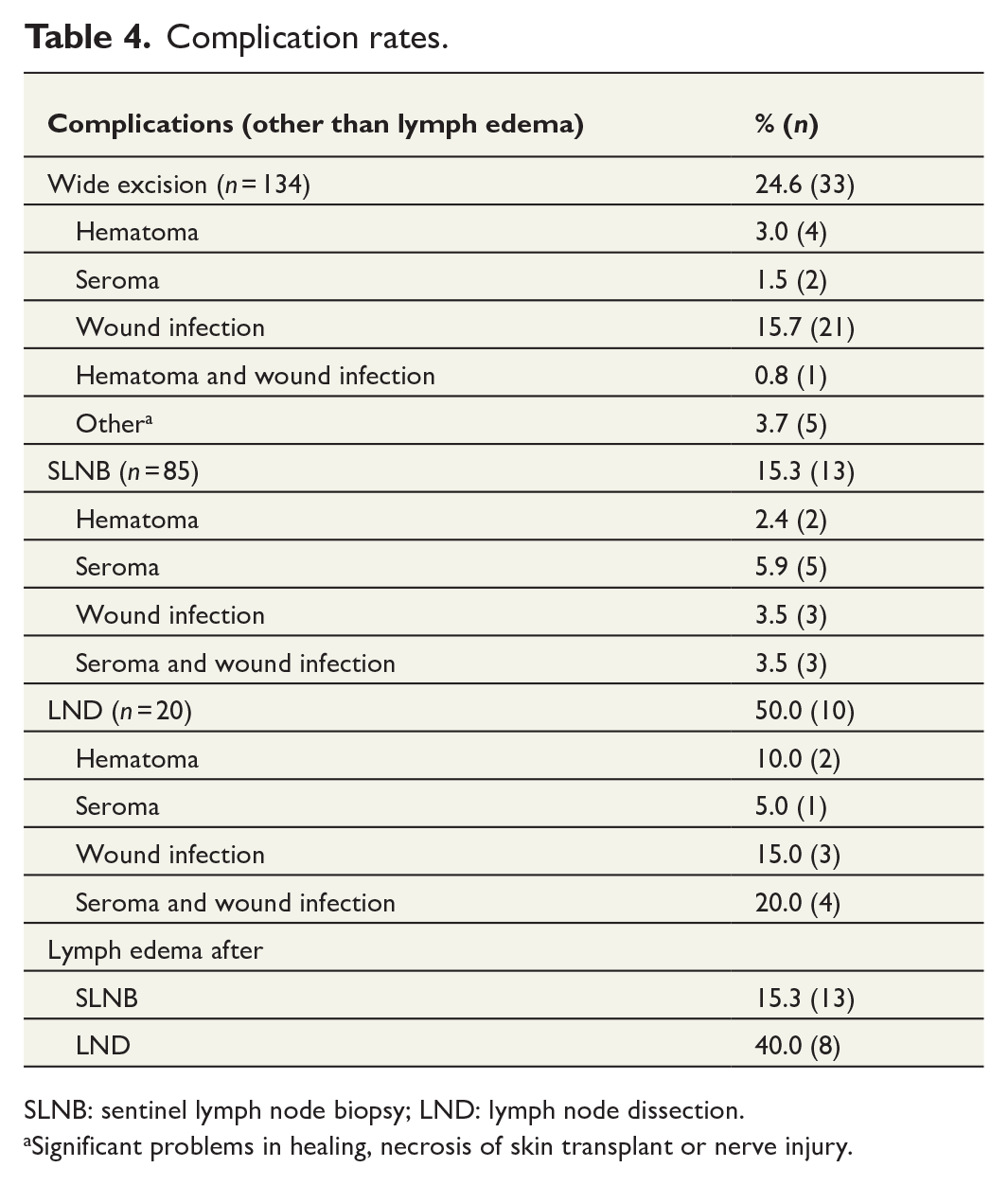
SLNB: sentinel lymph node biopsy; LND: lymph node dissection.aSignificant problems in healing, necrosis of skin transplant or nerve injury.
Significant problems in healing, necrosis of skin transplant or nerve injury.
Discussion
Only a few studies have previously researched the prognostic significance and importance of PSLNs and their biopsy or presence. Karakousis et al. 8 had the study protocol closest to ours, and they did not routinely perform biopsy of PSLNs. In other studies on this subject, the PSLNs were routinely biopsied if they were present.9–11,13,14 Swords et al. 10 earlier suggested that pelvic SLNB could be omitted. However, Schuitevoerder et al. 9 recommended PSLNs to be biopsied when SSLNs are positive, or drainage goes straight to the PSLNs. Vuoristo et al. 11 proposed removal of PSLNs if they are more or equally radioactive to the most radioactive SSLN. It is important to note when not examining the PSLNs, the first-tier SLN determines whether the patient needs adjuvant therapies.
We did not find poorer disease-free survival or higher local (groin) nodal recurrence rates in patients with drainage to PSLNs. Swords et al. 10 and Vuoristo et al. 11 reported similar findings. Karakousis et al. 8 did not detect a difference in the pelvic recurrence rates, but found that drainage to PSLNs is associated with marginally lower disease-free survival if SSLNs were histologically negative. We found the disease-free survival to be similar with or without drainage to the PSLNs.
Furthermore, we did not find melanoma-specific 5-year survival to be significantly shorter in patient with drainage to the PSLNs (78.6% and 87.2%). Swords et al. 10 previously reported that melanoma-specific survival did not significantly differ in patients with either histologically negative or positive SSLNs. Vuoristo et al. 11 also found that the presence of PSLNs did not affect melanoma-specific survival.
The below-knee melanomas were found to be thicker in Breslow depth, as also demonstrated by Miranda et al. 14 in their study. Thicker plantar site melanomas may explain the difference in Breslow depth, and furthermore, more often underwent SLNB and had a longer follow-up time in our data. 17 Plantar foot melanomas are known having decreased and thigh melanomas increased melanoma-specific survival, but we did not find a significant difference between the below- (83.93%) and above-knee (92.11%) groups. 18 Survival rates are approximately in line with those that the American Joint Committee on Cancer (AJCC) have reported for stage IB to IIA melanomas, which were the average stages of our patients. 19
The difference in the incidence of PSLN drainage in the SLNB due to the anatomical location was not significant, being 36.21% in below- and 25.93% in above-knee melanomas. Vuoristo et al. 11 found that PSLNs were more likely to be present when the primary melanoma tumor was located below the knee. Other predictive factors have been found to be older age, greater Breslow thickness, and melanoma subtype.8–11 Regarding the melanoma positivity of PSLNs, it is associated with male sex, tumor thickness, melanoma subtype, location in feet, and higher radioactivity count of the node.9,11 However, 30.8% of patients with PSLN had positive SSLN in SLNB, when patients without PSLN, the rate was 17.5%. Although the difference was not statistically significant, it leads to the situation where patients with PSLN receive adjuvant treatments due to positive SSLN.
The data in the current study were collected mainly before the discovery of evidence that patients with low SLN tumor burden gain no benefit from undergoing CLND compared to careful observation.20,21 Consequently, the majority of our patients underwent CLND, as this was the standard of care at the time. Current guidelines recommend that careful observation should often be preferred with low-risk micro-metastatic disease.3
We found the incidence of the melanomas occurring in the lower extremities to be 19.7%, which has been earlier reported to be 23.0%–25.1%.17,18,22 The morbidity rate after groin SLNB has been estimated to be 14.9%.16 Seroma with or without wound infection was the most common complication, affecting 9.4% of patients, along with lymph edema, which affected up to 15.3% of patients in our study. Recently, even higher incidences have been reported, with 46.3% of patients developing seroma and 21.1% lymph edema after groin SLNB. 11
The relatively small number of patients limits our study, and as a retrospective study, it has certain limitations. Interpretations are based on medical reports made by others and the correctness of these cannot be proven. In addition, structured pathology reports were not available from the early years, so some data were lacking. TNM classification was not used during the early years of the research period, and accurate classification could not be performed afterward. The true follow-up time was longer than reported in many cases, because low-risk melanoma patients had their later melanoma check-ups performed in primary healthcare and we did not have access to their medical records.
Conclusion
We did not find shorter disease-free survival, more frequent recurrence, or worse melanoma-specific survival in patients with drainage to PSLNs. Our research findings strengthen the evidence that, when the melanoma locates in the lower extremities, PSLNs should not be routinely biopsied unless they are first-tier nodes. Although more extensive, multicentre studies are needed to confirm these findings and to make more definitive conclusions.
Footnotes
Authors contributions
J.L. contributed to collection of data, statistical analysis of the data, and drafting the manuscript. H.N. contributed to study design, collection of data, and editing the manuscript. O.K. and T.T. contributed to the review of the article and approving the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
