Abstract
Objective:
Sentinel lymph node biopsy is the standard of care for nodal staging in clinically node-negative melanoma patients. Our goal was to present 10-year results of sentinel lymph node biopsy at our institution and to evaluate the clinicopathologic factors as potential predictors of sentinel lymph node and non-sentinel lymph node metastatic involvement in patients with cutaneous melanoma.
Methods:
We have analyzed clinicopathologic and lymphoscintigraphic characteristics in 420 patients with cutaneous melanoma who underwent sentinel lymph node biopsy between 2010 and 2019. In addition, we have examined the results of group of patients with positive sentinel lymph node biopsy undergoing complete lymph node dissection.
Results:
The overall detection rate of sentinel lymph node biopsies was 97.1%, of which 18.8% was metastatic. Drainage to one regional basin was seen in 345 patients (83.1%) and to multiple drainage regions in 71 patients (17%). In-transit lymph nodes were detected in 20 patients. On univariate logistic regression analysis, male gender, primary tumor thickness with nodular histology, acral location, presence of ulceration, and the number of nodes harvested were significantly associated with sentinel lymph node biopsy status (p < 0.05). On multivariate analysis, the Breslow thickness was the only independent predictor of sentinel lymph node biopsy status. The metastases in non-sentinel lymph node found in 26 patients with positive sentinel lymph node (35.6%) correlated on univariate, as well as on multivariate logistic regression, with tumor subtype and number of sentinel lymph node harvested.
Conclusions:
In addition to the well-established primary tumor thickness as a predictor of sentinel lymph node biopsy positivity, we observed acral location and nodular melanoma subtype to significantly enhance the risk of metastases in sentinel lymph node(s). Primary tumor histology and number of nodes harvested were the only statistically significant variables predicting the non-sentinel lymph node status on multivariate analysis. Lymphoscintigraphy imaging characteristics were not significantly associated with sentinel lymph node status.
Introduction
The concept of the sentinel lymph node (SLN) is based on the assumption that the tumor status of the first node draining the tumor area indicates the tumor status of the entire nodal basin. Tumor negative SLN predicts that other nodes in the same drainage basin will also be tumor free, and a tumor positive SLN predicts that non-sentinel lymph nodes (non-SLN) might also harbor metastasis. In the majority of previous studies, in up to 27% of patients, non-SLN node metastases were found after complete lymph node dissection following a positive SLN biopsy.
Since its introduction in the early 1990s by Donald Morton, SLN biopsy has become a widely accepted procedure and the reference standard for determining regional lymph node metastasis and staging of the disease.1 –4 It is also considered to be a very important tool in treatment decisions regarding adjuvant therapy, which became a standard of care for patients with lymph node metastases.1 –4 Prognostic factors for SLN metastatic involvement were reported in the literature, in relation mainly to primary melanoma clinicopathologic characteristics. The Breslow thickness was the most consistently reported and well-established predictor of SLN metastasis.5 –7 Several other demographic, clinical, and histological characteristics have been found to be predictive of SLN positivity, such as sex, age, tumor mitotic rate, regression, histological type, ulceration, tumor-infiltrating lymphocytes (TILs), and lymphovascular invasion, but with few of them found to be reproducible predictive factors.5 –10 The patterns of lymphatic drainage identified by lymphoscintigraphy—such as number of draining basins, SLN diameter, and number of SLNs visualized—were also linked to SLN positivity in previous studies.10 –12
Until recently, complete lymph node dissection (CLND), that is, removal of the remaining regional lymph nodes after sentinel node excision, was recommended for patients with sentinel lymph node metastases. However, the impact of CLND after a positive SLN biopsy on overall survival rates was not proven in large prospective trials, and ultrasound surveillance and adjuvant therapy are now the standard of care for these patients.2 –4 In previous studies, 11%–27% of SLN-positive patients had metastatic involvement in non-SLN, while the rest of the patients underwent CLND with no clear benefit but with the risks associated with surgical procedures and complications.2 –4,12 –15 There was a reported benefit of local disease control with CLND. Also, patients with non-SLN involvement had a worse survival rate than non-SLN-negative patients, indicating CLND status as a significant prognostic factor in these patients. 16 In this context, examining the role of demographic, clinicopathologic, and immunological characteristics of primary tumor or SLN metastasis that could be highly associated with increased risk of metastases in non-SLNs is of great importance in order to identify patients who could still benefit from CLND. The aim of this study was to analyze 10 years of experience in SLN biopsy at our center in Southeast Europe and to evaluate the correlation of primary tumor characteristics with both SLN status and non-SLN metastatic involvement in patients with cutaneous melanoma.
Patients and Methods
After approval was obtained from the Institutional Ethics Committee, retrospective database review was conducted on patients with cutaneous melanoma who underwent lymphoscintigraphy, re-excision, and SLN biopsy between November 2010 and January 2019. Each patient provided written informed consent. A total of 457 patients presented at the interdisciplinary tumor board were identified to have the indication for sentinel lymph node biopsy (SLNB). Of those, 420 patients had the procedure at our institution, 21 patients were lost to follow-up, and in 16 patients procedure was not performed due to age, comorbidities, and periodical shortage of radiopharmaceuticals. Patients were selected for SLNB according to international guidelines.17,18 The indication for SLNB was the Breslow thickness greater or equal to 1.0 mm, and for thin melanoma presence of ulceration and more than 1 mitoses/mm2. Clinically non-palpable regional lymph nodes and absence of distant metastases were mandatory for patient selection.
Lymphoscintigraphy was performed in all cases (n = 420) using the radioactive tracer technetium (99mTc) colloidal rhenium sulfide injection (Nanocolloid). At the beginning of the study period, dual mapping was performed in 89 patients with blue dye 1% in addition to the lymphoscintigraphy. Patients underwent a wide excision of the primary lesion and SLN removal guided by a hand-held gamma probe (Europrobe). Evaluation of the lymph nodes obtained through the SLN biopsy consisted of hematoxylin and eosin (H&E) staining and serial sections, as well as immunohistochemical analysis of S100-B, Melan-A, and HMB-45 expression. Most of the patients underwent complete lymph dissection in case of metastatic involvement of SLN. The specimens were analyzed by the following standard procedures:
Ethics. This study was conducted as a part of the research project “Sentinel lymph node biopsy in melanoma and influence of local and systemic immune response parameters in sentinel lymph node on the disease course and outcome” (no. MFVMA/15/13-15) that was approved by the local ethics committee.
Statistics. The detection rate was calculated as the number of successful SLN detections divided by total number of procedures. The analysis of the sample included descriptive and analytic statistics. Univariate and multivariate logistic regression analyses were performed using SLN metastasis (yes/no) as the binary outcome, with odds ratios (ORs) representing effect size. The association of demographic and clinicopathological factors with non-SLN involvement in CLND specimen was evaluated using χ2 or Fisher’s exact test, as appropriate. The number of SLNs harvested and the number of positive SLNs were also explored as potential predictors of positive non-SLNs. Model covariates were characterized with 95% confidence intervals. The significance level was determined at p < 0.05. All reported p-values were two-sided. Analyses were performed with SPSS (version 20.0).
Results
Baseline characteristics of patients and primary melanoma
A total of 420 patients (244 males and 176 females), with a median age of 57 years (minimum 9 and maximum 83), undergoing SLN biopsy for cutaneous melanoma were included in the study. The site of the primary lesion was the trunk in 178 (42.7%) patients, the head and neck in 68 (16.9%), an upper limb in 58 (13.8%), and a lower limb in 90 (21.4%) patients. The baseline characteristics of all reviewed patients (n = 420) are shown in Table 1.
Clinicopathologic factors in SLN-positive and SLN-negative patients.
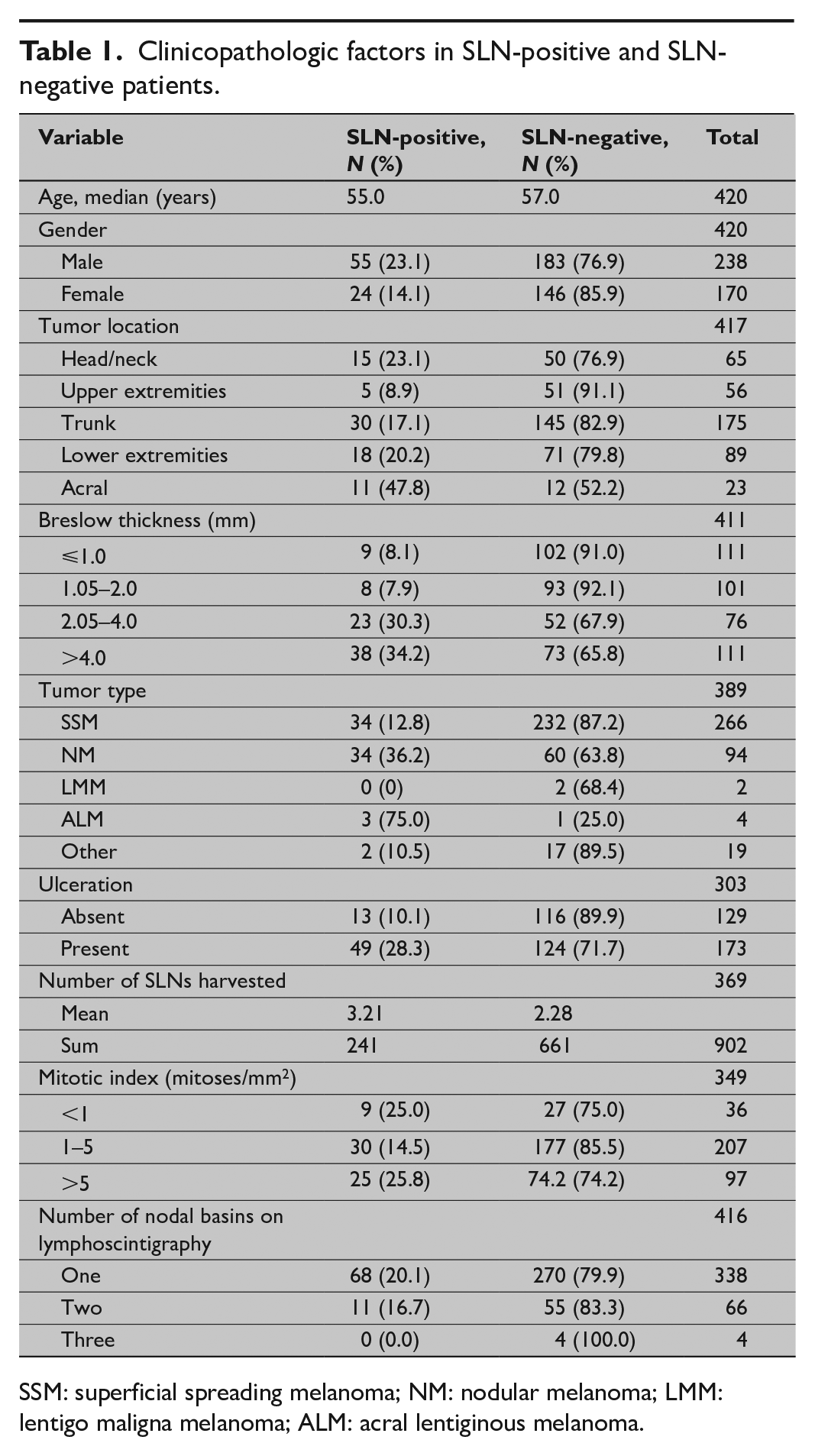
SSM: superficial spreading melanoma; NM: nodular melanoma; LMM: lentigo maligna melanoma; ALM: acral lentiginous melanoma.
Detection rate with slnb
The detection rate of SLNs was 97.1%, of which 18.8% were metastatic. Dual mapping was performed in the initial period after establishing the SLN biopsy technique at our institution in 89 patients. There was no difference in detection rate between the dual-mapping group of patients and the group in which blue dye was omitted from procedure (p > 0.05). In 12 patients, SLNs were not found, using imaging in four patients, and intraoperatively in eight patients. In total, 902 lymph nodes (mean 2.44 per patient) were removed from 369 patients (data were not available in 51 patients).
Lymphatic drainage patterns detected by nuclear imaging characteristics and slnb positivity
Drainage to the single regional lymph basin was found in 82.1% of patients. Drainage to multiple lymphatic basins was most common for trunk lesions (32.0%), followed by those on the head and neck (16.1%). The mean number of lymph nodes visualized on scintigraphy was 1.83. On univariate analysis, number of draining basins and number of SLNs visualized on scintigraphy were not predictive of SLN positivity (p > 0.05).
Clinicopathologic characteristics of primary melanoma associated with metastatic disease in slns
The mean and the median Breslow thickness of the primary tumors were 3.16 and 1.95 mm, respectively. On univariate analysis, the Breslow thickness (p < 0.001), nodular melanoma (NM) histology (p < 0.001), and ulceration (p < 0.001) were strongly correlated with SLN positivity. Male gender (p = 0.029) and the number of lymph nodes removed and analyzed in specimens (p = 0.016) were also significantly associated with SLN status (Table 2). Multivariate analysis of 408 cases revealed only Breslow thickness to be a significant independent predictor of SLN status (p < 0.05). The remaining variables were not significantly associated with SLN status.
Significant predictors of positive SLN by logistic regression analysis in 420 patients who underwent SLNB in Military Medical Academy, Belgrade, during 2010–2019.
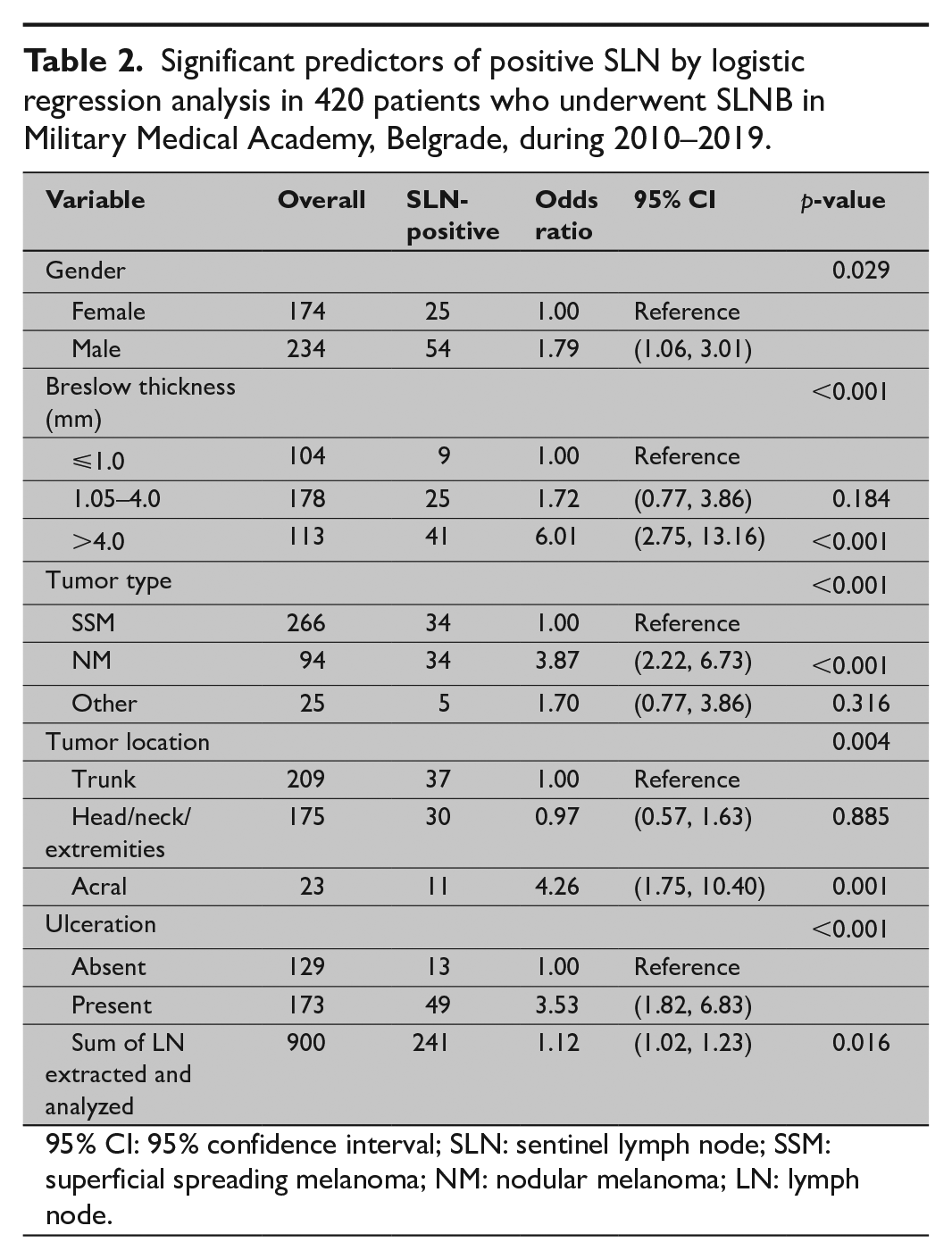
95% CI: 95% confidence interval; SLN: sentinel lymph node; SSM: superficial spreading melanoma; NM: nodular melanoma; LN: lymph node.
Clinicopathologic characteristics of primary melanoma associated with metastatic disease in non-slns
Out of 420 patients, 79 were diagnosed with metastatic SLNs. In patients with metastatic SLN involvement, CLND was indicated. Among the 73 patients who underwent CLND at our institution, additional metastatic nodes (positive CLND) were shown in 26 (35.6%) patients, while negative CLND was found in 47 (64.4%) patients. A total of 708 nodes were removed, of which 54 nodes were metastatic. CLND specimen analysis revealed one, two and three and more additional positive lymph nodes in 17, 4, and 4 patients, respectively. Positive non-SLNs were significantly more frequent in thicker tumors (p = 0.013). Out of 26 non-SLN-positive cases, 22 (84.6%) were related to Breslow thickness of >2.05 mm. We observed that nodular histology of primary melanoma was related to non-SLN metastatic involvement in 45.2% of cases compared with 21.2% in superficial spreading melanoma (SSM) melanoma subtype. Univariate analysis of 73 cases revealed only the histopathologic type of primary tumor to be a significant independent predictor of non-SLN status (p = 0.013). Also, we found that the presence of any additional lymph nodes harvested during SLN surgical procedure increased the risk of non-SLN status positivity (Table 3).
Univariate and multivariate analyses to predict positivity of non-SLN in 73 SLN-positive patients with CLND.
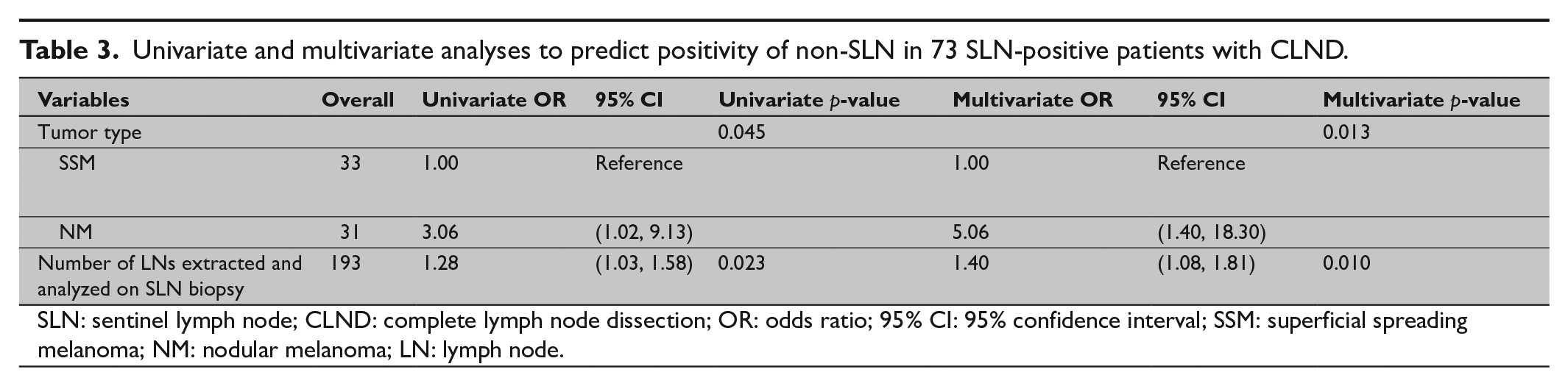
SLN: sentinel lymph node; CLND: complete lymph node dissection; OR: odds ratio; 95% CI: 95% confidence interval; SSM: superficial spreading melanoma; NM: nodular melanoma; LN: lymph node.
Discussion
The detection rate of SLNs in our group of melanoma patients was 97.6%. Although the dual mapping was performed in the initial period after establishing the SLNB technique at our institution, our study showed no difference in detection rate between the dual-mapping group of patients (n = 89) and the group in which blue dye was omitted from the procedure (p > 0.05). This is in line with published results that have suggested blue dye had no additional value in finding the sentinel node and could be safely omitted from the standardized technique.19,20
Failure to visualize the SLN with planar and single-photon emission computed tomography was evident in four cases (0.9%). Despite a positive lymphoscintigraphy, SLNs were not found in the operation room in eight patients. The majority of patients had the primary melanoma located on the head and neck region. Complex nodal basins in the neck area led to an increased risk of not identifying the correct SLN. 21 A review of de Rosa based on an analysis of 3442 patients from 32 studies reported a median sentinel node biopsy rate of 95.2% for head and neck melanomas, associated with an increased false-negative rate compared to the studies of non-head and neck lesions. 22
Multiple lymphatic basin drainage (MLBD) was most common in trunk lesions, followed by those on the head and neck. Only four patients showed three drainage basins, all with negative SLNs. No significant correlation between sentinel node positivity and MLBD was found in our case series.
In-transit (or interval) nodes were identified with lymphoscintigraphy in 30 out of 420 patients (7.1%). Based on the surgeon’s decision and depending on the location, in-transit nodes identified with lymphoscintigraphy were removed in 14 patients, revealing metastatic melanoma in four nodes (28.0%). In one patient with an identified in-transit node, a false-negative SLN biopsy was detected during follow-up for recurrence of the disease in the same nodal basin. This is in line with previous studies in which metastatic involvement of in-transit nodes varied from 8% to 38%. 23 The importance of excising and analyzing in-transit nodes whenever feasible is stressed by the finding that 30% of patients in whom in-transit nodes identified with lymphoscintigraphy were not removed later developed additional in-transit metastases. 23
According to univariate analysis, we have identified the Breslow thickness, male gender, NM, presence of ulceration, and acral localization to be significant predictors of metastatic involvement of SLNs (p < 0.05) just as it had been described by other groups.7 –14 NM was strongly associated with SLN positivity, increasing the risk of SLN metastasis by more than threefold (OR = 3.87, 2.22–6.73, confidence interval (CI) = 95%). These findings are in accordance with the data of Homolak et al. 23 and Bertolli et al. 24 , who also reported an increased risk of metastatic SLN in patients with NM. However, on multivariate analysis, clinicopathologic subtype was not associated with SLN positivity, probably due to the confounding factor of tumor thickness, which was higher in NM. The anatomic location of the primary tumor is generally considered to play a minor role compared to other factors. Previous data about primary tumor location as a risk factor for regional metastases have been reported quite variably and infrequently, providing conflicting conclusions.25,26 Acral melanoma shows significant differences from other clinical types, considering the biological, genetic, and clinicopathological aspects, although SLN biopsy is also widely applied in clinical practice. In our study, acral location was a strong predictor of SLN metastasis, although patients with acral primary tumors tended to present with thicker melanomas (a median Breslow thickness of 3.00 mm for acral vs 1.80 mm for non-acral melanomas), as has been shown in previous studies.25,26 The number of SLNs removed during operation was also significantly correlated with SLN positivity, enhancing the risk of metastases in SLNs up to 1.23-fold which is partially in line with existing literature. A study by Bertolli on 2013 patients found the number of positive sentinel nodes to be in statistically significant correlation with SLN metastatic involvement. 27
In 73 patients with positive SLNB, CLND was done at our institution. We found non-SLN positivity in 35.6% of patients. In previous studies, positive non-SLN metastatic involvement was found in 8%–27% of patients after CLND.13,14,27,28 In our case series, a higher percentage of thick melanomas was found, with 115/411(28%) tumors with the Breslow thickness of ⩾4 mm, and median Breslow thickness of 1.98 mm, which is higher than in the majority of reported series.13,14,28,29 Although no clear benefit on overall survival rates for CLND was found in recent studies, this high number of non-SLN positivity in our case series indicates the need to identify those patients who could still benefit from local disease control and additional prognostic information after CLND.
In this study, we evaluated the risk of non-SLN positivity on the basis of primary melanoma clinicopathologic factors. Among the variables tested, we found the subtype of primary melanoma to be significantly associated with metastatic non-SLN (p = 0.045). Comparing the proportion of positive non-SLNs between the group of patients with a nodular subtype (n = 31) and the group of SSM subtype (n = 31), we showed NM subtype to be more likely to harbor non-SLN metastases (45.2% vs 21.2%). As expected, univariant logistic regression showed that the risk of positive non-SLN increased 1.28-fold for every lymph node removed. Tumor type and number of nodes harvested on operation still correlated with non-SLN positivity on multivariate regression analysis. In recent studies of SLN tumor burden, especially the diameter and location of the largest metastasis were significantly associated with non-SLN positivity and overall survival.14,29,30 However, this analysis is reserved for the future studies on our series of patients with a longer follow-up.
Conclusion
In conclusion, our results confirm previous studies on SLN analysis of melanoma in terms of SLN identification rate (97.1%) and percentage of SLN-positive patients (18.8%), allowing us to exclude 81.2% of patients from unnecessary CLND. Using lymphoscintigraphy, we were able to identify SLNs out of predicted lymph basins as well as the SLNs localized between the primary lesion and regional lymph basin. Lymphoscintigraphy imaging characteristics were not predictive of SLN positivity in our study. In our case series, additional positive non-SLNs were found in 35.6% of patients, which points out to the need to identify those patients who could still benefit from CLND.
On multivariate analysis, the Breslow thickness was the only and strong independent predictor on SLN status. The metastases in non-SLN found in 26/73 patients with positive SLN correlated with tumor subtype and number of SLNs harvested. The association of SLN tumor burden as well as the expression pattern of immunomodulating factors with non-SLN metastatic involvement and survival will be validated in future studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was conducted after approval was obtained from the Institutional Ethics Committee as a part of the research project “Sentinel lymph node biopsy in melanoma and influence of local and systemic immune response parameters in sentinel lymph node on the disease course and outcome” (no. MFVMA/15/13-15).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
