Abstract
Background
Cutaneous head and neck melanoma (cHNM) has a high rate of false-negative sentinel lymph node biopsy (SLNB) and up to a 25% risk of recurrence despite negative SLNB. The aim of this study was to investigate the pattern of melanoma recurrence in patients with cHNM with negative SLNB.
Methods
A retrospective cohort study of consecutive cHNM patients at a tertiary care centre from 2014-2022. We included all cHNM patients with negative SLNB. All patients were categorized into Breslow thickness >2 mm and ≤2 mm and extracted information pertaining to histopathological characteristics and the presence and type of disease recurrences. We performed multivariable analysis using logistic and cox regression. We used an alpha of 0.05 and all statistical analyses were performed using R software.
Results
Overall, 167 patients met eligibility criteria and of these, 53.5% patients had cHNM ≤2 mm thick and 46.7% had lesions >2 mm thick. The overall recurrence rate was 29.3%. Multivariable analysis demonstrated that Breslow thickness [aOR: 5.89 (95% CI: 1.37, 32.3), P = 0.02] was associated with distant recurrence. Multivariable cox regression also identified that pathological ulceration [aHR: 3.17 (95% CI: 1.61, 7.66), P = 0.01] predicted time to distant recurrence. The SLNB false omission rate was 3.6% (95% CI: 1.3%, 7.7%).
Conclusion
SLNB-negative cHNM patients with high-risk pathological features may benefit from adjuvant immunotherapy.
Key Takeaways
• Cutaneous head and neck melanoma (cHNM) patients with negative sentinel lymph node biopsy (SLNB) have 25% risk of disease recurrence. • Breslow thickness >2 mm and pathological ulceration are associated with distant recurrence and shorter time to distant melanoma recurrence, respectively. • SLNB-negative cHNM patients with high-risk pathological features may benefit from consideration for adjuvant immunotherapy.
Introduction
Cutaneous head and neck melanoma (cHNM) has risen in incidence over the past four decades, yet survival outcomes steadily improve due to earlier detection and improved therapies. 1 High-risk melanoma recurs within two years in 13.4% of patients with risk factors for recurrence being head and neck primary site, positive sentinel lymph node biopsy, and rapid primary growth. 2 In cutaneous melanoma, sentinel lymph node biopsy (SLNB) quantifies lymph node metastasis and can detect clinically occult nodal disease in 20% of patients. 3 In cHNM, however, lymphatic drainage is often more complex with SLNB demonstrating a false negative rate upwards of 34%. 4 A recent SEER database analysis demonstrated that age, neck and scalp lesions, Breslow thickness, and mitotic index conferred worse prognosis in SLNB-negative cHNM patients. 5 The purpose of this study was to characterize melanoma recurrence patterns in SLNB-negative cHNM patients and therefore, help to inform future guidelines pertaining to oncological surveillance and adjuvant therapies in this challenging clinical population.
Materials and Methods
Patient Eligibility
We performed a retrospective review of patients with cHNM at a tertiary care center from 2014 to 2023. Study eligibility included any adult patients (≥18 years old) who underwent primary excision for cHNM and had a negative SLNB. Exclusion criteria included patients under 18 years of age, patients who refused treatment or who were undergoing palliative treatment, and patients diagnosed with melanoma in-situ, recurrent melanoma, more than one melanoma lesion, non-melanoma skin cancer, or mucosal melanoma of the head and neck. We categorized polyploid and nevoid melanoma as nodular melanoma and spindle cell melanoma as desmoplastic melanoma. All disease staging was performed according to the AJCC 8th edition staging guidance for cutaneous melanoma. 6 Surgical margin status, lymphovascular invasion (LVI), and perineural invasion (PNI) were extracted from surgical pathology reports. Radionucleotide injection (ie, 99m-Tecnetium) occurred within 6-to-8 hours of SLNB, which was performed under the guidance of an intraoperative gamma probe (Neoprobe®, Mammotome, Cincinnati, OH, USA). Prior to SLNB, all included patients also underwent intralesional patent blue injection (Guerbet, Paris, France). Adjuvant therapy modalities, agents, dosages, and schedules were coordinated by medical oncologists on a per-patient basis. In 2022, pembrolizumab was approved by Health Canada for adjuvant therapy in cutaneous melanoma and so, most included patients were not presented with this treatment option during this study period. 7 The study was approved by the Hamilton Integrated Research Ethics Board (2023-15828-C).
Primary Study Outcome and Data Extraction
The primary outcome was to identify the relationship between Breslow thickness and melanoma recurrence in SLNB-negative cHNM patients. We extracted demographic, clinical, pathological, and treatment information for all included patients. All treatment information was extracted from operating room notes and clinical documentation. All included patients were divided into Breslow thickness ≤2 mm (ie, T1a, T1b, T2a, and T2b) and >2 mm (ie, T3a, T3b, T4a, and T4b). We stratified patients to match DeCOG-SLT, which identified that distant recurrence-free survival, recurrence-free survival, and overall survival were worse in melanoma with >2 mm Breslow thickness. 8 Other studies have also used >2 mm Breslow thickness as a standard threshold since it is associated with an increased risk of melanoma recurrence and worse disease prognosis.9-11 We defined the SLNB false omission rate as all patients with a negative SLNB who developed regional recurrence within any nodal basin during the study period divided by the total number of negative SLNBs.12,13 We were unable to calculate a false negative rate since this calculation requires incorporating SLNB true positives (ie, SLNB false negatives/[SLNB true positives + SLNB false negatives]), which were not recorded or analyzed in this study. 14
Statistical Analysis
We performed descriptive analysis including mean, median, and standard deviation (SD) for continuous variables, and proportions for categorical variables. We did not perform propensity score matching so as to avoid reducing study power secondary to sample attrition. 15 We compared baseline clinical characteristics using students’ T-tests and chi-square tests for continuous and categorical variables, respectively. Multivariable logistic regression was used to evaluate the type of cHNM recurrence using predictor variables derived from demographic, clinical, and melanoma-specific characteristics. Furthermore, the removal of fewer than three lymph nodes during SLNB is associated with an increased risk of melanoma recurrence in N0 cutaneous melanoma and so, we included this cutoff in our analysis. 16 Multicollinearity was evaluated using the variance inflation factor (VIF), whereby we eliminated predictor variables with a VIF ≥5. Regression models were evaluated using the area under the curve (AUC) and model overfitting was prevented by ensuring 10-16 outcome events were included for each predictor variable. 17 We measured the time to melanoma recurrence using Cox proportional hazards regression after confirming proportional hazards assumptions were satisfied. Statistical significance was denoted as a type I error rate (alpha) of 0.05 (two-sided) and uncertainty was expressed as 95% confidence intervals (95% CI). All analyses and graphs were generated using R (version 4.3.1; R project, Vienna, Austria).
Results
Baseline Patient and Melanoma Characteristics
Baseline Demographic and Clinical Characteristics (N = 167).
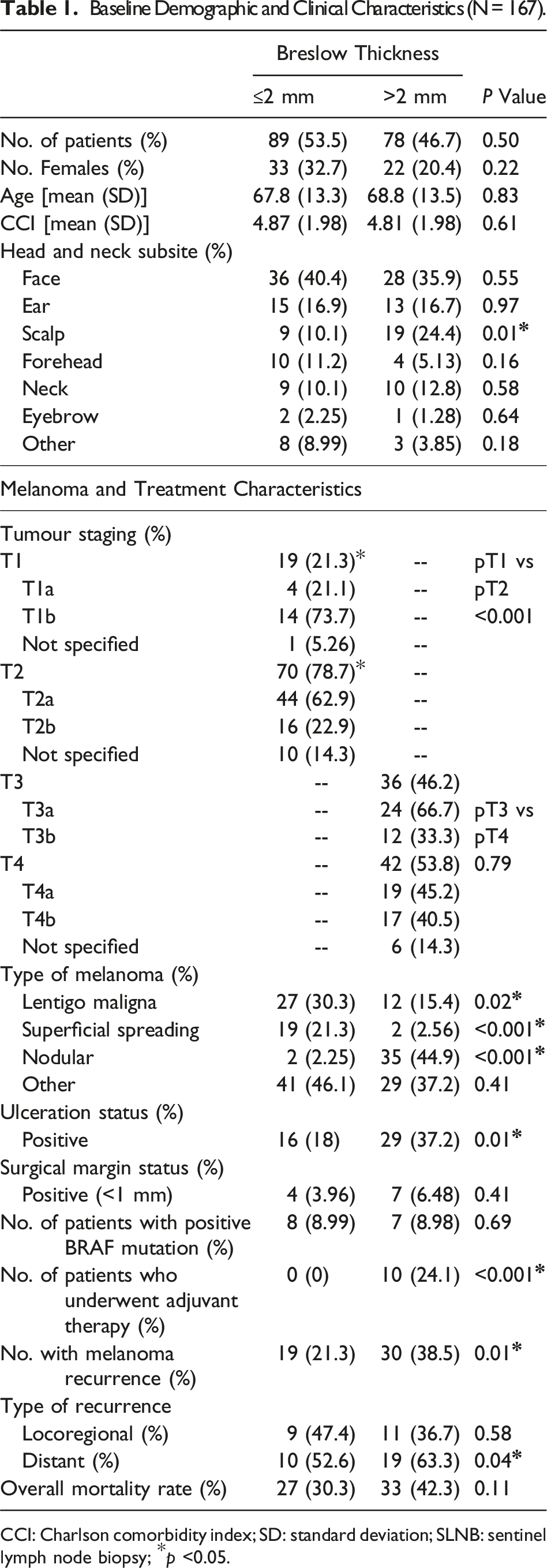
CCI: Charlson comorbidity index; SD: standard deviation; SLNB: sentinel lymph node biopsy; *p <0.05.
The overall melanoma recurrence rate was 29.3% (n = 49; 95% CI: 22.6%, 36.9%). The local or in-transit melanoma recurrence rate (ie, non-nodal recurrence) was 8.4% (n = 14; 95% CI: 4.7%, 13.7%). The regional melanoma recurrence rate (ie, false omission rate) was 3.6% (n = 6, 95% CI: 1.3%, 7.7%). The distant melanoma recurrence rate was 17.4% (n = 29, 95% CI: 12%, 24%). The mean number of lymph nodes sampled during SLNB was 2 (SD: 1.9; range: 0-15 lymph nodes). In ≤2 mm and >2 mm thick melanoma, the mean number of lymph nodes sampled was 2.3 (SD: 1.9) and 2.7 (SD: 1.9), respectively. We also identified that 75 patients (44.9%) had at least three lymph nodes sampled during SLNB. The most sampled lymph node basins were the parotid and perifacial sites, while more than one lymph node basin was sampled in 24 patients (14.4%). We found that the overall rate of melanoma recurrence was higher in >2 mm thick lesions when compared to ≤2 mm [30 (38.5%) vs 19 (21.3%), P = 0.01]. There was a significant difference in distant recurrence [18 (23.1%) vs 10 (11.2%), P = 0.04], but no significant difference in locoregional recurrence [10 (12.8%) vs 9 (10.1%), P = 0.58]. There was no significant difference in overall mortality between groups [33 (42.3%) vs 27 (30.3%), P = 0.11]. Median follow-up time for the entire cohort was 5.51 years (range: 0.1-14.7 years).
Breslow Thickness Predicts Distant Metastasis in SLNB-Negative cHNM
Multivariable Logistic Regression for Distant Melanoma Recurrence in SLNB-Negative Patients.
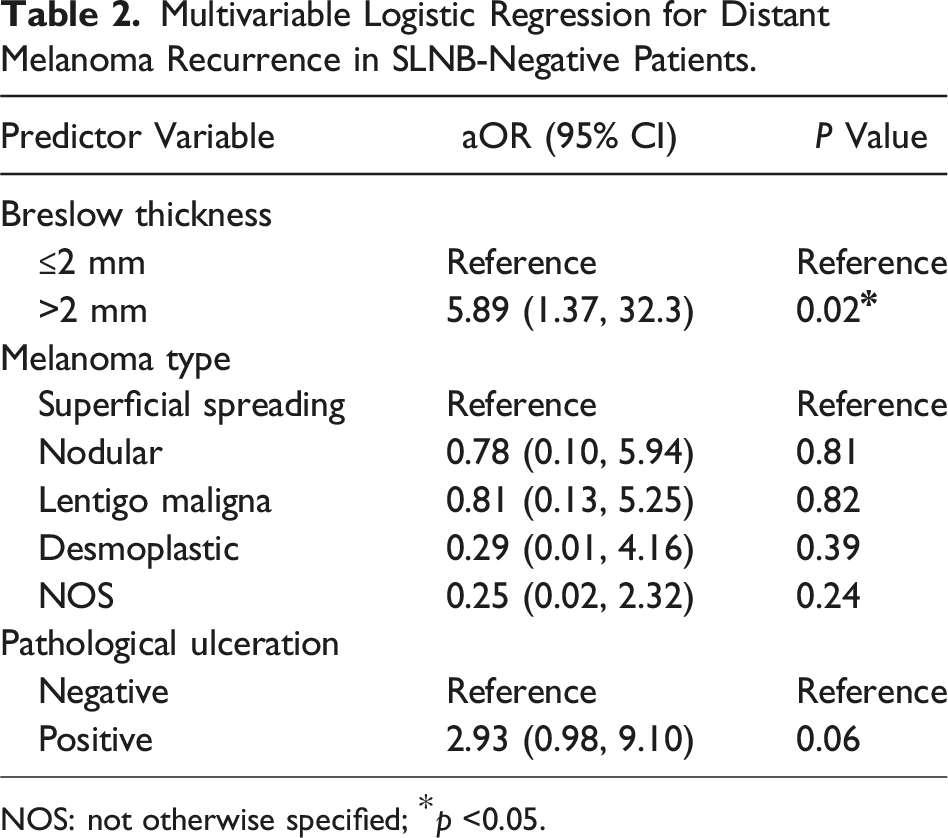
NOS: not otherwise specified; *p <0.05.
Multivariable Logistic Regression for Locoregional Recurrence in SLNB-Negative Patients.
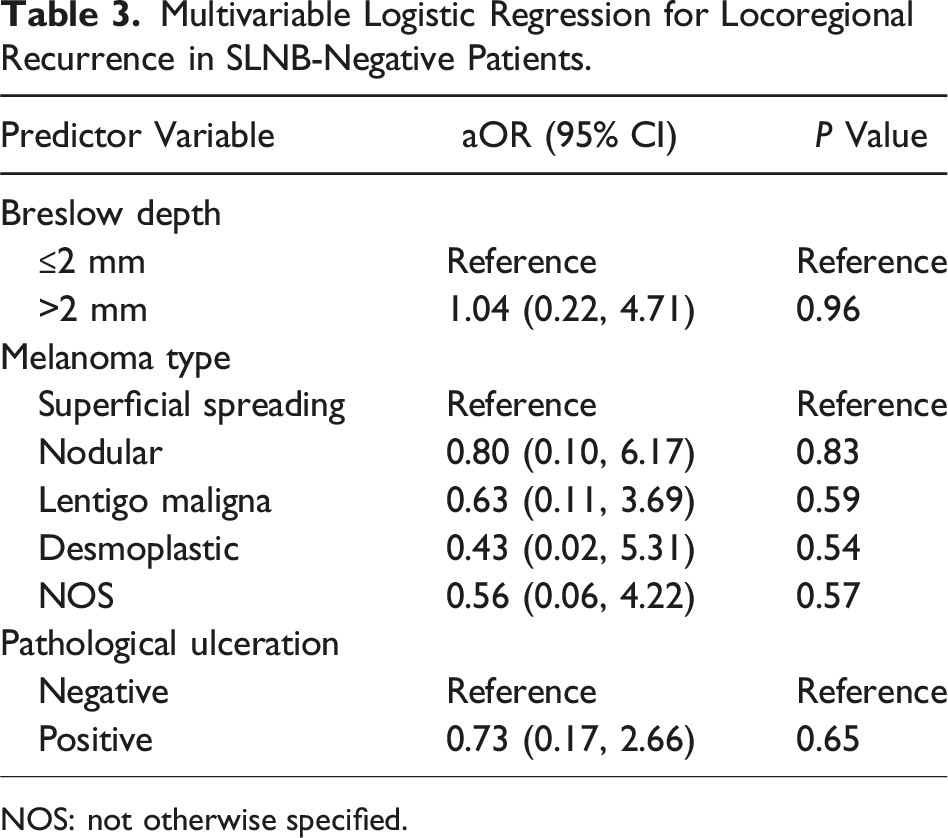
NOS: not otherwise specified.
Pathological Ulceration is Predictive of Time to Distant Recurrence in SLNB-Negative cHNM
Multivariate Cox Regression for Time to Distant Melanoma Recurrence in SLNB-Negative Patients.
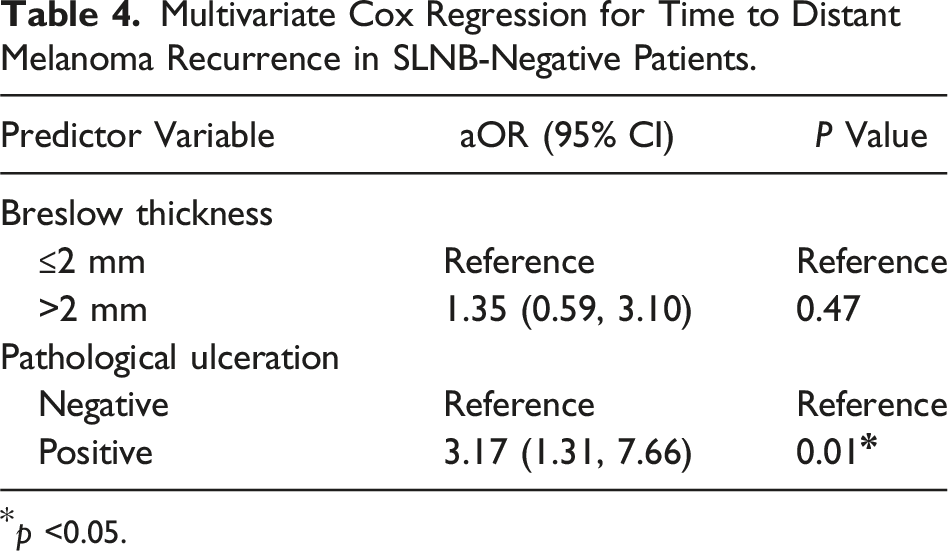
p <0.05.
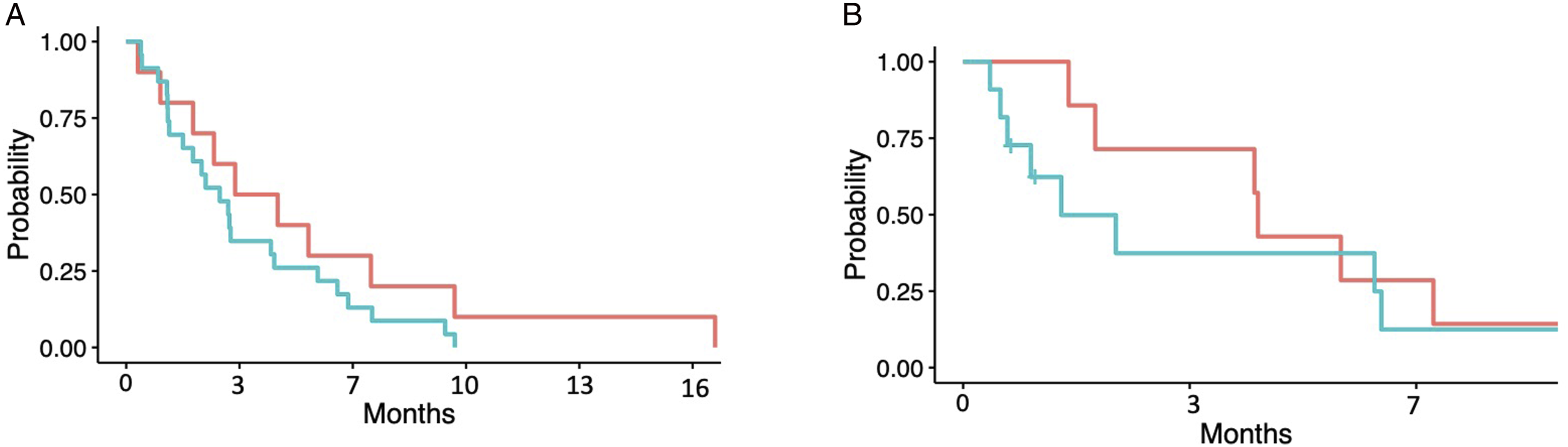
Kaplan Meier plot demonstrating relationship between Breslow thickness and time to (A) distant melanoma recurrence and (B) locoregional melanoma recurrence in months from surgery. ≤2 mm Breslow thickness (Red) and >2 mm Breslow thickness (Blue).
Discussion
To the best of our knowledge, this is one of the largest cohorts investigating disease outcomes in cHNM patients with a negative SLNB. We found that Breslow thickness >2 mm was associated with distant melanoma recurrence in SLNB-negative cHNM. Thomas and colleagues identified that Breslow thickness, pathological ulceration, and LVI predicted distant melanoma recurrence after negative SLNB. 18 Zogakis et al (2005) further identified that SLNB-negative patients were at an increased risk of developing distant melanoma recurrence. 19 Cutaneous melanoma patients with a negative SLNB may have an elevated risk of distant disease recurrence due to hematogenous spread, immunological clearance of lymph node basins, regression of the primary lesion, and/or obstruction of lymphatic drainage. 14 We further identified that pathological ulceration was predictive of shorter time to distant melanoma recurrence after negative SLNB. This is consistent with other studies suggesting that Breslow thickness >2 mm and pathological ulceration demonstrated worse disease-free survival in SLNB-negative patients. 19 Scalp and nodular melanoma were more common in ≥2 mm thick melanoma lesions, but we did not find any association with the type of melanoma recurrence; this finding is not consistent with other studies and may be secondary to our small sample size.20-22 Our study further suggests that high-risk pathological features in cHNM, such as Breslow thickness and pathological ulceration, are associated worse disease outcomes in SLNB-negative cHNM patients. KEYNOTE-716 and CHECKMATE-76K demonstrated that adjuvant pembrolizumab and nivolumab, respectively, in stage IIB and IIC cutaneous melanoma improved disease recurrence and mortality rates, while having manageable safety profiles.23,24 Hence, clinicians should consider having a low threshold for starting adjuvant immunotherapy in SLNB-negative cHNM patients with high-risk pathological features.
Our melanoma recurrence rates were consistent with other studies of SLNB-negative patients.14,18,20,25 Stewart and colleagues also reported that sampling fewer than three lymph nodes was associated with worse disease recurrence in SLNB-negative cutaneous melanoma, but we did not demonstrate this in our cohort. 16 Our false omission rate was 3.6%, which is consistent with studies suggesting the false omission rate for SLNB in melanoma ranges from 0.9% to 8.9%. 14 False negative rates in SLNB for melanoma range from 3.4% to 34%.26-28 Worse false-negative SLNB rates in cHNM may be due to occult lymphatic drainage pathways or an inability to achieve complete resection margins due to aesthetic considerations in the head and neck.26,29 Median time to recurrence in cHNM ranges from 9.5 to 19 months following negative SLNB. 14 Our accelerated time to disease recurrence may be secondary to the paucity of patients in this cohort who underwent adjuvant immunotherapy with BRAF inhibitors and/or MEK inhibitors, or pembrolizumab.
Study Limitations
We were limited by selection bias, small sample size, and low outcome event rate. Most patients in this cohort underwent treatment prior to our institutional adoption of adjuvant immunotherapy and BRAF molecular testing. Furthermore, few patients had PNI or LVI and as such, we were unable to explore their impact on melanoma recurrence. This analysis focused on dichotomizing patients based upon Breslow thickness, which limits us from drawing conclusions based on AJCC melanoma staging strata. We were also limited by lapses in data recording within the electronic medical record. Despite these limitations, however, this study represents one of the largest cohorts of SLNB-negative cHNM patients and further suggests that disease outcomes are worse in SLNB-negative patients with cHNM characterized by high-risk pathological features.
Conclusions
In conclusion, SLNB-negative cHNM patients with high-risk pathological features, including >2 mm Breslow thickness and pathological ulceration, have worse disease outcomes. Future large-scale prospective studies are needed to further stratify cHNM patients who may benefit from adjuvant immunotherapy despite having a negative SLNB.
Supplemental Material
Supplemental Material - Patterns of Failure in Cutaneous Head and Neck Melanoma Following Negative Sentinel Lymph Node Biopsy: A Retrospective Cohort Study
Supplemental Material for Patterns of Failure in Cutaneous Head and Neck Melanoma Following Negative Sentinel Lymph Node Biopsy: A Retrospective Cohort Study by Phillip Staibano, Michael Xie, Zahra Abdallah, Sofia Nguyen, Michael Au, Kelvin Zhou, Hailey Bensky, Michael K. Gupta, David L. Choi, Trevor A. Lewis, J.E.M. (Ted) Young, and Han Zhang in Journal of The American Surgeon™.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
