Abstract
Background and objective:
Obesity may increase the risk of adverse events after total knee arthroplasty (TKA). Although body mass index (BMI) is commonly used in categorizing obesity, its accuracy is limited. Body fat percentage (BFP) might indicate adiposity status and predict arthroplasty-related outcomes better than BMI. We investigated whether BFP is predictive of TKA-related outcomes.
Methods:
In this secondary analysis, BFP was measured preoperatively from 294 participants of a randomized trial that investigated the effects of tourniquet and anesthesia methods on TKA. Data concerning in-hospital assessments and events were collected. Knee range of motion (ROM) was measured, the Brief Pain Inventory–short form and Oxford Knee Score questionnaires were used to collect data on patient-reported pain and function, and the 15-dimensional health-related questionnaire was used to assess quality of life preoperatively and 3 and 12 months postoperatively. The patients reported satisfaction to TKA 3 and 12 months postoperatively. Data concerning infectious and thromboembolic events within 90 postoperative days and revision surgery, manipulation under anesthesia, and mortality within 1 year were collected. A separate post hoc analysis was performed for 399 participants to assess the effects of BMI on the respective outcomes.
Results:
A 1-unit increase in BFP affected the ROM by −0.37° (95% confidence interval (CI) = −0.60 to −0.13) 12 months after surgery. BFP was not significantly associated with the operation time or adverse events. However, the number of most adverse events remained too low for adjusted analysis. A 1-unit increase in BMI increased the operation time by 0.57 min (95% CI = 0.10 to 1.04) and affected the ROM by −0.47° (95% CI = −0.74 to −0.20) 12 months postoperatively. Neither BFP nor BMI was significantly associated with acute pain, pain management, length of stay, or with pain, function, quality of life, or satisfaction to TKA at 12 months after surgery.
Conclusions:
BFP seems to be a poor predictor of in-hospital results and of patient-reported outcomes 1 year after TKA.
Twitter handle
In this secondary analysis of a randomized trial, body fat percentage was poorly predictive of clinical outcomes during hospital stay and of patient-reported outcomes 1 year after TKA.
Keywords
Context and relevance
Results of two previous studies indicated that BFP could be predictive of adverse events, pain, and function after total joint arthroplasty. Thus, information on BFP could be used preoperatively for improving risk stratifications and for projecting patient-reported outcomes. In our results, however, BFP was poorly predictive of clinical outcomes during the hospital stay and of patient-reported pain, function, health-related quality of life, and satisfaction 1 year after TKA. In addition, data concerning the predictive performance of body mass index on TKA related outcomes have been partly conflicting. Our results suggest that, in nonmorbidly obese patients, body mass index has no effect on the patient-reported outcomes 1 year after TKA.
Introduction
Obesity may negatively affect the outcomes of total knee arthroplasty (TKA). Operation times seem to increase when body mass index (BMI) increases.1–3 Similarly, the risk of some complications such as surgical site infections increases, especially when the BMI surpasses 40 kg/m2.3–6 In addition, obese patients have a higher risk for revision surgery.4,5 Furthermore, obesity might reduce the improvements in patient-reported outcomes such as function, pain, and quality of life.6–9
Data on the effects of obesity on TKA-related outcomes, however, are partly conflicting. Some follow-up studies have found no differences in the improvements of patient-reported outcomes between nonobese and obese patients.10–13 In addition, some results suggest that obese patients may benefit from TKA more than normal-weight patients.14–16
Although BMI is commonly used in assessing adiposity status, its accuracy is limited.17,18 BMI does not differentiate adipose tissue from other tissues. Thus, investigating the associations between TKA and other indicators of body structure is well grounded. One such indicator is body fat percentage (BFP), which demonstrates obesity more precisely than BMI. 17 In addition, the results from two studies with combined total hip and knee arthroplasty cohorts suggest that BFP could be a better predictor of adverse events, function, and persistent pain than BMI.19,20
Methods for measuring BFP include bioelectrical impedance analysis (BIA), dual-energy X-ray absorptiometry (DXA), hydrostatic weighing, air displacement plethysmography, computed tomography, and magnetic resonance imagining. 21 From a clinical perspective, BIA is the most applicable although it does not provide information concerning fat distribution, unlike DXA which is one of the reference methods. However, many devices used in BIA have comparable accuracy with DXA in assessing BFP.22–25 In addition, BIA measurement is easy, fast, and noninvasive, and devices are usually portable and inexpensive.
Owing to limited data, we investigated the predictive performance of preoperative BFP on TKA-related outcomes up to 1 year after operation. Based on earlier reports, we hypothesized that increasing BFP increases the risk for poor outcomes.19,20 In addition, owing to the strong status of BMI and prior conflicting data, we extended our investigation post hoc to examine the predictive ability of BMI on TKA-related outcomes.
Methods
This prospective, single-center study followed the World Medical Association Declaration of Helsinki and was conducted as a secondary analysis of a randomized trial approved by the Finnish Medicines Agency Fimea (reference no. KL72/2016) and the ethics committee of HUS Helsinki University Hospital, Surgery (reference no. HUS1703/2016). The trial was registered to EudraCT (2016-002035-15). Every participant provided written informed consent.
The trial was conducted at the Arthroplasty Center of HUS Helsinki University Hospital, Finland. Patients were recruited and underwent operation between October 2016 and December 2018. Follow-up visits took place at the orthopedic outpatient clinic 3 and 12 months after operation. The 12-month follow-up period was completed in December 2019.
Patients
We investigated patients who participated in a randomized trial that compared the effects of spinal and general anesthesia and tourniquet use on outcomes after TKA. A detailed study description and results concerning the anesthesia and tourniquet comparisons have been published.26–29
Patients referred for TKA due to Kellgren–Lawrence grade 3 to 4 osteoarthritis, aged 18–75 years, and with a BMI of ⩽40 kg/m2 were eligible for the trial. Patients who had undergone prior major surgery, such as osteotomy, or had a severe valgus or varus malalignment (>15°), or flexion or extension deficit (maximum flexion of <90° or flexion contracture of >20°) of the study knee were excluded. 26 In addition, we excluded patients who had American Society of Anesthesiologists (ASA) physical status class ⩾IV, ongoing use of strong opioids, a contraindication to spinal or general anesthesia or to the study medications, or a need for bridging anticoagulation and those who underwent a bilateral operation. 26
From the present secondary analysis, we further excluded patients with a prior metallic endoprosthesis or cardiac pacemaker because their BFPs were not measured in accordance with the institutional and manufacturer’s instructions regarding the safety and reliability of BIA. However, we included patients without BFP measurements in the post hoc analyses of BMI.
Intervention
A single type of cemented total knee system (Triathlon CR; Stryker, Kalamazoo, MI, USA) with patellar resurfacing was used. Based on the randomization, patients were operated either under spinal or general anesthesia and with or without a tourniquet. 26 In-hospital pain management was multimodal, including local infiltration analgesia, oral acetaminophen and ibuprofen three times daily, and patient-controlled analgesia with intravenous oxycodone for the first 24 postoperative hours followed by one oral dose of extended-release oxycodone and repeated doses of immediate-release oxycodone given on request. 26 From the second postoperative morning, the patients were given oral tramadol or a combination of acetaminophen and codeine for up to three times daily. 26 Pregabalin and peripheral nerve blocks were allowed as rescue pain-relieving methods if oxycodone was insufficient. Physiotherapists examined and guided the patients preoperatively and postoperatively and gave instructions for postdischarge exercises. For postoperative thromboprophylaxis, the patients self-administered subcutaneous enoxaparin 40 mg once daily for 14 days, unless their comorbidities required other anticoagulation methods.
Data collection
Detailed information on patient characteristics was collected at the preoperative clinic. BFP was measured using a tetrapolar BIA device (Omron BF-500; Omron Healthcare, Kyoto, Japan) that uses eight electrodes and generates comparable results with DXA.22,23 During the measurement, the patients stood barefoot on metal footpads and held a handle with electrodes with both hands in front of the chest, and with arms extended to 90°.
The collected in-hospital data included information on the operating room events, use of analgesics and antiemetics, patients’ assessments of their postoperative pain and nausea, adverse events, and length of hospital stay (LOS). The patients reported preoperative and 3- and 12-month postoperative pain using the self-administered and validated Brief Pain Inventory–short form (BPI-sf) and Oxford Knee Score (OKS; pain subscale) questionnaires.30–32 Knee function was assessed at respective time points with the OKS function subscale 32 and by measuring knee range of motion (ROM) using a goniometer. Measurements were conducted by orthopedic surgeons and physiotherapists. Similarly, the patients reported health-related quality of life using a generic, self-administered 15-dimensional (15D) questionnaire. 33 Furthermore, they reported their satisfaction with TKA using a numerical rating scale (NRS) at 3 and 12 months after surgery.
We collected data concerning thrombotic and infectious complications after discharge and knee-related readmissions for any reason up to 90 postoperative days. Data concerning manipulation under anesthesia, revision surgery, and mortality were collected up to 1 year after surgery.
Statistical analysis
Before launching the primary trial, power calculation was calculated separately for the present secondary analysis. We used two-tailed tests with a power of 0.80 and an alpha of 0.05 to address correlations. For detecting a possible correlation (Pearson’s correlation coefficient (r) ⩾ 0.5) and the threshold for weak correlation (r = 0.3), the minimum samples were 29 and 85 patients, respectively.
Continuous outcome variables were analyzed using the Pearson and Spearman correlation for normally and non-normally distributed data, respectively. Multivariable analyses were performed for continuous data using the general linear model adjusted for age, sex, ASA classification (class I–II or III), anesthesia (spinal or general), tourniquet use (yes/no), and primary diagnosis (primary osteoarthritis or other). In addition, the preoperative value of the respective postoperative value was set as a covariate for applicable variables. We used natural logarithm transformation for significantly positively skewed data. We conducted a sensitivity analysis by removing outliers or by using square root transformations for slightly positively skewed data and square transformations for slightly negatively skewed data. Dichotomous outcomes were analyzed using unadjusted and adjusted binary logistic regression. To assess possible overparameterization, we set 10 events per variable as a limit for conducting a sensitivity analysis with a reduced number of adjusting variables.
Descriptive statistics were presented as mean values with standard deviations for normally distributed data, medians with interquartile ranges for non-normally distributed data, and number of patients with percentages for dichotomous data. Results concerning correlations were expressed as correlation coefficients, and results from the general linear model were presented as regression coefficients (95% confidence interval (CI)) for 1-unit increases in BFP or BMI and as coefficients of determination (R2). The logistic regression results were presented as odds ratios (95% CI) for 1-unit increases in BFP or BMI and as Nagelkerke R2.
Missing values concerning the OKS subscales were imputed with the mean value of other responses for that subscale if only one value was missing. The BPI-sf mean pain severity and interference scores were included in the analyses if at least three of four pain severity variables and four of seven pain interference variables were reported. Missing values in the 15D questionnaire were imputed using a multiple imputation procedure if no more than 3 of 15 values were missing. The patients were considered satisfied with TKA if they reported satisfaction scores of 9–10 (NRS, 0–10; 0 = extremely dissatisfied, 10 = extremely satisfied).
We considered results with p-values < 0.05 as statistically significant. All analyses were conducted using IBM SPSS Statistics 27 (IBM Corp., Armonk, NY, USA).
Results
During the primary trial, a total of 2783 patients were assessed for eligibility (Fig. 1). Of the 404 randomized patients, two received a nonprotocol prosthesis (i.e. a more constrained model because of ligament imbalance and insufficient stability) and two had Kellgren–Lawrence grade 2 osteoarthritis. These patients were excluded from the analyses. The data of two patients who underwent operation without patellar resurfacing for an unknown reason were included. Erroneously, data of one patient whose spinal anesthesia was insufficient were not collected. BFP was not measured from 104 patients and was clearly tabulated incorrectly for one patient. Thus, 294 patients were included in the BFP cohort, and 399 patients were included in the post hoc BMI cohort (Table 1). In both cohorts, two patients canceled their participation prior to the 3-month follow-up, and another three patients did not attend the 12-month follow-up.
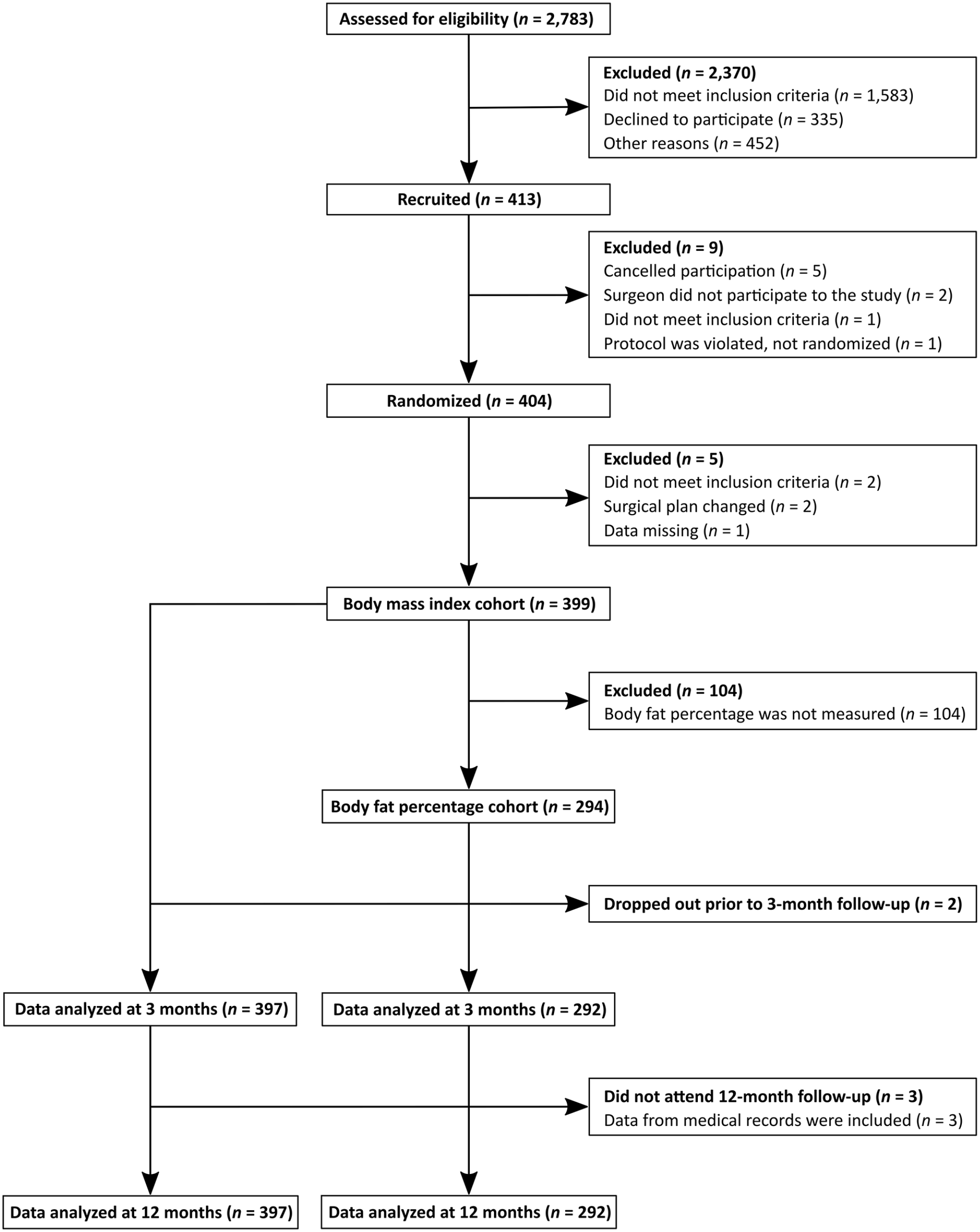
Flow chart of the patients.
Patient characteristics.
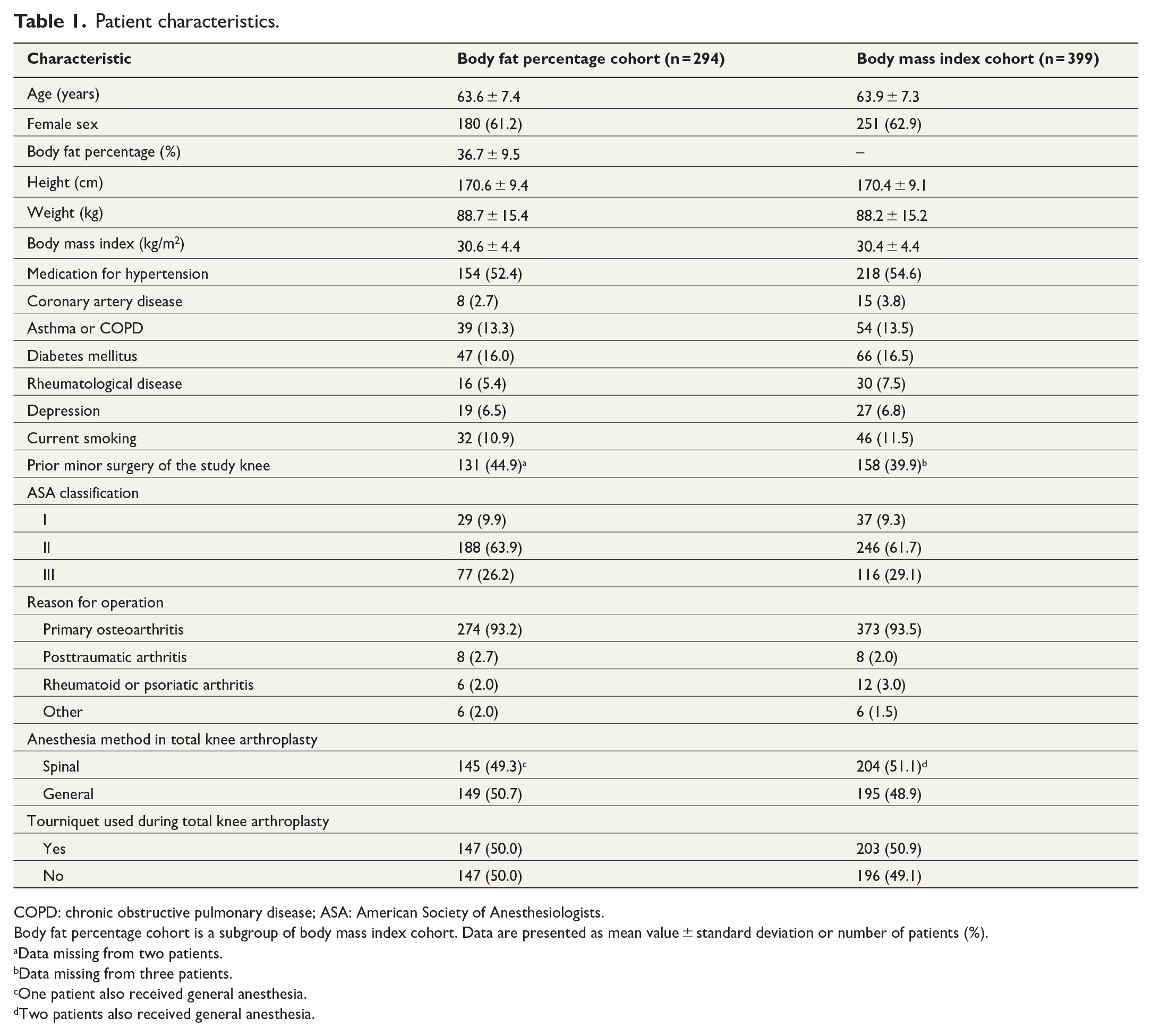
COPD: chronic obstructive pulmonary disease; ASA: American Society of Anesthesiologists.
Body fat percentage cohort is a subgroup of body mass index cohort. Data are presented as mean value ± standard deviation or number of patients (%).
Data missing from two patients.
Data missing from three patients.
One patient also received general anesthesia.
Two patients also received general anesthesia.
BFP and TKA-related outcomes
In the multivariable analyses, BFP was not significantly associated with operation time or LOS (Table 2). The effects of BFP on pain management and pain during hospital stay and at 3 and 12 months after TKA were also nonsignificant (Tables 2 and 3). Similarly, BFP was not predictive of patient-reported function, health-related quality of life, or satisfaction with TKA at 3 or 12 months after surgery (Tables 2 and 3). However, a 1% increase in BFP affected the ROM by −0.37° (95% CI = −0.60 to −0.13; p = 0.003) 12 months postoperatively. The sensitivity analysis results concerning two pain and two function variables were consistent with the main analysis results (Supplementary Table 1).
Associations between body fat percentage and the continuous outcomes of total knee arthroplasty (n = 294).
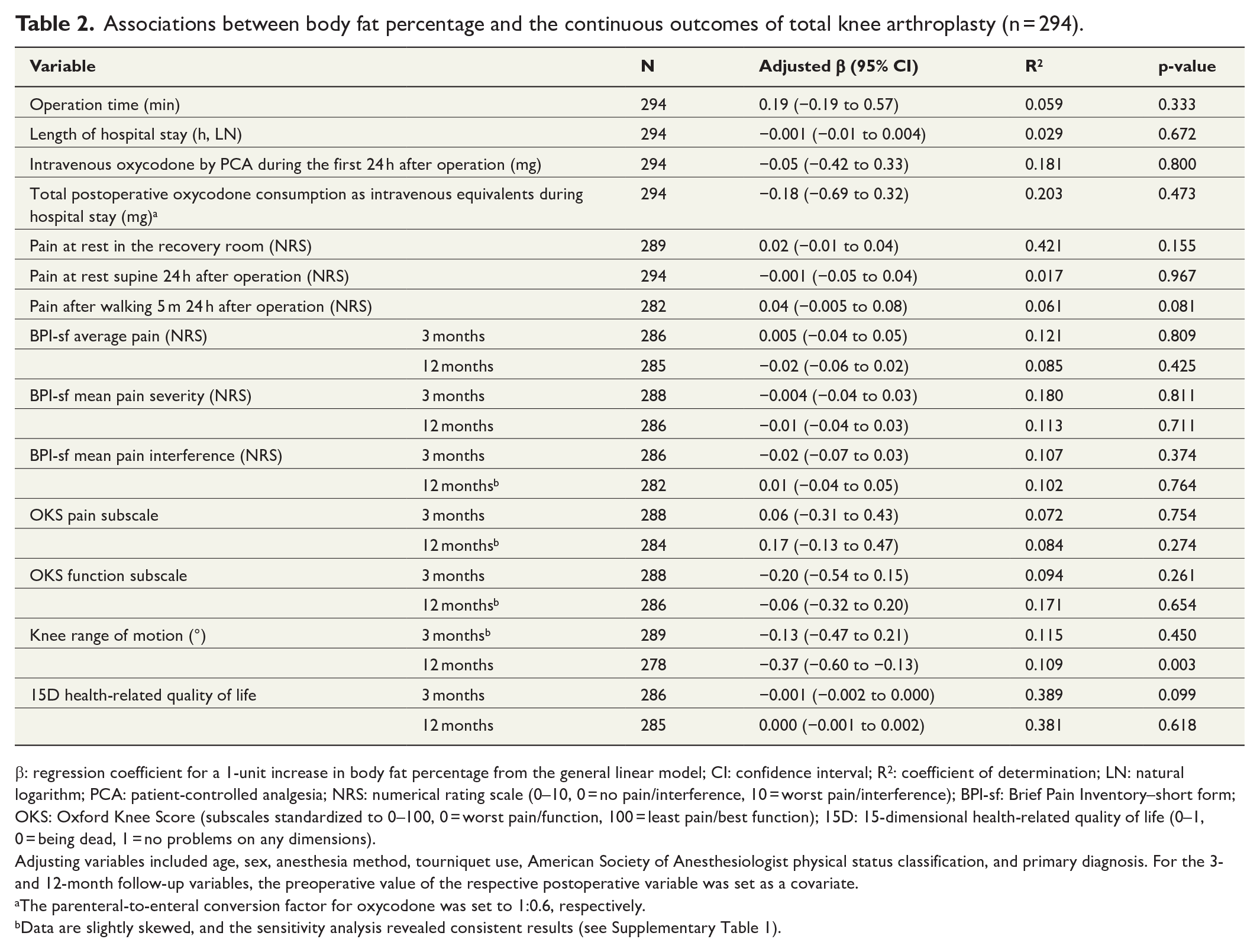
β: regression coefficient for a 1-unit increase in body fat percentage from the general linear model; CI: confidence interval; R2: coefficient of determination; LN: natural logarithm; PCA: patient-controlled analgesia; NRS: numerical rating scale (0–10, 0 = no pain/interference, 10 = worst pain/interference); BPI-sf: Brief Pain Inventory–short form; OKS: Oxford Knee Score (subscales standardized to 0–100, 0 = worst pain/function, 100 = least pain/best function); 15D: 15-dimensional health-related quality of life (0–1, 0 = being dead, 1 = no problems on any dimensions).
Adjusting variables included age, sex, anesthesia method, tourniquet use, American Society of Anesthesiologist physical status classification, and primary diagnosis. For the 3- and 12-month follow-up variables, the preoperative value of the respective postoperative variable was set as a covariate.
The parenteral-to-enteral conversion factor for oxycodone was set to 1:0.6, respectively.
Data are slightly skewed, and the sensitivity analysis revealed consistent results (see Supplementary Table 1).
Associations between body fat percentage and the dichotomous outcomes of total knee arthroplasty.
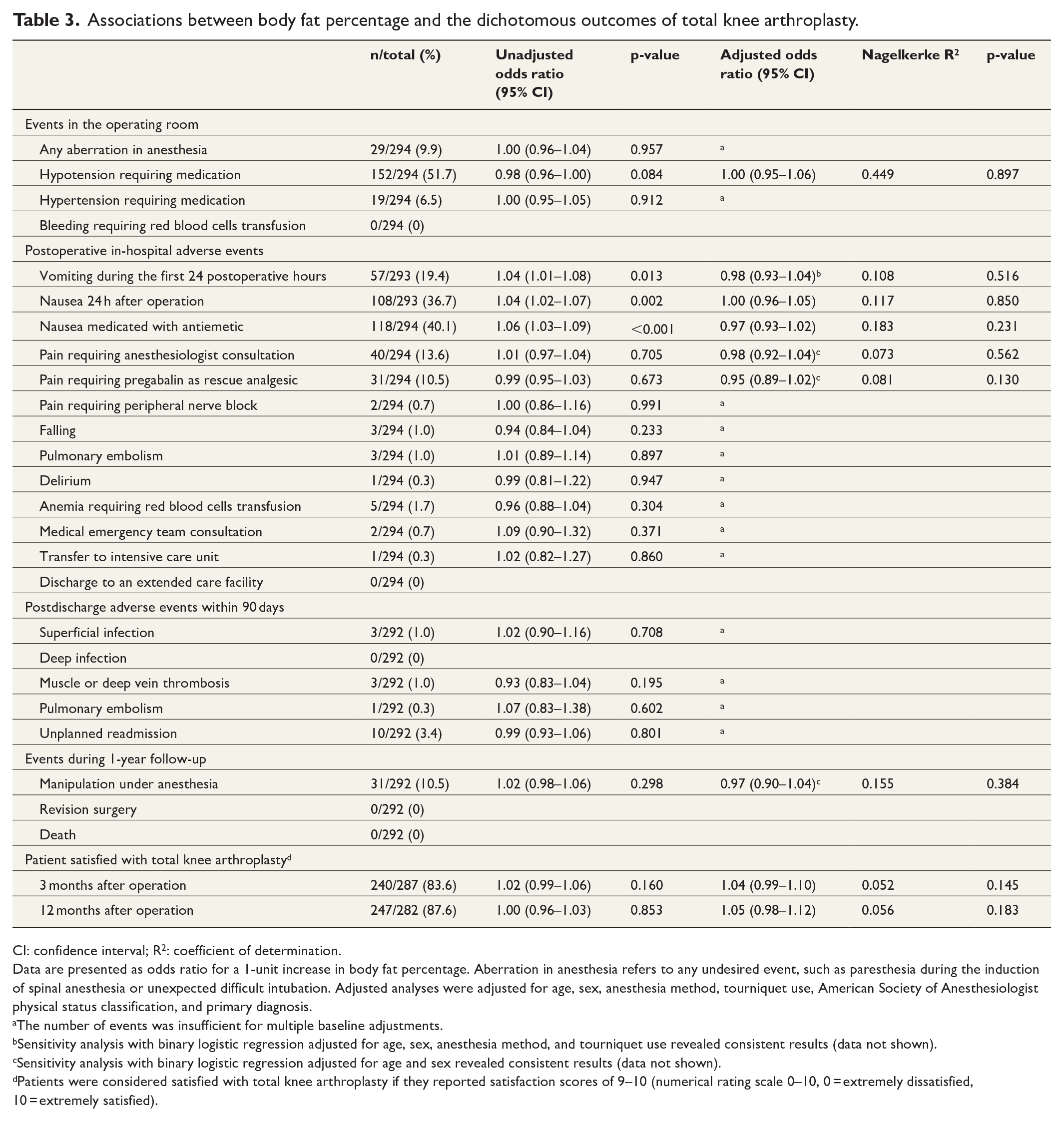
CI: confidence interval; R2: coefficient of determination.
Data are presented as odds ratio for a 1-unit increase in body fat percentage. Aberration in anesthesia refers to any undesired event, such as paresthesia during the induction of spinal anesthesia or unexpected difficult intubation. Adjusted analyses were adjusted for age, sex, anesthesia method, tourniquet use, American Society of Anesthesiologist physical status classification, and primary diagnosis.
The number of events was insufficient for multiple baseline adjustments.
Sensitivity analysis with binary logistic regression adjusted for age, sex, anesthesia method, and tourniquet use revealed consistent results (data not shown).
Sensitivity analysis with binary logistic regression adjusted for age and sex revealed consistent results (data not shown).
Patients were considered satisfied with total knee arthroplasty if they reported satisfaction scores of 9–10 (numerical rating scale 0–10, 0 = extremely dissatisfied, 10 = extremely satisfied).
In the unadjusted analyses, the increase in BFP increased the odds of postoperative nausea and vomiting; however, these associations were not significant in the adjusted analyses (Table 3). The numbers of most adverse events were too low for conducting an adjusted analysis (Table 3). None of the correlations between BFP and the continuous outcome variables reached the predefined threshold (r = 0.3) of weak correlation (Supplementary Table 2).
BMI and TKA-related outcomes
In the multivariable analysis, a 1-unit increase in BMI increased the operation time by 0.57 min (95% CI = 0.10 to 1.04; p = 0.018). The associations between BMI and in-hospital pain, pain management, and LOS were not significant (Tables 4 and 5). Increasing BMI was associated with improvements in pain scores at 3 months but not at 12 months after TKA (Table 4). The effects of BMI on patient-reported function, health-related quality of life, and satisfaction with TKA were not significant at 3 and 12 months after surgery (Tables 4 and 5). Nevertheless, a 1-unit increase in BMI affected the ROM by −0.47° (95% CI = −0.74 to −0.20; p < 0.001) at 12 months. In the sensitivity analyses concerning one pain management, two pain, and two functional variables, the results were consistent with the main analysis (Supplementary Table 3).
Associations between body mass index and the continuous outcomes of total knee arthroplasty (n = 399).
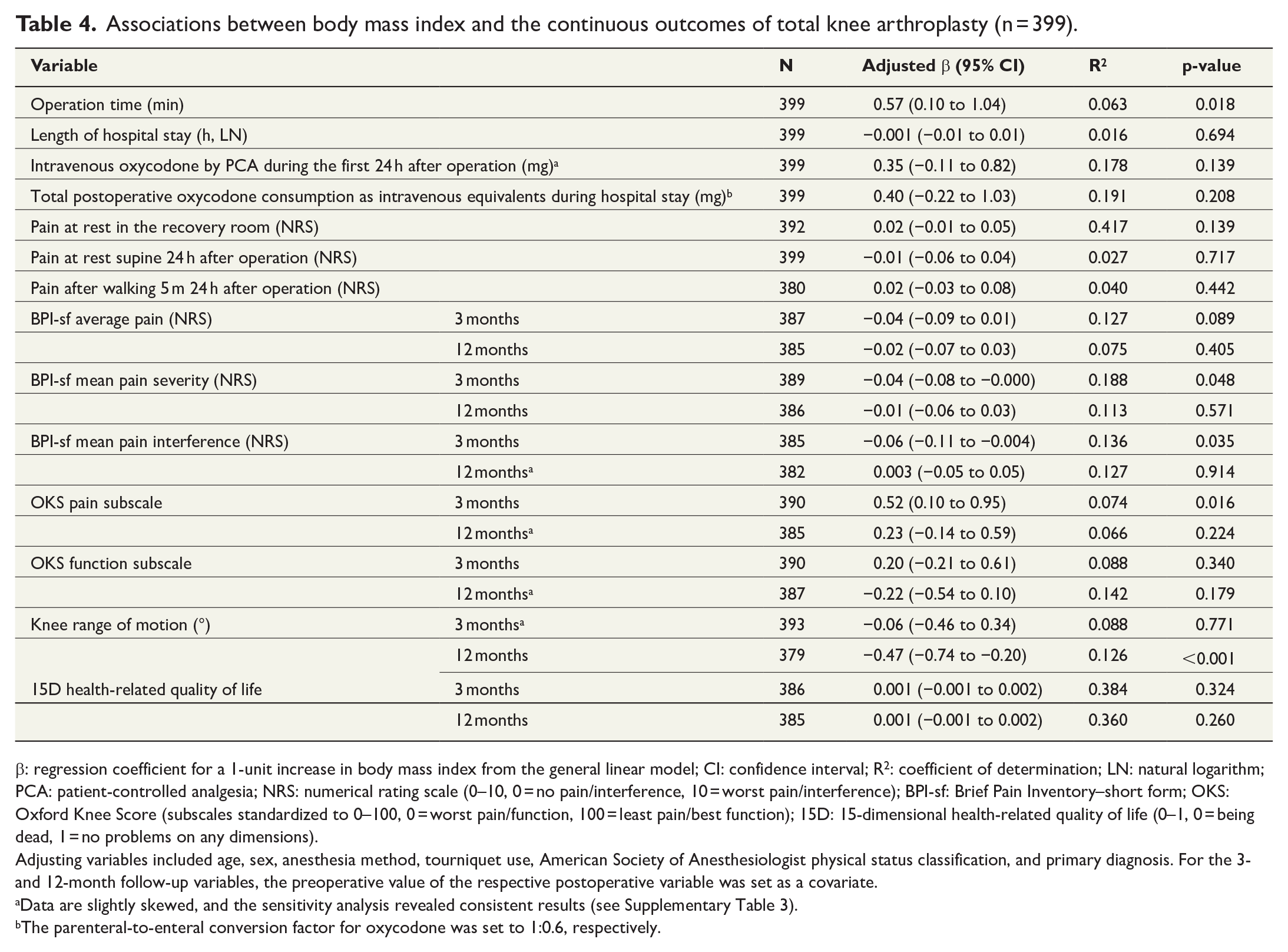
β: regression coefficient for a 1-unit increase in body mass index from the general linear model; CI: confidence interval; R2: coefficient of determination; LN: natural logarithm; PCA: patient-controlled analgesia; NRS: numerical rating scale (0–10, 0 = no pain/interference, 10 = worst pain/interference); BPI-sf: Brief Pain Inventory–short form; OKS: Oxford Knee Score (subscales standardized to 0–100, 0 = worst pain/function, 100 = least pain/best function); 15D: 15-dimensional health-related quality of life (0–1, 0 = being dead, 1 = no problems on any dimensions).
Adjusting variables included age, sex, anesthesia method, tourniquet use, American Society of Anesthesiologist physical status classification, and primary diagnosis. For the 3- and 12-month follow-up variables, the preoperative value of the respective postoperative variable was set as a covariate.
Data are slightly skewed, and the sensitivity analysis revealed consistent results (see Supplementary Table 3).
The parenteral-to-enteral conversion factor for oxycodone was set to 1:0.6, respectively.
Associations between body mass index and the dichotomous outcomes of total knee arthroplasty.
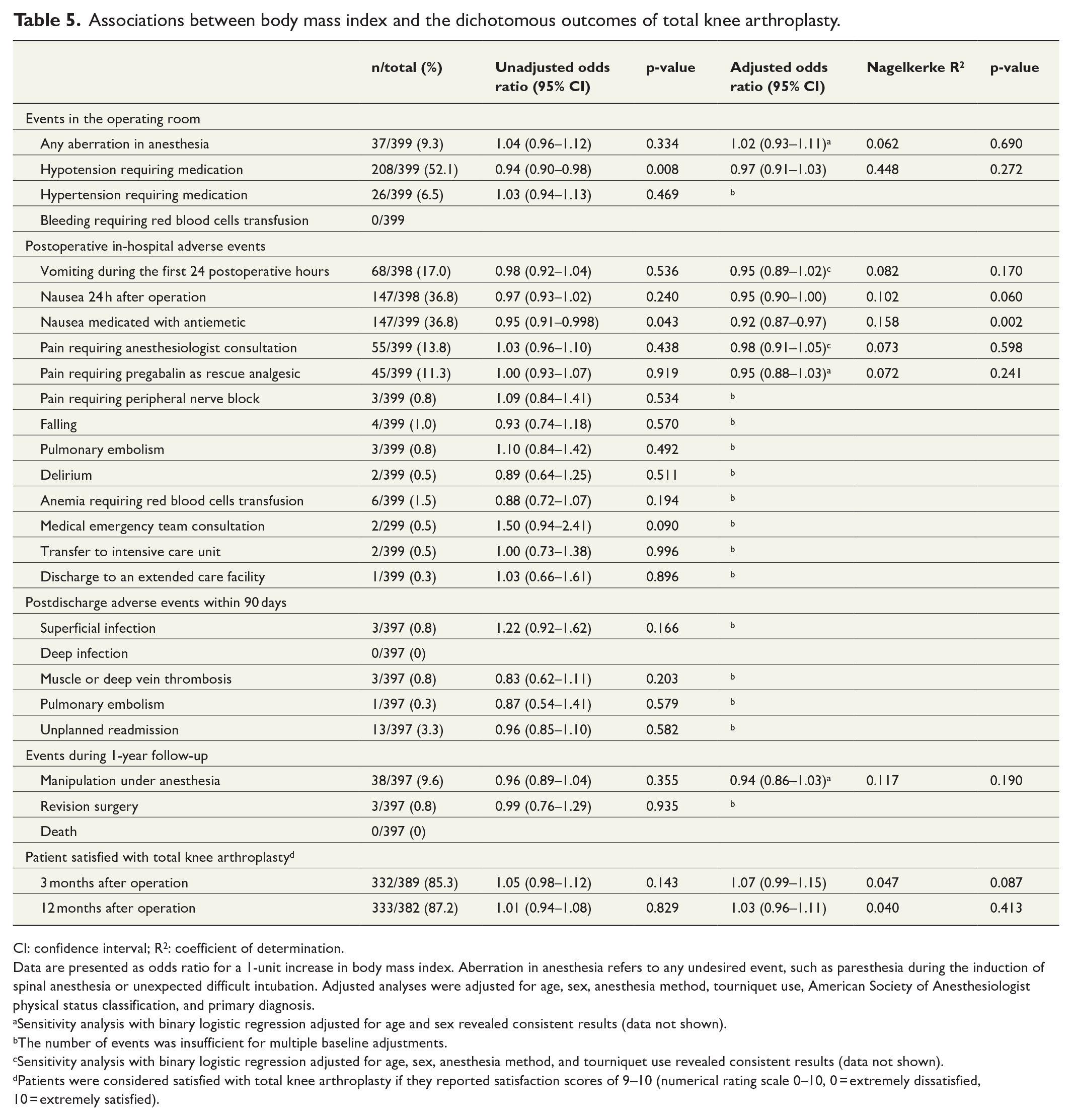
CI: confidence interval; R2: coefficient of determination.
Data are presented as odds ratio for a 1-unit increase in body mass index. Aberration in anesthesia refers to any undesired event, such as paresthesia during the induction of spinal anesthesia or unexpected difficult intubation. Adjusted analyses were adjusted for age, sex, anesthesia method, tourniquet use, American Society of Anesthesiologist physical status classification, and primary diagnosis.
Sensitivity analysis with binary logistic regression adjusted for age and sex revealed consistent results (data not shown).
The number of events was insufficient for multiple baseline adjustments.
Sensitivity analysis with binary logistic regression adjusted for age, sex, anesthesia method, and tourniquet use revealed consistent results (data not shown).
Patients were considered satisfied with total knee arthroplasty if they reported satisfaction scores of 9–10 (numerical rating scale 0–10, 0 = extremely dissatisfied, 10 = extremely satisfied).
In the unadjusted analyses, increasing BMI reduced the odds of intraoperative hypotension and postoperative nausea requiring medication (Table 5). However, in the adjusted analyses, only the association between BMI and the use of antiemetics remained statistically significant (Table 5). Other statistically significant associations between increasing BMI and adverse events were not detected. The correlations between BMI and the continuous outcome variables remained below the threshold of weak correlation (Supplementary Table 2).
Discussion
In this secondary analysis of a randomized trial with a 1-year follow-up, our hypothesis concerning the negative effects of increasing BFP on TKA-related outcomes appeared mostly incorrect. In the multivariable analyses, BFP was not predictive of operation time, LOS, in-hospital pain management, acute or persistent pain, adverse events, or patient-reported function or satisfaction 1 year after surgery. Thus, our results were inconsistent with those of previous studies which suggested that BFP is predictive of adverse events, pain, and function.19,20 However, some of the differences in the results might have originated from differences in study designs, sample sizes, outcome questionnaires, and follow-up times. Increased BFP was associated with decreased baseline-adjusted ROM 1 year after TKA; however, the clinical relevance of this finding remains questionable.
In the post hoc analyses, increasing BMI predicted increasing operation time, as reported previously.1–3 However, increasing BMI was not predictive of LOS, acute pain, pain management, or patient-reported outcomes 1 year after operation. Thus, our results support previous findings that indicated no association between BMI and patient-reported function and pain.10–12 The increase in BMI was predictive of statistically significant decrease in baseline-adjusted ROM 1 year postoperatively, which is consistent with a previous study. 16 However, we consider this finding clinically irrelevant owing to non-significant results in patient-reported outcomes which seem more important in assessing knee function than ROM. 34 Our results do not support the earlier report that indicated that increase in BMI might decrease the quality of life, but the previous study used a different questionnaire and follow-up time, which might explain the differences. 8 Even though increase in BMI did not predict postoperative nausea and vomiting during the first 24 postoperative hours in this study, it predicted decreasing odds for the need of postoperative antiemetics during the hospital stay. This might be regarded clinically relevant.
Although we investigated the predictive performance of both BFP and BMI on TKA-related outcomes, a comparison of these two body structure indicators as predictors was not the objective of the study. Thus, the cohorts differed, and the results were presented separately.
The strengths of this study include its prospective design. The patients were participants in a randomized trial in which treatments were thoroughly documented and standardized and followed modern fast-track protocols. We investigated the effects of BFP on multiple outcomes used to weigh the benefits of TKA. This study focused strictly on patients who underwent primary TKA, which increased the validity of our results compared with mixed surgery cohorts. Furthermore, the sample size of our BFP cohort was more substantial than that of a previous study that investigated associations between BFP and patient-reported outcomes. 19
This study also has some noteworthy limitations. First, the inclusion criteria, such as age ⩽75 years and preoperative BMI ⩽40 kg/m2, limit the generalizability of the results. Second, this was a single-center study. Third, the patients were not required to prepare themselves for the BIA, although dietary intake and previous exercise may affect the analysis. This might have biased the results. However, 271 (93.4%) of 290 patients had fasted for at least 2 h before BIA. In addition, most patients arrived at the preoperative clinic in the morning, and apart from short transitions and examinations, no physical stress was induced. Fourth, the inter-tester accuracy of the ROM measurements was not controlled which should be noted in interpreting the results. Fifth, this study was not powered to address rare adverse events.
The 1-year follow-up time might also be regarded as too short. However, a recent meta-analysis revealed no significant differences in patient-reported outcomes between 1 and 2 years after TKA. 35 In addition, numerous studies have reported that most patient-reported benefits could be measured within 1 year after operation. 36 In terms of OKS, the improvements do not seem to increase from the first to the fifth postoperative year.37,38 Similarly, ROM seems to remain at the level achieved during the first postoperative year. 16 Furthermore, the prevalence of moderate to severe persistent postsurgical pain at 1 year does not appear higher than at 2–5 years after TKA.11,29 Studies concerning health-related quality of life with 5-year follow-up durations have reported that after the first postoperative year, the quality of life might remain at the same level, improve, or deteriorate.38–40
In conclusion, our results suggest that BFP is poorly predictive of in-hospital results and of outcomes concerning patient-reported pain, function, and health-related quality of life 1 year after TKA. For nonmorbidly obese patients, increase in BMI seems to have only minor effects on TKA-related outcomes.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969221139722 – Supplemental material for Fat tissue is a poor predictor of 1 year outcomes after total knee arthroplasty: A secondary analysis of a randomized clinical trial
Supplemental material, sj-docx-1-sjs-10.1177_14574969221139722 for Fat tissue is a poor predictor of 1 year outcomes after total knee arthroplasty: A secondary analysis of a randomized clinical trial by Riku Palanne, Mikko Rantasalo, Anne Vakkuri, Klaus T. Olkkola, Tero Vahlberg and Noora Skants in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969221139722 – Supplemental material for Fat tissue is a poor predictor of 1 year outcomes after total knee arthroplasty: A secondary analysis of a randomized clinical trial
Supplemental material, sj-docx-2-sjs-10.1177_14574969221139722 for Fat tissue is a poor predictor of 1 year outcomes after total knee arthroplasty: A secondary analysis of a randomized clinical trial by Riku Palanne, Mikko Rantasalo, Anne Vakkuri, Klaus T. Olkkola, Tero Vahlberg and Noora Skants in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-3-sjs-10.1177_14574969221139722 – Supplemental material for Fat tissue is a poor predictor of 1 year outcomes after total knee arthroplasty: A secondary analysis of a randomized clinical trial
Supplemental material, sj-docx-3-sjs-10.1177_14574969221139722 for Fat tissue is a poor predictor of 1 year outcomes after total knee arthroplasty: A secondary analysis of a randomized clinical trial by Riku Palanne, Mikko Rantasalo, Anne Vakkuri, Klaus T. Olkkola, Tero Vahlberg and Noora Skants in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank Rita Linko, MD, PhD; Elina Reponen, MD, PhD; and Rami Madanat, MD, PhD for their advice concerning the study planning. The authors also thank Arja Mäkinen, RN, and Katarina Lahtinen, MD, for their invaluable work in data collection.
Author contributions
All authors contributed to the conceptualization and methodology. R.P., M.R., T.V., and N.S. contributed to the data curation. R.P. and T.V. contributed to the formal analysis. K.T.O., R.P., and A.V. contributed to the funding acquisition. R.P., M.R., and N.S. contributed to the investigation. N.S., A.V., and K.T.O. contributed to the project administration and supervision. R.P. contributed to the writing the original draft. All authors contributed to the reviewing and editing the draft.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.R. declared the following potential conflict of interest: research support from Zimmer Biomet and Smith & Nephew. Other authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the HUS Helsinki University Hospital Finnish government science grants (grant nos TYH 2017239 and TYH 2019113) and the HUS Helsinki University Hospital (grant no. Y102011095). Open access article processing charge was funded by Helsinki University Library. In addition, R.P. received personal grants from the Paulo Foundation, the Finnish Medical Foundation, the Research Foundation for Orthopaedics and Traumatology, the Finnish Arthroplasty Society, the Finnish Society of Anaesthesiologists, the Finnish Medical Society Duodecim, and the Orion Research Foundation sr. The funders had no role in the study design; in data collection, analysis, or interpretation; in writing the manuscript; or in the decision to submit the article for publication.
Supplemental material
Supplemental material for this article is available online.
Availability of data and materials
The raw data that were analyzed in this study are available from the corresponding author upon reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
