Abstract
Background and objective:
The ideal margin width for surgical resection of colorectal liver metastases has been extensively studied, but not sufficiently in accordance with other pathological factors. The aim of this study was to assess for the first time the prognostic impact of margin widths according to different prognostic pathological factors in colorectal liver metastasis.
Methods:
We evaluated 101 patients with a single resected metastasis. Slides stained by HE were assessed for the presence of poorly differentiated clusters, peritumoral inflammatory infiltrate, tumor pseudocapsule, and tumor borders pattern. Overall survival, disease-free survival, and hepatic recurrence were evaluated. The pathologic factors prognostic impact was evaluated according to a (< or ⩾) 10-mm margin size.
Results:
Factors independently associated with a shorter overall survival were absence of tumor pseudocapsule (p < 0.001) and infiltrative tumor border pattern (p = 0.019). The absence of tumor pseudocapsule was the only factor independently associated with shorter disease-free survival (p < 0.001). Hepatic recurrence was associated with infiltrative tumor border and absence of pseudocapsule. Margin width ⩾10 mm did not impact overall survival independently of the studied histological prognostic factors.
Conclusions:
In colorectal liver metastasis resection, the absence of tumor pseudocapsule was significantly associated with shorter overall survival and disease-free survival and hepatic recurrence. However, margins larger than 10 mm did not offer survival benefit when other pathologic negative prognostic factors were concomitantly analyzed, reinforcing the idea that biology, rather than margin size, is crucial for prognosis.
Introduction
The liver is the most frequent site of metastatic disease from colorectal cancer (CRC); 1 25% to 50% of patients with CRC present liver metastases at the diagnosis or during follow-up. 2 Management of colorectal liver metastases (CLMs) relies on a multidisciplinary approach, where liver resection develops a key role as the optimal curative modality, leading to 5-year overall survival (OS) rates ranging from 45% to 58%.2 –5
During the last years, several histopathological features in CLM had been reported as prognostic factors. Some of these factors can be easily identified by routine staining with hematoxylin and eosin (HE), as tumor budding, poorly differentiated clusters (PDCs), peritumoral inflammatory infiltrate, tumor pseudocapsule, and tumor borders pattern.2,3,6
The ideal margin width for CLM resection has been extensively studied in the last years. Historically, a 10-mm free margin was accepted as standard of care.2,7 –9 However, in an era of modern chemotherapy and aggressive surgical approaches, studies have shown that smaller margins—such as 5 mm and, most commonly, 1 mm—do not have a detrimental impact on local recurrence and survival rates.2,4,10
As histopathological prognostic factors occur in the tumor–normal liver interface, we hypothesized that the presence of these factors could impact the prognosis according to the classical 10-mm margin width. The aim of the current study was to assess the prognostic impact of pathological prognostic factors as PDC, peritumoral inflammatory infiltrate, tumor pseudocapsule, and tumor borders pattern according to a margin width shorter or larger than 10 mm.
Methods
After approval by the Institutional Ethics Committee, we reviewed medical records of patients who underwent the first resection of CLM in our institution between January 2007 and December 2015. All data were collected and managed using the REDCap electronic data capture tools. 11
Since our goal was to determine the impact of margin width according to the presence of different pathological factors, we chose to evaluate only patients with a single metastatic nodule to avoid bias because multinodular patients may present different pathological findings and different margin sizes. Exclusion criteria were patients with incomplete macroscopic resection (R2), postoperative death within 90 days, pathological complete response, and absence of slides for complete pathological evaluation.
All cases were discussed in a weekly multidisciplinary meeting prior to surgery. Parenchyma preserving techniques were routinely employed for CLM resection.12,13 The following data on clinicopathologic variables were evaluated: age, gender, body mass index (BMI), primary tumor site (colon or rectum), T staging of primary CRC, primary tumor lymph node status, timing of CLM diagnosis (synchronous when up to 12 months from CRC resection), preoperative serum carcinoembryonic antigen (CEA) level, and tumor size.
After liver resection, patients were followed according to our institutional protocol with the assessment of CEA and imaging evaluation every 4–6 months. Criteria for recurrence were the presence of histological confirmation or radiologic evidence of progression with subsequent clinical progression, and/or increasing serum levels of CEA. Disease-free survival (DFS) was defined as the time interval between liver resection and the date of the first documented recurrence at any site. Hepatic recurrence was considered when the first evidence of relapse was the liver, independently of other sites of recurrence. OS was defined as the time interval between liver resection and the date of death or the most recent date of follow-up if the patient was alive. Deaths from other causes were treated as censored events.
Pathological evaluation
All resected specimens were submitted to a standardized histological evaluation by the same experienced pathologist who was blind to clinical data (S.F.F.). Tumor size was measured in centimeters before fixation of the specimen. Formalin-fixed paraffin-embedded tissue blocks were cut and used for histological examination with HE staining. Surgical margin width was considered the closest distance between the tumor edge and the transected liver surface, measured microscopically in millimeters. Slides were evaluated for the presence of PDC, tumor borders pattern, presence of tumor pseudocapsule, and peritumoral inflammatory infiltrate. PDC was defined as cancer clusters of five or more cancer cells in the stroma and/or tumoral border 14 after evaluation of the whole tumor including its advancing edge and classified as present or absent. For the assessment of mucinous carcinoma, malignant clusters with these features that infiltrated the stroma with minimal extracellular mucin formation were classified as PDC, while cell clusters within a large mucin pool or mucinous lake were not. 15 Tumor borders pattern was defined according to Jass et al.’s 16 classification as either infiltrative, when the tumor spreads through the surrounding tissue, dissecting between normal hepatocytes, or expansive, when the tumor edges expanded in a well-delineated fashion, pushing the adjacent liver tissue. Tumor pseudocapsule was considered when there was a fibrous tissue between the tumor and the liver parenchyma. Peritumoral inflammatory infiltrate was defined as the presence of lymphocytes in the periphery of the tumor, being classified as absent to mild and moderate to severe. Fig. 1 illustrates different subtypes in pathological evaluation.
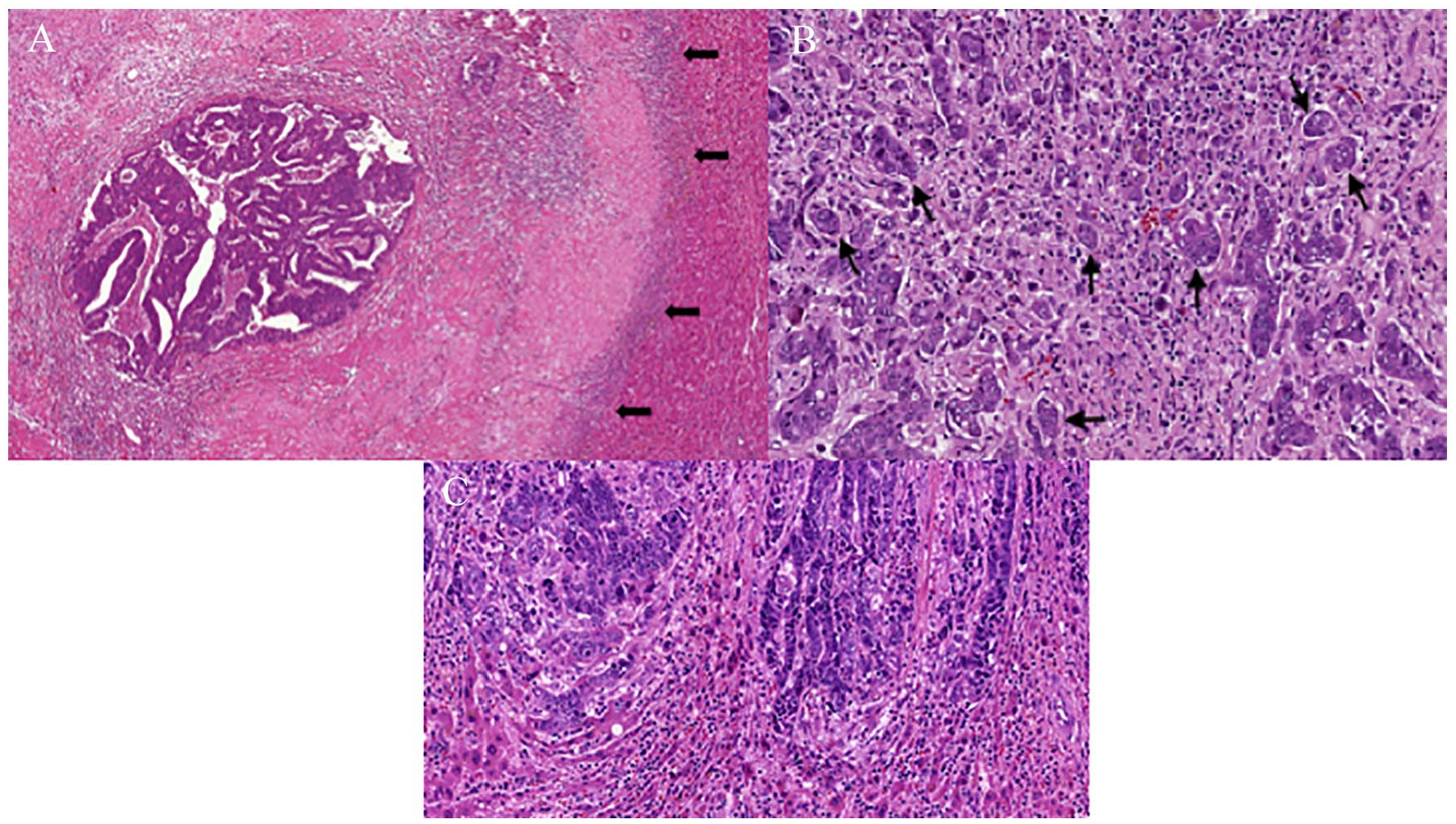
(A) Illustration of histopathological findings in colorectal liver metastasis in slides stained by hematoxylin and eosin. (A) Expansive borders exhibiting a thick fibrous pseudocapsule and surrounded by a moderate inflammatory infiltrate (arrows) (×2); (B) poorly differentiated clusters characterized by clusters comprised of five or more tumor cells within the tumor, without glandular structures (×20); (C) infiltrative borders characterized by tumor cells infiltrating the adjacent hepatic parenchyma (×20).
Statistical analysis
OS and DFS were evaluated using the Kaplan–Meier method and a comparison between curves was performed with the log-rank test. The stepwise backward Cox proportional hazards regression model was used to identify factors independently associated with OS and DFS. Variables with a p< 0.2 on univariate analyses were selected for multivariate logistic regression analysis. Hazard ratio (HR) values with 95% confidence interval (95% CI) were calculated. Factors associated with hepatic recurrence were identified by logistic regression analysis and summarized with odds ratio (OR) and respective 95% CI. For tumor pseudocapsule and hepatic recurrence, Fisher’s exact test was applied since no patient with tumor pseudocapsule presented hepatic recurrence.
A p value <0.05 was considered as statistically significant. Statistical analysis was performed with SPSS for Mac, version 26 (IBM, Armonk, NY).
Results
During the period of study, 552 liver resections for CLM were performed. After applying inclusion and exclusion criteria, 101 patients with a single CLM were included in the analytic cohort. Thirty-six (35.6%) underwent major hepatectomies (any resection of three or more contiguous liver segments 17 ), and 65 (64.4%) underwent minor hepatectomies.
Clinicopathological and histopathological features
The median age was 63 years (range 28–79), and 55 (54.5%) patients were men. Median BMI was 26.1 kg/m2 (range 18.3–34.2). The site of primary CRC was the colon in 57 (56.4%), and rectum in 44 (43.6%) patients. Synchronous CLM was observed in 62 (61.4%) patients. Concerning the primary tumor, 45 (44.6%) patients had lymph node metastasis and 86 (85.1%) were classified as T3 or T4. Median preoperative CEA was 10.3 ng/mL (range 0.8–6058 ng/mL). Regarding the nodule size, the median diameter was 3 cm (range 2.4–22 cm). The margin width was <10 mm in 66 and ⩾10 mm in 35 patients. All patients received perioperative oxaliplatin-based chemotherapy (pre and/or postoperative) during the study period.
On pathological evaluation, PDC was observed in 52 (51.5%) patients. Absent-to-mild peritumoral inflammatory infiltrate was seen in 71 (70.3%) patients and moderate or severe in 30 (29.7%). Tumor pseudocapsule was absent in 75 (74.3%) patients and present in 26 (25.7%). Tumor border was defined as expansive in 28 (27.7%) patients and infiltrative in 73 (72.3%).
Follow-up and long-term outcomes
Median follow-up after liver resection was 65 months (mean 62 months, range 12–156 months). The recurrence rate was 62.4% (n = 63), with a mean time between resection and recurrence of 13 months (median 9 months, range 1–86 months). Liver-only recurrence was observed in 18 (28.6%) patients, whereas liver plus extrahepatic recurrence in 13 (20.6%) patients; 32 (50.8%) patients had isolated extrahepatic recurrence. OS was 94.9%, 73.8%, and 57.6%, at 1, 3, and 5 years, respectively. DFS was 63%, 38.8%, and 36.6% at 1, 3, and 5 years, respectively.
Factors associated with overall survival, disease-free survival, and hepatic recurrence
On univariate analysis, factors associated with a shorter OS were CEA levels >20 ng/mL, presence of PDC, absence of tumor pseudocapsule, and tumor infiltrative borders. Margin width <10 mm was not related to a decrease in OS. On multivariate analysis, factors independently associated with shorter OS were absence of tumor pseudocapsule (HR 102.42, 95% CI 9.08–1154.85, p < 0.001) and infiltrative tumor border pattern (HR 13.15, 95% CI 1.53–112.72, p = 0.019). Table 1 summarizes factors associated with OS.
Univariate analysis of prognostic factors for overall survival in patients underwent to liver resection for uninodular colorectal liver metastases.
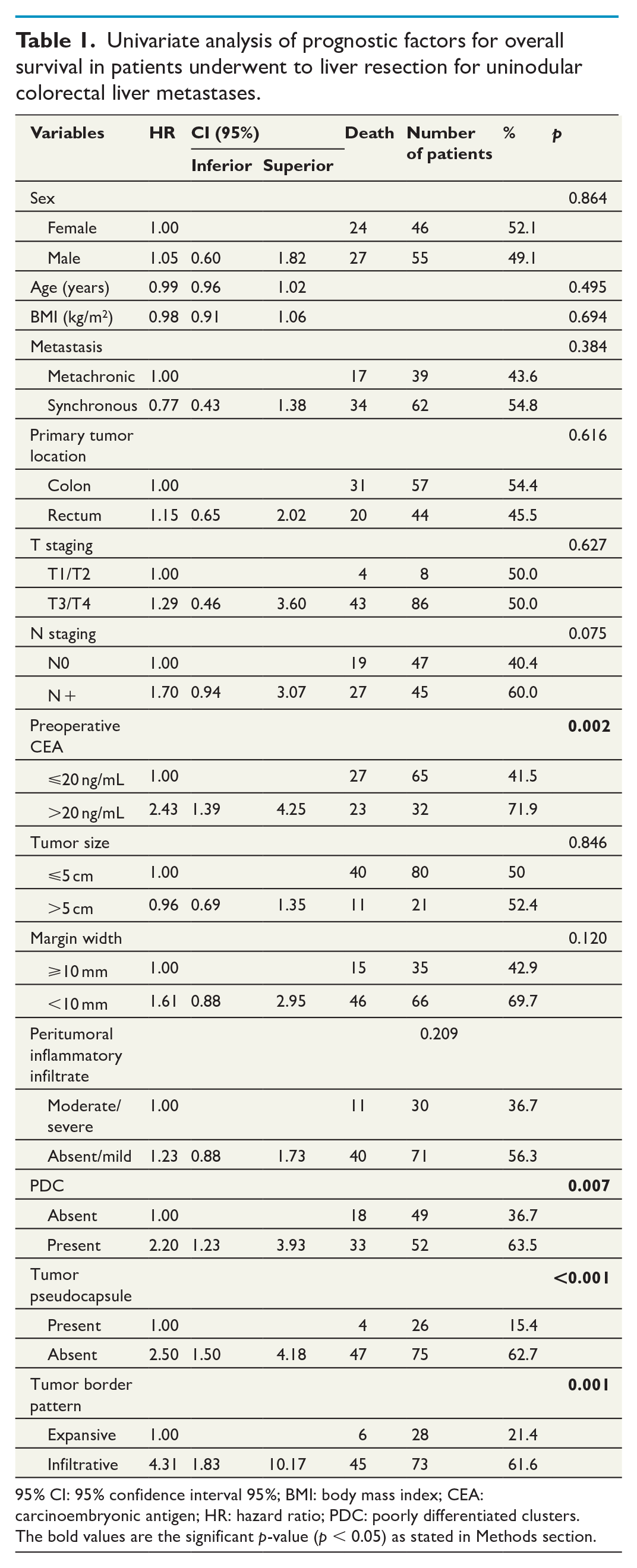
95% CI: 95% confidence interval 95%; BMI: body mass index; CEA: carcinoembryonic antigen; HR: hazard ratio; PDC: poorly differentiated clusters.
The bold values are the significant p-value (p < 0.05) as stated in Methods section.
On univariate analysis, factors associated with a shorter DFS were CEA levels >20 ng/mL, presence of PDC, absent-to-mild peritumoral inflammatory infiltrate, absence of tumor pseudocapsule, and tumor infiltrative borders. Margin width < 10 mm was also not related to a shorter DFS. On multivariate analysis, the only factor independently associated with a decrease in DFS was the absence of tumor pseudocapsule (HR 2.42, 95% CI 1.53–3.84, p < 0.001). Table 2 summarizes variables associated with DFS.
Univariate analysis of prognostic factors for disease-free survival in patients underwent to liver resection for uninodular colorectal liver metastases.
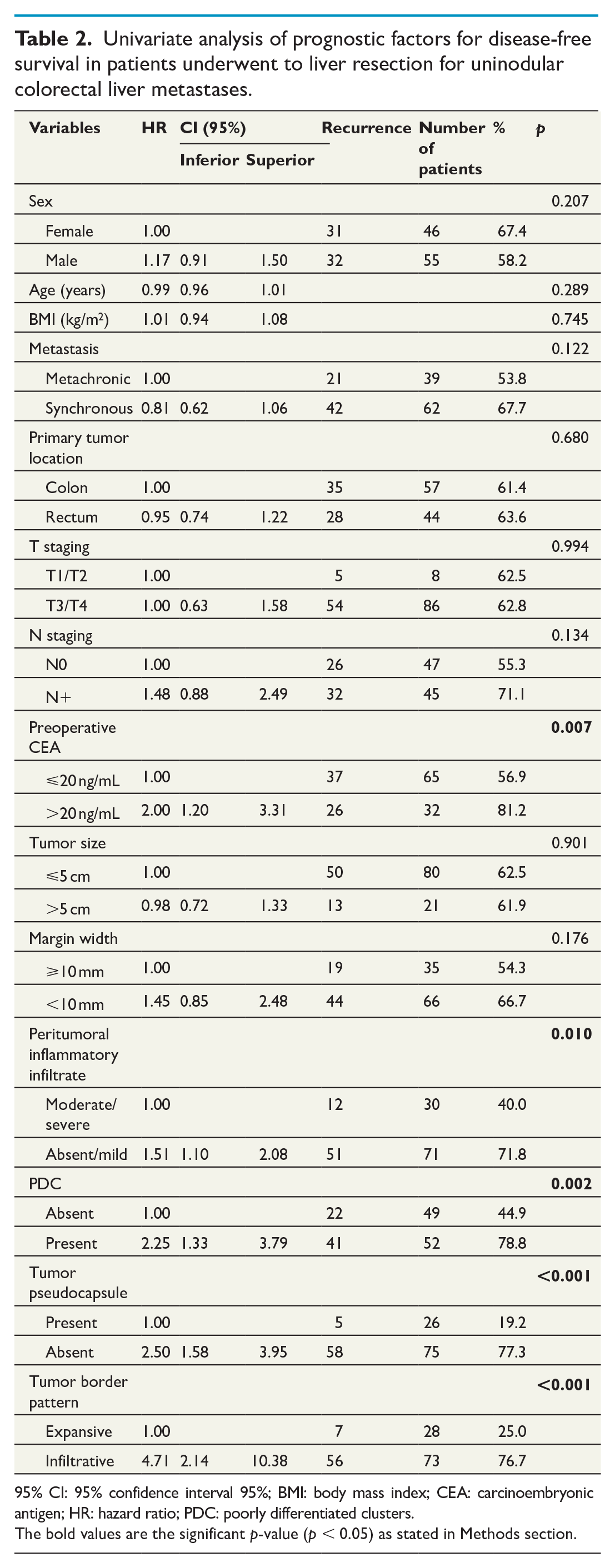
95% CI: 95% confidence interval 95%; BMI: body mass index; CEA: carcinoembryonic antigen; HR: hazard ratio; PDC: poorly differentiated clusters.
The bold values are the significant p-value (p < 0.05) as stated in Methods section.
Finally, the pathological factors associated with hepatic recurrence on univariate analysis were absence of tumor pseudocapsule and infiltrative tumor borders. None of the patients with the presence of tumor pseudocapsule had hepatic recurrence, the reason why it was not evaluated by multivariate analysis. Table 3 summarizes factors associated with hepatic recurrence.
Univariate analysis of factors associated to hepatic recurrence in patients underwent to liver resection for uninodular colorectal liver metastases.
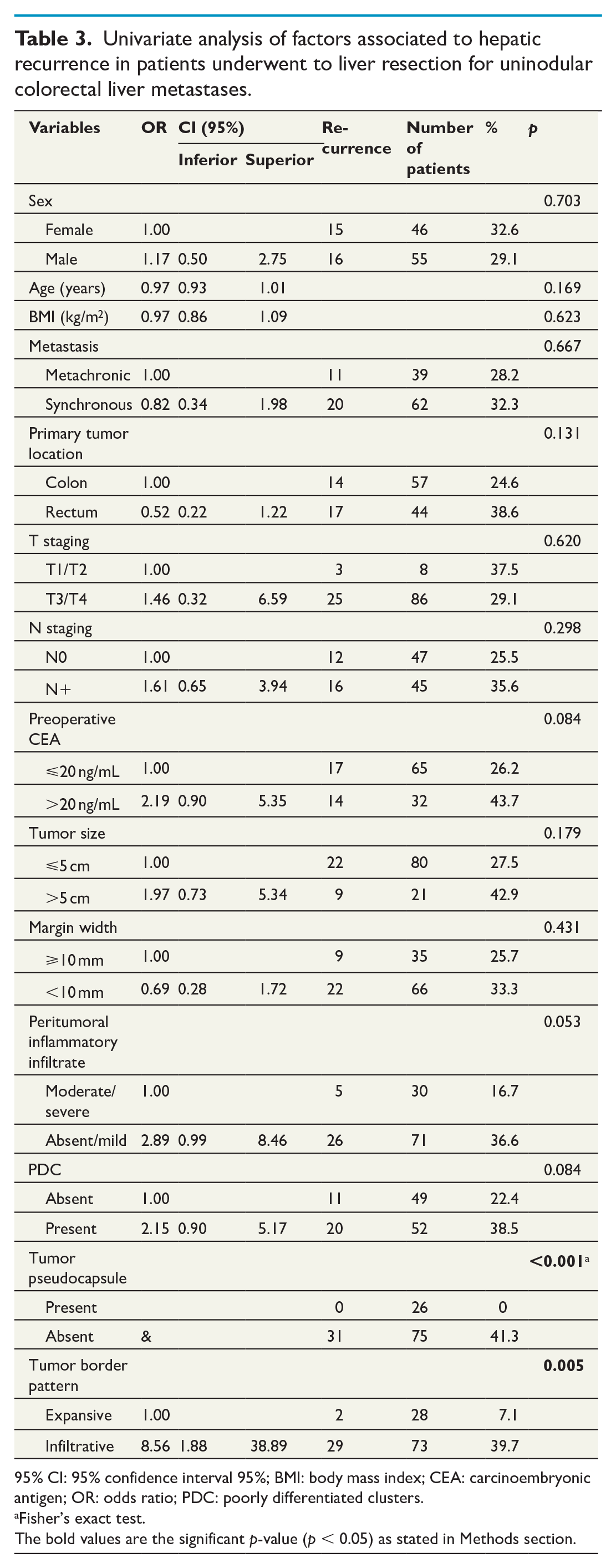
95% CI: 95% confidence interval 95%; BMI: body mass index; CEA: carcinoembryonic antigen; OR: odds ratio; PDC: poorly differentiated clusters.
Fisher’s exact test.
The bold values are the significant p-value (p < 0.05) as stated in Methods section.
Margin width and histopathological factors
OS and DFS were evaluated according to the presence of different pathological factors and a cutoff of 10-mm margin width. Subgroups of patients with presence or absence of PDC or pseudocapsule or expansive or infiltrative borders were not associated with shorter OS and DFS according to a <10-mm margin width. Table 4 summarizes OS and DFS for these subgroups of pathological factors according to margin width.
Overall and disease-free survival for different pathological factors according to the margin width in patients submitted to liver resection for colorectal liver metastases.
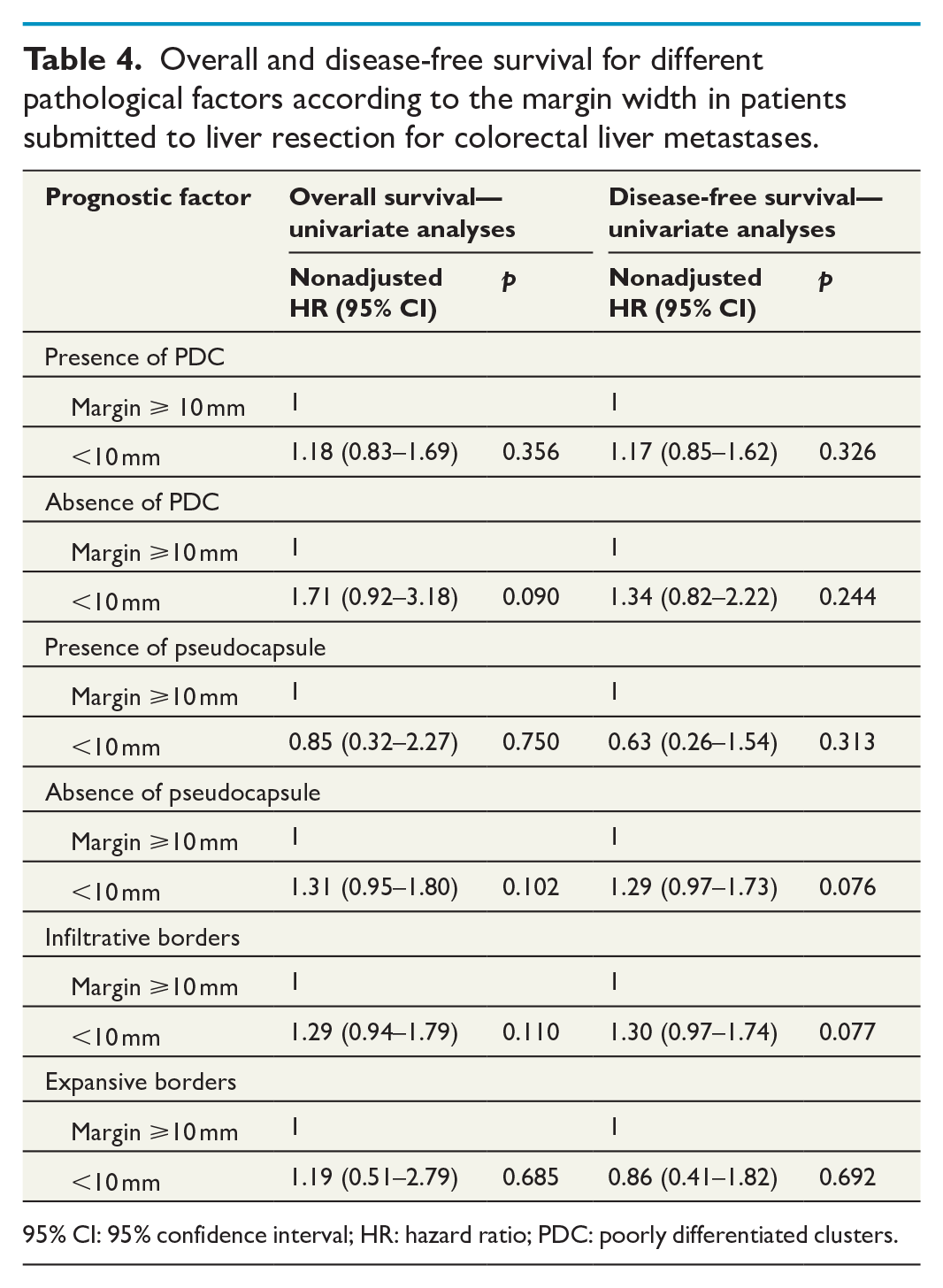
95% CI: 95% confidence interval; HR: hazard ratio; PDC: poorly differentiated clusters.
Discussion
Our study was motivated by the need for a deepening on the understanding of the impact of margin width on OS in patients with CLM. From the pathological prognostic factors evaluated, the surgical margin is under the control of the surgeon and can be changed, at least partially, for an optimal result. 18 However, we understand that surgical margin width alone could be a partially informative measure; therefore, margin size should be evaluated in association with other prognostic factors. For several years, a 10-mm surgical margin was recommended, based on the observation that up to 95% of microsatellite tumors were located within 1 cm of the tumor border in the absence of neoadjuvant chemotherapy. 8 After the advent of more effective systemic therapy regimens, it was postulated that malignant cells at tumor borders could be treated, allowing narrower margins.4,19 More recently, authors raised the hypothesis that oncological outcomes were not directly associated with the margin width or surgical technique, but a consequence of the underlying tumor biology.20,21
Our group has recently reported the prognostic impact of infiltrative borders as an independent factor associated with shorter OS and DFS, and higher hepatic recurrence rates. 22 A margin size larger than 10 mm did not impact prognosis in patients with infiltrative or pushing border patterns, reinforcing the preponderance of tumor biology over the margin width.
A question to be addressed in CLM is which margin width should be considered the “real margin” in multinodular patients. Traditionally, the closest margin of any of the metastatic nodules is considered the real margin. However, Sasaki et al. 23 proposed that the most relevant margin is the closest margin of the largest tumor, based on the hypothesis that the largest lesion is more likely to harbor occult microscopic disease beyond the grossly visible edge of the tumor. Another question is that multinodular patients could express different histopathological factors between nodules, hardening the interpretation of results. To avoid a biased result, we decided to evaluate only patients with single metastatic nodules. Thus, a small group of patients with more reliable and precise data was studied.
Beyond tumor border pattern, we chose to evaluate the presence of tumor pseudocapsule, PDC, and peritumoral inflammatory infiltrate for the following reasons: these factors are easily recognizable with HE staining used for routine histopathological examination without any additional cost; these factors are markers of tumor biology being present at the tumor–normal liver surface, which could be counterbalanced by increasing the dimensions of surgical margins; and, to the best of our knowledge, excepting tumor border pattern, none of these factors had been compared with the margin width.
Tumor pseudocapsule is characterized by myofibroblasts-stimulating collagen production and may act as a mechanical and chemical barrier, limiting tumor expansion and aggressiveness. It was considered as an independent favorable prognostic factor in CLM.2,3,24,25 PDC occurs at tumor borders and/or stroma, whose presence has also been recently reported as an independent negative prognostic factor in CLM.3,26,27 Peritumoral inflammatory infiltrate represents a host response to the tumor, composed predominantly by T-lymphocytes whose maximum cytotoxic activity against tumor cells occurs at tumor border and is described as a favorable prognostic factor in CLM.3,21,28
Our results highlight the prognostic impact of the absence of tumor pseudocapsule in CLM, as an independent prognostic factor for shorter OS and DFS. Finally, our study reinforces the concept that patients’ survival is determined predominantly by tumor aggressiveness rather than surgical margins width, since any of the studied pathological factors did not have an OS and DFS influenced by a margin width ⩾10 mm.
Nowadays, the evaluation of these pathological factors is only available after surgical resection. Nevertheless, recent studies about radiomics methods have demonstrated the possibility of predicting preoperatively pathological factors such as a tumor border pattern in CLMs.29,30 If these results are replicable and other pathological factors are predicted by radiomics, maybe we could assess prognosis more precisely before surgery and improve selection criteria for surgical resection and neoadjuvant therapy.
The present study has limitations including the small number of patients and its retrospective character. Moreover, we did not evaluate the impact of neoadjuvant chemotherapy. The reason why we opted not to evaluate the impact of chemotherapy is that there is a clear selection bias since the patients who underwent neoadjuvant chemotherapy were those supposedly with a more aggressive tumor. Our findings open a perspective for a deeper understanding of the effect of chemotherapy on these pathological factors. It is described concentric tumor reduction induced by chemotherapy and, in patients submitted to chemotherapy, no tumor cells were found 4 mm beyond tumor periphery. 19
Despite limitations, to the best of our knowledge, this is the first study demonstrating the impact of different pathological prognostic factors in CLM, especially PDC and tumor pseudocapsule, compared to the margin width, and the first one to use a restrictive selected evaluation of uninodular metastasis, minimizing bias.
Conclusion
In patients with a single CLM, factors independently associated with a shorter OS were the absence of tumor pseudocapsule and infiltrative tumor border pattern; the only factor independently associated with a shorter DFS was the absence of tumor pseudocapsule. Most importantly, margins ⩾10 mm did not independently impact OS, reinforcing the concept that prognosis is predominantly determined by tumor biology rather than by margin width.
Footnotes
Author contributions
G.M.F., E.S.d.M., and P.H. designed the study; S.F.F. performed pathological evaluation; G.M.F. performed statistical analysis; G.M.F., J.A.P.K., and P.H. wrote the paper; and E.S.d.M., V.B.J., F.F.C., and V.A.F.A. reviewed the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
The study was approved by the Institutional Ethics Committee. Informed consent was waived due to its retrospective.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by grant #2015/06551-4, São Paulo Research Foundation (FAPESP).
