Abstract
Background and Aim:
Lymph node involvement is the strongest prognostic factor for patients with gastric cancer; patients with node-negative gastric cancer present with better survival. However, some patients develop recurrent gastric cancer. The aim of this study was to review the clinicopathological characteristics and factors for prognosis in patients with node-negative gastric cancer who underwent curative resection.
Methods:
Between January 2004 and December 2015, the clinicopathological characteristics of 302 patients with node-negative gastric cancer who underwent curative gastrectomy in our hospital were retrospectively analyzed.
Results:
The 1-, 3-, and 5-year overall survival rates for patients with node-negative gastric cancer were 80.0%, 69.0%, and 63.0%, respectively. Univariate analysis showed that tumor size, histologic type, and depth of invasion had significant effects on survival (p < 0.05). Multivariate analysis showed that tumor size (hazard ratio: 1.83%, 95.0% confidence interval: 1.13–2.96, p = 0.014), histologic type (hazard ratio: 1.57%, 95.0% confidence interval: 1.01–2.44, p = 0.042), and depth of invasion (hazard ratio: 1.38%, 95.0% confidence interval: 1.14–1.67, p = 0.001) were independent prognostic factors.
Conclusion:
Tumor size, histologic type, and depth of invasion are important prognostic factors in patients with node-negative gastric cancer. These parameters should be considered to stratify patients for therapy and follow-up strategies.
Introduction
Gastric cancer remains one of the most frequent malignancies and is the third leading cause of cancer-related death worldwide. Although the survival of patients with gastric cancer has been significantly improved due to advances, such as more reasonable lymph node dissections, chemotherapy regimens, and targeted therapy, the prognosis of gastric cancer patients is still not optimistic because of postoperative recurrence and metastasis (1). As an important cause of tumor relapse, the presence of lymph node metastases is a well-known predictor of poor outcome (2). Despite the fact that patients with node-negative gastric cancer have better overall survival (OS) rates compared to those with nodal metastases, a proportion of patients remain at risk of recurrence. Therefore, there is an urgent need to identify adverse prognostic factors. While many previous studies have clarified the relationship between the depth of tumor invasion and OS (3, 4), the role of other factors, including tumor size, tumor location, and the presence of lymphovascular invasion, remains uncertain (5, 6). This information could be significant for decisions about the administration of adjuvant therapy. Importantly, many node-negative patients with an increased risk of recurrence do not undergo chemotherapy. Therefore, if we identify useful factors that more accurately predict prognosis, more specialized strategies could be prepared for follow-up examination after surgery. These strategies will optimize existing treatment options and enable patients with node-negative gastric cancer to obtain better curative outcomes.
Methods
Patients
We retrospectively analyzed the data collected from gastric cancer patients who underwent curative gastrectomy in our hospital from January 2004 to December 2015. All patients underwent D1+ or D2 lymph node dissection with more than 15 nodes harvested in accordance with the Japanese Gastric Cancer Association Guidelines. Among them, D1+ resection was usually performed for T1 stage patients, while D2 resection was usually performed for patients with a higher stage, there were 131 and 171 cases, respectively. Lymph node metastasis status was determined by routine pathological examination. None of the patients were given chemo-radiation therapy before the operation. Patients who simultaneously suffered from other synchronous primary tumors and underwent an emergency operation for gastric cancer were excluded. Patients with lymph node metastases and distant metastases were also excluded. Totally, electrocardiogram (ECG) with node-negative was 73.6% (131/178) and advanced gastric cancer (AGC) with node-negative was 25.4% (171/674). Finally, data from 302 patients were included: 131 cases of early gastric cancer and 171 cases of AGC.
Data Collection
Patient data, including demographic features, operation procedures, pathological findings, postoperative adjuvant therapy, and postoperative survival, were noted. Demographic data included age and gender. Operation procedures included proximal subtotal gastrectomy, distal subtotal gastrectomy, and total gastrectomy. Pathological findings included tumor location, tumor size, depth of tumor invasion, the presence of lymphovascular invasion, and histologic classification. Depth of tumor invasion was based on the seventh edition of the Union for International Center Control/American Joint Committee on Cancer Classification System. According to the World Health Organization Classification, the histological types of gastric adenocarcinoma were divided into a highly differentiated group and a poorly differentiated group. The highly differentiated group included papillary adenocarcinoma, highly differentiated tubular adenocarcinoma, and moderately differentiated tubular adenocarcinoma. The poorly differentiated group included poorly differentiated adenocarcinoma, signet-ring cell carcinoma, mucinous adenocarcinoma, and anaplastic carcinoma. With regard to adjuvant chemotherapy post-operation, advanced gastric carcinoma patients were further divided into a chemotherapy group and a non-chemotherapy group. Adjuvant chemotherapy scheme followed National Comprehensive Cancer Network (NCCN) guidelines for gastric cancer (7). For postoperative outcomes, we assessed patient OS. OS was defined as the duration from the date of surgery to the date of death or the last follow-up visit. The follow-up periods ranged from 1 to 117 months, and the median follow-up period was 39 months. This study was approved by the Regional Ethics Committee of our hospital.
Statistical Analysis
All statistical analyses were performed by SPSS 19.0. Patient survival was estimated using the Kaplan–Meier method, and the differences between subgroups were assessed using the log-rank test. Potentially important factors obtained by univariate analysis (p < 0.05) were included in the multivariate analysis. Prognostic factors were analyzed using the Cox proportional hazards model. Values of p < 0.05 were considered to be statistically significant.
Results
Clinicopathologic characteristics
Of the 302 patients, there were 217 males and 85 females. Patient age ranged from 27 to 85 years old with a median age of 60 years. In total, 35 patients (11.6%) had a tumor located in the upper third, 63 (20.9%) in the middle third, and 204 (67.5%) in the lower third of the stomach. The median tumor size was 3.7 cm (0.5–13 cm). The patients were divided into two groups according to tumor size, a <4 cm group and a ⩾4 cm group. From stage T1 to T4, there were 131 (43.4%), 60 (19.9%), 32 (10.6%), and 79 (26.1%) patients, respectively. Highly differentiated tumors were observed in 177 (58.6%) patients, while poorly differentiated tumors were observed in the remaining 125 (41.4%) patients. Only 15 (5.0%) patients had lymphovascular invasion. In node-negative patients with AGC, 98 (57.3%) patients underwent postoperative chemotherapy, while 73 (42.7%) did not. Patients’ data are provided in Table 1.
Clinicopathologic characteristics of node-negative gastric cancer patients.
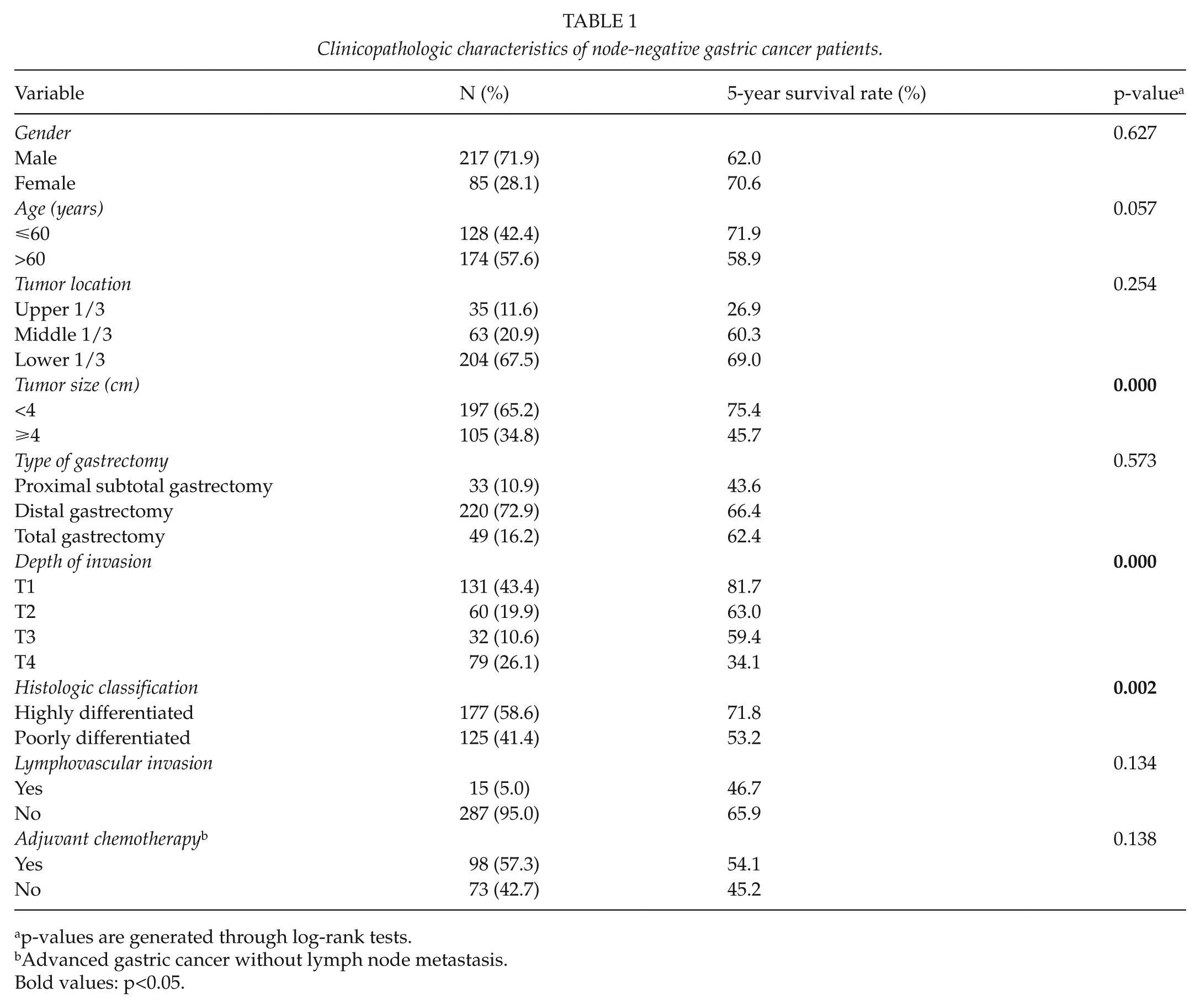
p-values are generated through log-rank tests.
Advanced gastric cancer without lymph node metastasis.
Bold values: p<0.05.
Survival
The median OS was 108 months. The overall 1-, 3-, and 5-year survival rates were 80.0%, 69.0%, and 63.0%, respectively. Univariate analysis showed that gender, age, operation procedure, tumor location, presence of lymphovascular invasion, and postoperative chemotherapy had no relation to 5-year OS rates (Table 1). However, the following variables were associated with improved OS: smaller tumor size (<4 cm, p < 0.001), highly differentiated pathological classification (p = 0.002), and low tumor T stage (p < 0.001). Multivariate analysis of these variables revealed that they were the only independent prognostic factors for better OS. Data are provided in Table 2. The survival curves of patients according to these three factors are presented in Figs 1 to 3.
Hazard ratio by age, tumor size, depth of invasion, histologic type, lymphovascular invasion, and adjuvant chemotherapy calculated by the Cox proportional hazard model.
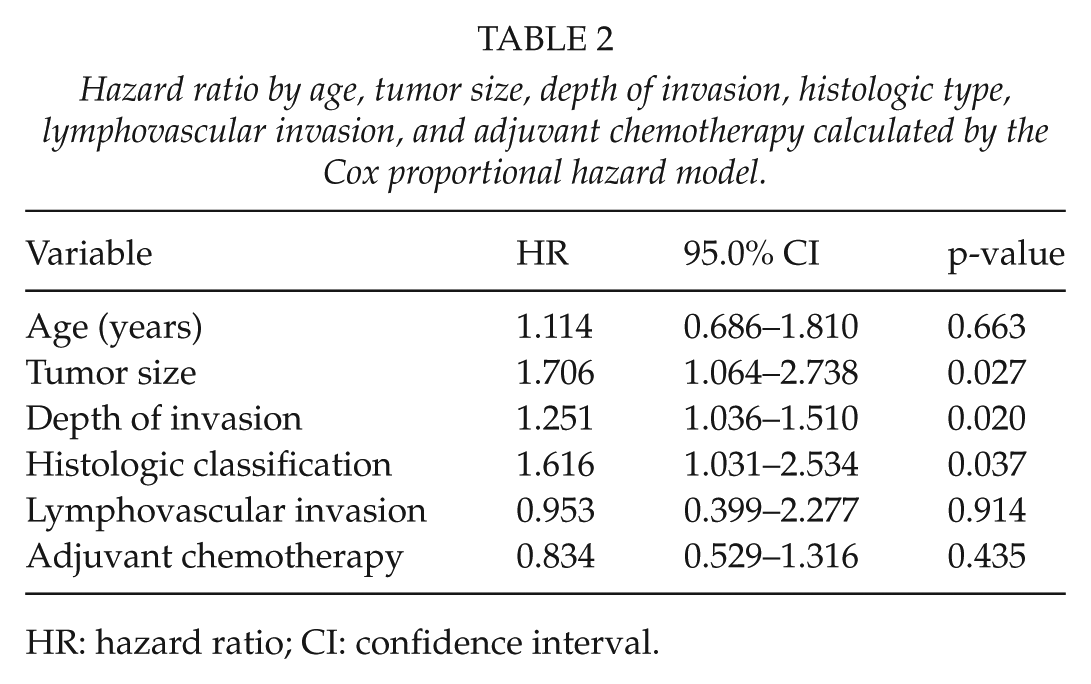
HR: hazard ratio; CI: confidence interval.
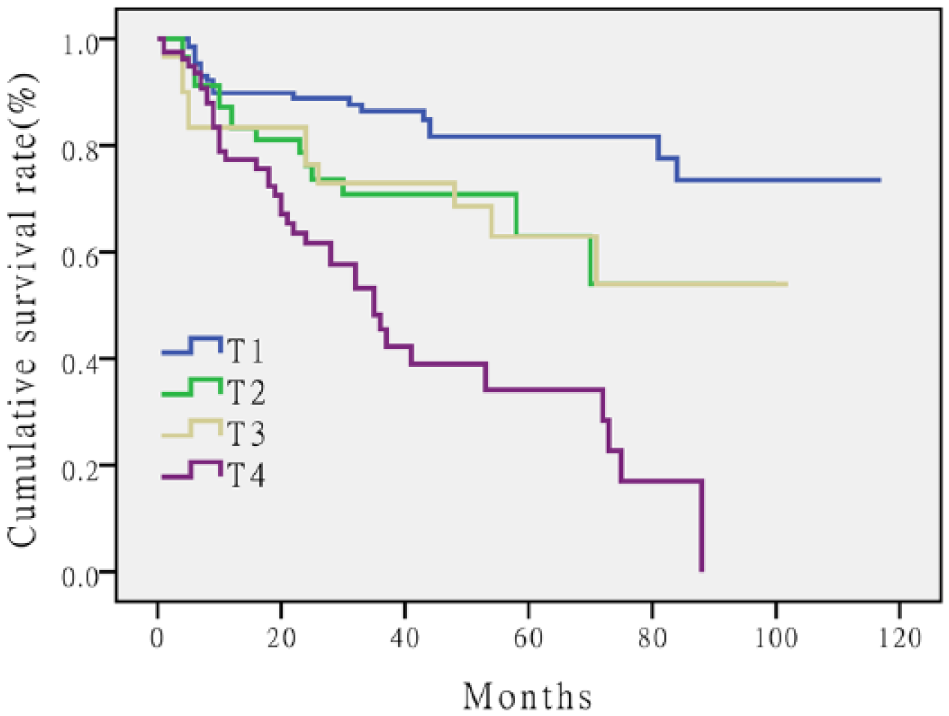
Survival curves were generated by the Kaplan–Meier method for gastric cancer patients who were divided into four groups, pT1 (n = 131), pT2 (n = 60), pT3 (n = 30), or pT4 (n = 79), according to the depth of tumor invasion. The log-rank test was used to analyze statistical significance.
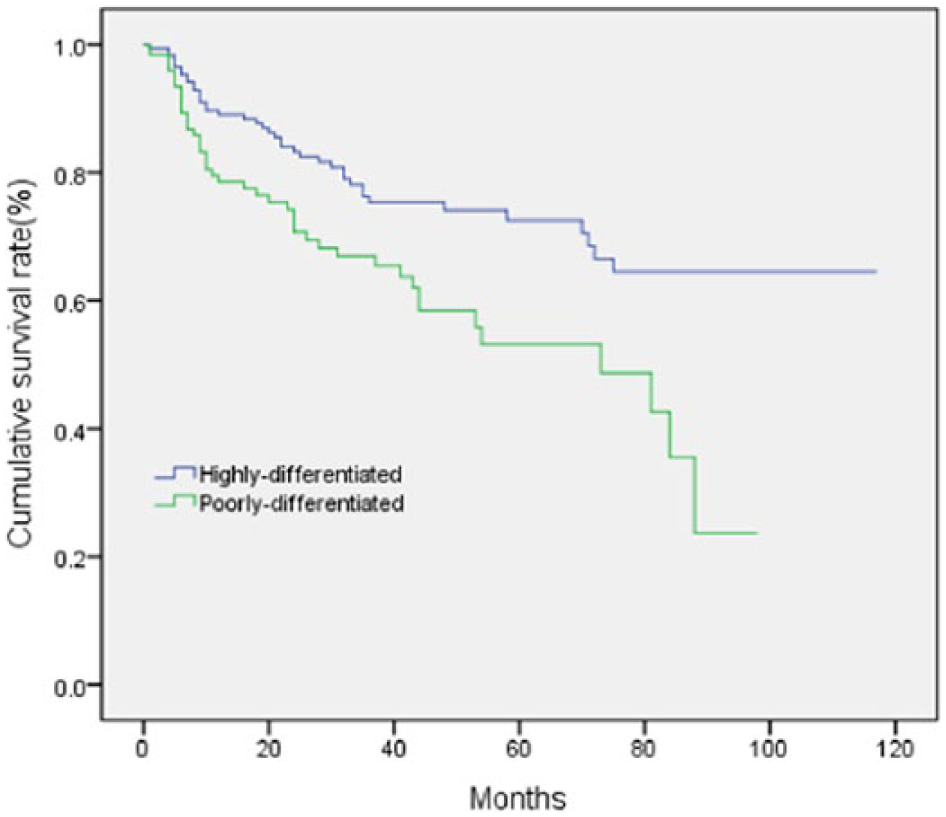
Survival curves were generated by the Kaplan–Meier method for gastric cancer patients who were divided into two groups, differentiation group (n = 175) and undifferentiation group (n = 125), according to histological types of tumor.
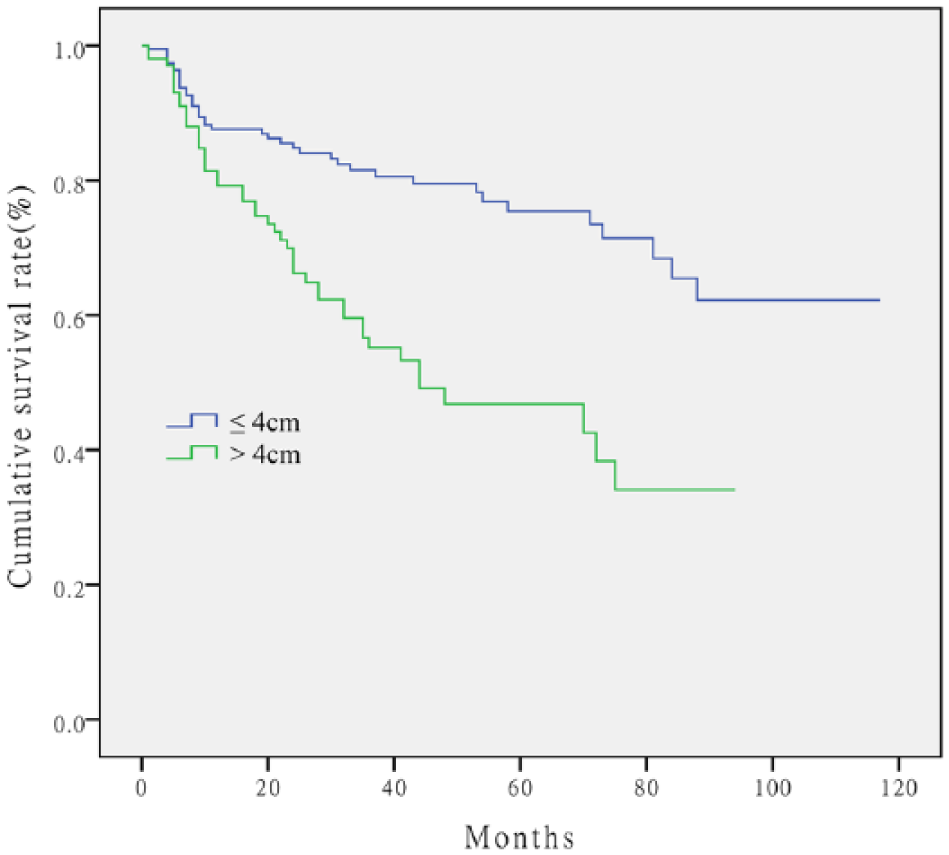
Survival curves were generated by the Kaplan–Meier method for gastric cancer patients who were divided into two groups, <4 cm group (n = 197) and ⩾4 cm group (n = 103), according to the tumor size.
Discussion
Identifying high-risk factors for the recurrence of gastric cancer helps to accurately distinguish between high-risk and low-risk patients. In this context, individual interventions could be performed to maximize individualized therapy effectiveness. Lymph node metastases are generally recognized as one of the most significant predictors of poor outcome, which is closely related to tumor recurrence. However, this fact does not suggest that node-negative patients are safe. It was reported that the 5-year OS varied from 72% to 97.1% in node-negative patients. Although this survival was significantly better than node-positive patients (8), some patients still experienced recurrence and metastasis.
A growing number of researchers have reported that the depth of tumor invasion and lymph nodes metastases were crucial prognostic factors in gastric cancer. In particular, the depth of tumor invasion has been commonly regarded as an independent predictor related to the survival rate, which is independent of lymph node metastasis (9, 10). The consensus was that the depth of tumor invasion was related to local recurrence. The probability of tumor cells shedding into the peritoneal cavity is increased when the depth of invasion is beyond the serosa and can simultaneously promote tumor cell metastasis. Kim et al. (11) argued that the prognosis of patients would be aggravated by lymph node micrometastasis, which was closely related to advanced T stage. We also found that advanced T stage was a negative predictor; patients with early disease had a longer OS than those patients with an advanced T stage.
Tumor size is another predictor of poor outcome in patients with lung cancer, thyroid cancer, and breast cancer, and tumor size is included in the tumor-node-metastasis (TNM) staging system (12). However, the relationship between tumor size and gastric cancer patient prognosis is still controversial. Using univariate survival analyses, Kim et al. (11) identified tumor size as a significant prognostic factor affecting 5-year OS. However, Baiocchi et al. (3) reported that some parameters associated with a better survival excluded tumor size. In our study, the 5-year OS of patients with tumor <4 cm (75.4%) was significantly better than those patients with tumor ⩾4 cm (45.7%). Multivariate analysis also confirmed that tumor size was also useful for predicting prognosis. Yokota et al. (13) found that tumor size was a predictor of survival according to univariate, but not multivariate, analysis. Still, other studies have reported that tumor size is an independent prognostic factor (14). The effects of invasion depth and lymph node metastasis on tumor size make it difficult to identify the most important prognostic factors. Thus, to eliminate lymph node effects, Xu et al. (15) analyzed the effects of tumor size on prognosis in patients with node-negative gastric cancer. The results showed that tumor size and depth of tumor invasion were independent prognostic factors for patient survival. With increasing tumor size, the probability of micrometastases migrating from the tumor through the lymph vessels is also increased (4). At the same time, tumors that penetrated over the submucosa would be more likely to invade the blood vessels, thus enhancing the likelihood of metastatic disease.
In addition, histological classification was also considered a prognostic factor, but with quite different conclusions. Seshadri et al. (16) reported that the tumor histology did not correlate significantly with survival. Nevertheless, Kim et al. (11) published a report in which histologic differentiation was found to be a statistically significant prognostic factor affecting 5-year OS. Our analysis showed that the 5-year OS of patients with poorly differentiated gastric cancer (53.2%) was significantly lower than that of those patients with highly differentiated gastric cancer (71.8%). We believe that more malignant poorly differentiated gastric cancer tumor cells were more aggressive and more likely to develop peritoneal metastasis.
At the same time, this study indicated that the patient’s gender, age, surgical method, tumor location, lymphovascular invasion, and postoperative chemotherapy were not independent predictors of prognosis. For patients with AGC, surgery combined with chemotherapy or radiotherapy was considered to be an effective treatment option (17, 18), especially in countries where D2 resection is the standard surgical procedure. Numerous meta-analyses have also suggested a survival benefit associated with adjuvant chemotherapy in patients with AGC (19, 20). However, for patients with node-negative gastric cancer, the role of chemotherapy was not recognized in some previous studies (21, 22). The results of our study showed that the 5-year OS of the chemotherapy group and the non-chemotherapy group were 54.1% and 45.2%, respectively, and Log-rank univariate survival analysis showed these differences were not statistically significant (p = 0.138).
This study has some limitations. First, our study was performed on 302 gastric cancer patients, who were negative for lymph node metastasis. Hence, the 5-year OS (63.0%) was slightly less than that reported in the literature. Second, the objective detection rates of early gastric cancer in China were low. Consequently, some patients with early gastric cancer who underwent endoscopic submucosal dissection (ESD) treatment instead of gastrectomy were excluded. As such, further studies with multiple centers that enroll more patients are needed to confirm our results.
Conclusion
This study demonstrated that large tumor size, poorly differentiated histologic classification, and deep tumor invasion depth were associated with an unfavorable 5-year OS rate for patients with node-negative gastric cancer. Such cases have an increased risk of tumor recurrence and should be considered for individual follow-up and radical treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
