Abstract
Background and Aims:
Osteoarthritis (OA) is the world’s most common joint disease and there is currently no cure. Glenohumeral osteoarthritis (GHOA) accounts for an estimated 5%–17% of patients with shoulder complaints. The etiology of GHOA is multifactorial, and we review the various non-specific and specific risk factors and further sub-classify them into local and systemic factors.
Materials and Methods:
Data for this review article were identified by searches of MEDLINE, PubMed, and references from relevant articles using search terms such as “glenohumeral,” “osteoarthritis,” “epidemiology,” “etiology,” “imaging,” and “pathophysiology.” Only articles published in English, German, and Finnish between 1957 and 2017 were included.
Results:
The prevalence of radiological shoulder OA has been estimated to be as high as 16%–20% in the middle-aged and elderly population, but the concordance between structural findings and symptoms seems to be weak, as many of these individuals are asymptomatic. The vast majority of GHOA is related to non-specific factors, namely advancing age, while specific risk factors are commonly found in young patients. Diagnosis of GHOA is made when typical clinical features and defined radiological findings overlap in an individual.
Conclusion:
Conclusion: Ultimately the determinants of shoulder pain in GHOA remain incompletely understood. Improved understanding of the etiology and diagnosis of GHOA will enable clinicians to better determine which patients will benefit from different treatment modalities, as well as provide new avenues to potential treatments.
Keywords
Introduction
Degenerative changes of the glenohumeral (GH) joint are found in up to 17% of patients with shoulder pain, a patient group that has tripled in the last 40 years1, 2. Although symptomatic glenohumeral osteoarthritis (GHOA) is not as common as osteoarthritis (OA) of the weight bearing hip and knee joints, it can be just as debilitating due to the functional importance of the upper limbs. Pain and functional impairment can affect physical as well as psychological well-being, causing limitations in occupational and leisure activities in younger individuals and threatening physical independence in the elderly 3 .
The purpose of this article is to give a narrative review of the literature on the factors influencing the development of GHOA and outline its basic diagnostic process.
Defining GHOA
OA is a degenerative joint disease involving the articular cartilage, subchondral and periarticular bone, and periarticular soft tissues including ligaments, muscles, and synovium. Symptoms of OA include joint pain, stiffness, and limitation of movement, while radiological changes include joint space narrowing, osteophyte formation, periarticular cysts, and subchondral sclerosis. GHOA is defined by these features of joint damage affecting the glenohumeral joint.
Clinical Definition
Clinically OA can be defined using clinical history and physical examination findings, such as those developed by the American College of Rheumatology (ACR) for knee and hip OA4, 5. However, no such generally accepted clinical criteria for GHOA exist. Defining GHOA solely on clinical grounds is problematic because of the anatomical complexity of the shoulder girdle and clinical similarities with differential diagnoses (capsulitis and to some extent rotator cuff syndrome).
Radiological Definition
Radiological definitions of OA rely on identifying the radiological features of joint damage listed above. However, up to six different radiographic classification systems have been applied to GHOA 6 . The Samilson-Prieto classification, despite having been originally developed for dislocation arthropathy, has become the most commonly used, while modifications have been published by both Allain and Gerber. The Kellgren-Lawrence classification, advocated by the World Health Organization (WHO) for epidemiological OA studies, has also been applied to the shoulder joint. More recently, the Weinstein and Guyette classifications have also been introduced. Radiological findings consistent with GH joint damage are, however, commonly found in asymptomatic individuals, making purely radiological definitions inadequate.
Pathophysiology of GHOA
GHOA is diagnosable when anatomical changes of joint damage are identifiable as radiological features and when clinical symptoms led by joint pain occur in a patient. The pathogenesis of GHOA is a complex and still incompletely understood process with various factors affecting joint damage and joint pain. We focus on the prior as these are more concrete and the target of surgical treatment, while not underestimating the importance of the latter.
Joint Damage
A commonly held view is that joint damage is caused by either abnormal mechanical stress on normal cartilage or normal stress on structurally weakened cartilage. These opposites represent the ends of a spectrum, with patient factors influencing the proportional effect of mechanical stress and cartilage health on the patient’s risk of developing OA. Joint damage, irrespective of cause, leads to a common biochemical cascade resulting in ongoing joint wear and synovitis (see Fig. 1).
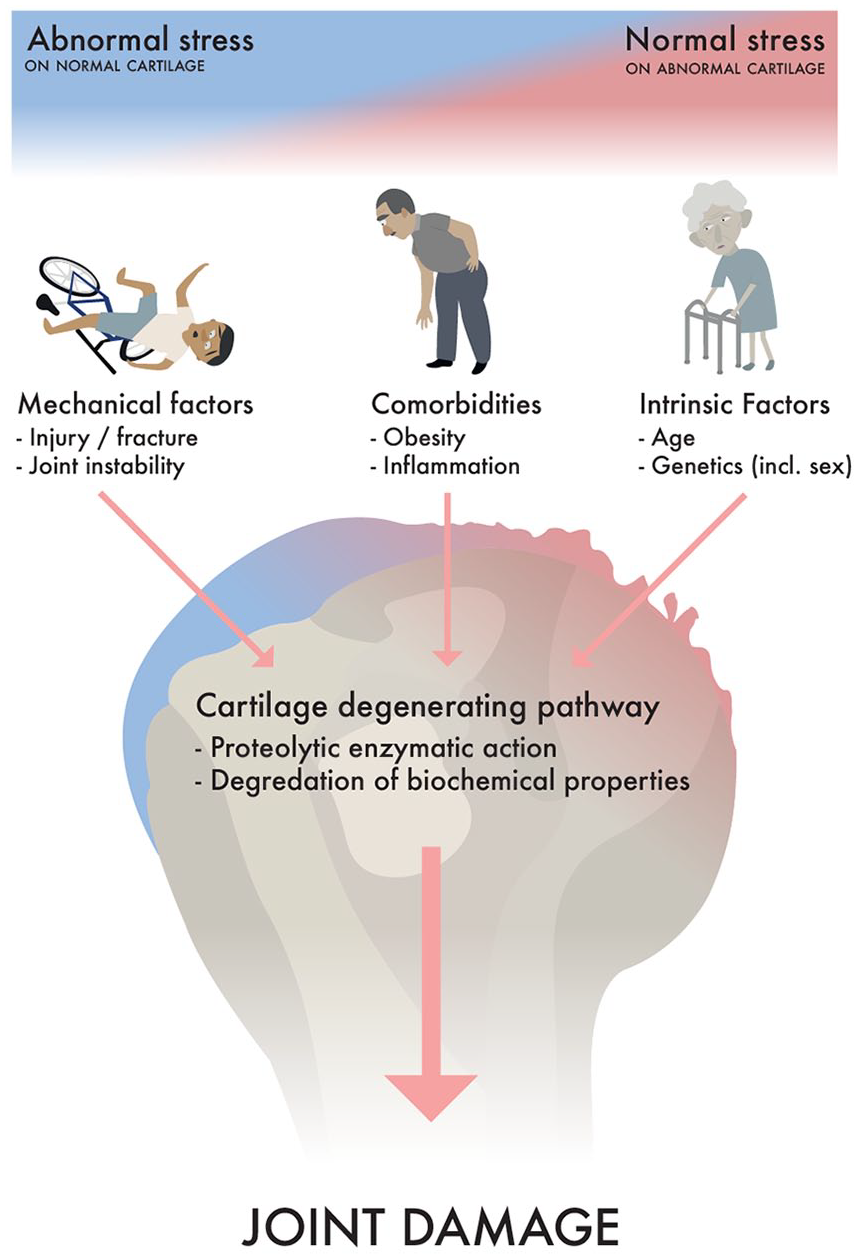
Pathogenesis of OA: A continuum of proportional effect from specific factors with normal cartilage under abnormal mechanical loading and abnormal cartilage under normal loading at opposite ends of the spectrum. These effects lead to a common biomechanical cascade leading to joint damage.
In the glenohumeral joint, focal or global cartilage loss and subsequent subchondral bony sclerosis are most pronounced in the upper two-thirds of the humeral head. This area is in contact with the glenoid between 60 and 100 degrees of abduction 7 .
Joint Pain
Articular cartilage is relatively insensitive, but periarticular tissues like synovium and subchondral bone are richly innervated and are the likely sources of nociceptive stimuli 8 . Increased intra-osseous pressure in subchondral bone from altered biomechanics or cartilage damage could also explain symptoms such as night and rest pain. In addition to local anatomical factors in and around the joint, an individual’s pain sensation is modified by local and central pain pathways as well as contextual psychosocial and financial factors 8 (see Fig. 2). Contextual factors such as depression, anxiety, coping mechanisms, and the patient’s level of education may explain some of the commonly seen discordance between subjective symptoms and objective radiological findings of joint damage, as is sometimes seen in workers’ compensation cases, where compensation claims are commonly related to poorer outcomes9 –11.
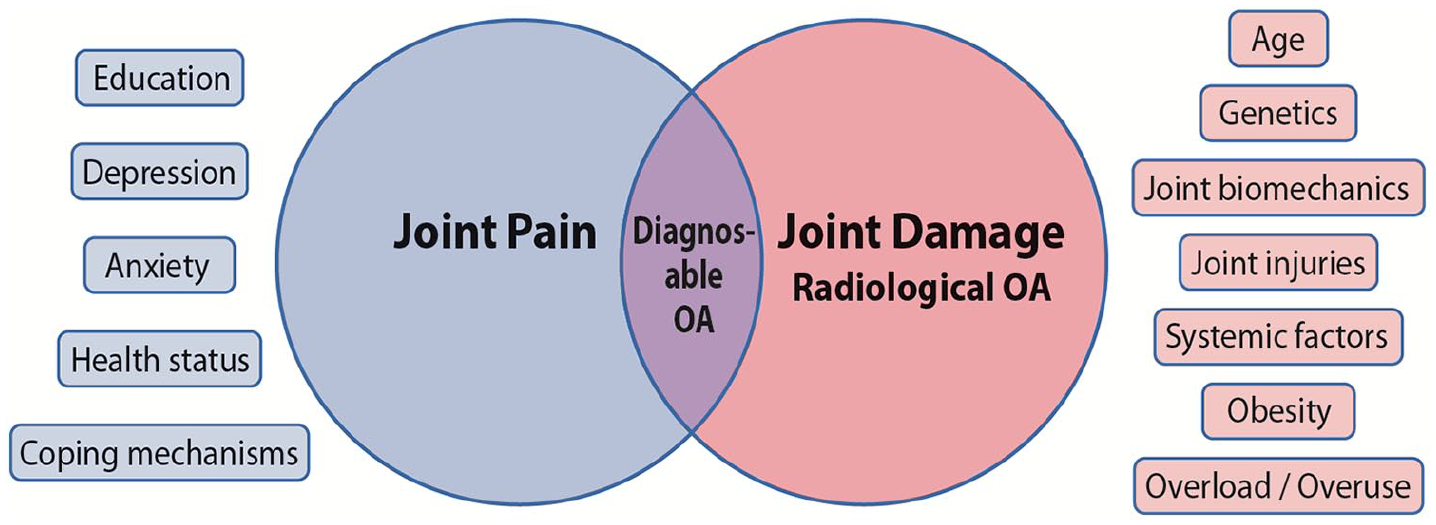
Illustration of the factors affecting the subjective experience of joint pain and the radiological changes seen in osteoarthritis. Only the portion of patients for whom joint pain and radiological changes overlap could be considered as having clinically relevant osteoarthritis.
Pathoanatomy
The most frequent bony change in GHOA is the formation of osteophytes, due to chondrocyte stimulation and enchondral ossification in the transition area of the hyaline cartilage and synovial membrane. Osteophytes are predominant in the anterior-inferior aspects of the humeral head and inferior aspects of the glenoid and are thought to result from mechanical capsular traction 12 . Humeral osteophytes are often accompanied by enthesopathic changes at ligament attachment sites, typically involving the anatomical neck and tuberosities as well as the bicipital groove, which can be narrowed by bony proliferation 13 . Although periarticular osteophytes have been regarded as pain generators, it is possible that they instead attempt to provide pain relief. Osteophytes and joint capsule thickening may serve as natural splints immobilizing a painful joint and potentially even leading to spontaneous arthrodesis 14 (see Picture 1).
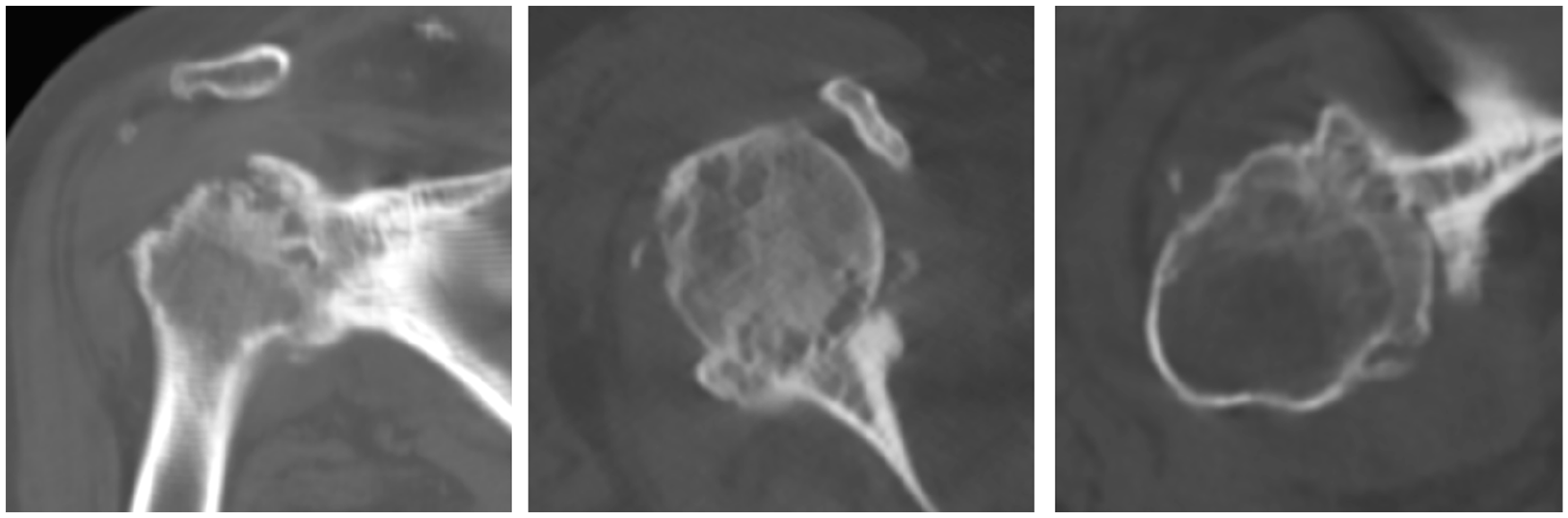
Spontaneous arthrodesis: Computer tomography images of end-stage osteoarthritis showing spontaneous arthrodesis of the glenohumeral joint in a 61-year-old woman.
In GHOA, the humeral head may be subluxed posteriorly This can be either as a result of posterior glenoid wear and labral degeneration or the cause of those changes12, 13. Glenoid cartilage wear patterns have been classified by Walch, with type A showing central wear, while type B results from posterior glenoid wear. Type C is defined by glenoid retroversion of more than 25° and subsequent posterior humeral subluxation 15 .
Glenoid retroversion has traditionally been attributed to ossification abnormalities (such as glenoid dysplasia, Walch Type C glenoid), but this seems to have overlooked the wide range of normal variation in glenoid anatomy due to generic factors. Attention has more recently been shifted to the causes of secondary posterior glenoid wear (Walch Type B glenoid) such as soft tissue imbalance, due to internal rotation contracture or muscular strain and functional considerations 16 . Typical shoulder positions during manual work and daily activities—with the arm in forward flexion and adduction—significantly increase posteriorly directed forces across the glenohumeral joint leading to strain on the posterior glenoid cartilage 17 . Although the Walch classification tries to divide the causes of glenoid retroversion into primary (osseous, Type C) and secondary (wear-related, Type B) forms, a clear division may not always be possible 18 . Early neuromuscular imbalance (e.g. obstetric brachial plexus palsy), for example, may lead to both primary osseous maldevelopment and capsular contracture and muscular imbalance causing secondary posterior glenoid wear. Furthermore, making distinctions between causes and effects on radiological imaging in clinical practice can be challenging.
Epidemiology of GHOA
The glenohumeral joint is the third most common large joint to be affected by OA. As there is no one accepted definition of GHOA, authors of epidemiological studies have used differing definitions, making comparison of the published data problematic. However, epidemiological studies of OA in general suggest prevalence rates are rising 19 .
A recent Korean study using the Kellgren-Lawrence classification identified GHOA (K-L ⩾ 2) in 5.2% of the population age 65 and above 20 . Two imaging studies using the Samilson-Prieto classification showed GHOA prevalence rates in the middle-aged and elderly (age above 40 and 65 years respectively) to be as high as 17%–19%, while bilateral disease was identified in 3.1%–7.7% of the population21, 22. Prevalence rates of GHOA due to a specific identifiable cause (secondary OA) was reported at only 1.3%–1.7%, making age-related non-specific GHOA 10 times more common21, 22. However, GHOA, due to a specific identifiable cause, has been shown to be significantly more common in patients age less than 50 23 .
Etiology of Glenohumeral Joint Damage
Our understanding of the etiology of GHOA is derived from advancements made in understanding OA in general. Theories have evolved from age-related cartilage wear and tear to a disorder that affects the entire joint and its surrounding tissues. Some consider OA a common endpoint of a heterogeneous group of disorders that lead to degenerative joint damage 8 .
Traditionally OA has been classified into primary (no known cause) and secondary (resulting from an identifiable cause) forms, but this dualistic division has been criticized. The pathogenesis of joint damage, seen as part of a common pathological process, is influenced by multiple factors 24 . These can be divided into non-specific and specific factors as well as into systemic and local factors. Joint damage develops from the interplay between these factors, where local or systemic factors, or non-specific or specific factors, may dominate8, 25 (see Fig. 3). Disease progression is, however, typically affected by a combination of genetic, behavioral, and environmental factors.
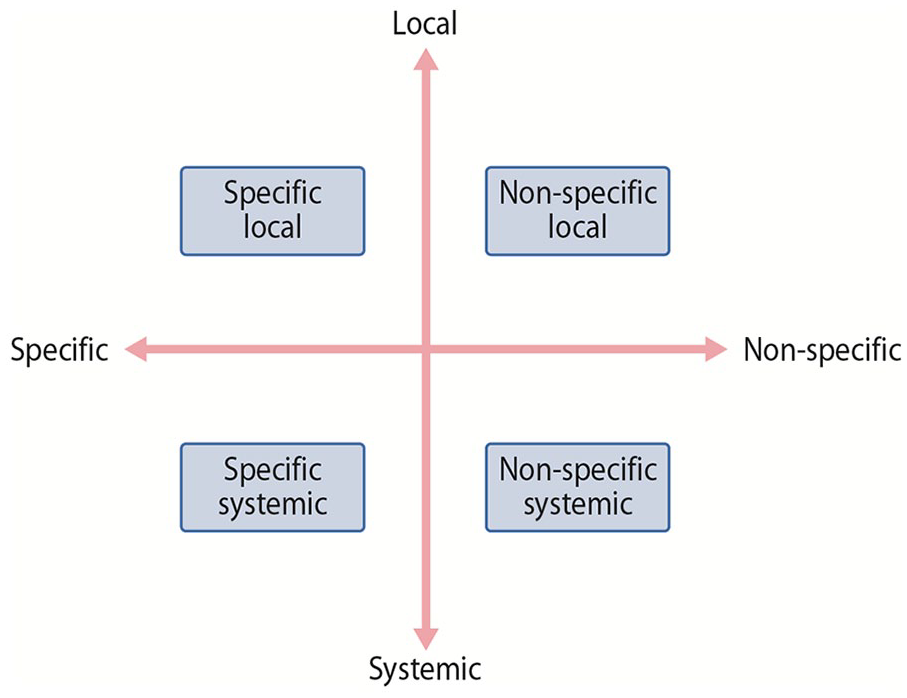
The pathogenesis of joint damage is influenced by non-specific and specific factors as well as local and systemic factors. A division of GHOA according to these factors might be more appropriate than the traditional division into primary and secondary forms.
Non-Specific Systemic Risk Factors for Glenohumeral Joint Damage
Advancing age
Aging is the principal etiological risk factor in the development of GHOA. Chondrocyte density in the hyaline cartilage and responsiveness to growth factors diminish with age, leaving the joint cartilage more vulnerable to damage (26). GHOA prevalence rates increase with age from 1.8% in people in their 40s to 27.5% in those in their 80s22, 27.
Genetics
Hereditary factors have long been known to play a key role in the development of degenerative joint damage. Inheritance patterns are complex and polygenic, with an estimated 35% to 65% of OA being attributed to genetic influence 28 . Recent studies have identified genetic risk loci and genetic variations that are directly associated with OA and others have been found to be associated with contributing factors such as excessive inflammation, congenital malalignment of joints, and obesity29, 30.
Obesity
Obesity is known to be a risk factor for hip and knee OA, which is thought to be due to excessive joint loading 31 . This mechanical theory, however, does not entirely explain the association of obesity with OA in non-weight bearing joints of the hand and wrist 32 . Although these findings are not specific to the shoulder, they suggest a more complex etiology, with research recently focusing on the role of adipose tissue and its endocrine functions. Cytokines released by adipose tissue can promote systemic low-grade inflammation, which in addition to dyslipidemia, may play an important role in obesity-related OA32, 33.
Non-Specific Local Risk Factors for Glenohumeral Joint Damage
Exercise and occupation
Excessive mechanical loading poses a risk for increased joint surface wear, but an appropriate amount of loading can maintain or even improve the biomechanical properties of healthy hyaline cartilage and subchondral bone 34 . Insufficient stimulus through a sedentary lifestyle propagates the opposite, predisposing joints to damage and wear. Muscle weakness and neuromuscular dysfunction may compromise innate protective mechanisms, making joints more susceptible to abnormal motion 35 . Conversely, excessive muscle strength or joint laxity may also promote the progression of OA36, 37. Heavy construction work and overhead sports are identified risk factors for GHOA with elite tennis players showing GHOA changes in their dominant shoulders38, 39. Other athletes at risk of developing GHOA include weight lifters and throwing athletes such as baseball players 40 . Interestingly, although high rates of shoulder pain are reported in wheelchair users, GHOA seems to play an insignificant role in this group of patients41, 42.
Specific Local Risk Factors for Glenohumeral Joint Damage
Glenohumeral instability and instability repair
Prevalence of radiological GHOA changes—following both dislocations and instability surgery—have been reported as high as 56%–68%, but symptoms are typically infrequent or mild43 –45 (see Picture 2A). Direct joint damage as well as altered biomechanics can be responsible for these radiological changes, but the degree to which these changes are caused by dislocation versus subsequent surgery is unclear.
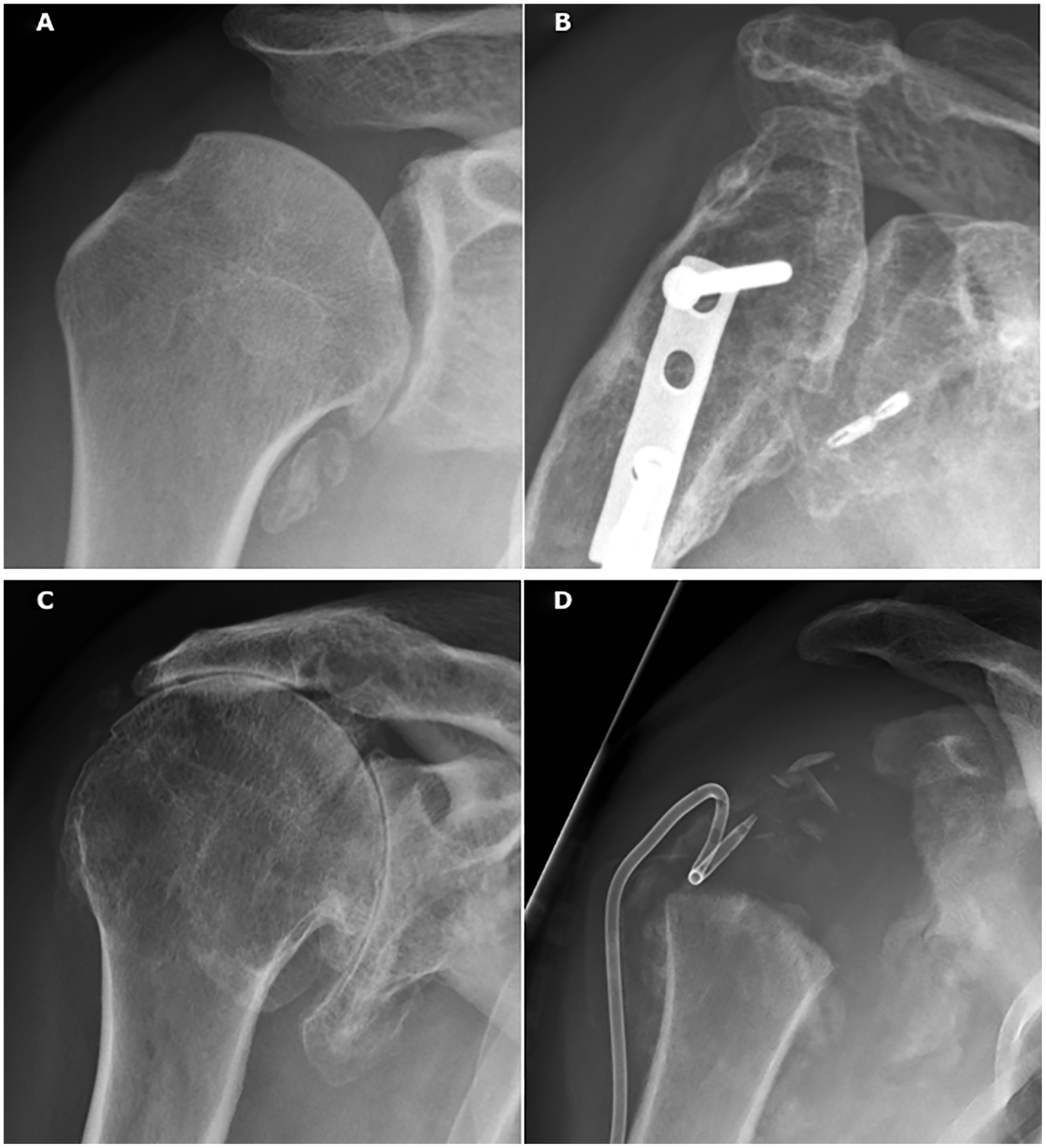
Specific causes of GH joint damage. (a) 37-year-old male 12 years following labral repair for traumatic anterior shoulder dislocation. Clinically he was pain free with full range of motion (ROM) despite radiographic changes consistent with Samilson-Prieto grade 2 OA. (b) 39-year-old female who sustained a proximal humerus fracture from a fall and underwent anatomic reduction and fixation with a non-locking plate. Seventeen years later, radiographs show severe posttraumatic GHOA and avascular necrosis of the humeral head, while clinically she has pain at rest with very limited ROM. (c) 69-year-old male showing typical changes of cuff arthropathy: Cranialization of the humeral head, diminished subacromial space, and acetabularization of the coracoacromial arch. He presented with good function with active flexion and abduction of 170°, but progressive pain. (d) 41-year-old male who developed swelling of his right shoulder and presented 5 days later with pain and fever. Joint aspirate and cultures remained negative and at one month radiographs showed total loss of the humeral head. Past medical history revealed surgery for L1 burst fracture 17 years previously and a new MRI of the spine revealed a massive syringocele from conus to C0, confirming suspicion of Charcot arthropathy. The patient was referred to neurosurgery for further treatment. Swelling and pain subsided, but function remained poor.
Although it is uncertain if laxity or joint instability per se is a risk factor for GHOA development, the association with shoulder dislocations (to which instability is a predisposing factor) has been established in numerous studies43, 44, 46. Patients with a single shoulder dislocation have a 10- to 20-fold increased risk of developing radiological GHOA compared to persons without acute shoulder injuries 47 . Recurrent dislocations, older age at primary dislocation, and greater trauma energy further increase the risk of GHOA development43, 44, 46.
Fractures
Fractures can cause macroscopic damage to the articular cartilage and drive the degenerative cascade in an exaggerated manner, leading to rapid joint destruction, commonly known as posttraumatic OA. In addition to direct damage to the articular cartilage, proximal humeral as well as glenoid fractures can lead to altered biomechanics and subsequent GHOA through bony malalignment, non-union, or joint incongruity. Iatrogenic joint damage can also occur from hardware penetration following surgical fracture fixation (see Picture 2B).
Rotator cuff arthropathy
GHOA, due to traumatic or degenerative rotator cuff tears, displays three characteristic changes. These are rotator cuff insufficiency, cranial migration of the humeral head, and subsequent radiographic degenerative changes. Radiographs typically show bony erosion of the superior glenoid, resulting in acetabularization of the coracoacromial arch, and rounding off of the humeral greater tuberosity 48 (see Picture 2 C). Cuff arthropathy is seen more commonly in women and mostly affects the shoulder of the dominant arm.
Glenohumeral chondrolysis
Glenohumeral chondrolysis is a rare iatrogenic process resulting from intra-articular local anesthetic agents, bioabsorbable anchors, and thermal damage during arthroscopy49, 50. It particularly affects younger patients and results in symmetric hyaline cartilage loss and periarticular osteopenia.
Scapular morphology
Several factors of scapular morphology have been associated with GHOA development. These include the lateral extension of the acromion as well as glenoid inclination. These two measurements have recently been combined to give the
Milwaukee shoulder
Milwaukee shoulder is a rare joint-destroying disease seen typically in older women that is associated with intra- and periarticular hydroxyapatite crystal deposition. Identification of these crystals by Alizarin Red staining is a hallmark of this rapidly progressive disease often showing marked erosion of the humeral head and surrounding soft tissues 59 .
Specific Systemic Risk Factors for Glenohumeral Joint Damage
Inflammatory arthritis
Autoimmune mediated inflammatory causes include rheumatoid, psoriatic, and juvenile idiopathic arthritis, as well as spondyloarthropathies and systemic lupus erythematosus (SLE). Of these, rheumatoid arthritis (RA) is the most common, with more than half of patients showing involvement of the GH joint 60 . The worst affected patients typically have bilateral disease and central glenoid wear patterns, but the associated pain may be more related to the inflammatory synovitis than the joint destruction itself. Crystalline arthropathies, including gout and pseudogout, caused by sodium urate and calcium pyrophosphate crystals in the synovial fluid, respectively, also lead to inflammatory joint destruction. They characteristically cause glenohumeral cartilage wear without prominent secondary radiological OA changes.
Avascular osteonecrosis
Avascular osteonecrosis (AVN) of the humeral head leads to bony collapse and loss of joint congruity and has been estimated to account for 5% of all GHOA 61 . It is the second most common site of AVN following the hip 62 . Common etiological factors include trauma, corticosteroid use, and alcoholism, while rare causes include Gaucher disease, sickle cell anemia, Caisson disease, and certain pharmaceuticals 62 .
Endocrine diseases and neuropathic arthropathy
Neuropathic arthropathy, also known as charcot arthropathy, is another rare degenerative disorder characterized by rapid destruction of the joint with extensive involvement of the bone and soft tissue. Cervical syringomyelia accounts for 75% of neuropathic shoulder arthropathy, while other causes of insensitivity, such as diabetes mellitus, chronic alcoholism, or end-stage renal disease, are less common 63 . The underlying pathophysiology is not well understood, but neurovascular and neurotraumatic theories have been developed, with the loss of nociception and proprioception playing important roles (see Picture 2D).
Diagnosis of GHOA
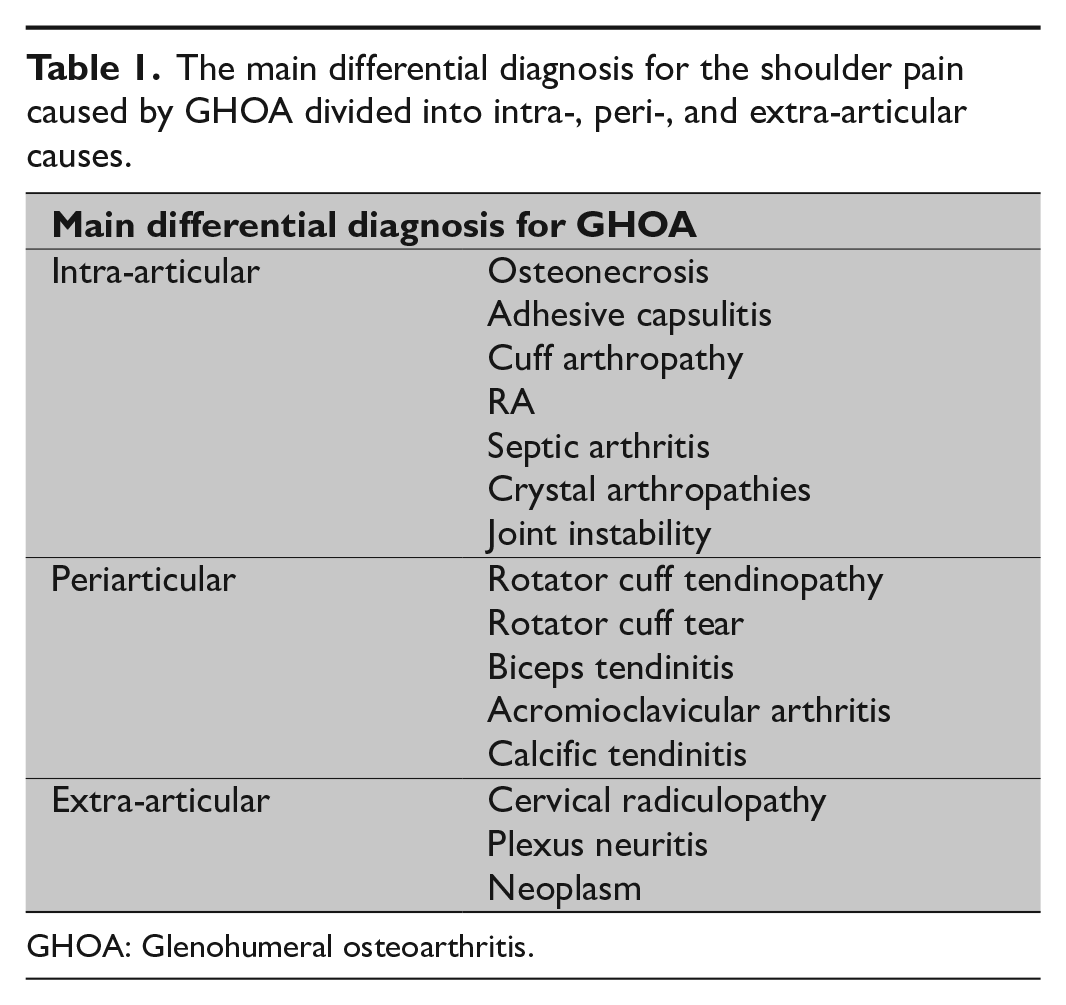
Diagnosing GHOA relies on patient history supported by clinical findings and corroborated by radiological examinations. The shoulder is affected by a complex interaction of potential intra- and extra-articular pain generators, which may complicate the diagnosis of GHOA (see Table 1). GHOA is not the most common cause of shoulder pain and the diagnosis is often one of exclusion, after other more frequent conditions of the shoulder are ruled out 2 . Loss of passive ROM is the most reliable clinical indicator. However, this must be correlated to plain radiographs to differentiate GHOA from its primary differential diagnosis of adhesive capsulitis and to rule out glenohumeral joint dislocation.
The main differential diagnosis for the shoulder pain caused by GHOA divided into intra-, peri-, and extra-articular causes.
GHOA: Glenohumeral osteoarthritis.
Traditionally, radiographic changes indicative of joint damage were held responsible for patient symptoms, but this link has been shown to be weaker than expected with radiological changes being very common also in the asymptomatic population22, 43, 64. With anatomic changes failing to translate into symptomatology and vice versa, appreciation for a more complex interplay between anatomic and psychological factors has developed 8 (see Fig. 2). Thus, defining GHOA solely on clinical or radiological grounds seems inadequate and a more holistic approach is needed.
Medical History and Clinical Examination
The diagnostic process for shoulder pain starts with a thorough
Clinical examination findings in the early stages of GHOA can be elusive but become more pronounced as disease progresses. Clinical findings include joint line tenderness on palpation, crepitus, and pain during joint movement as well as reduced range of passive motion, particularly external rotation. Inspection might reveal muscle atrophy or fluid collection (“fluid sign or geyser sign,” escape of glenohumeral joint synovial fluid into the subacromial-subdeltoid bursa) around the shoulder girdle, both indicative of rotator cuff tear arthropathy.
Imaging studies provide important additional information in the diagnostic process. One must be aware, however, that radiological findings may be incidental64, 65. As the severity of joint degeneration seen on imaging studies has been shown to poorly correlate with pain, one should avoid attaching a disease label to a patient on imaging findings alone 66 (see Fig. 2).
Plain Radiography
The diagnostic workup of GHOA is completed with plain radiographs, which are sufficient in most cases and should include a true anteroposterior, lateral, and axillary view of the shoulder 67 (see Picture 3). A typical finding in early stage disease is an inferomedial humeral osteophyte spur (“goats-beard”) at the insertion of the joint capsule. Other changes include joint space narrowing, subchondral sclerosis, and/or cyst formation, as well as osteophytes that are best visualized in external rotation films. In advanced stages, humeral head deformities with loss of concentricity between the humeral head and the glenoid are seen along with posterior subluxation of the humeral head in axial projections 68 . Cranial migration of the humeral head is indicative of chronic rotator cuff tears and consequent cuff arthropathy.
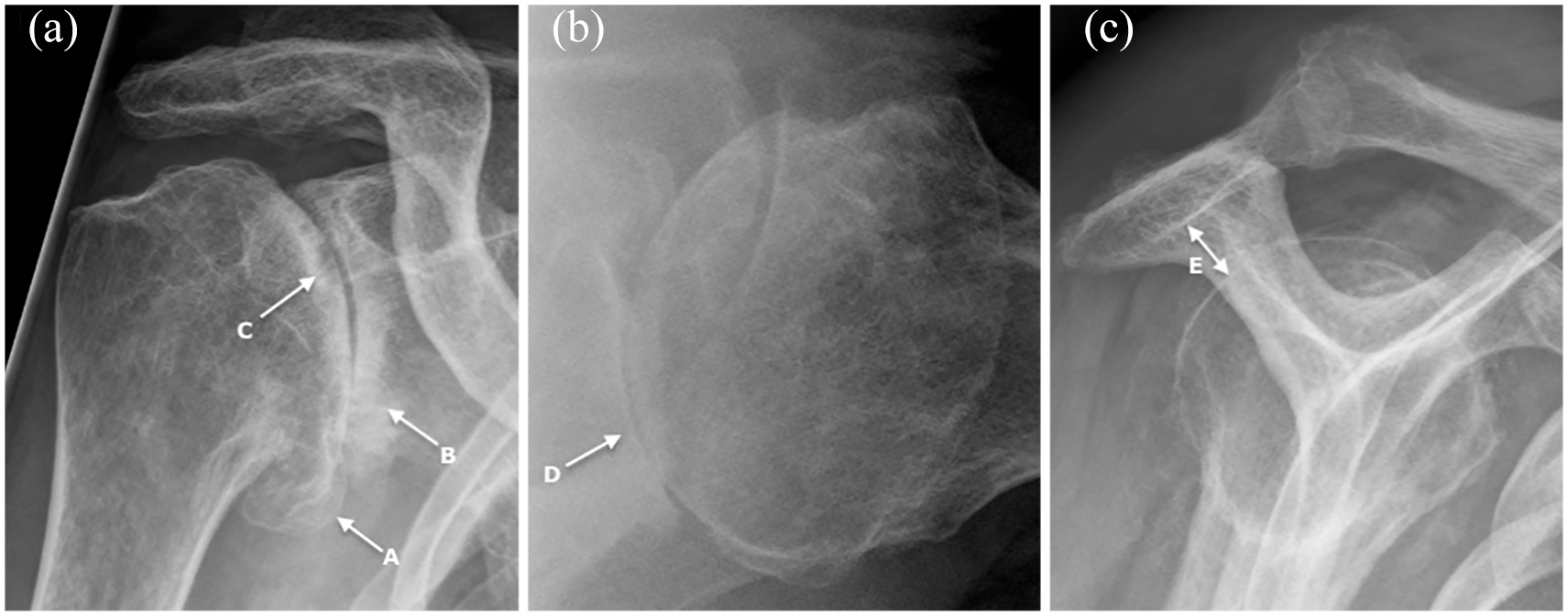
Plain radiographs showing typical osteoarthritic changes of the glenohumeral joint. (a) AP view showing inferomedial humeral osteophyte spur (“goats-beard”) at the insertion of the joint capsule (a), joint space narrowing, subchondral sclerosis (b), and cyst formation (c). (b) Axillary view showing joint space narrowing, subchondral sclerosis, and typical posterior glenoid wear (d). The axillary view allows assessment of glenoid bone stock and retroversion, although computer tomography is more accurate. (c) Y-view showing preserved subacromial space (e) (differentiating it from cuff arthropathy).
COMPUTER TOMOGRAPHY
Despite the associated radiation and inherent limitation in soft tissue imaging, computer tomography (CT) has gained an increasingly significant role though the use of intra-articular contrast agent. These CT arthrography studies, which are faster and more widely available, have shown comparable results to magnetic resonance imaging (MRI) in identifying rotator cuff, labral, and glenoid cartilage pathologies69, 70. The utilization of 3D image reconstruction makes them superior to MRI in evaluating glenoid morphology, bone stock, and joint centricity, which makes CT the imaging modality of choice whenever shoulder arthroplasty is considered71, 72. The condition of rotator cuff muscles can be evaluated using the Goutallier classification, which was originally used for quantifying fatty infiltration on CT scans.
MRI
MRI, on the other hand, is superior to CT in imaging the soft tissues surrounding the glenohumeral joint and particularly in picking up early changes, such as joint effusion, synovial proliferation, or subtle chondral alterations. However, the latter are typically underestimated when compared to arthroscopic findings 73 . MRI is not routinely used to diagnose GHOA per se, but it can play a role in a select group of patients with undetermined diagnosis or suspicion of periarticular tumors. MRI does, however, have a significant role in the diagnostic work-up of shoulder pain, and thus can exclude other causes of pain such as rotator cuff pathology. MRI can, in these instances, reveal findings of GHOA, including subtle intrinsic signal abnormalities of the labrum or overt generalized signs of joint degeneration.
Ultrasound
Ultrasound has increasingly been used in the diagnosis and treatment of inflammatory joint diseases. Due to improvements in image quality, it has lately also been studied in the evaluation of degenerative joint disease. It is accurate in assessing joint effusion and synovial thickening, but reliability in gauging cartilage or bony changes is questionable 74 .
Conclusion
With aging populations, GHOA represents an increasing burden to individuals as well as healthcare systems. The inconsistencies in defining GHOA make its epidemiological study challenging. While specific risk factors are found in young patients, the vast majority of GHOA is related to non-specific factors, namely advancing age. We have divided the principal risk factors for GHOA into specific and non-specific as well as local and systemic to reflect the interplay between the various factors responsible. Symptoms are not directly related to radiographic changes, but result from a complex interaction between structural changes, biological processes, and psychosocial factors. Ultimately the determinants of shoulder pain in GHOA remain incompletely understood. Improved understanding of the etiology and diagnosis of GHOA will enable clinicians to better determine which patients will benefit the most from different treatment modalities, as well as provide new avenues to potential treatments.
Footnotes
Author contributions
Both T.I. and T.S. contributed equally to the writing and preparation of this article. A.L. and M.P. gave support, advice, and editorial review for the article.
Declaration of conflicting iInterests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Thomas Ibounig, Tomi Simons, Mika Paavola, and Antti Launonen certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Search strategy and selection criteria
Data for this review article were identified by searches of MEDLINE, PubMed, and references from relevant articles using search terms such as “glenohumeral,” “osteoarthritis,” “epidemiology,” “etiology,” “imaging,” and “pathophysiology.” Abstracts and reports from meetings were included only when they related directly to previously published work. Only articles published in English, German, and Finnish between 1957 and 2017 were included.
