Abstract
Introduction:
There has been a rapid development in minimally invasive pancreas surgery in recent years. The most recent innovation is robotic pancreatoduodenectomy. Several studies have suggested benefits as compared to the open or laparoscopic approach. This review provides an overview of studies concerning patient selection, volume criteria, and training programs for robotic pancreatoduodenectomy and identified knowledge gaps regarding barriers for safe implementation of robotic pancreatoduodenectomy.
Materials and methods:
A Pubmed search was conducted concerning patient selection, volume criteria, and training programs in robotic pancreatoduodenectomy.
Results:
A total of 20 studies were included. No contraindications were found in patient selection for robotic pancreatoduodenectomy. The consensus and the Miami guidelines advice is a minimum annual volume of 20 robotic pancreatoduodenectomy procedures per center, per year. One training program was identified which describes superior outcomes after the training program and shortening of the learning curve in robotic pancreatoduodenectomy.
Conclusion:
Robotic pancreatoduodenectomy is safe and feasable for all indications when performed by specifically trained surgeons working in centers who can maintain a minimum volume of 20 robotic pancreatoduodenectomy procedures per year. Large proficiency-based training program for robotic pancreatoduodenectomy seem essential to facilitate a safe implementation and future research on robotic pancreatoduodenectomy.
Keywords
Introduction
Robotic pancreatoduodenectomy (RPD) aims to minimize the impact of this extensive surgical procedure and hence enhance patient recovery. Several studies have suggested superior outcomes as compared to open and laparoscopic pancreatoduodenectomy. A Pan-European propensity score matched study showed superior outcomes to open pancreatoduodenectomy (OPD) in terms of time to recovery and to laparoscopic pancreatoduodenectomy in terms of conversion levels (1). A single-center propensity-matched analysis reported a lower rate of clinically relevant pancreatic fistulas after RPD as compared to OPD (2).
However, several questions arise regarding the potential for safe implementation of a highly complex procedure as RPD. First, which patients should be selected for RPD. Are there contraindications or is RPD safe/superior for all indications? (3–8). Second, is there a minimum annual volume for safe implementation of RPD. There have been several studies describing volume criteria for RPD (3, 9–15). This review sets this out to describe the outcomes related to volume. Third, how long is the learning curve for RPD and how to minimize patient risk during this period? Multiple studies describe a considerable learning curve for RPD (9, 12–15). This highlights the importance of specific training programs for RPD to shorting the learning curve and minimize or even exclude any patient risk during this phase. This review describes the training programs for RPD and other robot pancreas surgery found in the literature (4, 16–18).
This review sets out to give insight about patient selection, volume criteria, and training programs for RPD and what needs to be studied for safe implementation of RPD.
Materials and Methods
We conducted a systematic literature search using PubMed, using MESH terms and free text related to patient selection, volume criteria, or training programs for RPD. In addition, studies describing training programs for other robotic pancreas surgery were included. We included randomized controlled trials, retrospective and prospective cohort studies, and systematic reviews. Only full text articles were included. Inaccessible articles, non-English articles, and publications before 1998 were excluded. Studies describing RPD but not in relation to patient selection and volume criteria were excluded. Finally, studies not describing robotic surgery were excluded except for training programs. The initial screening was performed on title and abstract in PubMed. Articles that were selected after title and abstract screening were screened on full text. The literature search is described in a Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart under “RESULTS” (Fig. 1).
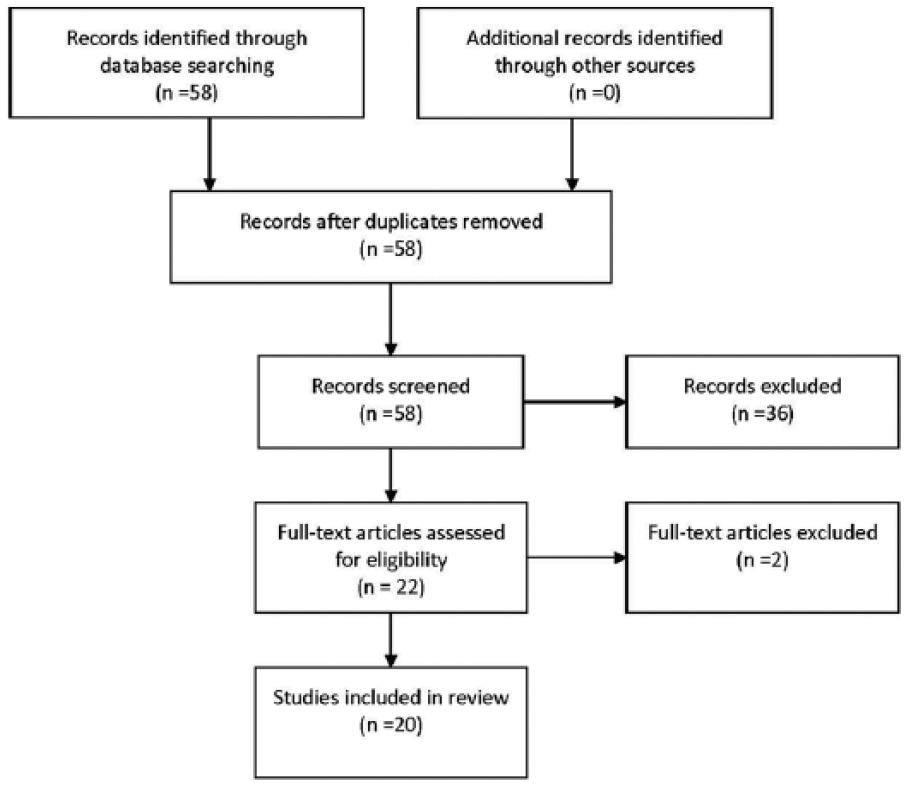
Flow chart.
Results
Overall, we identified 58 studies. After title and abstract screening, we were left with 22 studies to be further assessed for eligibility. We conducted a full text screening as stated in “MATERIALS AND METHODS”. After this we included 20 studies in our review. We reported these steps in the following flow chart (Fig. 1).
Patient Selection
We researched literature and outlined noteworthy recommendations for patient selection for RPD (Table 1).
Patient selection.
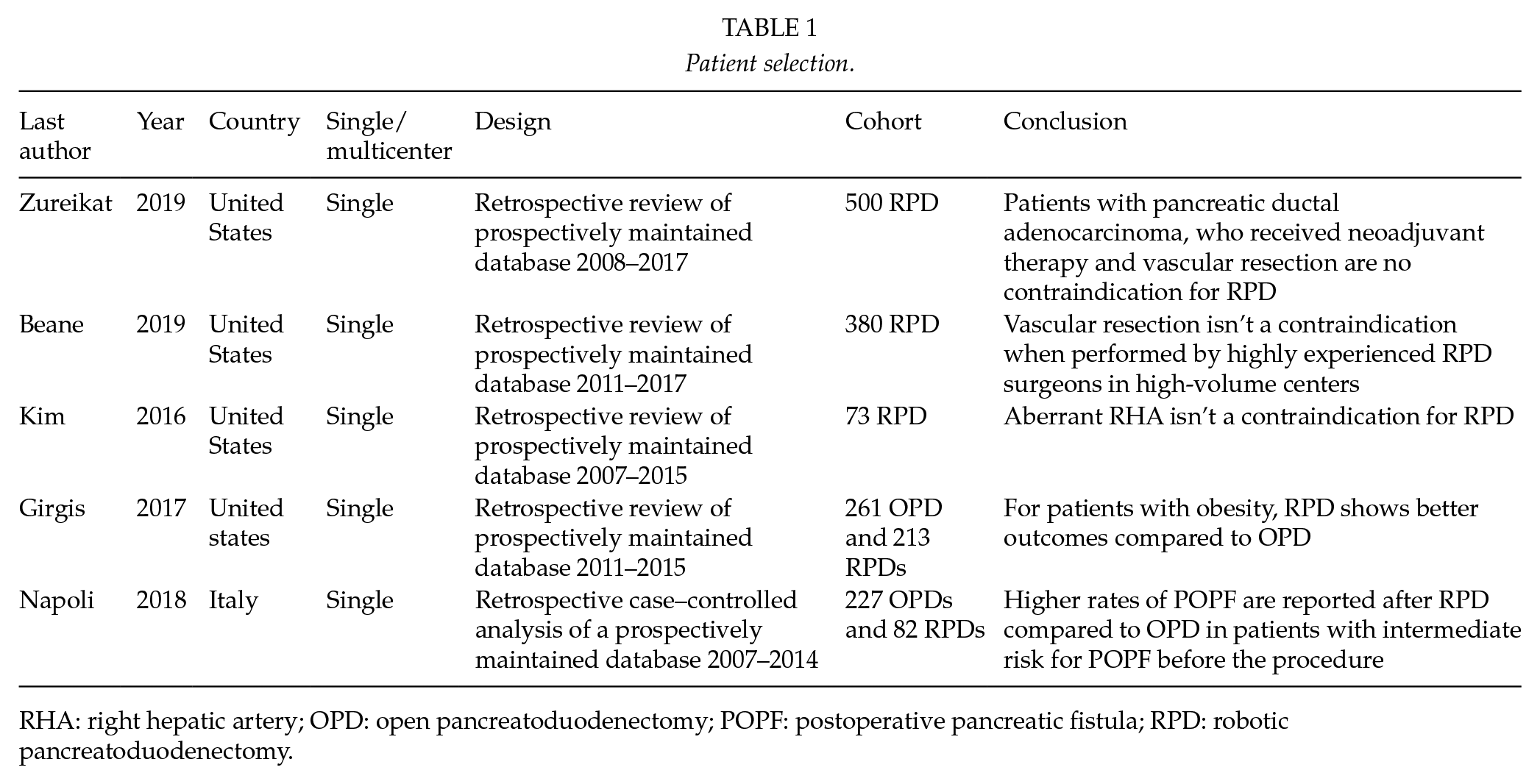
RHA: right hepatic artery; OPD: open pancreatoduodenectomy; POPF: postoperative pancreatic fistula; RPD: robotic pancreatoduodenectomy.
First, the evidence-based Miami guidelines set out some recommendations for patient selection (3). They do however state that there is limited comparative data available and further investigation is needed. These guidelines recommend that vascular resection should only be performed in high volume centers by highly experienced surgeons. Furthermore, they state that patients shouldn’t be excluded from RPD based on obesity, patient age, and previous abdominal surgery (3). Zureikat et al. (4) describes the first 500 RPDs of a single center and showed a continued improvement of outcomes after 200 cases. It also details that the expansion of patient selection criteria did not impact outcomes such as operative time, conversion to open, blood loss, and clinically relevant postoperative pancreatic fistula (POPF). There was an expansion of patients with higher Charlson comorbidity scores, patients with pancreatic ductal adenocarcinoma, patients who received neoadjuvant therapy, and vascular resections (4). Beane et al. researched the outcomes and learning curve of RPD with vascular resection. They showed a decrease in operation time after eight procedures and a learning curve around 35 cases. The group that performed this study first performed 80 RPDs before performing vascular resection. The results show comparable postoperative outcomes compared to RPD without vascular resection but did show an increase in mean estimated blood loss, conversion rate, and mean operation time (5). Kim et al. (6) demonstrated that an anatomical variation of the right hepatic artery (RHA) isn’t a contraindication for RPD with no significant differences in the pre- and postoperative outcomes compared to the normal RHA group. Obesity also isn’t a contraindication for RPD according to Girgis et al. (7) since they found a decrease in operative time, estimated blood loss, and POPF rate when compared to OPD with obesity. An increased number of POPF was seen after RPD compared to OPD in patients that were classified as intermediate risk for POPF. This study did report a limitation based on limited sample size (8).
Volume Criteria
The following studies reported on volume criteria (Table 2).
Volume criteria.
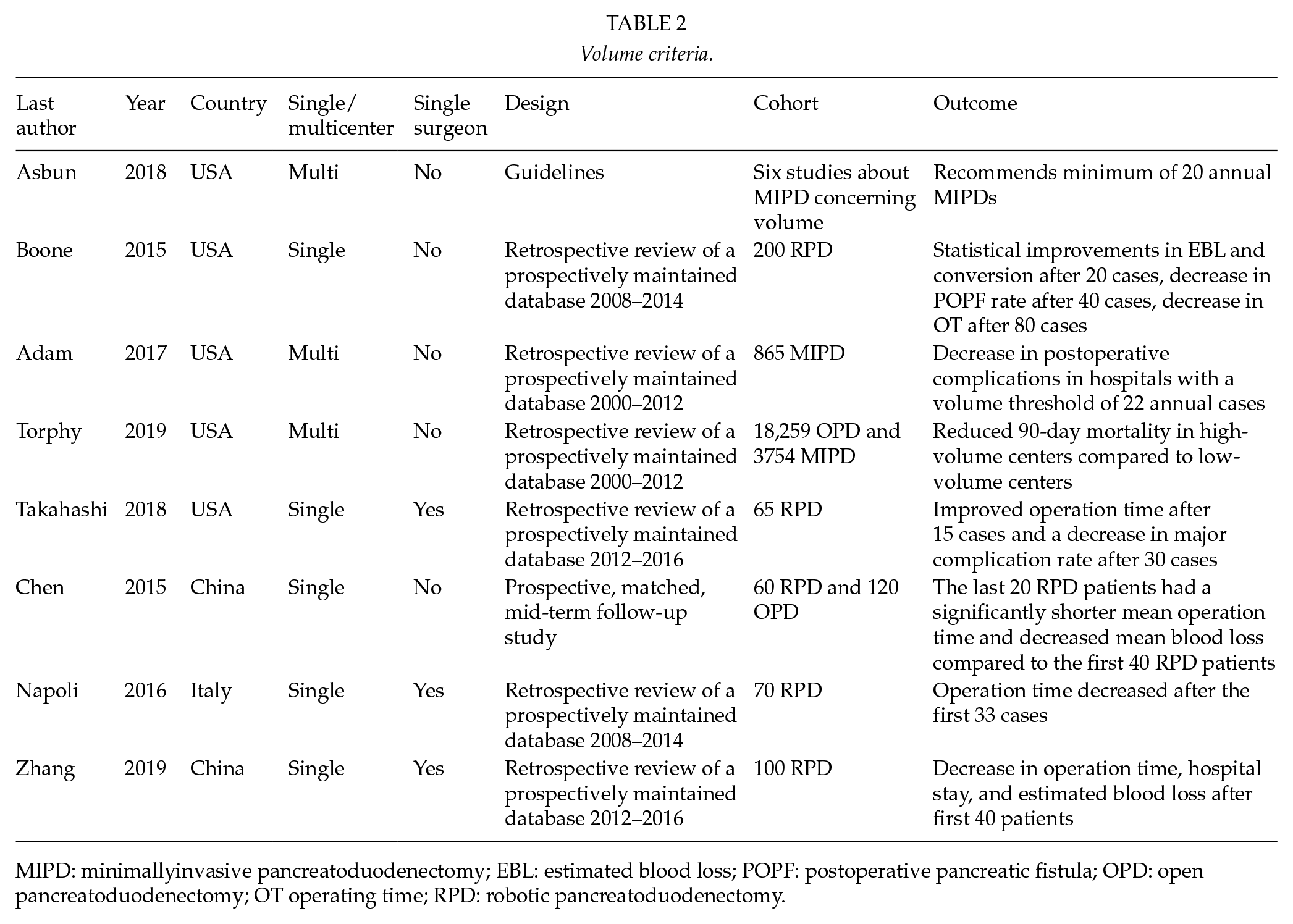
MIPD: minimallyinvasive pancreatoduodenectomy; EBL: estimated blood loss; POPF: postoperative pancreatic fistula; OPD: open pancreatoduodenectomy; OT operating time; RPD: robotic pancreatoduodenectomy.
The previously mentioned Miami guidelines describes a decreased complication rate when the annual volume per center is less than 20 RPD procedures and an increased mortality rate when this annual volume is less than 10 RPD procedures. It recommends an annual volume of 20 RPDs per center per year (3).
A retrospective study of 200 consecutive RPDs in an expert center reported improvements in estimated blood loss and conversions after overall 20 procedures, less pancreatic fistula after overall 40 procedures and reduced operative time after overall 80 procedures (9).
Adam et al. (10) found a higher annual hospital volume of RPD is associated with improved outcomes, especially with annual center volumes exceed 22. Torphy et al. (11) describe reduced 90-day mortality in high-volume centers compared to low-volume centers and a higher risk of conversion for low-volume centers. Takahashi et al. (12) reported an improved operation time after 15 cases and a decrease in major complication rate after 30 cases. A single-center study studied 60 patients and divided them in two groups, the first 40 and the last 20. The last 20 RPD patients had a significantly shorter mean operation time and decreased mean blood loss compared to the first 40 patients (13). In a single surgeon study, operation time decreased after 33 procedures and this was associated with a decrease in delayed gastric emptying rate. After 40 procedures readmission rate decreased (14). Finally, a single surgeon study describes a significant decrease in operation time, hospital stay, and estimated blood loss after first 40 patients (15).
Training Program
In the literature, we identified three training programs for minimally invsive pancreas surgery. One national training program for minimally invasive distal pancreatectomy (16) and one national training program in laparoscopic pancreatoduodenectomy (17).
This review focuses on RPD and there are two training programs for RPD described in the literature, Knab et al. (18) predicts that a proficiency-based training program can decrease the learning curve and therefore improve outcomes of RPD, outlined in (Fig. 2). Nota et al. (19) extrapolates the training program from Knab et al. to a nationwide training program in the Netherlands but outcomes have not been reported yet.
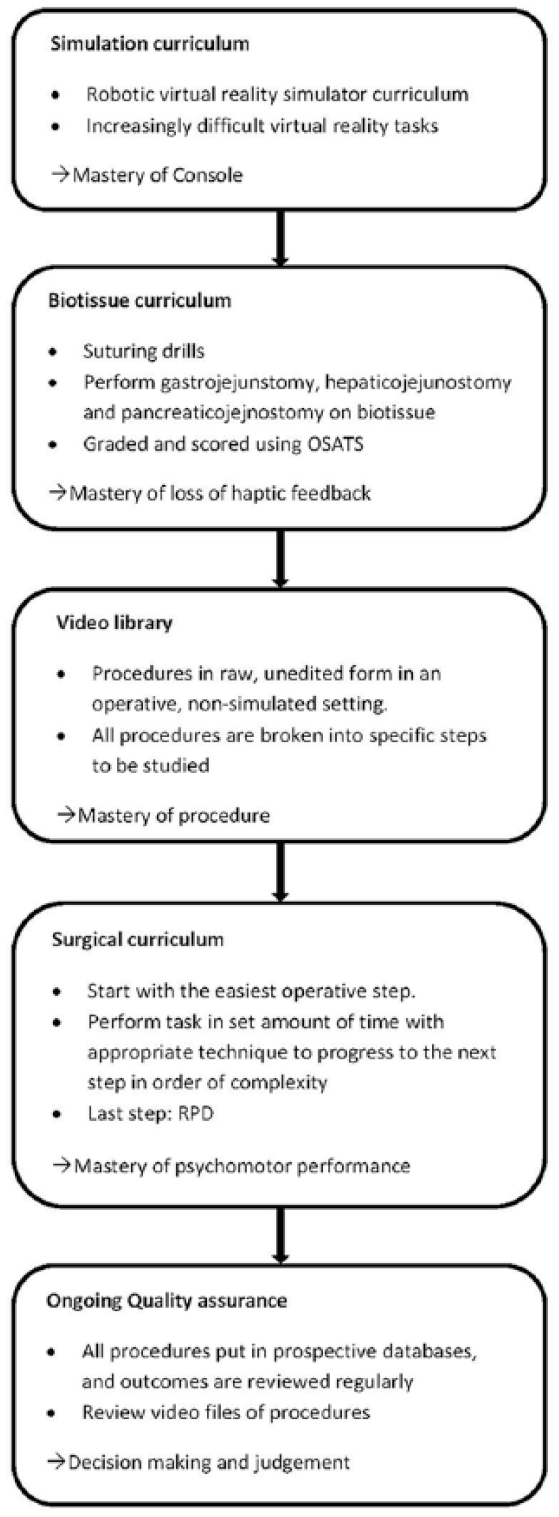
Training program according to Knab et al.
Zureikat et al. describes the effects of the training program of Knab et al. The integration of resident and fellow training in RPD did not negatively affect outcomes. Outcomes even continued to improve (18).
Summary
This review highlights current selection criteria, evidence on volume cut-offs and the learning curve in RPD and summarized evidence on training programs on RPD.
As described, there are no contraindications such as obesity, vascular resection, patients with pancreatic ductal adenocarcinoma, patients who received neoadjuvant therapy, patients with an anatomical variation of the RHA for RPD, as long as these procedures are performed by specifically trained and experienced RPD surgeons, working in high volume centers. A minimum annual center volume of 20 RPDs is recommend by several studies and seems reasonble in our view. Because approximately 40%–50% of patients will initially be eligible for RPD in centers as vascular resection will initially not be performed this means that the average annual PD volume should be around 50 procedures per year.
Improved outcomes and a shortened learning curve were reported after a training program for RPD. This calls for proficiency-based training program implemented in all international centers performing RPDs. Knab et al. and Zureikat et al. proved that a sufficient training program for RPD can significantly reduce learning curve and improve patient safety.
This review highlights the relevance of good patient selection, surgeon training, and sufficient annual volume. Based on this criteria we can built toward quality registries for RPD, such as recently started in Europe by the European Consortium on Minimally Invasive Pancreatic Surgery (E-MIPS; www.e-mips.com). Such quality registries, which already existed in separate countries (20) as Germany and the Netherlands can report on the outcomes of RPD and allows centers to reflect on their own outcomes in relation to those of others. These systems are highly supported by both the Miami guidelines and international organizations for hepato-pancreato-biliary (HPB) surgery.
Footnotes
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MJW Zwart received funding from the board of directors from the Amsterdam UMC (location AMC) for studies on safe implementation of robot-assisted and laparoscopic pancreatic surgery. He also received funding from the Dutch Digestive Foundation, for studies on the before mentioned topics.
*
Shared senior authorship.
