Abstract
Purpose:
Robotic-assisted urologic stone removal and any potential histopathological effects of this approach have not been well studied. Here we assess safety and user experience of the novel MONARCH™ Platform, Urology, in performing both robotic-assisted ureteroscopy (URS) and percutaneous nephrolithotomy (PCNL) within a preclinical setting, alongside conventional devices.
Materials and Methods:
Two endourologists conducted six simulated robotic-assisted URS and six robotic-assisted PCNL in a porcine model, comparing these with conventional approaches. The primary objectives were (1) perceived ease of completion as rated by the endourologists using a numerical 1–4 rating scale; (2) occurrence of adverse safety events as determined by the operating endourologists; (3) assessment of contrast extravasation seen on post-operative retrograde pyelogram; and (4) histopathological evaluation of the porcine models’ urinary tracts. Mantel-Haenszel chi-square test was used, with p < 0.05 considered significant.
Results:
Tasks within URS and PCNL exhibited comparable ease of completion score means (median of all scores 4 in conventional URS and 4 in robot-assisted URS, p = 0.131; 3.17 in conventional PCNL and 4 in robot-assisted PCNL, p = 0.258). No safety events were observed by the endourologist during URS or PCNL procedures for either the robotic-assisted or conventional devices. In post-procedure pyelograms, none of the robotic-assisted cohort had more than minimal contrast extravasation, statistically similar to conventional devices (URS: median [range]; conventional, 0 [0–2] vs. robotic-assisted, 0 [0–1], p = 0.337; and PCNL: conventional, 0 [0–2] vs. robotic-assisted, 0 [0–1], p = 0.379). Blinded pathology assessment demonstrated no biologically significant nor clinically relevant differences between robotic-assisted and conventional devices in any category.
Conclusions:
This study demonstrates that the MONARCH™ Platform in Urology has a safety profile comparable with conventional devices and a trend of easier completion of some tasks. This foundational study establishes the feasibility of a single platform to complete both robotic-assisted URS and PCNL, as a new treatment paradigm for urologic stone management.
Keywords
Introduction
The increasing prevalence and incidence of urologic stone disease has driven advancements in technologies aimed at improving the treatment of kidney stones. In recent years, technologies utilizing suction to assist with evacuating stone fragments and dust have emerged to improve stone clearance.1–3 In addition, though not in widespread use, additional technologies have been developed to simplify gaining percutaneous access for percutaneous nephrolithotomy (PCNL). 4 Novel ureteroscopes and other devices also have been developed with advanced navigation and steering technologies to improve control and precision in stone clearance procedures.3,4 Despite the promising potential of these technologies, there are still limitations in adoption, evaluations of usability, and clinical outcomes dissemination.
One technology that aims to address these gaps is the MONARCH™ Platform, Urology (Johnson & Johnson MedTech Surgery, Santa Clara, CA), an United States Food and Drug Administration (FDA) 510(k)-cleared first-of-its-kind robotic-assisted system that enables urologists to perform ureteroscopy (URS) and also a PCNL procedure that combines retrograde lithotripsy with antegrade suction-driven stone removal. 5 For ureteroscopic procedures, the MONARCH platform allows urologists to articulate a flexible ureteroscope, basket, and laser fiber, as well as manage fluid through the system’s irrigation and aspiration system with a hand-held controller. The system incorporates a novel electromagnetic guidance system to facilitate gaining percutaneous access into the kidney, enabling urologists to use this platform to remove kidney stones percutaneously. Altogether, this platform combines elements of PCNL with ureteroscopic lithotripsy for kidney stone clearance.
Prior reports have described the clinical use of an earlier (non-robotic) version of this system and also the clinical use of the current MONARCH Platform in a small cohort of patients for the combined mini-PCNL with ureteroscopic lithotripsy procedure.5,6 These studies established safety in a clinical environment. This investigation aims to add histopathological analyses of samples to assess safety at a microscopic level within a live porcine animal model. We directly compare use of the MONARCH robotic-assisted system with that of traditional devices used for URS and PCNL for an evaluation of safety and usability.
Materials and Methods
Study design
Within this study we evaluated ease of use and safety of the MONARCH system while performing simulated URS and PCNL procedures, in comparison with that with conventional manual devices. These simulated URS and PCNL procedures were conducted in a porcine animal model, under protocols approved by the Johnson & Johnson Institutional Animal Care and Use Committee. In total, 12 female Yorkshire-cross swine, 55–75 kg, were utilized for these assessments. Six animals were used for assessing the simulated URS procedures, and a different six animals were used to assess the simulated PCNL procedures. The simulated procedures were conducted such that a robotic-assisted procedure and a conventional procedure were both evaluated on contralateral kidneys within the same animal, where sidedness was randomized between technologies. For PCNL, caliceal access selection was at the discretion of the urologist. The study workflow is depicted in Figure 1. As these models did not possess, nor were implanted with, kidney stones, the simulated procedures encompassed the workflow steps associated with URS and PCNL procedures, without the removal of any kidney stones. Ureteral access sheaths (12/14 F and 11/13 F, lengths 36–46 cm) were selected according to animal size and were placed into the non-pre-stented ureters prior to URS procedures (both conventional and robotic-assisted) and also for robotic-assisted PCNL procedures. All procedures were performed by board-certified urologists and endourologists (authors T.C. and M.L.S.), who each received approximately 3 hours of didactic and hands-on training on the robotic system prior to its use in the study. This study was conducted, and data integrity maintained, under FDA Good Laboratory Practice regulations (21 CRF 58).
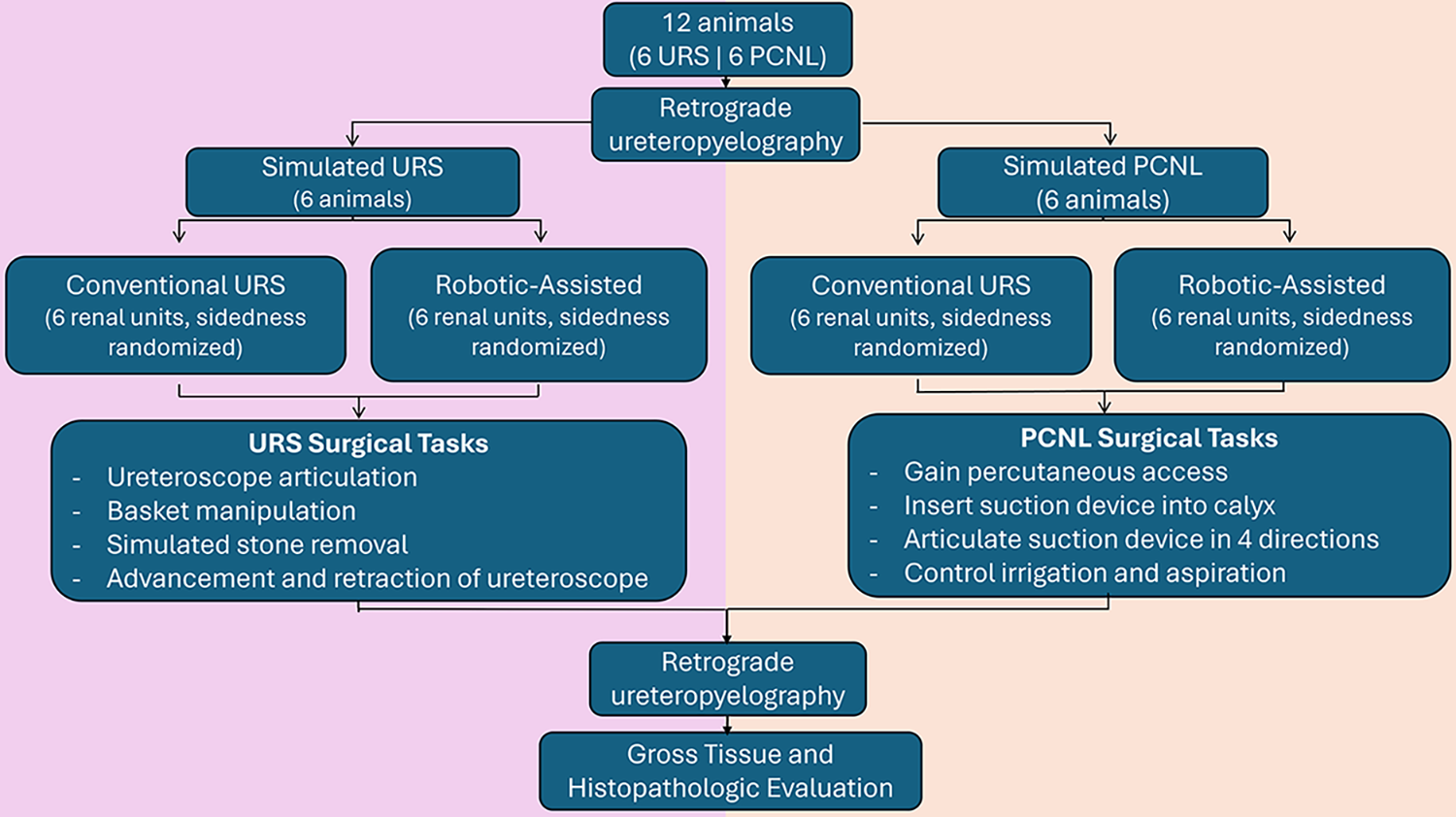
Schematic of the study workflow and assessments comparing robotic-assisted and conventional procedural approaches for URS and PCNL. PCNL = percutaneous nephrolithotomy; URS = ureteroscopy.
Simulated ureteroscopy procedures
Conventional approach
Ureteroscopy with conventional devices was performed using LithoVue™ Ureteroscope (Boston Scientific, Marlborough, MA) and ZeroTip™ Nitinol Stone Retrieval Basket (Boston Scientific, Marlborough, MA). Irrigation was managed with FluidSmart Fluid Management System (Thermedx LLC, Cleveland, OH).
Robotic-assisted approach
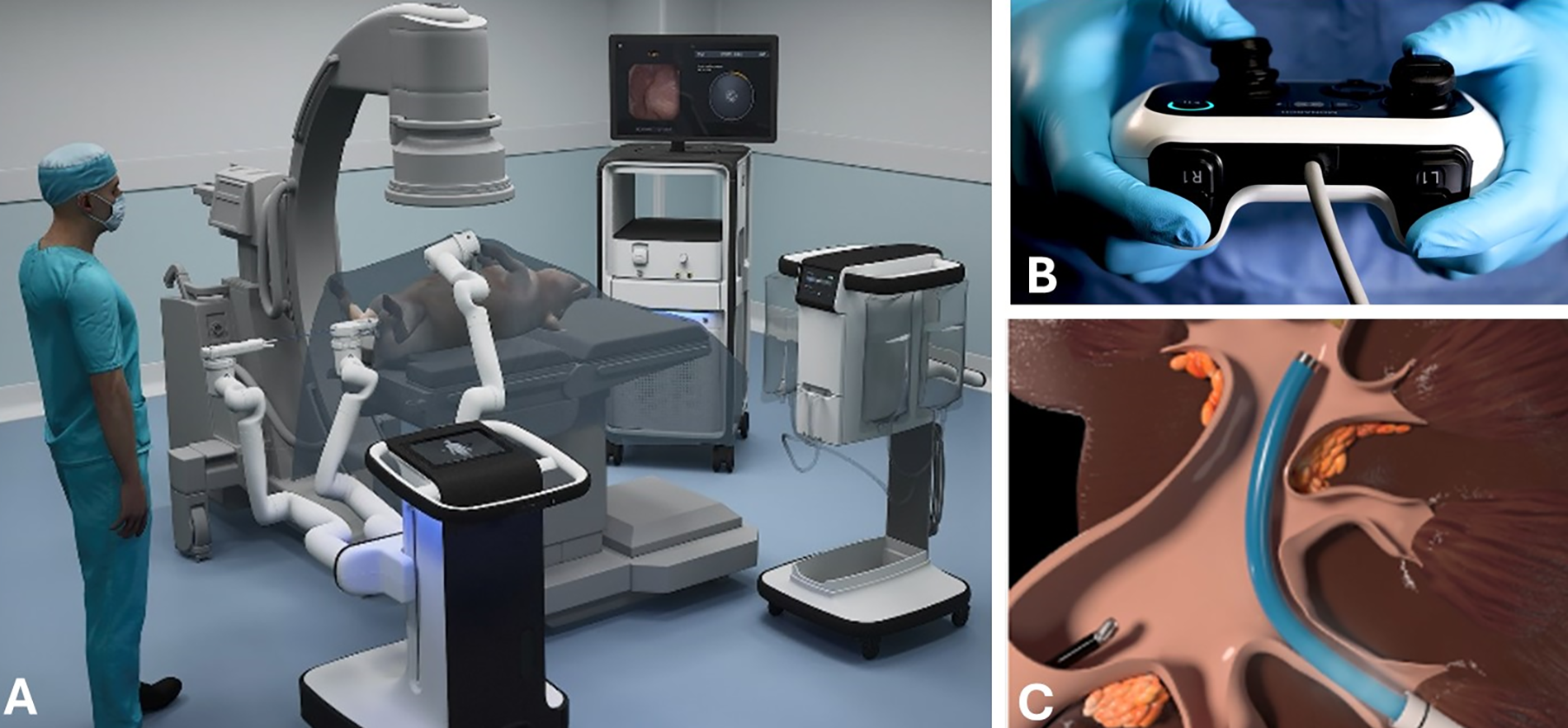
Robotic-assisted ureteroscopy was performed with the MONARCH platform, as shown in Figure 2. Within this platform, a flexible ureteroscope was docked into two robotic arms. The working channel of the ureteroscope accommodated a basket for simulated stone removal. The endourologist used a handheld controller to manipulate the ureteroscope, basket, and the proprietary irrigation system to complete the procedure.
Surgical tasks
The endourologist performed the same series of tasks using both conventional and robotic-assisted approaches. These tasks included systematically articulating the ureteroscope through the renal collecting system, advancing a basket through the working channel while in upper and lower poles, retracting the scope from the renal pelvis to exit the urinary system (simulating stone removal), and advancing the ureteroscope back to the renal pelvis through the ureteral access sheath, repeating the retraction and advancement 10 times.
Simulated percutaneous nephrolithotomy
Conventional approach
The endourologist obtained percutaneous access to the renal collecting system using the Percutaneous Introducer System (Boston Scientific, Marlborough, MA), which contained an 18-gauge needle, using a combination of fluoroscopy and ultrasound guidance. The percutaneous tract then was dilated to 30 F via a conventional technique over a guidewire. An Amplatz renal sheath then was placed into the collecting system. The FluidSmart Fluid Management System (Thermedx LLC, Cleveland, OH) supplied irrigation through a 26 F nephroscope (Karl Storz, Tuttlingen, Germany), and aspiration was controlled with a LithAssist suction device (Cook Medical, Bloomington, IN).
Robotic-assisted approach
First, the endourologist set up the MONARCH platform similarly to that for a conventional ureteroscopy procedure, as described above, where retrograde renal access was established by driving the MONARCH ureteroscope through the ureteral access sheath into the ipsilateral kidney. Then, the endourologist used the guidance provided by the MONARCH platform’s electromagnetic field generator (visualized from a graphical use interface), endoscopic visualization, and a combination of fluoroscopic and ultrasound guidance to position the needle for percutaneous access into the kidney. The tract was then sequentially dilated, followed by placement of the platform’s 23.5 F percutaneous access sheath. A third robotic arm at the percutaneous site was used to robotically steer a suction catheter. During the procedure, all instruments, as well as fluid irrigation and aspiration, are manipulated by the endourologist using the handheld controller. The setup is shown in Figure 2.
Surgical tasks
Using both robotic-assisted and conventional devices, the endourologist performed the following tasks: inserting the respective suction device into an adjacent calyx, then articulating the suction catheter in four directions (cranial, caudal, anterior, and posterior) within the calyx and demonstrating control of the suction by switching to maximal pressure.
Assessments
Ease of completion
After each task performed by the endourologist, they were asked to complete a 1–4 numerical rating scale judging the ease of completion of the task. Scoring was as follows: (1) unable to complete task; (2) able to complete task with significant workaround or inconvenience; (3) able to complete task with minor workaround; or (4) able to complete the task with no workaround. Workaround was defined as an alternate workflow when the intended steps in the protocol could not be followed.
Adverse safety events
The endourologist performing the procedure evaluated safety events. The endourologist graded safety events as negligible to severe and noted whether the safety issue was caused by the device and whether the event occurred during a specific task or during transition time.
Ureteropyelogram
Before and after the simulated procedures, retrograde ureteropyelography was performed. Post-procedure pyelograms were evaluated by the endourologist in assessment for extravasation on a scale of 0–3: (0) no contrast extravasation; (1) minimal contrast extravasation from calyces and/or ureter; (2) moderate/significant contrast extravasation from calyces and ureter; or (3) ureteral avulsion or severe collecting system perforation.
Histopathological evaluation
After completion of procedures, animals were administered heparin to prevent postmortem clots and then euthanized. A veterinary pathologist, blinded to treatment groups, performed postmortem necropsy and histopathological assessments. The peritoneal cavity and retroperitoneum were evaluated for signs of effusion or hemorrhage on gross necropsy. The kidneys and ureters were examined for contusion, laceration, perforation, or mucosal trauma. Tissue samples of ureter, renal pelvis, and kidney (including additional tissue sections at the puncture site for animals who underwent PNCL) were fixed, sectioned, mounted onto glass slides, and stained with hematoxylin and eosin. Light microscopy histopathological examination focused on acute pathology attributed to the test procedure. Semiquantitative scoring of relevant and/or significant acute pathological features (inflammation, hemorrhage, necrosis, perforating injury, and ureter erosion) was performed on each histology section using the grading criteria outlined in Table 1.
Histopathological Scoring System Used by Pathologists Reviewing Urologic Tissue After Procedure

Statistical analysis
Comparisons between task completion scores for the robotic device and conventional devices were made using a Mantel-Haenszel chi-square test, with p < 0.05 considered statistically significant.
Results
Ease of completion
The results of the ratings (1–4) of each task’s ease of completion as performed by participating endourologists are collated in Table 2. There was no statistically significant difference between the MONARCH™ or conventional devices for any task (p > 0.05 for all tasks).
Score Results of Ease of Use as Rated by Participating Endourologists
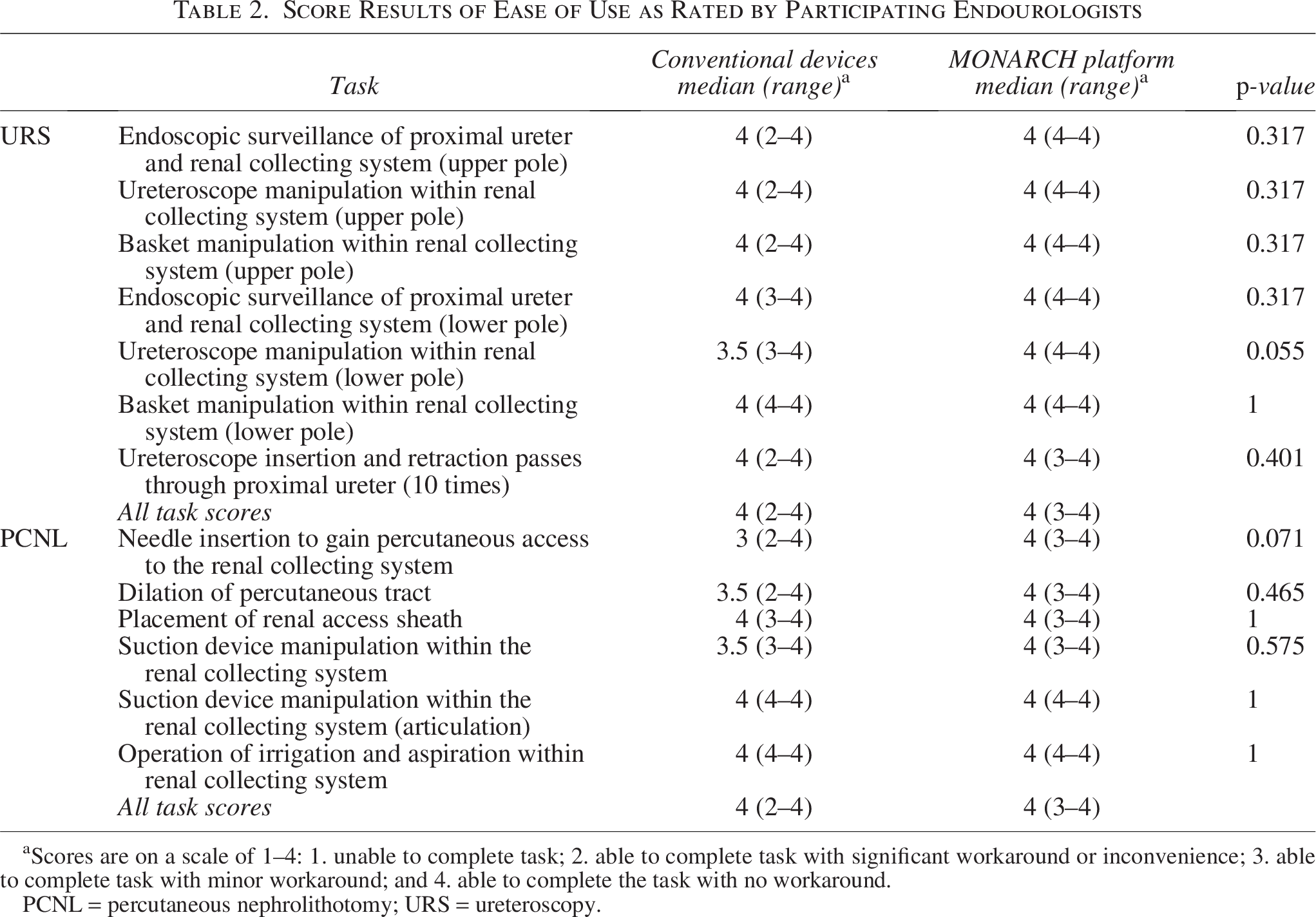
Scores are on a scale of 1–4: 1. unable to complete task; 2. able to complete task with significant workaround or inconvenience; 3. able to complete task with minor workaround; and 4. able to complete the task with no workaround.
PCNL = percutaneous nephrolithotomy; URS = ureteroscopy.
Safety
Adverse safety events
There were no adverse events during the URS procedures using the MONARCH or conventional devices. Similarly, there were no device-related adverse events during the PCNL; however, one animal was excluded from the study due to a protocol deviation in which the endourologist did not use fluoroscopy or ultrasound guidance while obtaining percutaneous access. To meet the minimum number of animals required by the protocol, a replacement animal was subsequently included.
Ureteropyelogram
All renal units were found to have no contrast extravasation on fluoroscopic retrograde pyelography prior to conducting the simulated procedures. There were no significant differences regarding contrast extravasation between the two device groups following URS (median [range]; conventional, 0 [0–2] vs. robotic-assisted, 0 [0–1]; p = 0.337).
In PCNL, similarly, there was no significant difference between conventional devices or the MONARCH platform (median [range]; conventional, 0 [0–2] vs. robotic-assisted, 0 [0–1]; p = 0.379).
Gross Tissue and Histopathology Evaluation
Following URS, assessments of all gross tissue and histopathologic findings examined (hemorrhage, erosion, inflammation, necrosis, injury to the muscular wall, or perforating injury) were biologically and clinically similar between treatment groups (Table 3). The most pronounced finding was the presence of mild to moderate erosion in the ureters (Fig. 3), which was comparable between treatment groups. In addition, one ureter of the conventional device group and one ureter of the robotic-assisted device group both exhibited compression of the muscular wall, which was not considered clinically significant and was thought to be associated with the use of ureteral access sheath rather than the ureteroscopy devices themselves. Within the kidneys the most significant finding was the presence of mild to moderate hemorrhage, which was not considered clinically significant nor different between device groups.
Results of Semiquantitative Histopathological Scoring
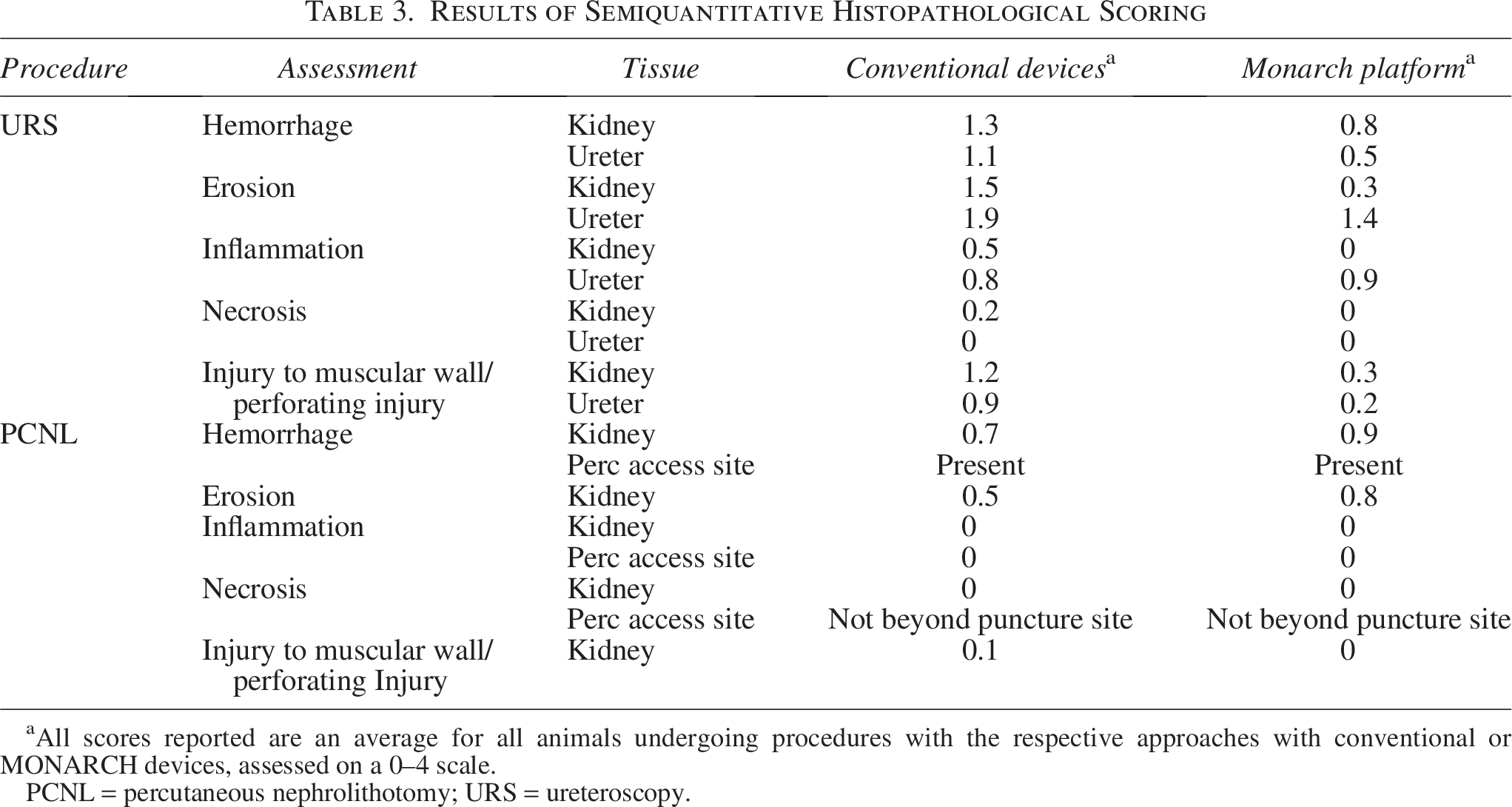
All scores reported are an average for all animals undergoing procedures with the respective approaches with conventional or MONARCH devices, assessed on a 0–4 scale.
PCNL = percutaneous nephrolithotomy; URS = ureteroscopy.
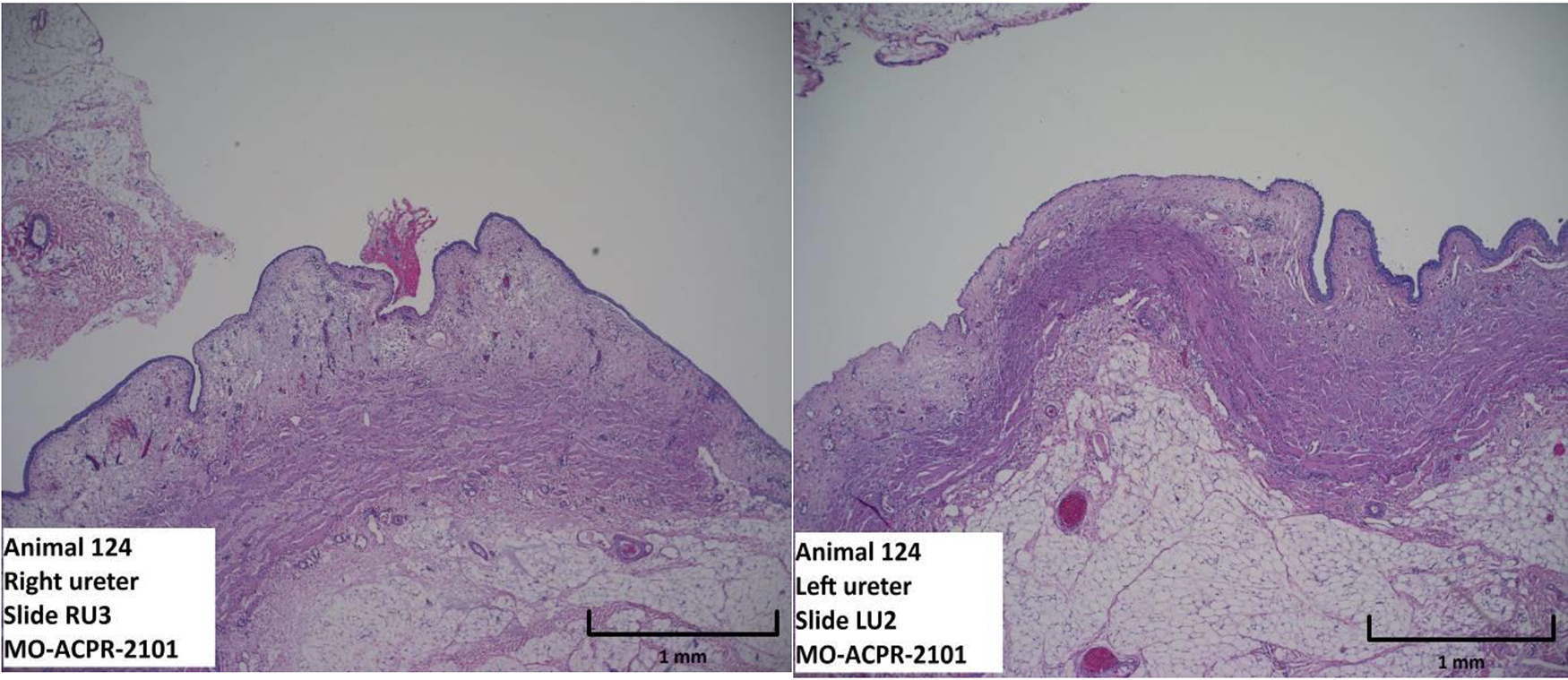
Photomicrograph with hematoxylin and eosin stain representing comparable minimal erosion of the ureter following ureteroscopy (URS) with the MONARCH™ Platform, Urology (left image), and following URS with conventional devices (right image).
Similarly, following PCNL, no biologically significant nor clinically relevant differences between kidneys were observed between the MONARCH and conventional devices. Percutaneous access sites demonstrated similar acute pathology in both groups and were associated with an anticipated degree of hemorrhage (blood clot) that filled the tract and entered the renal pelvis. There was minimal to mild hemorrhage and trauma beyond the percutaneous access puncture tract in both groups. In both groups, the pathology findings were reported as expected from a PCNL and expected to heal spontaneously without clinical complications or sequela. For the semiquantitative histopathological scoring, the average score for any category of pathological finding was less than 1, indicating minimal injury at greatest (Table 3).
Discussion
In this study, we demonstrate comparable safety between the MONARCH Platform and conventional devices, based on the collective observations of adverse events, urothelial extravasation, and gross and histopathological examination of harvested tissues. This is noteworthy in the context that the use of robotic-assisted technologies for stone removal is emerging in a field where endourologists are largely accustomed to the tactile feedback that comes with manual control of surgical instruments. Although the MONARCH Platform does not provide this tactile feedback, we find comparable safety—even at the gross and microscopic tissue levels—between this technology and conventional devices. This study is the first known assessment of gross and histopathological tissue following the use of robotic-assisted instruments for stone removal procedures and demonstrates safety of the MONARCH Platform.
High intrarenal pressures are associated with retrograde flow and endothelial damage and are a topic of recent interest within the field. 7 The MONARCH Platform has an integrated irrigation and aspiration system that is intended to help endourologists manage fluid level. Although this investigation did not measure intrarenal pressures directly, we did not observe clinically significant endothelial damage to tissues following the use of the MONARCH Platform.
This study also finds comparable ease of completion of both simulated URS and PCNL procedures between MONARCH and conventional devices. This is remarkable, considering the endourologists had only approximately 3 hours of training on the robotic system prior to its use to conduct the simulated procedures. This suggests that, despite the differences in user interaction with the MONARCH user controls compared with that with traditional methods, the learning curve is small and the users’ ability to perform the procedure remains unaffected. The MONARCH Platform incorporates several technologies that may facilitate stone removal procedures. These include providing a single urologist simultaneous control of retrograde and antegrade instruments, guided percutaneous access, and management of irrigation and aspiration throughout the procedures.
In recent years, the field of endourology has expanded to include multiple robotic-assisted technologies: flexible ureteroscopy, endoscopic combined intrarenal surgery, and percutaneous access to successfully navigate the urinary upper tracts to treat urinary stones.4,8,9 Robotic assistance for percutaneous renal access, with usage of a robotic arm or electromagnetic navigation, has separately been described in preclinical models in the past.10,11 Recent novel steerable stone aspiration systems have shown promise in removing residual fragments as a part of a laser lithotripsy procedure. 3 The MONARCH platform innovatively incorporates several of these technologies into one robotic-assisted system that can perform both URS and a PCNL procedure that combines ureteroscopic lithotripsy with suction-based percutaneous stone removal.
This investigation has several limitations. Porcine models may not fully translate the range of human anatomies found in a clinical setting, as porcine model exhibits variation in anatomical structures, with smaller kidneys and a more acute angle between the caudal infundibulum and renal pelvis. 12 Furthermore, as these animal models did not have any kidney stones, our comparisons between device groups did not directly assess the stone removal portion of the workflow step. Expanded, or additional, study in a clinical setting that includes this portion of the workflow would allow for further assessment of procedural metrics (e.g., procedure duration, radiation use, etc.). Despite these differences, the porcine kidney still serves as a suitable animal model for endourologic research, and the swine model is one of the most commonly used experimental models within the field. 12
This study was also limited by assessments from only two endourologist operators, with each only performing either only the URS or only the PCNL procedures. Further study with additional endourologists, as well as endourologists with varying levels of experience or practice techniques, may provide additional nuances to the comparisons between device groups, especially for subjective assessments related to ease of use. In addition, future assessments of ureteral injury should incorporate the Post-Ureteroscopy Lesion Scale 13 to provide comparability to injury assessments more commonly performed in a clinical setting or otherwise in a chronic animal study. 14 However, these findings from two experienced endourologists support the relative comparability of device groups and the histopathological safety of the MONARCH Platform. While there are early data regarding the safety of the MONARCH platform in a clinical setting, future directions should be aimed at larger studies further evaluating the MONARCH Platform across real-world variation of human anatomy, with specific outcomes of interest such as stone clearance rates, percutaneous access success, procedure duration, and radiation usage.
Conclusions
The MONARCH Platform is a novel robotic-assisted device that may provide advantages for stone removal over conventional devices, including the use of a robotically steerable flexible suction catheter and built-in guidance for obtaining percutaneous access. However, the use of robotics for stone removal is still in its infancy. This preclinical study finds comparable safety outcomes and ease of use assessment in performing simulated URS and PCNL procedures with MONARCH platform and conventional manual devices. Importantly, this study underscores histopathological safety following the use of the MONARCH Platform.
Authors’ Contributions
T.C.: Conceptualization, investigation, and writing—review and editing. B.C.: Formal analysis and writing—original draft, review and editing. C.G.: Formal analysis and writing—original draft, review and editing. N.L.S.: Supervision and writing—review and editing. M.L.: Investigation and writing—review and editing. P.M.: Formal analysis and writing—review and editing. M.L.S.: Conceptualization, investigation, and writing—review and editing.
Footnotes
Acknowledgments
The authors also acknowledge Lauren Hathaway and Rachel Chok for their contributions to the development, investigation, and initial reporting of this laboratory study, as well as veterinary pathologist Taylor Spangler, DVM, DACVP.
Funding Information
This study was funded by Johnson & Johnson MedTech Surgery.
Author Disclosure Statements
Thomas Chi is a paid consultant at Johnson & Johnson MedTech Surgery. Brandon Cowan and Camilla Gomes: All support for time spent preparing this article was funded by Johnson & Johnson MedTech Surgery via salary; no other incentives such as stock or stock options, receipt of equipment, gifts, or other financial or other nonfinancial incentives were obtained. Nancy Sehgel is a full-time employee of Johnson & Johnson MedTech Surgery. Maggie Lin is a full-time employee of Johnson & Johnson MedTech Surgery. Paul Morris is a full-time employee of Johnson & Johnson MedTech Surgery and has stock options per compensation agreement as an employee. Marshall Stoller is a paid consultant of Johnson & Johnson MedTech Surgery.
